Abstract
Keywords
Introduction
Globally, thalassemia is a serious medical issue, it is a genetic disorder in which the synthesis of globin chain of hemoglobin is defective. It is more prevalent in the Mediterranean region but now it is found all over the world particularly in Africa, America, Pakistan, India, and Nepal. According to an estimation, around 270 million carriers are found with hemoglobinopathies and different kinds of thalassemia. Out of which 80 million are carriers of β-thalassemia (β-T), whereas more than 23 000 births occur annually with β-Thalassemia Major (β-Tm) and up to 90% of these occur in developing countries. 1 Pakistan has the largest number of transfusion-dependent thalassemia (TDT) children, with about 8 million carriers whereas about 80 000 children are suffering from β-Tm. This imposes an immense emotional, psychological, and financial burden on a large population. Patients with β-Tm need lifelong management to prevent and control the effects of long-term adherence to treatment. According to these guidelines management, strategies for β-Tm comprise regular blood transfusion, iron chelation therapy (ICT), splenectomy, and, for affording patients, hemopoietic stem cell transplantation (HSCT). 2
Now thalassemia is emerging as a syndrome, regular blood transfusion and effective ICT enable long-standing control of the disease. Iron overload (IOL) is the most common complication of TDT. Each unit of packed red blood cells (RBCs) comprises 200 to 250 mg iron. In TDT, iron from transfusion usually contain to .3 to .6 mg/kg per day with an expected 2 to 4 U packed RBCs transfused monthly. Senescent RBCs are phagocytized by the reticuloendothelial system. Consequently, labile cellular iron is released into the plasma to bind transferrin. Transferrin binding is saturated due to repetitive blood transfusion, non–transferrin-bound iron (NTBI) is transported that subsequently developed several complications by producing Reactive Oxygen Species (ROS) which has devastating effects on vital organs; heart, liver, brain, and endocrine glands by producing cellular functional impairment, apoptosis, and necrosis. Impaired general growth, decreased calcium absorption, absence of secondary sexual characteristics, cirrhosis of the liver, cardiac failure, and cancer are the major causes of quality-adjusted life years (QALY) and ultimately early death and high mortality rate. Therefore, management of iron overload due to repetitive blood transfusion is the main challenge in the care of β-Tm and other TDT. 3 In this regard, the current available iron-chelating drugs for thalassemia patients, that is, Deferoxamine, Deferasirox, and Deferiprone showed bad compliance and significant adverse effects which also lead to increase in complications and ultimate death.
In this scenario, a safe, effective, and convenient iron chelator is the biggest demand of thalassemia patients. Therefore, a polyherbal iron chelator - “Irochel,” was developed, which is safe, effective, and palatable for lifetime use. This product is an extract-based polyherbal iron chelator made up of 6 herbs; Fagonia cretica L. (Whole plant), Triticum aestivum L. (Wheatgrass), Emblica officinalis L. (Fruit pulp), Cucurbita pepo L. (Seeds), Momordica charantia L. (Fruit Pulp), and Tribulus terristris L. (Fruit). The selected herbs possesses vast range of pharmacological actions and used in various disease conditions. As well as these herbs have a great nutritional values. The wide range of pharmacological activities, that is, ferric reducing antioxidant potential, and iron chelating ability, medicinal uses and rich amount of nutrients support to manage comorbidities in patients with thalassemia.
Herbs have a rich amount of active phytochemical constituents such as alkaloids, anthocyanins, flavonoids, glycosides, phenols, resins, saponins, tannins, etc. which have accountable biological and pharmacological actions. These bioactive compounds have been accredited to secure toxicological potential along with beneficial therapeutic effects. The toxicity of the herbs has been attributed to the nature, quantity, and strength of their bioactive compounds. As the use of traditional medicine is rising globally, the recognition of community health and safety issues are also progressively established. 4
AIM of the Study
The rationale of this study was to investigate the phytochemical, antioxidant, and cytotoxic (Brine shrimps lethality assay) activities and to analyze the toxicological studies and acute and subacute toxicity on Wistar rats based on Organization of Economic Co-operation and Development (OECD) guidelines 423 and 407, respectively. The aim of this study is to formulate a safe drug for the management of IOL in thalassemia patients. The results were analyzed through SPSS v. 23 by applying paired sample t-test and the P-value was obtained.
Materials and Methods
Investigational Herbs
Fagonia cretica L
(FC) is a tropical, small, spiny, and erect, under shrub that belongs to the family Zygophyllaceae. It is found in the Asia and Africa. In Pakistan, it is abundantly found in desert areas of Sindh, Baluchistan, and Khaiber Pakhtoon Khwah. It is also known as Sachchi buti (Urdu); Dhamasa (Tibbi name); Dhamaasaa (Unani); Shoka, Shakai (Arabic), and Khrason thorn (English). 5
It is reported that FC is heavenly rich in alkaloids, coumarins, 6 phenolic compounds, saponins, 7 flavonoid glycosides and triterpenoids, 8 amino acids, carbohydrates, proteins, 9 vitamins and minerals, 10 and sterols. 11
Its pharmacological actions reported as antioxidant, analgesic, anti-inflammatory and wound healing, anti-allergic, neuroprotective, cytotoxic and anti-tumour, 12 dermatological, anti-hemorrhagic, 13 hepatoprotective, antidiabetic, antipyretic, hematological, 14 free radical scavenging, ferric reducing and iron chelation, antimicrobial, and anti-fungal 15 activities.
It is used in the hepatic, neurological, and hematological problems, dysentery, fever, thirst, vomiting, gastritis, inflammation, and skin diseases, 16 thalassemia, 17 and iron chelation. 18
Emblica officinalis L
(EO) is a plant of the family of Euphorbiaceae, well recognized as Phyllanthus emblica (Scientific name) or Emblic Myrobalan (English), Gooseberry (Indian), Amla, and Amalaki (Urdu). The fleshy fruit is edible, small, globular, and yellowish-green. The fruit has an astringent, sour, and bitter taste. It is abundantly found in Sri Lanka, China, Malaysia, India, Pakistan, and Bangladesh. It contains the richest amount of vitamin C, also contains flavonoids, carbohydrates, tannins, saponins, glycosides, phenolic, and many other compounds. 19
The pharmacological activities of EO are well reported such as analgesic, anti-inflammatory, antimicrobial, 20 antidiabetic, hypolipidemic, 21 antioxidant, cryo-protective, 22 antiulcerogenic, 23 hepatoprotective activities against iron, 24 and iron chelating activity. 25
The EO is widely used for the management of various ailments ie diarrhea, inflammatory disorder, jaundice, headaches, nausea, vomiting, hair fall etc. 26
Triticum aestivum L. (Wheatgrass)
WG is a member of the family Poaceae (Gramineae). It is also known as Hanta (Arabic), Gandum (Persian), and Wheatgrass (English), Triticum aestivum (Scientifically). 27 It is cultivated on large scale in the Middle East and Europe. It is the best source of chlorophyll. 28 WG has an astounding phytochemical profile as it contains alkaloids, flavonoids, carbohydrates, proteins, amino acids, and other bioactive compounds. It also contain rich amount of nutrients, vitamins, minerals etc. 29 Because of its rich chemical composition, it possesses a wide range of pharmacological actions like antioxidants, 30 blood generators, 31 blood purifiers, anti-inflammatory, 32 anti-arthritic, 33 analgesic, iron-chelating, 34 anticancer, 35 hepatoprotective, cardioprotective, 36 and many more. It is used in the treatment of neurological, gastrointestinal, hepato-biliary, endocrinological, hematological, cardiovascular, musculoskeletal, diabetes, 37 rheumatoid arthritis, 38 and dermatological disorders. 39 It is used in form of powder and fresh juice for thalassemia patients. 40
Cucurbita pepo L
(CP) is also known as Kadoo (Urdu, Hindi, and Saraiki) and squash (English). Seed, also known as pepitas, are edible, small, flat, and green in color, belong to the Cucurbitaceae family, and are cultivated extensively all over the world such as China, India, Pakistan, Western America, Northern Mexico, and Europe. 41 CP seeds are mostly used as a dietary supplement as they contain proteins, polyunsaturated fatty acids, 42 phenolics, 43 phytosterols, tocopherols, and squalene. 44 As well as it has a tremendous amount of elements and minerals, that is, copper, zinc, manganese, phosphorus, magnesium, etc. 45 CP seeds exhibited great pharmacological actions such as antioxidant including free radical scavenging and iron chelation activity, 46 anti-inflammatory, 47 antidiabetic, 48 cardio-protective, 49 anticancer, 50 and antimicrobial 51 activities. It is used in various medicinal disorders like intestinal inflammation, infectious conditions, diabetes, urinary disorders, and analgesia, 52 beneficial to treat bronchitis, burns, gastritis, enteritis, neuralgia, headaches, fever, irritable bladder, and prostatic hyperplasia. 53
Momordica charantia L
(MC) is a climbing herb that is belong to the family Cucurbitaceae. The typical MC fruit is emerald green, 20–30 cm long with an undulating warty surface, oblong with tapering ends. It is also known as bitter melon (English), Kareela (Urdu, Hindi), and Qasa-ul-hamar (Arabic). It is widely grown in Bangladesh, China, India, other parts of Southeast Asia, and Pakistan. 54 It has an excellent phytochemicals and nutrients profile that includes amino acids, proteins, carbohydrates, fatty acids, alkaloids, flavonoids, phenolic compounds, phytosterols, saponins, and tannins. 55 MC has a vast array of pharmacological actions, like antioxidant, anti-inflammatory, anti-leukemic, antiviral, hypotriglyceridemic, 56 hypoglycemic and antidiabetic, 57 antihyperlipidemic, 58 and antiulcer 59 effects. It is beneficial to treat various diseases for example diabetes mellitus, anemia, malaria, carminative, jaundice, anthelmintic, cholera, and ulcers. 60
Tribulus terristris L
(TT) belongs to the family Zygophyllaceae; is a well-known small prostrate shrub, 10–60 cm in height. Its fruits are faint greenish-yellow with spines. It is an annual shrub found in Africa, Australia, Bulgaria, China, India, Mexico, Europe, USA, Spain, and Pakistan. It is commonly known as Caltrops (English), Gokharu (Hindi), and Khar-e-Khasak Khurd (Urdu).61,62 It contains flavonoids, glycosides, resins, alkaloids, saponin kaempferol, and quercetin. 63 It is rich in proteins and minerals, phenols, terpenes, and steroids (saponins) present in its extracts. 64 This excellent phytochemical profile exhibited antioxidant, 65 anti-inflammatory, antimicrobial, 66 cardioprotective 67 aphrodisiac diuretic, 68 and iron-chelating 69 properties.
Traditionally, all these herbs are used in various single and compound pharmacopeial herbal formulations such as Jawarish, Maajoon, Khameera, Kushta, Atrifal, syrups, and powder.
Plant Materials
Identification of Plants Part Used
All the plant materials were purchased, collected, and grown authenticated by the taxonomist at Herbarium, University of Karachi. The voucher specimens were deposited there for future reference. The herbs identification voucher numbers are Fagonia cretica L. (H-02), Wheatgrass (H-08), Emblica officinalis L. (M-04), Momordica charantia L. (M-05), Tribulus terristris L. (SB-31), and Cucurbita pepo L. (SB-43)
Macroscopic (Organoleptic) Evaluation
Organoleptic evaluation (sensory and morphological) of all medicinal plants were performed as described earlier. 70
Handling of Plants
Irochel is a polyherbal preparation consist of the following herbs which were acquired or grown as described below: 1. The whole plants of FC and TT (10 kg each) were collected from uncultivated and desert areas near to Madinat-al-Hikmah, Hamdard University, Karachi, Pakistan. The fruits of TT were separated. All the plant materials were washed, dried under shade, crushed, and used for extraction. 2. Wheatgrass (10 kg) was grown in the herbal garden of the Faculty of Eastern Medicine, Hamdard University, Karachi (FEM, HU, KHI). Sprinkles water twice a day for 10 days. After the young grass reached the 5–7 inches height it was collected, washed with tap water followed by distilled water, dried in mild sunlight, and at room temperature of 37°C. 3. EO, MC, and CP (10 kg each) were procured from the vegetable market at highway and local herbs markets at Joriya Bazar in Karachi. The fresh fruits of EO and MC were washed, cut into pieces, and deseeded. The resultant pulp was cut into small pieces, and dried in the air. The CP (seeds) were cleaned, washed, and shade dried.
Extraction Method
All the plant materials were grounded coarsely and soaked in 98% ethanol 1:6 w/v %. After a week it was decanted and the filtrate obtained was 94% for each plant. The material was soaked with the same ratio and decanted as described above. Three such percolates were mixed and evaporated on low-pressure 40oC using rotary evaporator (Buchi R-200). The extracts of all plants were dried in the dryer at 40oC, weighed and stored in the glass bottles, kept at room temperature 37°C until further used. 71
Formulation Preparation
Formulation of Irochel.
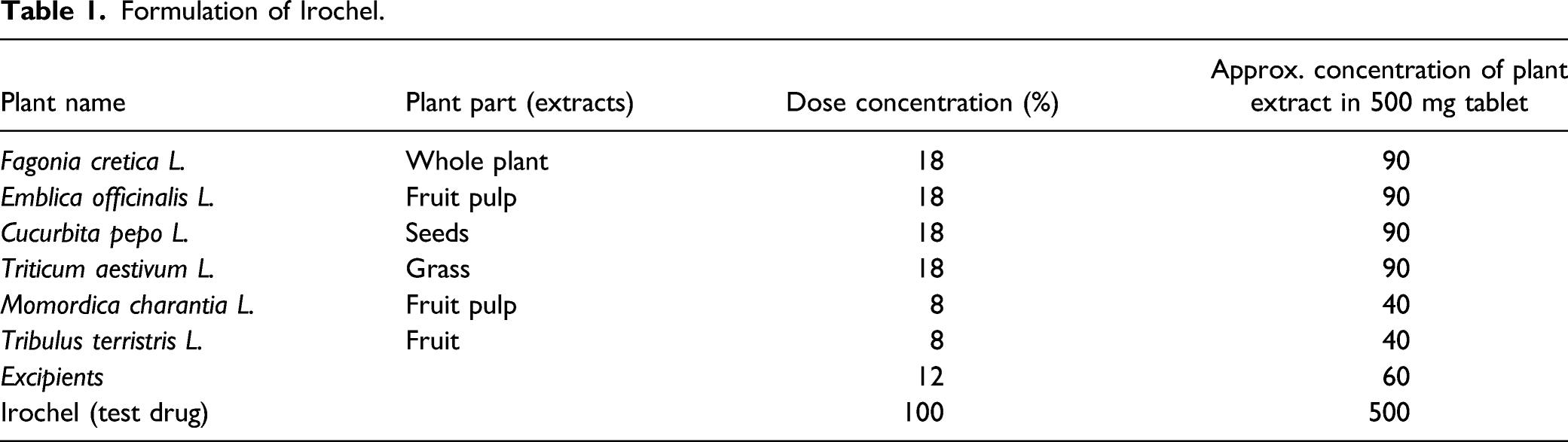
Phytochemical Analysis
The phytochemical screening of each plant’s extracts separately was done for the determination of proteins, carbohydrates, flavonoids, alkaloids, steroids, glycosides, terpenoids, phenols, tannins, saponins, and resins by using standard protocols as described by PK Mukharjee, (2019). 70
Antioxidant Assay
The extract (Irochel) was analyzed for free radical scavenging activity by 2, 2- diphenyl-1-picrylhydrazyl (DPPH). Gallic Acid was used as standard. Different concentrations of the extract were prepared, mixed with DPPH solution (.5 mg), and allowed to react (for 30 min) at 37°C, and measuring the absorption at 517 nm. The radical scavenging activity (RSA) was calculated using the following equation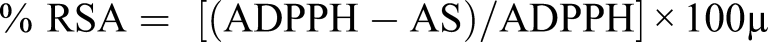
AS: absorbance of the solution when the extract (Irochel) has been added at a particular level and, ADPPH: absorbance of the DPPH solution. 72
Cytotoxic Activity (Brine Shrimp Toxicity Assay)
The BSLA was conducted in ICCBS followed by the standard procedure as described earlier by Meyer’s et al, (1982).
73
The process was carried out in triplicate (3 test/Concentration) as 10, 100, and 1000 μg/ml of extract in 3 vials, added seawater (2 mL), shrimps (n=30) were subjected, and the vials were placed under the incubator at temp (28° + 1°C). After the incubation period of 24 hours, the vials were inspected and the number of dead larvae was counted, and the mortality percent was calculated by using the equation given below
Acute and Sub acute Toxicity in Wistar Rats
The experimental study protocol on animals was approved by the Ethical Review Board (ERB) of HU (reference number ERB-18-05). Animal studies were conducted in the Faculty of Pharmacy (FOP), HU, in July–August 2019. The animals of both gender (n = 36) weighted (200–250 g). The animals n = 12 and n = 24 were used for the ACT and SAT, respectively. All the animals had free access to a standard rat pellet diet and water. They were housed in the polyvinyl chloride cages in the animal house of FOP, HU, and KHI. The temperature was maintained at 23 ± 2°C with a light/dark cycle of 12 hours each. The OECD guidelines were used to carry out the whole experiment. 74
Acute Toxicity Study
(ACT) study was carried out according to OECD guideline-423. All WRs (n = 12) were divided into 2 main groups. Group A: Control (n = 6; 3 females and 3 males) and group B: treated group (n = 6; 3 females and 3 males), age 8 months, weighted 200–250 g.
The WRs were fasted for 12 hours before dosing. The control group received normal saline orally through gavage. The treated group received Irochel in the highest dose of 5000 mg/kg constituted in distilled water (1 mL). WRs were observed continuously for 4 hours, and then after 24 hours, daily, for a total of 14 days. The observations were included skin, eyes, signs of nervous system toxicity; convulsions, spasticity, tremors, back tonus, posture, ptosis, motor activity, ataxia, pinna, exophthalmos, grasp, piloerection, lacrimation, salivation, writhing, and unconsciousness, signs of gastrointestinal and renal disturbances; diarrhea, urination, signs of respiratory disturbances; respiratory rate, dyspnea, and mortality. The water and food intake and weight of the animals were recorded daily.
Collection of Blood and Tissue Sampling
On day 15, after overnight fasting, the rats were anesthetized using ketamine (50 mg/kg) and xylazine (5 mg/kg), and blood was drawn through cardiac puncture for the hematological and biochemical profile. All the animals were sacrificed by cervical dislocation. The body’s vital organs, that is, heart, liver, and kidneys were excised, cleaned with saline water, and weighed. Thereafter, gross examination for any abnormality was conducted and samples were preserved in 10% formaldehyde. The ROW was calculated as follows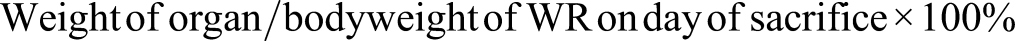
Hematological Analysis
Complete blood count comprises hemoglobin (Hb),RBCs, packed cell volume (PCV), red cell indices; mean corpuscular volume (MCV), mean corpuscular hemoglobin (MCH), mean corpuscular hemoglobin concentration (MCHC), as well as white blood cells (WBCs), differential leukocyte count (DLC), and platelets were carried out by an auto hematology analyzer (Sysmex KX-21).
Serum Biochemistry
The blood samples were centrifuged (4000g) for 10 minutes to obtain serum. The biochemical analysis includes LFT comprises on Serum Glutamic-Pyruvic Transaminase (SGPT), Total Bilirubin (TB), Direct Bilirubin (DB), Alkaline Phosphatase (ALP), LP consists of Total Cholesterol (T. Chol), High-Density Lipoprotein (HDL), Low-Density Lipoprotein (LDL), as well as S. Urea, S. Uric Acid, and S. Creatinine was carried out using a biochemistry analyzer (Merck Microlab 300), and fasting blood sugar (FBS) by glucometer.
Histopathological (Histo) Examination
The slide preparation was carried out in the histopathological laboratory, Dow University of Health Sciences, Karachi. The preserved organ tissues (heart, liver, and kidneys) of control and treated groups were processed and embedded in paraffin wax and sections (4–6 μm) were prepared. After staining with hematoxylin and eosin. The slides were examined under a microscope and photographed.
Subacute Toxicity Study
The SAT study was conducted according to the OECD guideline 407 to analyze the toxicity of Irochel, in WRs for 28 days. The hematological and biochemical parameters (LFT, LP, S. Uric Acid, S. Urea, and S. Creatinine) were analyzed as described in the ACT.
Experimental Design
The WRs (n = 24 of both gender), aged 8 months, (weighed 200–250 g) were randomly distributed into 2 groups. Group A-control group (n = 6; 3 males and 3 females) received normal saline, and Group B-treated group (n = 18; 9 males and 9 females). Group B-further categorized into 3 sub-groups as per the dose administered. The Irochel was constituted in distilled water (1 mL) administered at 3 doses of 50 mg/kg (n = 6; 3 males and 3 females), 300 mg/kg (n = 6; 3 males and 3 females), and 2000 mg/kg (n = 6; 3 males and 3 females) per day for a consecutive 28 days orally through gavage to overnight fasted WRs whereas the control group.
The rats were observed for behavioral and morphological changes, toxic manifestations, and mortality for 28 days. Observations included the same parameters as described in the ACT. All the rats were sacrificed on day 29, blood samples and vital organs (heart, liver, and kidneys) were collected for hematological, biochemical, gross, and histopathological analysis.
Gross necropsy and Histopathological Examination
Necropsy of both groups (treated and control) was conducted and the weights of the organs (heart, liver, and kidneys) were measured and fixed in 10% formaldehyde for histopathological examination.
Relative Organ Weight (ROW)
A careful dissection of selected organs liver, kidneys, and heart was carried out and ROW were obtained by the following calculation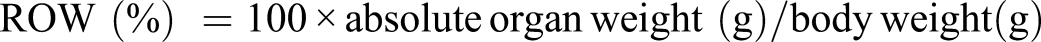
Hematology, Biochemistry, and Histopathology
The CBC, serum biochemistry, and histopathological examination were carried out the same as mentioned in the ACT study. 75
Manufacturing of the TEST DRUG (Irochel)
Procedure
The Irochel 500 mg tablets was formulated through the standard procedures, that is, Mixing, Granulation, Drying, and Tablet Compression as described by the Gad, S.C., 2008. Granules were prepared by using a sieve (#40) and then set for drying in a dryer (Memmert ULP-500, Germany) at 40°C. After the dry granulation process, compression of tablets was undergone a single rotary press machine in which the punch size was 11 mm×8 mm. Talcum powder 100 mg/kg as a lubricant, glucose syrup 20 g/kg of Irochel extract as a binder and sodium benzoate 1.5 gm/kg as preservative were used for the preparation of the 500 mg tablets. 76
Pharmaceutical Analysis of the Irochel Tablet
The quality control analysis of Irochel (500 mg Tablet) was carried out as per the WHO guidelines for standardization of medicine (Ghimire P et al, 2020). The general appearance; color, odor, and taste of the tablets, the diameter and thickness were measured on 20 tablets with the help of a thickness tester such as Vernier Caliper (Mitutoyo 530-101), the hardness (kg/cm2) of 20 tablets was measured by using a manual tablet hardness tester (Vinsyst VMT-1), the variation of weight (gm) was tested by weighing 30 tablets separately, calculate the mean ± SD, and compare each tablet’s weight to the mean weight.
The friability test was performed by using a friability (%) test apparatus (Model: Curio Tech- Fb0498) following standard procedure, and the disintegration time of tablets was measured in water (37°C) using the USP disintegration test apparatus. Place a basket in .1 HCl for 1 hour and then in phosphate buffer (KH2PO4 and NaOH .2 N) of pH 6.8, maintaining the temperature at 37±2°C. Note down the disintegration time. 77
Statistical Analysis
The data were analyzed by applying compare means and SD, probit analysis, and paired sample t-test (SPSS version 23) and the P-value was obtained.
Results
Organoleptic Evaluation
Organoleptic Evaluation of Selected Medicinal Herbs.

Organoleptic features of all herbs (FC= Fgonia cretica L., WG=Wheatgrass, EO= Emblica Officinalis, CP= Cucurbita pepo L., MC= Momordica charantia L., TT= Tribulus terristris L.) used in Irochel formulation.
Phytochemical Analysis of Plants
Phytochemical Analysis of Medicinal Plants of Irochel.

Phytochemical analysis of all herbs (FC= Fagonia cretica L., WG=Wheatgrass, EO= Emblica officinalis, CP= Cucurbita pepo L., MC= Momordica charantia L., TT= Tribulus terristris L.) used in Irochel formulation. + = Present, - =Absent.
Antioxidant Activity (DPPH- FREE Radical Scavenging Activity)
Physical Characteristics of Irochel Tablets.

Physical characteristics of Irochel tablets consist of color, shape, odour, taste and Size; Mean ± St. Deviation of thickness and diameter of tablets (n = 20).
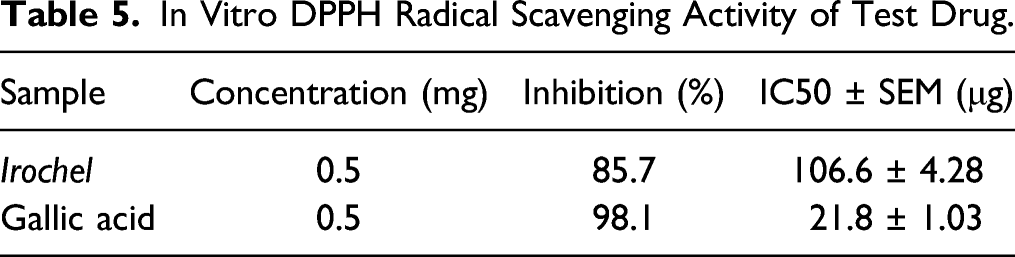
In Vitro DPPH Radical Scavenging Activity of Test Drug.
Cytotoxicity
Cytotoxicity of Irochel (Test Drug) in 24 Hours and LC50 Value.

The Probit Mortality Analysis.

Chi-square value is 286a.
Probit Model Using Log Estimation.

PROBIT model: PROBIT(p) = Intercept + BX (Covariates X are transformed using the base 10.000logarithm.).
Cytotoxicity.
In Table 7, the result exhibited significant cytotoxicity having LC50 124.327 μg/ml, whereas the standard error of the mean was obtained .196 which has been analyzed by SPSS v.23.
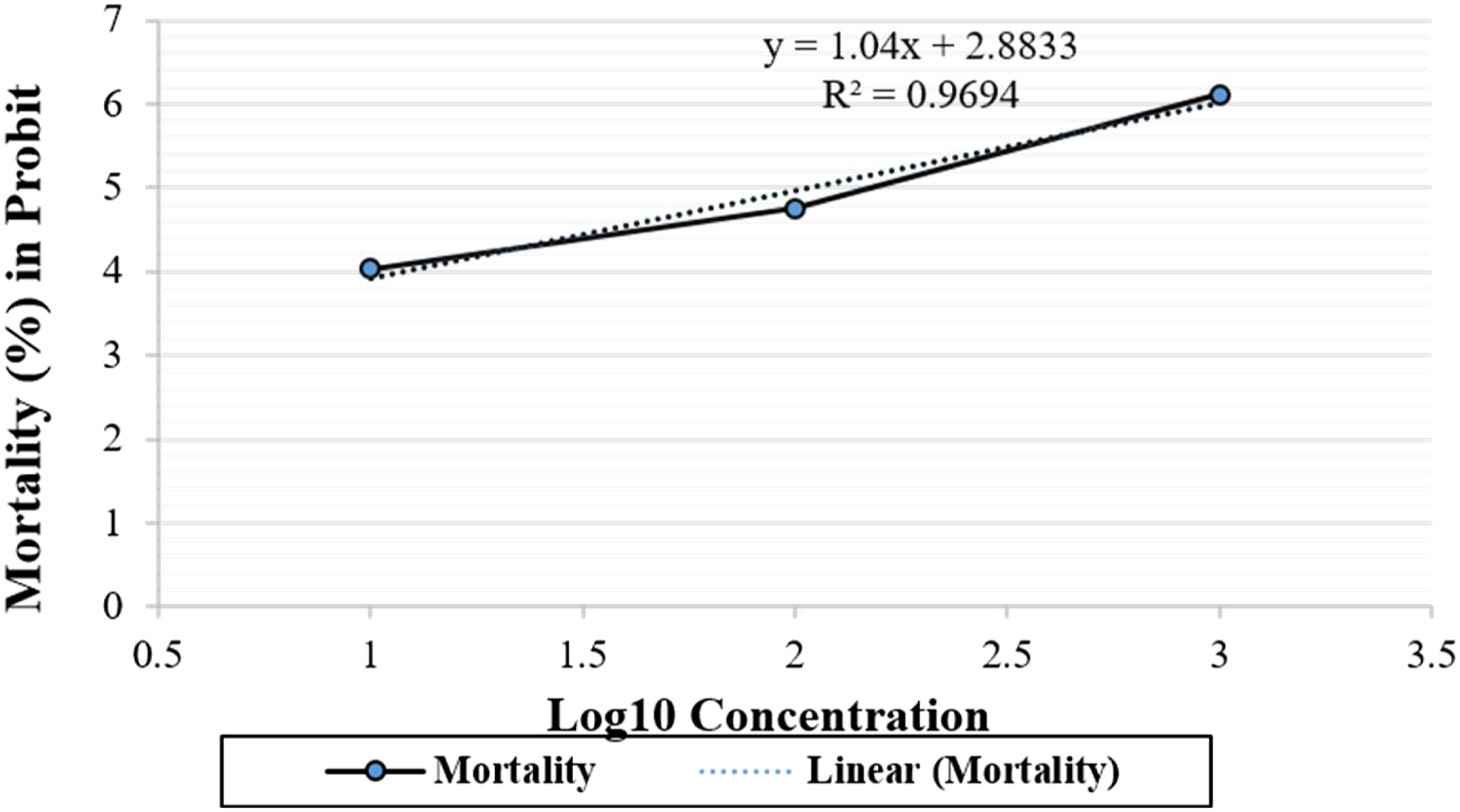
Table 8 exhibited log 10 concentration of different concentrations of extract and observation of response. The log estimation showed that the 95% CI was obtained as .649–1.424 upper and lower limit of dose concentration. In Figure 1, the probit mortality equations for Irochel were measure as Y= 1.04x + 2.8833 log conc. The graphical representation of probit and regression values; series 1 represents plotted line between log concentration and probit values while series 2 denotes that the line was plotted between log concentration and mortality fitted equation using regression analysis. An approximately linear correlation was observed among log of concentration and mortality (%) in the probit and LC50 was obtained from the best-fitted line method.
Acute Toxicity Study
The ACT effects of the Irochel at the dose of 5000 mg/kg were analyzed according to the OECD guideline 423. No treatment-related toxic manifestations, behavioral changes, or mortality was observed during the course of the study. Therefore, the test drug was considered safe at the highest dose of 5000 mg/kg for human use.
Body weight measurement
The bodyweight of the rats was observed weekly till the completion of the study and mean ± standard deviation (SD) was calculated for both groups. The results showed no significant variation between treated and control groups. See Figure 2(a). (a) Body weight of wistar rats in acute toxicity, (b) Body weights of animals in subacute toxicity.
Subacute Toxicity Study
The SAT study was conducted following OECD guideline 407. The Irochel at 3 doses (50, 300, and 2000 mg/kg) was administered to the treated group after an overnight fast for consecutive 28 days orally through gavage, whereas normal saline was administered to the control group. There was no sign of toxicity and lethality observed in the treated and control groups during the experimental period.
Body weight measurement
The bodyweight of rats was calculated daily until the completion of the study and the mean ± SD of all treated and control groups were obtained weekly for 28 days. The results showed a gradual rise in body weight of the treated groups at the highest doses during the course of the study but no significant difference in comparison with the control group. See Figure 2(b).
On day 29, all the animals were sacrificed after anesthesia, blood samples and body organs (heart, liver, and kidneys) were collected for CBC and biochemical analysis and histopathological examination as mentioned in methodology. Necropsy of dissected out organs was followed by the absolute and ROW.
Relative organs weight (ROW)
Relative organs weight (mg) of Heart, Liver, and Kidneys of Treated and Control Groups.

Mean and SD. n = 24 (n = 6; 3 ♂ and 3 ♀ in all Test and control groups).
Laboratory Analysis
The laboratory analysis was conducted in the same manner as described in the ACT.
Hematology
Complete Blood Count (CBC) and ESR of Different Doses of Test and Control Groups.

Mean and SD (n = 6 of each group). Normal Values: Erythrocytes: Hemoglobin (mg/dl): 11.5-16.4, RBCs (10^6/μl): 3.9- 5.6, PCV (%); 40.7%–50.3% for men and 36.1%–44.3% for women, MCV (fl): 76-96, MCH (pg): 28-32, MCHC (g/dl): 32-36. Leucocytes: TLC (10^3/μl): 4.0-11.0, Neutrophils (%): 40-75, Lymphocytes (%): 20-45, Eosinophil (%): 1-6, Monocytes (%): 2-10, Basophils (%): 0-1. Platelets (10^3/μl): 150-400, ESR (mm/1st hr): In male 0-10, in female 0-18.
The P-value in all CBC parameters were found to be >.05 and exhibited no significant differences in treated and control groups except WBCs and platelet counts. The P-value of WBCs count was obtained as .004 and .000 for the dose of 300 mg/kg and 2000 mg/kg, respectively, which is <.05 thus found to be significant in comparison with the control group, whereas at dose 50 mg/kg P-value was obtained as > .05 which showed no significant difference between the treated and control groups. Similarly, in platelet counts the P-value was found to be .000 for the doses of 300 mg/kg and 2000 mg/kg which exhibited a highly significant difference between treated and control groups while at the dose of 50 mg/kg the P-value was found to be > .05 which demonstrated no significant difference. All other parameters were found to be in normal ranges. The mean ± SD values of RBCs and Hb were found slightly increased in the rats receiving Irochel at the doses of 300 mg/g and 2000 mg/kg in comparison with the control group. Similarly, the P-value of ESR at dose 50 mg/kg was obtained as .043 which showed a significant difference whereas at doses of 300 mg/kg and 2000 mg/kg the P-value was found to be insignificant in comparison to the control.
As per Oslon, (2000), the study of hematological parameters is applicable to risk assessment because blood has great prognostic importance for human toxicity (91%). 78 As blood provides a transport medium and carries many drugs and xenobiotics due to which the RBCs, Hb%, WBCs, and platelets are forced to expose to harmful substances in the body. Any damage to the blood cells will consequently affect the normal physiological functions of the body. 79 Therefore, the herbs which do not have adverse effects on parameters of CBC suggesting the non-toxic nature of the drug. As far as Irochel is concerned, a significant increase in WBCs suggested that this polyherbal combination can be used safely as an immune booster due to significant effects on the production of WBCs which provides defense systems in the body. Increased platelets count in a higher concentration of doses represents its effectiveness regarding coagulation; therefore, it may recommend in thrombocytopenia and other conditions in which platelets count becomes decreased such as in TDT. As the drug was administered to the normal WRs not diseased, therefore, as the effects of test drug it increases the production of blood cells including platelets count due to the increase production of thrombopoietin hormone from the liver.
Serum Biochemistry
The mean ± SD of the LFT, LP, S. Uric Acid, S. Urea, S. Creatinine, and FBS were calculated and the P-value was obtained by applying paired sample t-test through SPSS v. 23.
Liver Function Test of Different Doses of Test and Control Groups.

Mean and SD (n = 6 of each group). Liver Function Test: TB (mg/dl): ‹ 1, DB (mg/dl): ‹ .30, SGPT (U/L): In male up to 41, in females up to 33, ALP: male 40-130, females 35-105.
Lipid Profile of Different Doses of Test and Control Groups.

Mean and SD (n = 6 of each group). Lipid Profile: T. Chol (mg/dl): ‹ 200, TGs (mg/dl):‹ 150, HDL (mg/dl): 45-65, LDL (mg/dl): ‹ 130.
Serum Urea, Uric Acid, Creatinine, and Blood Glucose Levels of Different Doses of Treated and Control Groups.

Mean and SD (n = 6 of each group). Uric Acid, S. Urea (mg/dl): 10-50, S. Uric Acid (IU/l): male 3.4-7.0, female 2.4-5.7, S. Creatinine (mg/dl): male .6-1.5, female. 5-1.5, FBS (mg/dl): 70-110.
In the evaluation of FBS level, the mean ± SD exhibited as 88.80 ± 7.259, 87.20 ± 15.610, and 82.80 ± 8.642 at the doses of 50, 300, and 2000 mg/kg, respectively, which delineated a slight decrease in all treated groups in dose-dependent manner when compared to the control. However, the P-value for all groups was found to be .391, .583, and .168, respectively, which is considered insignificant. This slight decreasing effect on the blood glucose level may be appeared due to the presence of MC which has a significant effect in lowering the blood glucose level. Shuo Jia et al, (2017) delineated that numerous pharmacological researches revealed that cucurbitanes from MC are responsible for hypoglycaemic and anti-diabetic activity. Moreover, Cucurbitane-type bioactive principles have been isolated from the methanol extract of MC which could lower blood glucose in diabetic animals. 80 Therefore, Irochel could provide good support for preventing or managing diabetes mellitus in patients with thalassemia.
Histopathology
The results of histopathological examination of vital organs (heart, liver, and kidney) was exhibited no pathological findings in morphology under the light microscope. See Figure 3. Histological slides of heart, liver and kidneys of test and control groups.
Pharmaceutical Analysis of Irochel
Irochel was subjected for the evaluation of characteristic parameters according to WHO guidelines such as physical parameters (form, size, shape, color, and texture), uniformity of weight, disintegration time, hardness, and friability. These standard parameters were also found to be acceptable as per the British and US Pharmacopoeias specifications.
The quality control pharmaceutical analysis of the polyherbal formulation comprising physical characteristics of tablets which showed that Irochel is a round, shiny black colored and bitter taste tablet having a characteristic odor. The tablet average weight is 500.83 ± 2.245 mg with a diameter of 4.830 ± .320 mm, thickness 11.17 ± .123 mm, hardness 4.70 ± .923 kg, friability .929%, and disintegration time was 32 minutes. See Table 4
Discussion
Herbal medicines are considered safe because they are natural. Besides this increasing global popularity, a lack of evidence on the pharmacologically important actions of the polyherbal formulations, has created an apprehension over the quality, efficacy, and safety of the plant products. Therefore, it is necessary to evaluate the presence of phytochemicals, significant pharmacological activities, and toxicity of these formulations to avoid adverse or lethal effects. This study was focused on the organoleptic and phytochemical analysis of each plant and plant extract as well as antioxidant, antimicrobial, cytotoxicity, and ACT and SAT investigation of a polyherbal formulation based on plants extracts.
Ascorbic acid (vitamin C) was discovered as the first antioxidant molecule. In plants, such acid is involved in the regeneration of tocopherol (vitamin E) and carotenoids. The latter can also act as an antioxidant and liposoluble redox system, protecting against lipid peroxidation. Many types of research studies showed that β-carotene could be regenerate vitamin E from the tocopherol radical; consequently, the resulting carotenoid radical cation would repair by ascorbic acid. The studies establish that polyphenols and carotenoids are the 2 main kinds of antioxidant phytochemicals, and they contribute the most to the antioxidant properties of foods/plants. 81 Hence, the medicinal herbs of this polyherbal formulation comprises both food and medicinal plant. Therefore, exhibiting significant antioxidant activity.
The medicinal herbs of the test drug are rich in vitamin C, vitamin E, polyphenols, and carotenoids. That is why it could be a strong antioxidant and play a significant role against ROS due to IOL.
The Irochel exhibited significant antioxidant activity as IC50 was found 106.6 ± 4.28 which help in decreasing the production of ROS in TDT. In subjects with TDT, ROS formed due to free and excess iron in the body cause tissue damage ultimately results in injurious effects on organs manifested by malfunctioning of those organs, consequently leads to death. This is the main cause of mortality in subjects with TDT. Due to the significant antioxidant potential, it removes free radicals from the body and preventing deposition of ROS in the tissues, thus ultimately protect organs from damaging effects of ROS and enhance their functional capability. Moreover, ROS causes damage to RBCs membrane components which increases the rigidity and fragility of the membrane of RBCs that leads to hemolysis of RBCs and the release of Hb into the plasma. Therefore, the antioxidant potential of Irochel not only protects body tissues and organs from injurious effects of ROS but also prevent hemolysis of RBCs by strengthening membrane stability.
Epidemiological studies have shown that flavonoids are linked to reduced incidence or mortality from CVD globally. The excessive production of oxidants is 1 of the prime etiology of CVD. Oxidative damage can cause endothelial cell injuries and deleterious vasodilator effects. Studies have proven that polyphenols could modify molecular events and improve endothelial function. Therefore, it has a significant role in the prevention of CVD. Moreover, polyphenols also protect the cardiovascular system by reducing blood pressure and inflammatory conditions besides oxidative damages. 82
The herbs used in this polyherbal formulation showed significant phytochemicals and nutraceutical importance; therefore, acquired strong antioxidant, and free radical scavenging activity. Thus, Irochel is a natural antioxidant based on plants that could be used to reduce oxidative damage, comprises not only foods but also contain medicinal herbs. It may play an important role in delaying the oxidation process initiated by free radicals and other oxidizing reactions and subsequently inhibit polymerization chain reactions. This might protect against diseases. Due to its significant antioxidant activity through reduction of the oxidation of unsaturated fatty acids, it is also beneficial in preventing and treatment of diabetes mellitus in these subjects as it is 1 of the consequences of ROS due to IOL. Thus, it could be very much helpful in preventing co-morbidities caused by oxidative damages.
The BSLA characterize a fast, cost-effective, and easy to conduct bioassay for testing natural medicine extracts’ cytotoxicity and anti-tumor properties. This is the most suggestive test for quality control of herbal products as well. LC50 of a given plant extract in BSLA depends on the potential of bioactive constituents in it. 83
The results of cytotoxicity by BSLA proven that the test drug has significant cytotoxicity showing LC50 124.327 μg per ml in dose-dependent manner. This indicated that herbs in this product have rich bioactive compounds that can play a significant role in maintaining and preservation of health. This can be evaluated for anticancer activity in the future. As far as thalassemia is concerned a study indicated that the incidence of cancer was 3.96 and 2.60 per thousand subjects—years of thalassemia. There was about a 52% higher incidence of cancer in TDT subjects, which was found in children below the age of 10 years. 84 A research study has proven that bioactive compounds such as flavonoids, steroids, triterpene, saponin, and glycosides have significant cytotoxic activity, particularly flavonoids and triterpenoids can be the main compound that possesses anti-cancer activity. 85 Based on the research studies on individual herbs of Irochel, it can be considered as a cancer-protective drug for thalassemia subjects. These results open a door to analyze the anti-cancer activity of the test drug deeply in the future.
In toxicity evaluation of plant extracts by BSLA LC50 values lower than 1000 μg/mL are considered toxic. The degree of lethality was found to be directly proportional to the dose concentration of the drug, ranging from the lowest concentration to the highest concentration. The results of this study manifested that this extract-based polyherbal formulation showed significant cytotoxicity in response to greater concentration.
Polyherbal formulations are used in traditional medicines with a vast variety of dosage forms and have great importance in Unani medicine for the management of numerous diseases. In this study, the ACT and SAT of a polyherbal formulation were analyzed in Wistar rats. The ACT study was carried out for 14 days at the highest dose of 5000 mg/kg in which no morbidity and mortality was observed. This proves that the Irochel is safe for acute management up to the dose of 5000 mg per kg. Thus, this polyherbal formulation, with LD50 > 5000 mg/kg is considered to be safe for acute use in TDT patients for the management of IOL.
In SAT, the Irochel was administered at the doses of 50, 300, and 2000 mg/kg/day, the results exhibited no abnormal behavior or any other sign of toxicity during the course of the study. There was a gradual increase in body weight of the treated groups in the highest doses relative to that of control (Figure 2(a)and(b)). These results proposed that this polyherbal formulation did have a positive effect on the bodyweight of the rats due to its significant nutritional value.
However, no significant changes in the absolute weight of the vital organs (heart, liver, and kidneys) of the rats indicate no gross toxic effects of the test drug on these organs. Demma et al (2007) described that “relative organ weight is more indicative of toxicity than absolute organ weight.” 86 Moreover, the results also exhibited that there were no significant variations in the ROW (Table 9), no gross and microscopic toxicity (Figure 3) of these organs in both groups was found. The gross and microscopic findings also suggested that there was no injurious effect of the test drug on these organs.
The results exhibited significant changes in some parameters of CBC, that is, RBCs, WBCs, Platelets, and Hb%. The biochemical parameters of LFT, LP, and renal markers in different concentrations of doses 50, 300, and 2000 mg/kg exhibited no abnormal findings.
The results of CBC showed different effects of Irochel on WBCs and platelets count significantly increased in a dose-dependent manner in comparison with control. Increased WBC is referred to as leukocytosis while increased platelet count is called as thrombocytosis. The increase in mean ± SD levels of RBCs and Hb % in comparison with control indicates that the product either contains erythropoietin hormone or a substance alike that stimulated the production of RBCs or Hb %, or it decreased the RBCs fragility. These findings were also supported by studies conducted individually on herbs that constitute this polyherbal formulation, that is, FC, 87 EO, 88 and WG 89 in which there is a significant rise in Hb%, RBCs, WBCs, and platelets count.
However, blood glucose level showed a dose-dependent decrease in comparison with control. This hypoglycemic action of the test drug is also very beneficial for thalassemia patients. As the global prevalence of diabetes mellitus in patients with thalassemia major was estimated at 9%, out of which the incidence of diabetes in patients with beta-thalassemia major (β-Tm) was reported 6–27% in some countries such as United Arab Emirates (10.5%), Oman (27%), Taiwan (26.8%), South America (14%), and Italy (6.5%). 90 Besides, insulin deficiency, chronic liver disease, viral infection, and/or genetic factors may also be contributory factors in the development of diabetes mellitus in patients with β-Tm. 91
These outcomes delineated that Irochel could be safely employed for the treatment of IOL in β-Tm patients at different doses as per OECD guidelines.74,75 The presence of EO as an ingredient in this formulation has proved to as vital organ protective also having strong chelating ability due to the significant amount of vitamin C in it. 26 The formulation also contains CP which has great nutritional value along with medicinal properties. 92 Likewise, WG, known as green blood also has remarkable nutritional importance and it contains chlorophyll which is considered to be helpful in the generation of Hb%. 93 The MC is also used as food although having great medicinal properties. 94 The FC is a famous herb for the management of chronic diseases and recent studies on β-Tm patients revealed significant results for ICT as well as maintenance of Hb level. 95 However, TT is used traditionally as a diuretic and analgesic. It also has a good nutritional composition. 96
These results of Irochel are supported by toxicological studies carried out on medicinal herbs of Irochel ie The toxicological studies on FC conducted by I Nazir, et al, (2017) 97 and L Badshah, F Hussain (2011), 16 on EO by Middha S. K. et al, (2015), 98 and Phetkate P. et al, (2020), 99 on CP by RCB. Cruz. et al, (2006), 99 and Malgwi et al, (2014), 99 on WG by Khan N, et al, (2015) 99 and Abed K.A.K., et al, (2017), 99 on MC by Abalaka, M. E., et al, (2009), 100 and Husna, R. N., et al, (2013), 101 and on TT by Zhu W. et al, (2017), 102 and Ruxandra . et al, (2020). 103
In the histopathological analysis, there were no changes found in the morphology of heart, liver, and kidney at a dose of 50 mg/kg/day, 300 mg/kg/day, and 2000 mg/kg/day.
The quality control pharmaceutical analysis of the test drug comprising physical characteristics of tablets which showed that Irochel is a round, shiny black colored and bitter taste tablet having a characteristic odor. The result exhibited that the size of the granules was uniformed, the flow of granules from the hopper was uninterrupted, lubricant was mixed well and particles were evenly distributed, and dies, punches, as well as the speed of the machine, was normal during manufacturing. The disintegration time indicates that Irochel will be disintegrated in the alkaline flora of the intestine. WHO guidelines (2019) and the drug regulatory authority of Pakistan have mentioned standard measures for polyherbal formulations are same as for conventional medicine. 104
Conclusion
Thus, Irochel is a natural antioxidant based on plants that could be used to reduce oxidative damage, comprises not only foods but also contain medicinal herbs. It may play an important role in delaying the oxidation process initiated by free radicals and other oxidizing reactions and subsequently inhibit polymerization chain reactions. Moreover, due to its significant antioxidant activity through reduction of the oxidation of unsaturated fatty acids, it is also beneficial in preventing and treatment of diabetes mellitus in these subjects as it is 1 of the consequences of ROS due to IOL. Thus, it could be very much helpful in preventing co-morbidities caused by oxidative damages and early death.
The findings of the ACT study revealed that the test drug is safe with a single oral dose of 5000 mg/kg/day. The 28 days of SAT study, also shown no toxic effects at all doses according to OECD guidelines. Thus, the Irochel is considered to be safe as it did not cause either any toxicity or lethality. Hence, it is concluded that this polyherbal formulation can be prescribed for therapeutic use in humans for long-term management of IOL in β-Tm patients.
This study is an acknowledgment with the traditional knowledge about these novel herbs and the results of earlier experimental investigations. The plant sources of food and drugs heavenly rich bioactive compounds that possessed astounding biological and phyto–pharmacological activities that play a crucial role not only in the cure of various diseases but also in maintaining health.
Footnotes
Acknowledgements
The authors are thankful to Prof. Dr. Ahsana Dar Farooq, Adjunct Professor, FEM, HU, for her support in writing the manuscript, Ms. Taba Rauf, Taxonomist, FEM, HU for identifications of plants and issue of the voucher, Dr. Shamim Akhtar. Dean, FOP, HU for providing the support to conduct this study in Research Lab-II, FOP, Dr. Farzana Sadaf, Assistant Professor, Department of Pharmacology, FOP, Tabiba Huma Sayeed Siddiqui, Hakim Amjad Ismail Lecturers, FEM, HU for their support in conducting the animal studies, Dr. Mohammad Asfar, Medical superintendent, for hematology and serum biochemistry tests in Pathology Lab, Shifa-ul Mulk Memorial Hospital for Eastern Medicine and all Research Lab-II, FOP staff to accomplish this work, Dr. Bushra Hina, Assistant Professor, Department of Pharmacognosy, Institute of Pharmaceutical Sciences, Jinnah Sindh Medical University, Mr. Ahmad Yar Sukhera and Rukhsar Javed for their assistance in proofreading the manuscript, designing graphical abstract, and video making.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Hamdard University Research Committee (HURC), Hamdard University, Karachi provided financial support for conducting this research study. But the decision to publish this research paper is not supported by HURC or any other funding organization.
