Abstract
In a pharmacokinetic study, high-performance liquid chromatography analysis of blood samples of Wistar female rats fed with partially saturated canthaxanthin (PSC) of Aspergillus carbonarius showed the presence of the carotenoid in the plasma within 6 hours of feeding. In another study for safety assessment of PSC fed to rats over a period of 28 days at 0.05%, 0.10%, and 0.25%, the rats showed no changes in food intake. There were no significant differences observed in body weight, hematological parameters, or serum clinical enzymes compared to the control group not fed with PSC. Deposition of PSC in the eyes of the rats was also not observed. The results showed that PSC-fed rats were not adversely affected as far as toxicological studies were concerned.
Introduction
Carotenoids, a group of fat-soluble pigments produced in plants, algae, fungi, yeast, and photosynthetic bacteria, comprise the largest group of pigments such as β-carotene, lycopene, β-cryptoxanthin, and so on. The major carotenoids that impart colors in food and feed are β-carotene, astaxanthin, zeaxanthin, bixin, canthaxanthin, and norbixin. Among these, canthaxanthin, a xanthophyll, is used widely as a drug or as a food and cosmetic colorant. Canthaxanthin is frequently given to patients with tumors as it can act as a strong antioxidant. 1 Administration of canthaxanthin have been shown to effectively reduce cancers induced chemically or physically. 2 –5 In vitro studies on inhibition of cancer cell growth by canthaxanthin have reported positive response in the case of murine melanoma, fibrosarcoma, and human squamous carcinoma. 6 Canthaxanthin has been shown to act as an antioxidant, 7,8 to potentiate immune response, 9 and to enhance gap junctional communication between cells directly 10 or through the formation of 4-oxo-retinoic acid, 11 they stimulate retinoic acid receptor. 12 In addition to its anticancer properties, canthaxanthin may function as antioxidants by quenching triplet sensitizers, singlet oxygen, and trapping free radicals. 7 ,13 Supplementation of canthaxanthin in fish diets help in pigmentation of muscles, thereby inhibiting lipid peroxidation thus preserving polyunsaturated fatty acids. 14
Regardless of the reported bioactive properties of this molecule, there are reports extensively reviewed on the effects of canthaxanthin in human chronic diseases including canthaxanthin retinopathy, retinal dystrophy, or aplastic anemia. 15 ,1 Of these, canthaxanthin retinopathy is connected with damage to the blood vessels around the place of crystal deposition, and the growth of new, abnormal vessels. The toxicity of canthaxanthin toward lipid membranes is due to the strong interaction between the pigment and lipid molecules and of the formation of crystalline aggregates of canthaxanthin in the membranes even at small concentration. 16 The above reports suggest the requirement of safety assessment of canthaxanthin for biological applications.
A mutant Aspergillus carbonarius containing partially saturated canthaxanthin (PSC) was isolated in this laboratory. 17 Acute and subacute toxicological studies showed that the fungal mycelia were not toxic when fed to Wistar rats. 18 When purified, it showed the structure of PSC and treatment to prostate cancer cell lines induced apoptosis, apparently by cell signalling through RAR/RXR receptors. 17 Since the above results were promising for using PSC of A. carbonarius as a drug, experiments were carried out to study its toxicity and pharmacokinetics after feeding the purified xanthins to Wistar rats. The results are described in this article.
Materials and Methods
Extraction of PSC From A. carbonarius Mutant
The biomass of A. carbonarius mutant (accession no. CFTRI UV-10046), containing carotenoid, was obtained as described earlier. 18 The carotenoids extracted from wet biomass, using acetone:methanol (7:2), was concentrated at 40°C using rotary evaporator (Buchi Rotovapor R-205, Switzerland). From the concentrated carotenoid extract, PSC was purified by phase separation using hexane and purity was confirmed by high-performance liquid chromatography (HPLC). 19
Experimental Animals
Female Wistar rats (CFT strain) weighing 100 ± 5 g were obtained from Animal House Facility of the Institute and housed in individual stainless steel cages at room temperature (25°C ± 2°C). The relative humidity of 65% ± 10% and 12-hour dark–light cycle were maintained. Permission from the Institutional Animal Ethics Committee (IAEC) was obtained (IAEC approval no. 142/09) for the study, and the experiments were conducted in accordance with the guidelines prescribed by Committee for the Purpose of Control and Supervision on Experiments on Animals (CPCSEA), Government of India.
Safety Assessment
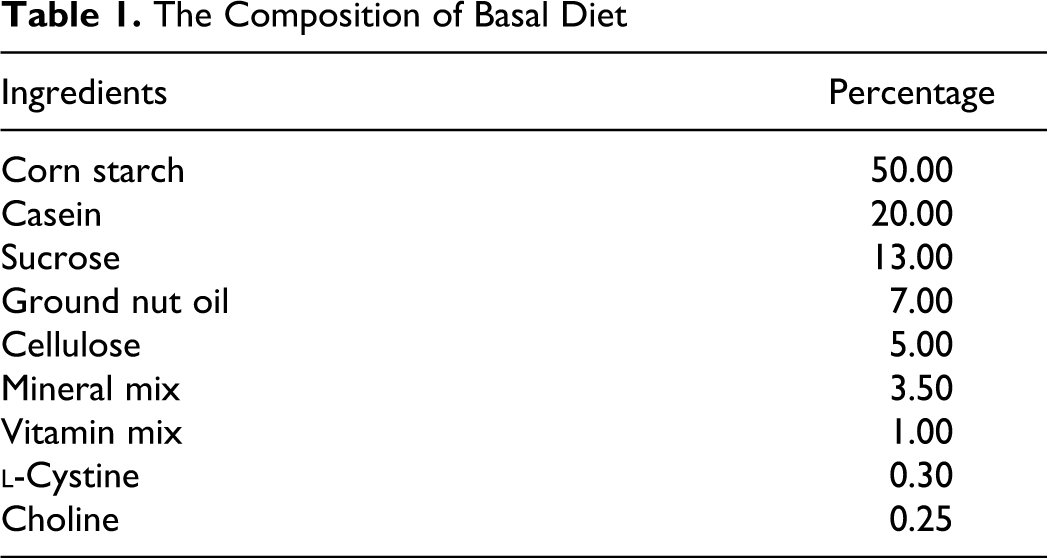
Twenty-four animals were randomly distributed into 4 groups (n = 6 per group). They were maintained for 4 weeks on various experimental diets which consisted of AIN-93G as the basal diet (Table 1) with or without PSC. Animals in control group were fed with only basal diet. Animals in groups I, II, and III received basal diet containing PSC at 0.05%, 0.10%, and 0.25%, respectively. Test feed material was made by mixing the diet with PSC dissolved in ground nut oil. To make the feed uniform, ground nut oil in the basal diet was replaced. All the animals were observed daily, for onset of any signs of toxicity, till the end of the experimental duration. Individual body weights and feed intake were recorded once a week and daily. Animals, fasted overnight, were killed under light ether anesthesia and liver, lungs, kidney, spleen, brain, adrenals, and heart from each rat were weighed and the organ:body weight ratio were calculated before histopathological analyses. Tissues fixed in Bouin fixative were processed to obtain sections of 5 µm and stained using hematoxylin and eosin. Eye samples were homogenized with 9 parts of ice-cold isotonic saline. Partially saturated canthaxanthin was extracted from eye samples according to the procedure of Baskaran et al. 20 The homogenate (0.8 mL) was added to 2.4 mL of dichloromethane: methanol (1:2 v/v) and mixed for 1 minute using a vortex mixture followed by the addition of 4.0 mL of hexane. After centrifugation at 1000g for 5 minutes, the upper hexane/dichloromethane layer was collected. Extraction was repeated twice with 1.2 mL of Dichloromethane (DCM) and 2.0 mL of hexane. The pooled extract evaporated to dryness under nitrogen and dissolved in 20 μL of methanol was used for HPLC analysis. 19 The PSC prepared for structural and cell culture studies, described earlier from this laboratory, was used as standard 17
The Composition of Basal Diet
Hematological Study
Blood samples were collected by heart puncture from each group for hematological profile, hemoglobin concentration (Hb), packed cell volume (PCV), erythrocytes (red blood cells), leucocytes (white blood cells), differential counts, mean corpuscular hemoglobin (MCH), mean corpuscular hemoglobin concentration (MCHC), and platelets (PLT). The determination procedure was as described by Bharucha et al. 21
Clinical Chemistry
Blood was allowed to stand at room temperature for 2 to 4 hours for serum separation. Serum glutamate oxaloacetate transaminase (SGOT) and serum glutamate pyruvate transaminase (SGPT) were estimated using commercially available biochemical kits (Aspen Laboratories Pvt Ltd, New Delhi, India) according to modified IFCC (International Federation of Clinical Chemistry and Laboratory Medicine) method. Glucose and alkaline phosphatase (ALP) were determined using commercially available biochemical kits (Span Diagnostics Ltd, India). Creatinine, creatine kinase, urea, bilirubin, and lactate dehydrogenase (LDH) were estimated using commercially available biochemical kits (Aspen Laboratories Pvt Ltd). Before use, the kits were calibrated per the manufacturer’s instructions.
Pharmacokinetics Study
Twelve animals randomly distributed into 4 groups (n = 3 per group) and maintained on basal diet ad libitum for 48 hours were used for the study. Animals were given a single dose of PSC at 5, 10, and 25 mg−1 kg body weight (bw) through gavage administration. Blood from capillary nerves of eye was collected from each group of animals at every 3 for 48 hours using heparinized capillary tube. After transfer into heparinized test tubes, centrifugation was carried out at 1000g for 15 minutes at 4°C to obtain plasma. The plasma (0.8 mL) was used for PSC extraction according to the procedure described earlier. 20
Statistical Analysis
The results of weekly gain in body weights, feed intake, relative organ weights, bioavailability, biochemical, and hematological indices were analyzed using the SPSS statistical package (version 16.0) software. Statistical differences between means were determined by analysis of variance and Duncan test. P value <.05 was considered significant. To quantify the postprandial PSC in plasma over 48 hours, the area under the curve (AUC) was calculated by trapezoidal approximation using the software Prism 5 for Windows (Version 5.04, GraphPad Software, Inc, La Jolla, California, USA).
Results
Clinical Signs and Mortality
In the 28-day repeated toxicity study, dietary intake of PSC at 0.05%, 0.10%, and 0.25% did not result in any treatment-related deaths. Rats fed on diets containing PSC did not develop any clinical signs of toxicity either immediately or during the posttreatment, even at highest dose of 0.25%. No treatment-related changes were noted at the postmortem examination.
Organ Weights, Body Weight Gain, and Feed Intake
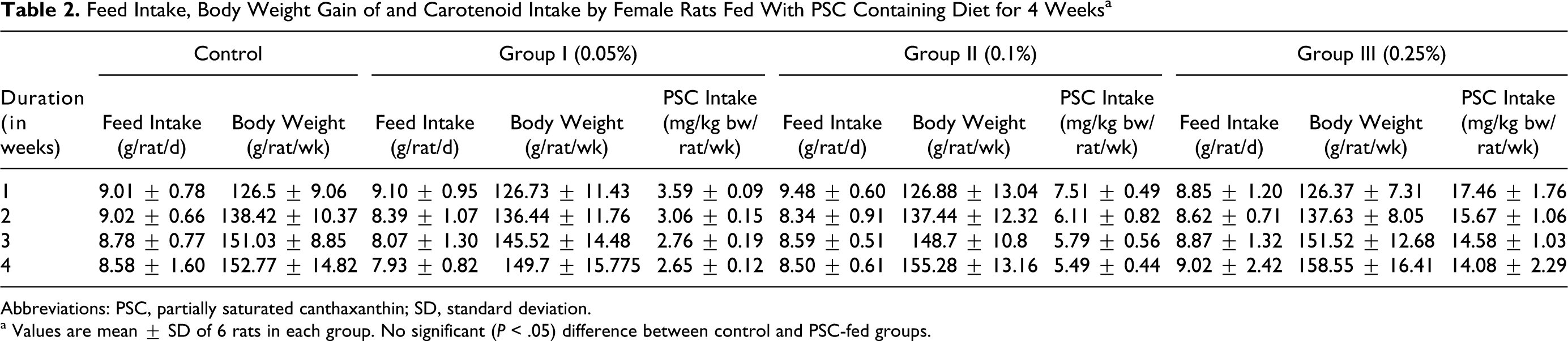
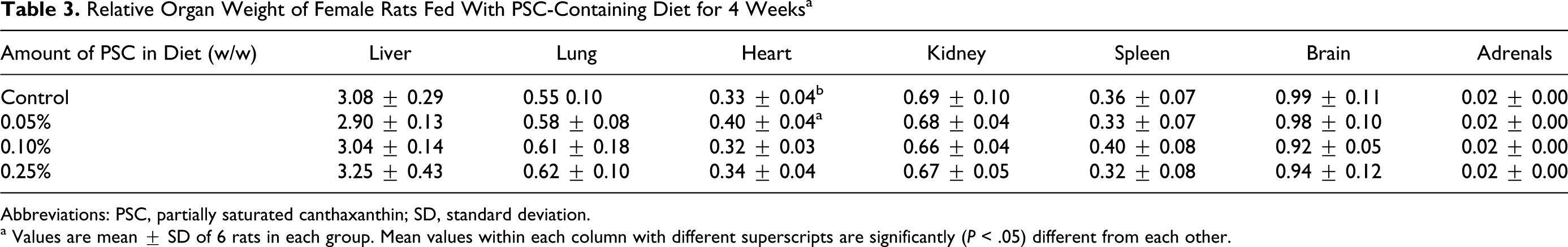
The actual feed intake, mean gain in body weight, and actual intake of PSC per kg body weight are presented in Table 2. There were no significant differences (P < .05) with respect to feed intake and gain in body weight of rats fed with PSC compared with control. There were no significant differences (P < .05) in the relative weights of various vital organs in the PSC-treated rats as well. A significant (P <.05) increase of 21% in relative heart weight was observed in rats fed with 0.05% PSC as compared to control (Table 3).
Feed Intake, Body Weight Gain of and Carotenoid Intake by Female Rats Fed With PSC Containing Diet for 4 Weeks a
Abbreviations: PSC, partially saturated canthaxanthin; SD, standard deviation.
a Values are mean ± SD of 6 rats in each group. No significant (P < .05) difference between control and PSC-fed groups.
Relative Organ Weight of Female Rats Fed With PSC-Containing Diet for 4 Weeks a
Abbreviations: PSC, partially saturated canthaxanthin; SD, standard deviation.
a Values are mean ± SD of 6 rats in each group. Mean values within each column with different superscripts are significantly (P < .05) different from each other.
Hematological Profile
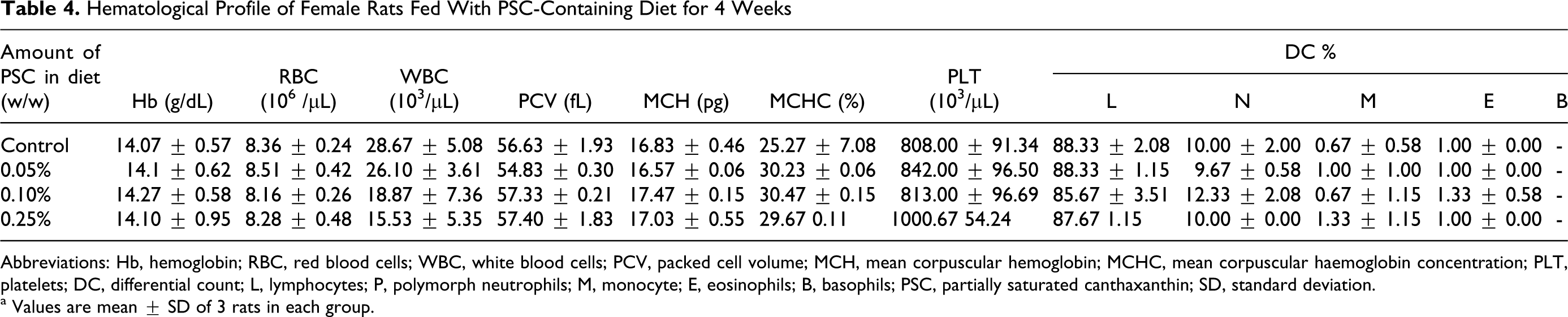
Data on the various hematological parameters are presented in Table 4. No treatment-related adverse effect of PSC on hematology parameters in female Wistar rats was observed. However, statistically significant differences (P < .05) were noted between groups. A significant reduction of 4.36% and 5.15% (P < .05) was observed, respectively, in PCV and MCH in rats fed with 0.05% PSC compared to that fed with 0.10% PSC. Similarly, significant increase of 1.85% and 2.62% (P < .05) was observed in mean corpuscular hemoglobin concentration (MCHC) in rats fed with 0.05% and 0.10% PSC when compared to rats fed with 0.25% PSC.
Hematological Profile of Female Rats Fed With PSC-Containing Diet for 4 Weeks
Abbreviations: Hb, hemoglobin; RBC, red blood cells; WBC, white blood cells; PCV, packed cell volume; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular haemoglobin concentration; PLT, platelets; DC, differential count; L, lymphocytes; P, polymorph neutrophils; M, monocyte; E, eosinophils; B, basophils; PSC, partially saturated canthaxanthin; SD, standard deviation.
a Values are mean ± SD of 3 rats in each group.
Histopathology
No abnormality observed between the control and PSC-fed rats was further evidenced by the absence of any histological changes in various tissues/organs examined. Partially saturated canthaxanthin had no adverse effect on cellular structure of these organs. Gross examination of vital organs during autopsy did not reveal any abnormalities that could be attributed to PSC feeding in rats. No visible deposition or crystal formation was observed in eyes and HPLC analysis revealed the absence of PSC deposition in the eyes of treated rats, suggesting no canthaxanthin-related retinopathy.
Clinical Chemistry
Analysis of various serum enzymes such as SGOT), SGPT, ALP, LDH are presented in Table 5. When compared to controls, no marked alteration in any of the specific activities of enzymes in PSC-fed rats was noticed. However, a significant decrease of 40.92% (P < .05) in creatinine content was found with the group of rats fed with 0.10% PSC.
Effect of Feeding of PSC-Containing Diet for 4 Weeks on Serum Enzymes of Female Rats a
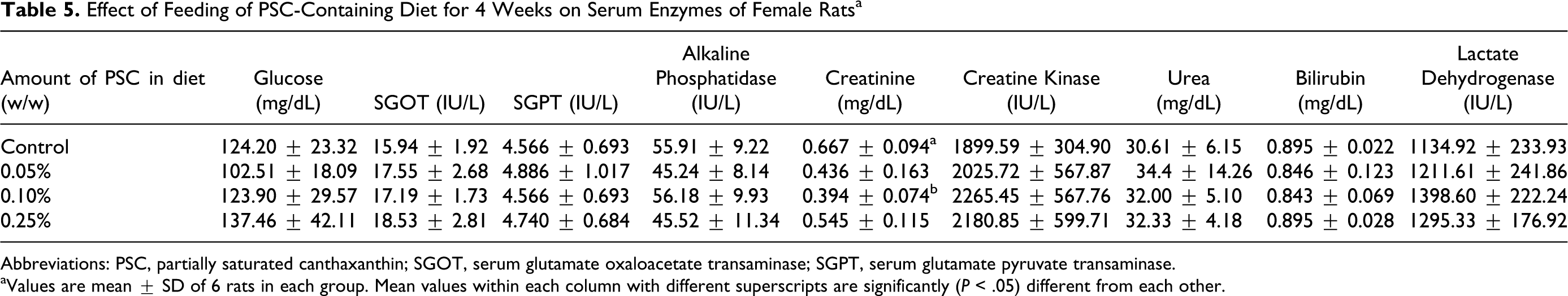
Abbreviations: PSC, partially saturated canthaxanthin; SGOT, serum glutamate oxaloacetate transaminase; SGPT, serum glutamate pyruvate transaminase.
aValues are mean ± SD of 6 rats in each group. Mean values within each column with different superscripts are significantly (P < .05) different from each other.
Pharmacokinetics
In the rats fed with dietary PSC, a gradual increase in PSC concentration was observed in plasma up to 6 hours followed by a gradual reduction at all the dose levels until 36 hours (Figure 3). Rats administrated with a single dose of PSC at 5 mg/kg bw showed 1.38 nmol/mL (Cmax) in the plasma in 3 hours (Tmax). This level decreased gradually to 0.03 nmol/mL at the end of 48 hours. The PSC concentration in the plasma of rats fed with 10 mg/kg bw was 2.93 nmol/mL at 3 hours and the maximal absorption (Cmax) of 4.24 nmol/mL was reached 6 hours (Tmax) of postdosing. Although after 36 hours it decreased to 0.08 nmol/L, there was an increase of 37.45% in the plasma PSC after 48 hours. At highest PSC dosage level (25 mg/kg bw), its plasma concentration was 5.48 nmol/mL which increased to 7.22 nmol/mL (Cmax) at 6 hours (Tmax) and the level decreased to 0.13 nmol/mL at 36 hours. As earlier, 33.94% increase in PSC was seen after 48 hours. Plasma of rats fed with PSC when analyzed by HPLC showed a unique peak corresponding to the xanthins (Figure 4). All other peaks were in common as detected in the control animals fed with basal diet. The AUC, elimination half-life, elimination rate, volume of distribution, and oral clearance for groups I, II, and III are described in Table 6. Based on the average plasma PSC levels, the AUC value of PSC in group III (105.20 nmol/mL/h) was highest followed by group II (63.37 nmol/mL/h) and group I (10.99 nmol/mL/h).
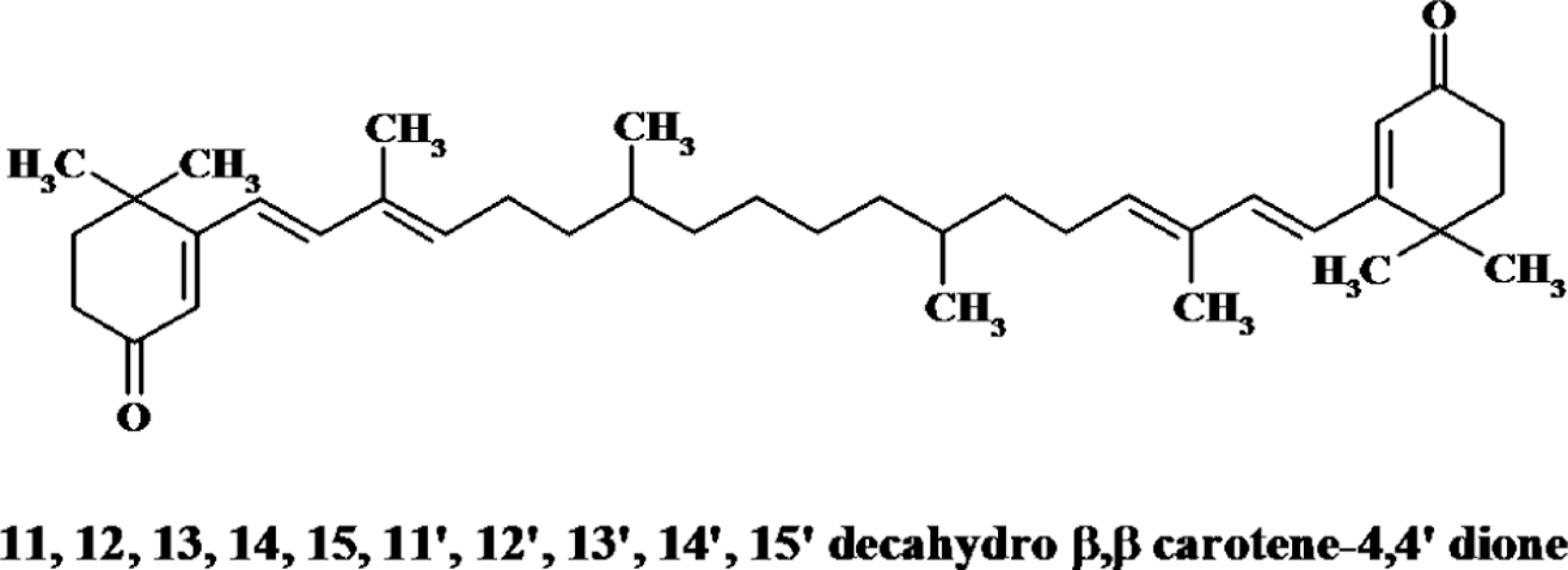
Structure of partially saturated canthaxanthin.
High-performance liquid chromatography elution of partially saturated canthaxanthin from Aspergillus carbonarius.
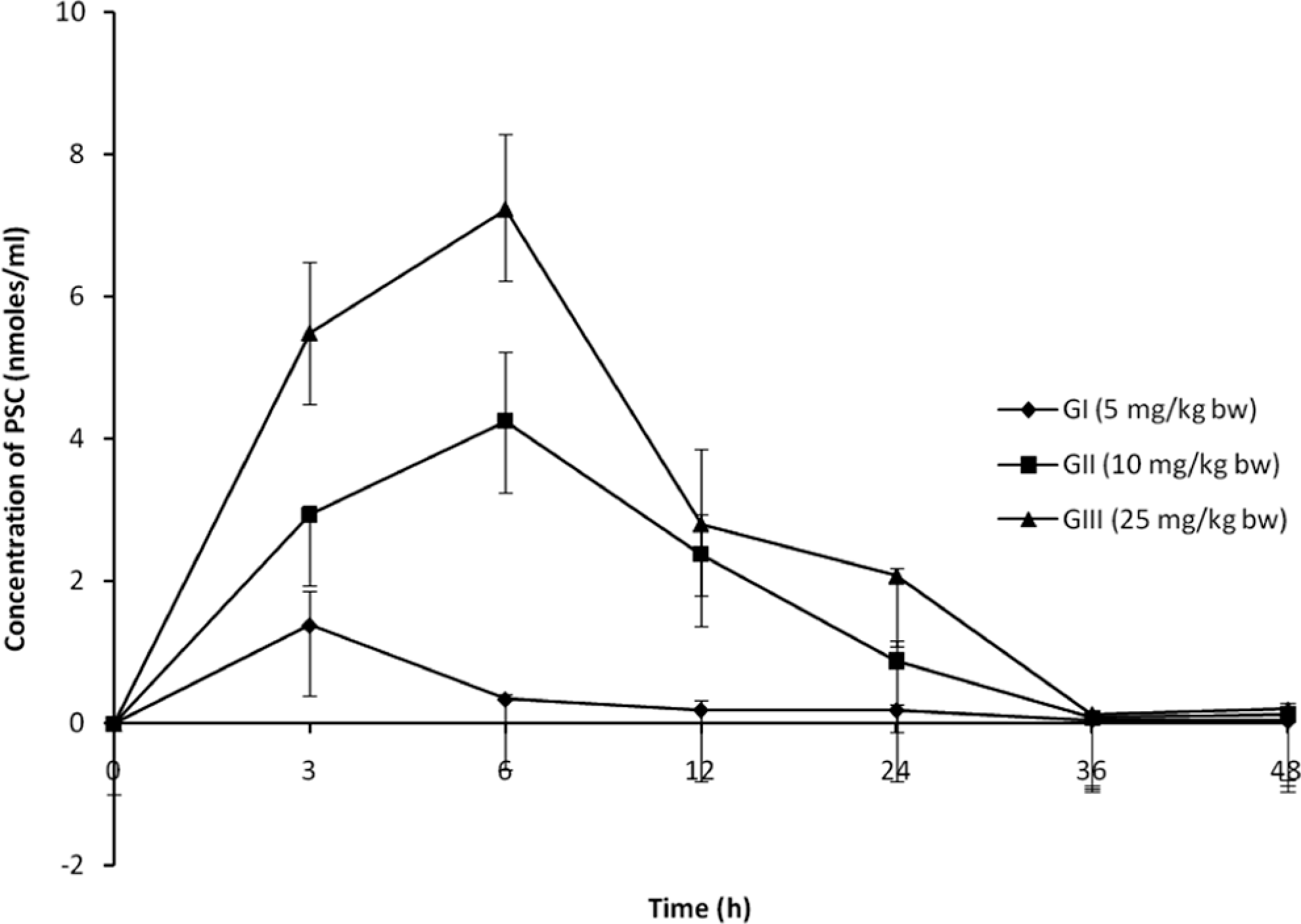
Partially saturated canthaxanthin concentration in plasma at different time intervals. Values are mean ± SD of 3 rats in each group. SD, standard deviation.
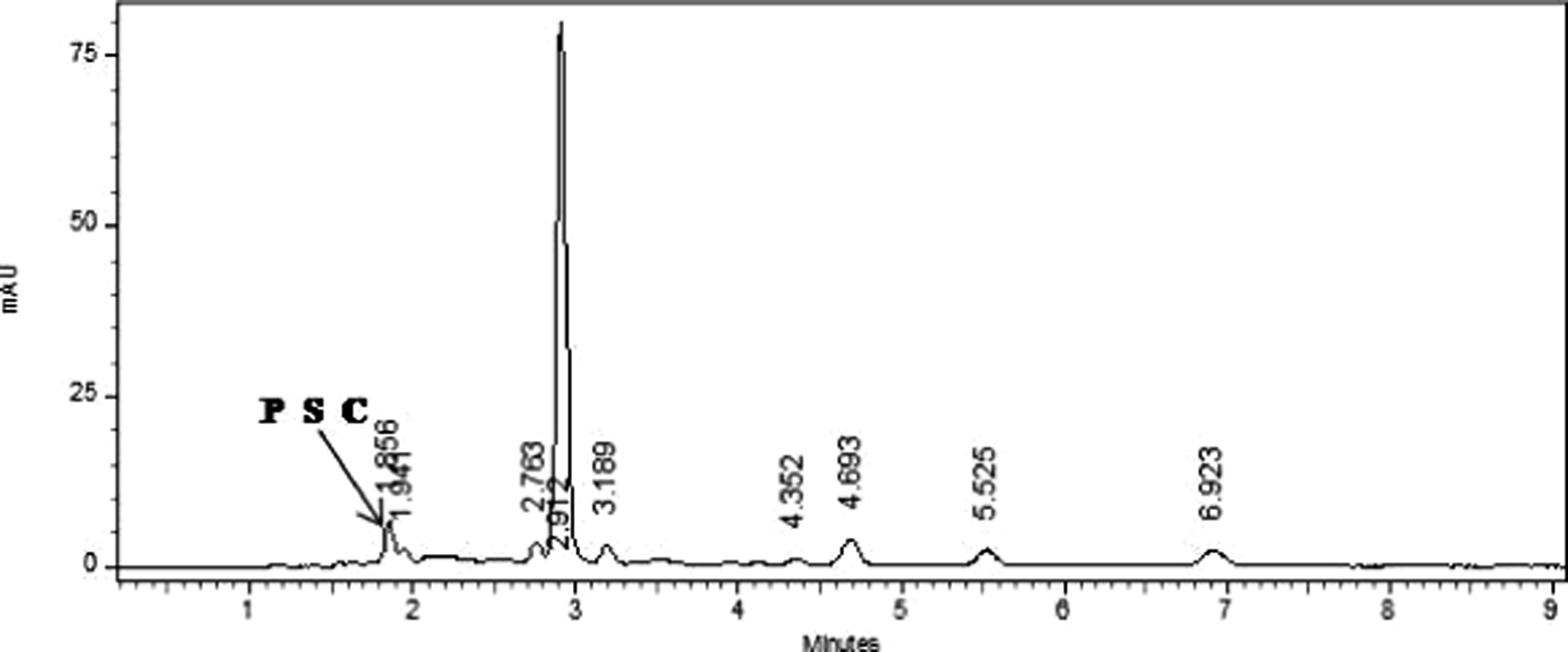
High-performance liquid chromatography of plasma sample showing the presence of PSC. PSC indicates partially saturated canthaxanthin.
Single-Dose Oral Pharmacokinetic Parameters for Plasma Levels of PSC
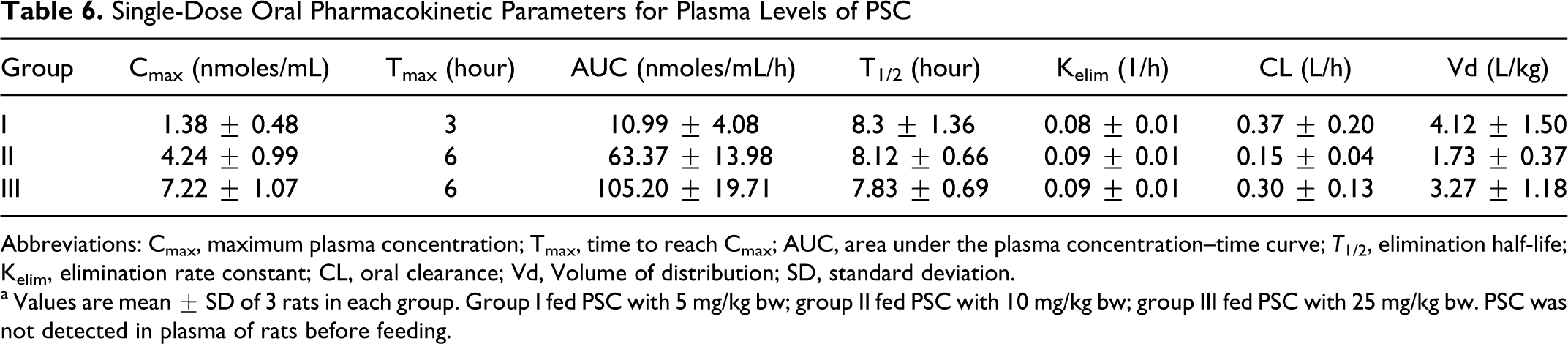
Abbreviations: Cmax, maximum plasma concentration; Tmax, time to reach Cmax; AUC, area under the plasma concentration–time curve; T 1/2, elimination half-life; Kelim, elimination rate constant; CL, oral clearance; Vd, Volume of distribution; SD, standard deviation.
a Values are mean ± SD of 3 rats in each group. Group I fed PSC with 5 mg/kg bw; group II fed PSC with 10 mg/kg bw; group III fed PSC with 25 mg/kg bw. PSC was not detected in plasma of rats before feeding.
Discussion
The results described in this study, using purified PSC, showed its safety and possible use as food. Determination of the PSC in plasma and nondeposition in the eye together with our earlier report on its ability to induce apoptosis in prostate cancer cell lines 17 suggested its possible application as a drug. Nonsignificant differences in hematological parameters and serum enzyme levels between groups indicate no adverse effect of PSC on liver and kidney functioning. These results were similar to the observation reported by Steiger and Buser 22 in albino rats. Short-term toxicity study conducted for 4 weeks by feeding PSC of A. carbonarius did not cause any mortality, change in body weight, food consumption pattern, organ weight, and other adverse effects in Wistar rats like the reports of lutein and its esters, 23 which are related carotenoids. It has also been shown that canthaxanthin fed to other animals, such as dogs, at dose levels of 0, 250, or 500 mg/kg bw/d for 13 weeks neither showed any difference in food and water intake/body weight gain nor on ophthalmoscopic examination. 24
Following oral administration, PSC appeared within 3 to 6 hours in the blood. While the maximum concentration of PSC in the plasma of rats reached after 3 hours in group I animals, group II and III animals recorded maximum PSC in the plasma in 6 hours. The time periods (range: 3-6 hours) was similar to the report of Mathews-Roth et al 25 who reported that the maximum absorption of radioactive canthaxanthin in the rats fed placebo diets occurred around 4 hours of postdosing. The differences in PSC peak time observed in other animals, such as rainbow trout (20 hours), Atlantic salmon (30 hours), and Rhesus monkeys (8 hours), 25 –27 may be due to the difference in structure of these carotenoids or difference in physiology of the animals. In canthaxanthin-fed rainbow trout, 28 a half-life for the terminal clearance of canthaxanthin was 12.6 hours. In this study, T1/2 for PSC was 8.30, 8.12, and 7.83 hours for groups I, II, and III, respectively. Although elimination of PSC was longer, it did not cause any effect to the rats.
In conclusion, canthaxanthin is the sole pigment of the carotenoid family registered for use in animal feed and human food. Canthaxanthin is a nonprovitamin A carotenoid authorized for use in animal feed as a coloring agent in poultry feeds and fish feeds in accordance with Council Directive 70/524/EEC as E-161 g. It is also a color additive for food (E-161). Canthaxanthin (E-161) is permitted as a coloring agent for medicinal products. Absence of toxicity of A. carbonarius PSC suggests its dietary application for anticancer and other applications.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: AK and AHS gratefully thank Council of Scientific and Industrial Research (CSIR), and Indian Council of Medical Research (ICMR) New Delhi, India, for supporting their research through Senior Research Fellowship awards.
