Abstract
Background
Oleanolic acid (OA) has notable anti-inflammatory and anti-tumor effects, but the role of OA in cerebral ischemia-reperfusion injury (CIRI) has not been reported so far.
Methods
Oxygen and glucose deprivation/reoxygenation (OGD/R) model was induced in PC12 cells. MTT assay was used to detect the cell viability of PC12 cells, while ELISA assay detected the expression of TNF-α, IL-1β and IL-6. The expression of superoxide dismutase (SOD), malondialdehyde (MDA) and reactive oxygen species (ROS) was detected by the appropriate kits, and cell apoptosis by Tunel technique. Western blot assay detected the expression of apoptosis-related proteins. The cell transfection technique overexpressed miR-142‐5p. After overexpressing miR-142‐5p by cell transfection technique, the expression of miR-142‐5p was detected by RT-qPCR.
Results
Besides the ability to promote cell acitivity, OA ameliorated OGD/R-induced inflammatory response and apoptosis in PC12 cells. Moreover, the capability of OA to alleviate OGD/R-induced inflammation and apoptosis in PC12 cells was observed to be related to the down-regulation of miR-142‐5p.
Conclusion
OA improved inflammatory response and apoptosis of PC12 cells induced by OGD/R through downregulating miR-142‐5P
As the most sensitive organ to hypoxia, the brain tissue can be regionally damaged and lose its function under the joint force of ischemia and hypoxia. It is acknowledged that ischemia per se is not the sole factor causing damage to brain tissue. But upon the restoration of blood supply, excessive free radicals attack cells in this part of the tissue that regained blood supply, which is called “cerebral ischemia-reperfusion injury (CIRI)”. 1 CIRI seriously endangers human health with the characteristics of high incidence, high disability rate and high mortality, 2 so it is very important to find new drugs to treat CIRI.
Oleanolic acid (OA) is a pentacyclic triterpene widely found in various fruits and edible plants. It has obvious anti-inflammatory, immunomodulatory, hypoglycemic, liver-protecting and anti-tumor effects. 3 -6 Study has shown that OA can prevent renal injury induced by ischemia and hypoxia through antioxidant, anti-inflammatory and anti-apoptotic activities in rat of acute renal injury models induced by ischemia and hypoxia. 7 In the model of hepatic ischemia reperfusion, OA pretreatment, which significantly reduces the apoptosis of hepatocytes, inhibits the release of HMGB1, and alleviates inflammation, shows the protective effect of OA on hepatic ischemia reperfusion injury. 8 However, the specific effect of OA on CIRI has not been reported so far.
MicroRNAs (miRNAs), a class of highly conserved non-coding single-stranded RNA, can regulate about 60% of human genes and therefore play an important role in human biology. 9 Study has reported that the expression of miR-142‐5p was up-regulated in OGD/R-induced hippocampal neurons and promoted cell injury, while the down-regulation of miR-142‐5p alleviated OGD/R-induced neuron injury by promoting Nrf2 expression. 10 Thus, miR-142‐5p might be used as an indicator for the diagnosis and treatment of CIRI or a target for new treatments of CIRI. In addition, Chen et al showed that OA restricted fibrosis induced by hyperglycemia of NRK-52E cell by inhibiting miR-142‐5P and then promoted expression of PTEN and autophagy level. 11 Therefore, we speculated OA could play a role in CIRI by targeting miR-142‐5P.
In this paper, we examined the effect of OA on OGD/R induced inflammatory and apoptotic damage in PC12 cells and investigated its therapeutic targets, which provided a solid theoretical basis for OA in the targeted therapy of CIRI.
Results
OA Promoted Proliferation of OGD/R Induced PC12 Cells
The chemical structure formula of OA is shown in Figure 1(A).To know the critical role of OA in CIRI, the cell viability of PC12 cells treated by OA at different concentrations was determined by MTT assay. The results showed that the viability of 80μM OA-induced cells began to decrease compared with the control group (Figure 1(B)). Subsequently, PC12 cells induced by OGD/R were treated with OA at different concentrations. We found that the cell viability of OGD/R-induced PC12 cells increased compared with OGD/R group, and 40μM + OGD/R group, which displayed the most obvious increase in the cell viability, rendered us choose 40μM OA for subsequent experiments (Figure 1(C)).

OA promoted proliferation of OGD/R induced PC12 cells.A. The chemical formula of OA. B.CCK-8 was used to detect the cell viability after the treatment of different concentrations of OA. C. CCK-8 was used to detect the cell viability of OGD/R induced PC12 cells after the treatment of different concentrations of OA. *P < 0.05, ***P < 0.001 vs Control; ##P < 0.01, ###P < 0.001 vs OGD/R.
OA Inhibited Inflammation of OGD/R Induced PC12 Cells
The results of ELISA showed that the expression of inflammatory cytokines TNF-α (Figure 2(A)), IL-1β (Figure 2(B)) and IL-6 (Figure 2(C)) increased after OGD/R induction compared with the control group. The expressions of TNF-α, IL-1β and IL-6 in the OA +OGD/R group were decreased compared with the OGD/R group. In addition, compared with the control group, the expressions of ROS (Figure 2(D)) and MDA (Figure 2(E)) in OGD/R group was significantly increased, while the expression of SOD (Figure 2(F)) was decreased. Compared with OGD/R group, ROS and MDA expression in OA +OGD/R group decreased, while SOD expression increased. These results suggest that OA inhibited inflammation induced by OGD/R in PC12 cells.

OA inhibited inflammation of OGD/R induced PC12 cells. ELISA assay were used to detect the expressions of TNF-α (
OA Inhibited Apoptosis of OGD/R Induced PC12 Cells
TUNEL results showed that compared with the control group, the apoptosis level in OGD/R group was increased (Figure 3(A)), accompanied by increased expression of Bax, cleaved caspase-3, cleaved caspase-9 proteins, and decreased bcl-2 (Figure 3(B) and (C)). Compared with the OGD/R group, apoptosis was decreased in the OA +OGD/R group, accompanied by decreased Bax, cleaved caspase-3, cleaved caspase-9 proteins, and increased bcl-2 proteins. The above results indicated that OA inhibited the PC12 cell apoptosis induced by OGD/R.
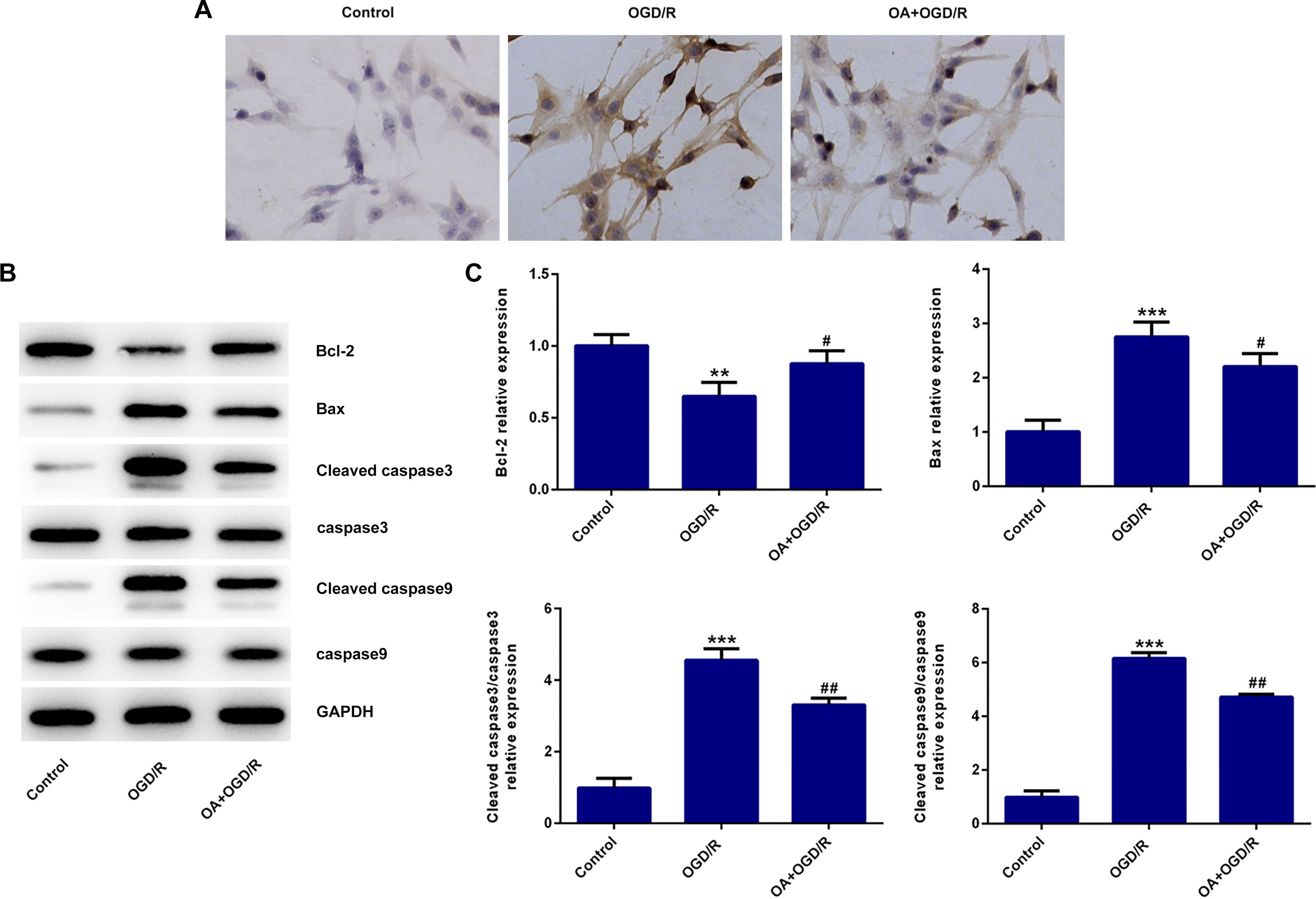
OA inhibited apoptosis of OGD/R induced PC12 cells. A. TUNEL assay was used to detect the apoptosis of cells. B. Western blot detected the expression of apoptosis related proteins. **P < 0.01, ***P < 0.001 vs Control; #P < 0.05, ##P < 0.01 vs OGD/R.
OA Inhibited Inflammation and Apoptosis of OGD/R Induced PC12 Cells by Down-Regulating miR-142-5p
During the experiment, we found that the expression of miR-142‐5p was abnormally increased after OGD/R induction compared with the control group. After the action of OA, the expression of miR-142‐5p in OA +OGD/R group was significantly down-regulated (Figure 4(A)). Therefore, we speculated whether miR-142‐5p was a target for OA to inhibit OGD/ R-induced inflammation and apoptosis in PC12 cells. To verify our conjecture, miR-142‐5p was overexpressed in cells by cell transfection technique and transfection efficiency was measured by RT-qPCR (Figure 4(B)). Cells were divided into Control group, OGD/R group, OA +OGD/R + mimic NC and OA +OGD/R + miR-142‐5p mimic. ELISA results showed that compared with OA +OGD/R + mimic NC, the expressions of TNF-α (Figure 4(C)), IL-1β (Figure 4(D)) and IL-6 (Figure 4(E)) in the OA +OGD/R + miR-142‐5p mimic group were significantly increased, the expressions of ROS (Figure 4(F)) and MDA (Figure 4(G)) were also increased, and the expressions of SOD (Figure 4(H)) were decreased. Subsequently, apoptosis level was detected. Compared with OA +OGD/R + mimic NC, apoptosis of the OA +OGD/R + miR-142‐5p mimic group increased (Figure 5(A)), accompanied by increased Bax, cleaved caspase-3, cleaved caspase-9, and decreased bcl-2 (Figure 5(B) and (C)). These results suggested that OA inhibited inflammation and apoptosis of OGD/R induced PC12 cells by down-regulating miR-142‐5p.

OA inhibited inflammation of OGD/R induced PC12 cells by down-regulating miR-142‐5p. A and B. RT-qPCR was used to detect the expression of miR-142‐5p. ELISA assay were used to detect the expressions of TNF-α (

OA inhibited apoptosis of OGD/R induced PC12 cells by down-regulating miR-142‐5p. A. TUNEL assay was used to detect the apoptosis of cells. B. Western blot detected the expression of apoptosis related proteins. ***P < 0.001 vs Control; ###P < 0.001 vs OGD/R; ΔP < 0.05, ΔΔP < 0.01, ΔΔΔP < 0.001 vs OA +OGD/R + miR-142‐5p-NC.
Discussion
CIRI is a complex pathological process, which can eventually lead to oxidative stress, cellular inflammation and apoptosis of brain neurons, thus aggravating the brain injury. In our paper, we selected the classic OGD/R cell model to simulate CIRI in vitro. We found that OGD/R induced decreased cell activity, inflammatory response and significantly increased apoptosis level, indicating the success of the model. We found that after OGD/R induction, cell activity decreased, aggravated inflammatory response and significantly increased apoptosis level, indicating the successful establishment of the model
OA is a broad-spectrum antimicrobial drug, which is widely used in the treatment of inflammatory diseases and infectious diseases. 12,13 Research showed that OA can improve diet-induced obesity by modulating fat preference and fever in mice. 14 OA can inhibit inflammatory cell infiltration, reduce oxidative stress response of cells, and inhibit cell apoptosis, thus playing a protective role in lung injury of mice. 15 However, the effects of OA on inflammation and apoptosis in CIRI have not been reported so far. In this paper, we found that OA can promote OGD/ R-induced cell proliferation and inhibit OGD/ R-induced cell inflammation and apoptosis, so as to achieve the therapeutic effect of CIRI.
During the experiments, we also found that the expression of miR-142‐5p was abnormally increased in OGD/ R-induced PC12 cells, which was reversed after OA was given. Likewise, previous eveidence noted that the expression of miR-142‐5p was also abnormally elevated in the lung ischemia-reperfusion injury model of pigs. 16 In addition, miR-142‐5p was up-regulated in OGD/ R-induced hippocampal neurons and promoted OGD/R-induced cell injury, while the down-regulation of miR-142‐5p alleviated OGD/ R-induced neuron injury by promoting Nrf2 expression. 10 These findings all suggested that miR-142‐5p played an important role in ischemia-reperfusion injury and it might be a potential diagnostic marker or therapeutic target for this disease. Moreover, OA can limit cell fibrosis by targeting the expression of miR-142‐5p. 11 Therefore, the query about the mechanism by which OA exerts therapeutic effects on CIRI was raised: Whether OA targets miR-142‐5p to affect the progression of CIRI? In this paper, it was found that overexpression of miR-142‐5p could reverse the inhibitory effect of OA on OGD/R-induced inflammation and apoptosis. This suggested that OA may inhibit OGD/R-induced inflammation and apoptosis in PC12 cells by down-regulating miR-142‐5p.
Conclusion
In this paper, we confirmed that OA can improve OGD/ R-induced inflammatory response and apoptosis injury in PC12 cells by down-regulating miR-142‐5p. Our paper provided a solid theoretical basis for OA in the targeted therapy of CIRI.
Materials and Methods
Cell Culture
PC12 cells were purchased from the Type Culture Collection of the Chinese Academy of Science and incubated in DMEM (Gibco; Thermo Fisher Scientific) added with 10% FBS (Gibco; Thermo Fisher Scientific) at 37 ˚C with 5% CO2.
Oxygen and Glucose Deprivation/reoxygenation (OGD/R) Model
The PC12 cells were incubated in a hypoxic condition containing 95% N2 and 5% CO2 in serum/glucose-free DMEM medium at 37 ˚C. 2 hours later, the medium was replaced by the high-glucose DMEM containing 15% FBS, and then the cells were transferred into the normoxic incubator with 95% air and 5% CO2 for 12 hours.
Transfection
Cells were transfected with 20 nmol/L of miR-142‐5p mimics or negative control (mimic-NC) duplex (GenePharma, Shanghai, China) using Lipofectamine 2000 transfection kit (Invitrogen, Carlsbad, California), according to the manufacturer’s protocol.
MTT Assay
A total of 5 × 103 cells/well were seeded into 96-well plates (Corning Incorporated). The cells were grown for 24 hours, and subsequently 20 µl of modified tetrazolium salt (5 mg/mL) was added to each well and incubated for 4 hours at 37 °C. The supernatant was subsequently removed and 100 µl dimethyl sulfoxide (Sigma-Aldrich; Merck KGaA, Darmstadt, Germany) was added to dissolve the formazan crystals. The absorbance was evaluated at a wavelength of 490 nm with a Bio-Rad Benchmark micro-plate reader (Bio-Rad Laboratories, Inc., Hercules, CA, USA).
ELISA
The levels of TNF-α, IL-1β and IL-6 in cell supernatant were measured using enzyme-linked immunosorbent assay (ELISA) kit (Shanghai Biovol Corp., Shanghai, China) in compliance with the manufacturer’s instructions.
Measurement of ROS, MDA, and SOD Activity
The levels of ROS, SOD, and MDA in the cells were detected using a corresponding assay kit (Nanjing Jiancheng Corp., Nanjing, China) according to the manufacturer’s instructions.
TUNEL Assay
The treated cells were fixed with 4% paraformaldehyde for 30 minutes then washed with PBS. Later 0.3% Triton-X 100 in PBS was added and incubated for 5 minutes. One step TUNEL apoptosis detection kit (Beyotime, Shanghai, China) was used to estimate the apoptosis efficiency according the manufacture’s instructions. Cells were incubated with 50 µL TUNEL reaction buffer in a 37 °C humidified chamber for 1 hours. The nucleus was counterstained with DAPI for 1 minutes at 25 °C in the dark later and then mounted with anti-fade mounting medium. Stained apoptotic cells were visualized at 20 × by LSM 710 Laser scanning confocal microscope (Carl Zeiss, German).
Western Blot
150 µg protein aliquots isolated from the cells were run on 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE). After blocking with 5% skim milk for 1 hours, the membranes were incubated with the primary antibodies at 4 °C overnight, and subsequently with alkaline phosphatase-conjugated secondary antibodies. The antigen-antibody products were measured by Thermo Scientific Super Signal West Pico Chemiluminescent Substrate (Thermo Scientific, USA). Image J software (version 146; National Institutes of Health, Bethesda, MD, USA) was used to analyze the fold-changes of protein levels. The primary antibodies including anti-Bax (1:1,000; cat. no. 14796S), anti-caspase3 (1:1,000; cat. no. 9953S), anti-caspase9 (1:1,000; cat. no. 9504T), anti-cleaved-caspase3 (1:1,000; cat. no. 9953S), anti-cleaved-caspase9 (1:1,000; cat. no. 9509T), anti-Bcl-2 (1:1,000; cat. no. 3498Tc), and anti-GAPDH (1:1,000; cat. no. 5174S) antibodies were obtained from Cell Signaling Technology, Inc.
RT-qPCR
Total RNA in PC12 cells was extracted by the Trizol reagent (Invitrogen). The purity and concentration of RNA in the extracts was tested by UV spectrophotometry. To test the expression of lncRNA GAS5 and TP53INP1, First Strand cDNA Synthesis Kit (Invitrogen). qRT‐PCR was performed using SYBR Green Mix (Takara, Dalian, Liaoning, China) with the following procedure: 95 °C for 10 minutes, 40 cycles of 95 °C for 10 s, 55 °C for 10 s, and 72 °C for 30 s. Relative expression level was calculated with the 2−ΔΔCt method. Primers used were as follows: miR-142‐5p forward: 5′-CATAAAGTAGAAAGCACTAC-3′; reverse: 5′-GAACATGTCTGCGTATCTC-3′; GAPDH forward: 5ʹ-GAAGGTGAAGGTCGGAGTC-3; reverse: 5ʹ-GAAGATGGTGATGGGATTTC-3’.
Statistical Analyses
All data are represented as the mean ± SD of 3 independent experiments. All data were evaluated using Student’s t-test or one-way analysis of variance (ANOVA) and Tukey’s post test. Values were considered significant when P < 0.05.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
