Abstract
Background
Guggulutiktam Kashayam (GTK) is a water decoction used in Ayurveda for the treatment of various diseases like skin diseases, ulcers, anemia, thyroid problems, stress, liver disease, and leucorrhoea. It is a polyherbal formulation prepared from specific parts of 28 medicinal plants.
Objectives
The study aimed at a comparative phytochemical evaluation of an important polyherbal formulation with its ingredient plants.
Materials and Methods
Chemical profiling of GTK has been carried out in comparison with its ingredient plants. Spectrophotometric estimation of major class compounds and high-performance thin-layer chromatography (HPTLC) profiling were done using a standardized protocol.
Results
The results of the study showed that the phytochemical contents of the finished formulation are different from those of the ingredient plants, which might be due to the numerous synergistic and antagonistic interactions of numerous chemical components extracted from different ingredient drugs. HPTLC-based comparative chromatograms showed the differences and similarities in certain compounds that are specific to some of the plant ingredients.
Conclusion
The study established the cumulative chemical interactions of various phytochemicals during the preparation of an important polyherbal formula used in Ayurveda.
Introduction
The majority of Ayurvedic formulations are polyherbal in nature and contain several ingredients in specific proportions, as prescribed in the ancient Ayurvedic classics. Polyherbal formulations (PHFs) are considered to be more active due to the presence of multiple ingredients, which is commonly known as polyherbalism (Sulaiman et al., 2021). Guggulutiktam Kashayam (GTK) is one of the well-known classical PHFs used to treat skin diseases, ulcers, anemia, thyroid problems, stress, liver disease, and leucorrhoea. Many phytochemicals, including phenolics, flavonoids, coumarins, and catechins, have been reported from this formulation (Sulaiman et al., 2020). The phytochemicals present in the finished PHFs are cumulative chemical entities after many synergistic and antagonistic chemical interactions. The chemical pattern of any finished formulation is different from that of its individual ingredient plants due to the herb–herb interaction that normally leads to the synergistic chemical transformation of many of the phytochemicals extracted from the various ingredient raw drugs. The range of such interactions depends on the nature of the raw drugs used and the method of preparation. In most cases, Ayurvedic formulations are prepared by prolonged heating of the different raw drugs in boiling water. Another phenomenon, antagonism, in which the effect of some compounds is nullified by other constituents, is also happening during the PHF preparation (Ministry of Health and Family Welfare, 2003).
The therapeutic efficacy of any herbal preparation depends on the various chemical constituents present in it that are capable of possessing the desired biological action. Chemical profiling of finished PHFs is one of the most reliable methods to correlate their active constituents with their pharmacological activities. Comparative phytochemical profiling with raw ingredient drugs may provide a preliminary understanding of the synergism and antagonism occurring during the preparation of the formulation. High-performance thin-layer chromatography (HPTLC) is a strong analytical tool for the comparative profiling of PHFs with their raw drugs. Chromatograms with good resolution can be used to distinguish the presence or absence of individual chemical constituents in PHF with respect to the individual ingredient plants. HPTLC techniques have been widely used for the comparative chemical profiling of plants and herbal products (Akhtar et al., 2022; Parasuraman et al., 2014; Singh et al., 2014). In this study, a comparative chemical evaluation of an Ayurvedic PHF has been carried out by various spectrophotometric and HPTLC analyses. The study has been conducted to ensure the quality of the selected finished formulation with respect to its ingredients. A comparative phytochemical evaluation of this formulation with its ingredient drugs has not yet been reported.
Materials and Methods
GTK was obtained from the Product Development Department of the institute. Aluminium-backed precoated HPTLC plates Si 60F254, and all the analytical-grade solvents were procured from Merck India Pvt. Ltd. Gallic acid was procured from Sigma Aldrich Chemicals.
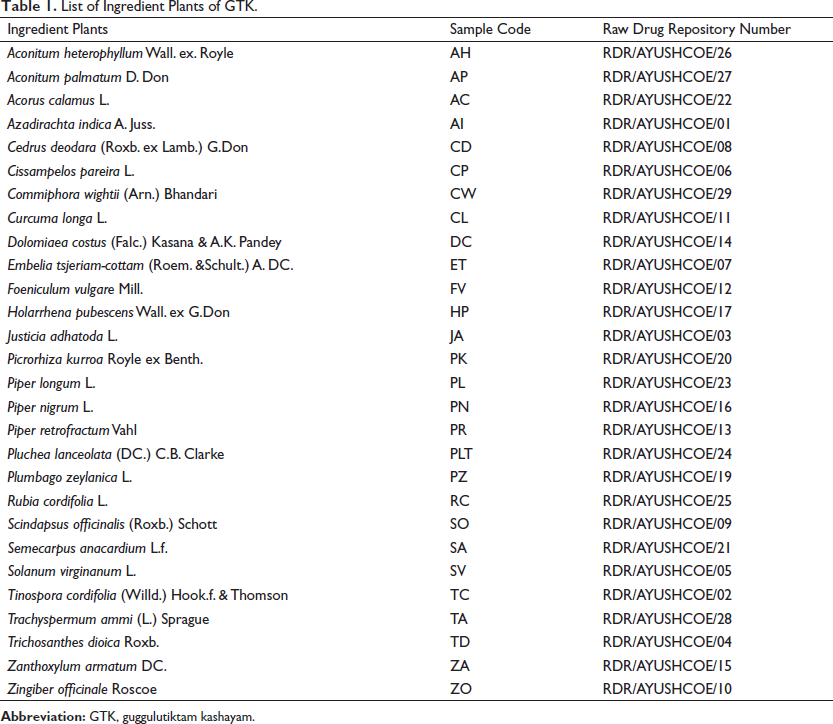
Ingredient plant materials for GTK were obtained from the Materials Department of the institute, and all the materials were authenticated by the Pharmacognosy Division of the institute. Voucher specimens were deposited in the Raw Drug Repository (Table 1).
List of Ingredient Plants of GTK.
Preparation of Extracts
Ten grams of each of the shade-dried materials were ground into coarse powder and refluxed with 100 mL of distilled water for 6 h. It was then filtered and kept in the refrigerator until various phytochemical analyses.
Physicochemical Screening
Physicochemical tests such as total ash, acid-insoluble ash, water-insoluble ash, sulfated ash, alcohol-soluble extractive, and water-soluble extractive of all the ingredient plants were done as per the standard procedures of the Ayurvedic Pharmacopeia of India (The Ayurvedic Pharmacopoeia of India, 2001).
Estimation of Phenolics
Major phytochemicals were estimated using an ultraviolet (UV)-visible spectrophotometer (Shimadzu, Japan). Total phenolic content was estimated for GTK and its ingredient plants. The Folin-Ciocalteu method was used for the total phenol estimation (Singleton & Rossi, 1965; Sulaiman & Balachandran, 2012; Sulaiman et al., 2014; Sulaiman et al., 2020). The values were calculated with respect to standard gallic acid.
HPTLC Profiling
HPTLC analysis was performed using a CAMAG HPTLC system (Switzerland) equipped with an automatic sample applicator (CAMAG ATS-IV) on an aluminium-backed, precoated silica gel 60F254 HPTLC plate (Merck India). The stock solution of GTK was prepared by sonicating 2 g in 10 mL of distilled water using an ultrasound bath. The mobile phase was optimized as per the previous method with slight modifications (Sulaiman et al., 2020; Sulaiman et al., 2022). Ideal separation was obtained with the mobile phase containing toluene, ethyl acetate, and methanol in the ratio 7:3:1. Comparative HPTLC profiling of aqueous extracts of ingredient plants along with GTK was done in triplicate with the same mobile phase. The chromatogram was developed in a twin-trough glass chamber presaturated for 5 min at room temperature with the optimized mobile phase. After development, the plates were dried and scanned using CAMAG HPTLC Scanner-III at UV-254, UV-366, and in visible light after derivatizing with anisaldehydesulphuric acid reagent. All the developed plates were documented by the CAMAG HPTLC photo-documentation system.
Results
This study was carried out to identify the major class of chemical compounds in the extracts and distinguish the presence or absence of individual chemical constituents in PHF with respect to the individual ingredient plants.
Physicochemical Screening
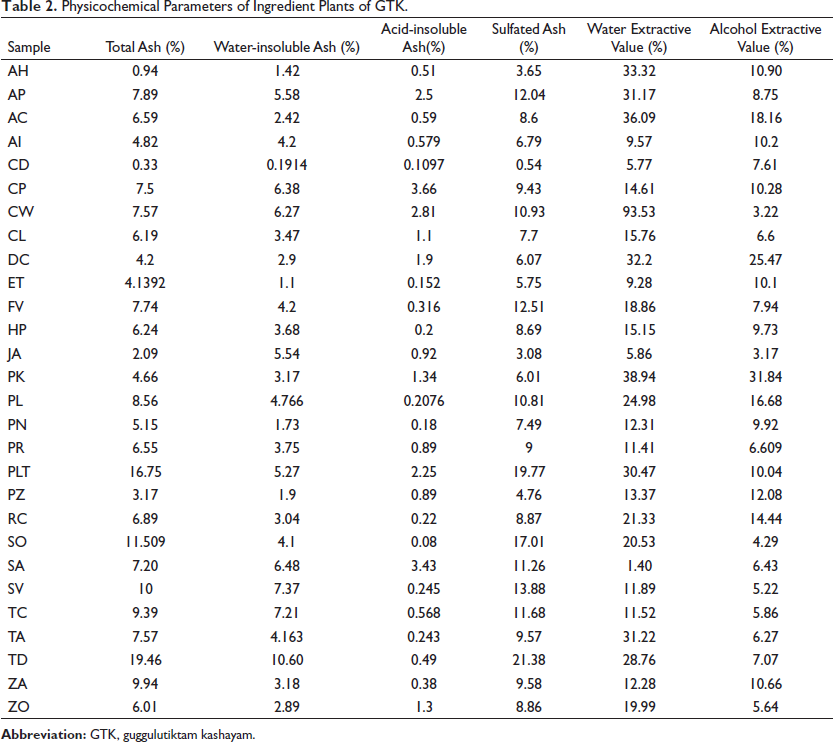
Physicochemical properties such as total ash, acid-insoluble ash, water-insoluble ash, sulfated ash, water-soluble extractive, and alcohol-soluble extractive were evaluated in all the ingredient drugs of GTK, and the results are presented in Table 2. The physicochemical parameters of raw herbal drugs are an important set of quality markers as they ensure the required quality of raw materials (Jeevitha et al., 2021). The Ayurvedic Pharmacopoeia of India (API) is the regulatory document that fixes the quality parameters of raw drugs. All the samples were analyzed to confirm their authenticity and quality.
Physicochemical Parameters of Ingredient Plants of GTK.
Estimation of Major Phytochemicals
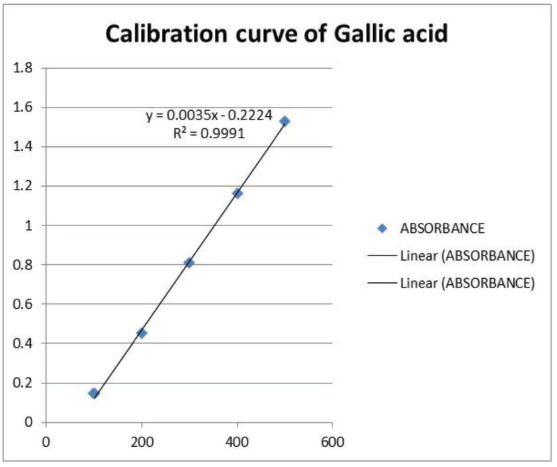
Medicinal plants are rich sources of polyphenols with diverse biological properties. Quantitative estimation of total polyphenols provides the preliminary chemical pattern of the plant extracts. Spectrophotometric methods have been employed for the estimation of total polyphenols in herbal drugs and finished formulations (Dinakaran et al., 2018; Sulaiman et al., 2021). The total phenolics (TPC) of all the ingredient plants, along with GTK, were estimated. The TPC was calculated from the calibration curve of gallic acid (R² = 0.999, Figure 1). The TPC contents of GTK and its ingredient plants are given in Table 3.
Calibration of Curve of Gallic Acid Standard.
Spectrophotometric Estimation of TPC in GTK and its Ingredient Drugs.
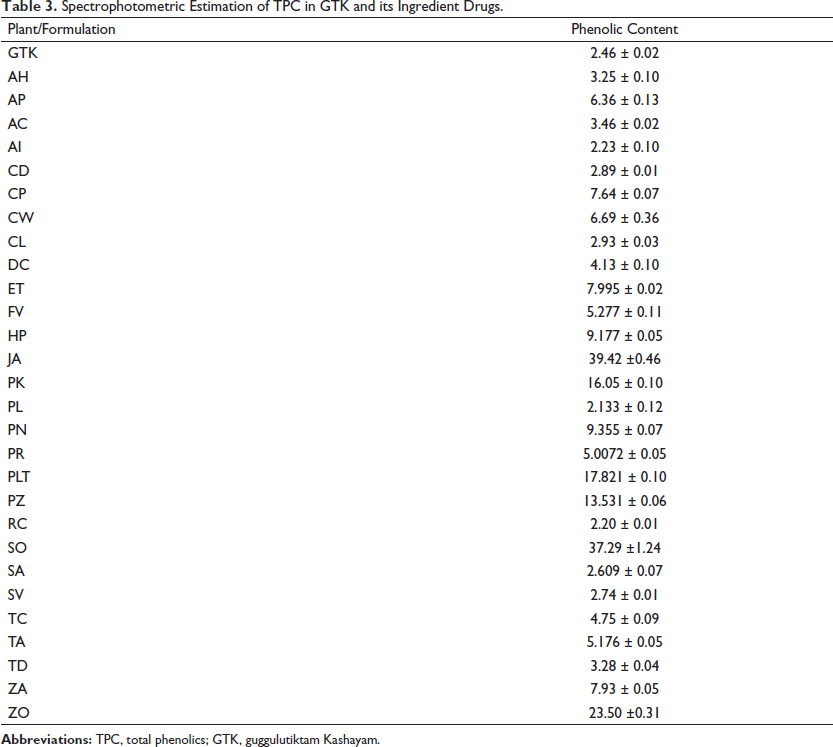
The highest TPC was shown by Justicia adhatoda (JA) (39.42 ± 0.468), followed by Scindapsus officinalis (SO) (37.29 ± 1.248), Zingiber officinale (ZO) (23.50 ± 0.316), and Pluchea lanceolata (PLT) (17.821 ± 0.1010). The lowest value was observed for Piper longum (PL) (2.133 ± 0.008), followed by Rubia cordifolia (RC) (2.20 ± 0.010), and Azadirachta indica (AI) (2.23 ± 0.104). The TPC of GTK was found to be 2.46 ± 0.028, and the value is much lower compared to that of other ingredient plants. This might be due to the chemical transformations that occurred due to the synergistic and antagonistic interactions of various polyphenols extracted from the ingredient plants. Variations in phenolics with respect to the herbal drug samples have been reported earlier (Mehmood et al., 2022; Siddiqui et al., 2017; Tungmunnithum et al., 2018); however, no previous reports are available on the comparative estimation of the polyphenols with respect to the finished formulation. Synergistic interactions of polyphenols with respect to the ratio of plant materials have been reported earlier (Aslam et al., 2017). More studies are required to establish the effect of various raw drugs on the total phenolic composition of finished PHFs.
HPTLC Profiling
HPTLC has emerged as an important analytical tool for the qualitative and quantitative phytochemical analysis of herbal raw drugs and PHFs (Lalhriatpuii, 2020). Comparative HPTLC profiling has been carried out for GTK with all the ingredient plants. The chromatogram documented under various UV visualizations was compared with respect to the R f values. Chemical quenching at UV-254 nm and fluorescence at UV-366 nm have complied, and the data is presented in Table 4. On evaluating under UV-254, bands with R f 0.03, 0.09, 0.11, 0.17, 0.22, 0.29, 0.47, 0.57, 0.70, and 0.77 have been observed in GTK, and out of these compounds with R f 0.03 (AP, AC, PN, RC), 0.09 (PK, SA, ZA, SO, TC, TD), 0.11 (AH, PK, JA, ZO), 0.17 (PN, RC), 0.29 (SA, CL), 0.70 (SA, ZA), and 0.77 (CD) are identified. On evaluating under UV-366 nm, bands with R f 0.06, 0.12, 0.20, 0.21, 0.28, 0.31, 0.37, 0.40, 0.47, 0.50, 0.53, 0.57, 0.64, 0.69, 0.77, and 0.82 have been observed in GTK. Out of these compounds with R f 0.06 (PN, CD, SA), 0.12 (PTA, TA), 0.28 (CL), 0.31 (CW), 0.37 (SA), 0.50 (SA), 0.64 (CD, PN, PR), 0.69 (PZ), and 0.82 (SA) are identified (Figure 2).
HPTLC R f Values of Various Compounds Separated from GTK and its Ingredient Plants.
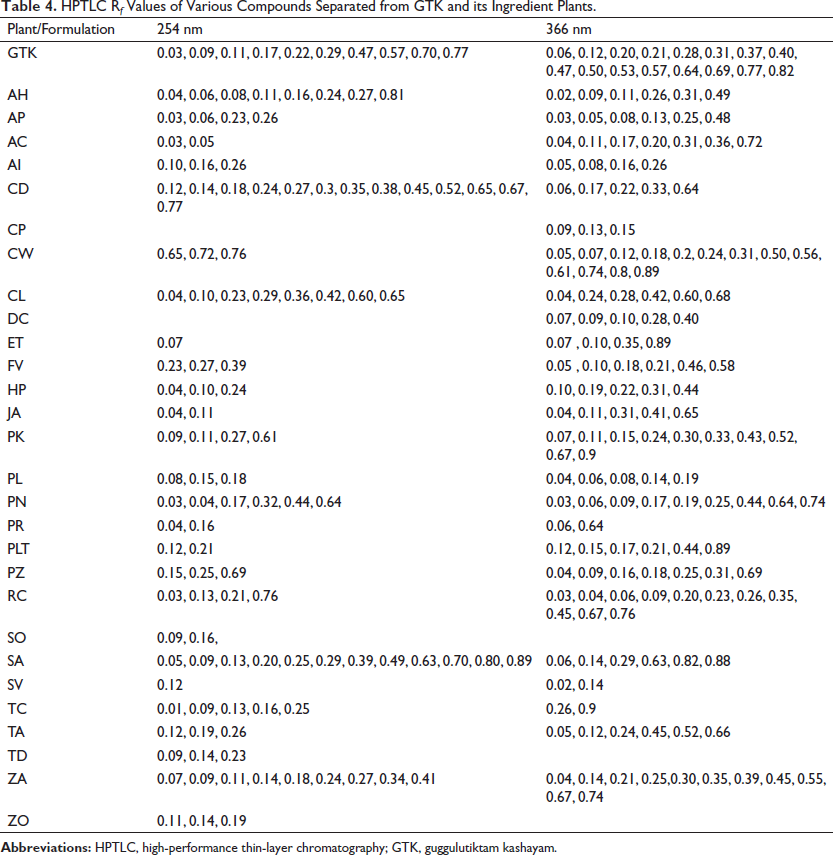
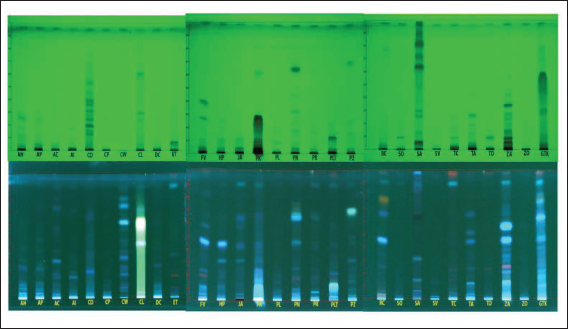
Comparative chemical profiling showed that there are differences in the chemical pattern of the finished formulation with respect to the chemical constituents of all the ingredient plants. The changes might be due to the chemical interactions of various phytochemicals extracted from the different raw drugs. Evidence is reported to support the occurrence of synergy within a total extract of a single herb as well as between different herbs in a formulation. Positive and negative aspects of interactions, including antagonistic reactions, have also been reported (Hemaiswarya et al., 2022; Williamson, 2001; Zhou et al., 2016). The present study also supports the previous reports on the herb-herb interaction and synergistic and antagonistic chemical reactions possibly happening during the preparation of PHFs.
Discussion
The majority of the herbal formulations used in Ayurveda are polyherbal in nature and contain multiple ingredient plants. Quality standardization of each ingredient drug is very important as it directly affects the quality and safety of the finished formulation. The phytochemical standardization achieved by multiple chemical profiling, along with preliminary screening and quantification of marker compounds, can assure the reproducibility of the activity of the PHFs. The clinically pertinent scientific data to support the asserted synergistic therapeutic effects of Ayurvedic PHFs needs to be established
Conclusion
The phytochemical evaluation of PHF is the very first step in the correlation of their biological properties with the active compounds identified. In the case of Ayurvedic formulations, all the compounds are important, irrespective of their abundance. Hence, comparative chromatographic fingerprinting is a useful tool to understand the chemical pattern of formulations along with ingredient drugs. In this study, comparative phytochemical studies have been carried out in an important Ayurvedic formulation with respect to all of the ingredient drugs. Quality standardization of all the raw materials was done as per the pharmacopoeial standards. Major groups of chemicals, such as total phenols and total flavonoids, have been estimated, and chromatographic comparison was done by HPTLC profiling. The result of the study showed the differences in the chemical constituents of the finished formulation when compared to its ingredient drugs, which provide a lead for further studies to confirm the synergy and antagonism in the selected formulation.
Footnotes
Summary
GTK is an important Ayurvedic PHF that contains a variety of phytoconstituents with numerous biological properties.
Abbreviations
GTK: guggulutiktam kashayam; HPTLC: high-performance thin-layer chromatography; API: ayurvedic pharmacopoeia of India; TPC: total phenolic content; GaE: gallic acid equivalent; PHF: poly herbal formulation; ATS: automatic TLC sampler; AH: aconitum heterophyllum; AP: aconitum palmatum; AC: acorus calamus; AI: azadirachta indica; CD: cedrus deodara; CP: cissampelos pareira; CW: commiphora wightii; CL: curcuma longa; DC: dolomiaea costus; ET: embelia tsjeriam-cottam; FV: foeniculum vulgare; HP: holarrhena pubescens; JA: justicia adhatoda; PK: picrorhiza kurroa; PL: piper longum; PN: piper nigrum; PR: piper retrofractum; PLT: pluchea lanceolata; PZ: plumbago zeylanica; RC: rubia cordifolia; SO: scindapsus officinalis; SA: semecarpus anacardium; SV: solanum virginianum; TC: tinospora cordifolia; TA: trachyspermum ammi; TD: trichosanthes dioica; ZA: zanthoxylum armatum; ZO: zingiber officinale.
Acknowledgment
The authors are thankful to the Ministry of AYUSH, Government of India for financial assistance.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work is based on the research funded by the Ministry of AYUSH, Government of India under the Centre of Excellence component of Ayurswasthyayojana.
