Abstract
Aim
To explore whether LPA5 was involved in the inflammatory responses in CI/R injury by regulation of NLRC4.
Method
The cerebral I/R model in rats was constructed with ischemia of 2h and different time points of reperfusion. After that, western blot was used to determine protein expression (LPA5, NLRC4, AIM2, caspase-1, cleaved-caspase-1, mature IL-1β, and precursor IL-1β). And LPA5 and NLRC4 expression were also detected by using immunofluorescence experiment. Afterward, two sequence of LPA5-siRNA were injected into rats via intracerebroventricular administration. TTC staining and HE staining were performed.
Result
As the reperfusion time was prolonged, LPA5 content was continuously increased, and the highest expression of NLRC4 was found at 4h of reperfusion. And protein expression of AIM2, cleaved-caspase-1, and mature IL-1β was also at highest level at 4h. And after reperfusion of 4h, LPA5 siRNA1# or 2# was injected into lateral ventricles. LPA5 silence markedly reduced the infract volume and improved the histological change of ischemic zone. And LPA5 silence significantly downregulated NLRC4, AIM2, and the ratio of cleaved-caspase-1/caspase-1 and mature IL-1β/precursor IL-1β. And compared with LPA5-siRNA2#, LPA5-siRNA1# exerted a more significant effect.
Conclusion
Low expression of LPA5 can protect against the inflammatory responses in CI/R model of rats through inhibiting NLRC4 inflammasomes.
Introduction
Stroke is the third leading cause of morbidity and mortality worldwide, and it is the most common reason of long-term disability. 1 To date, inflammation was considered to be crucial for the pathogenesis of stroke, such ischemia stroke and other forms of ischemia brain injury. 2 Currently, many clinical cases have proved poorer outcomes of stroke patients with systemic inflammation.3–5 In recent years, a large number of experimental strokes reported that cerebral ischemia stroke could lead to recruitment and activation of inflammation cells (such as T cells, neutrophils, and so on) with a time-dependent manner. And limiting inflammation could decrease infarct volume and promote neuro-function recovery.6,7 After ischemia, the inflammation responses in the brain mainly showed the activation of many inflammatory cells.8–13 Hence to develop more effective therapeutic interventions, it is necessary to further explore regulators associated with the inflammatory response and underlying mechanisms.
For neuroinflammation, inflammasomes are the crucial players in the initiation and maintenance of the inflammatory cascade. 14 Inflammasomes are referred to cytosolic multi-protein complexes including NLR Pyrin domain containing 1 (NLRP1) and 3 (NLRP3), NLR family apoptosis inhibitory proteins (NAIPs), and NLR CARD domain containing 4 (NLRC4). 15 NLRC4 emerged as both an NLR protein and inflammasome activator, which possesses the common three-domain structure: CARD (Caspase-Activation and Recruitment Domain) domain, a central nucleotide (ATP) binding domain, and a series of C-terminal LRRs. 16 NLRC4 is a cytosolic sensor to activate inflammasome by sensing stimuli. 17 Inflammasomes could lead to chronic inflammation in the infectious disease or cellular stress, which cause the maturation of interleukin-1β (IL-1β). 18 Currently, NLRC4 has been the hotspot for stroke research. After cerebral ischemic, NLRC4 expressed abundantly in neurons. 19 And the deletion of NLRC4 was protective after ischemic stroke. 20
Lysophosphatidic acid (LPA) could bind to specific G protein-coupled receptors (LPA1, LPA2, LPA3, LPA4, LPA5, and LPA6) to exhibit distinct biological functions.21,22 Additionally, we also found abundant LPA receptors in many types of cells,22–24 and LPA receptors also play an important role in central nervous system (CNS) disorders.25–28 When cerebral ischemia occurs, LPA receptors could lead to brain injury. 29 Among these receptor subtypes, LPA5 has been widely studied, indicating that LPA5 could be recognized as an important factor, promoting inflammatory response.30,31 In a word, during ischemia stroke, LPA5 plays an important role in brain damage. 32 However, it is unknown about the relationship between LPA5 and NLRC4. Whereafter, whether LPA5 silencing regulates NLRC4 to decrease the infarct size and improve histological change was explored further.
In this draft, we put forward the hypothesis: LPA5 is closely correlated to NLRC4 in brain tissues of SD rats bearing cerebral ischemia reperfusion. Downregulation of LPA5 disturbed formation of NLRC4 complexes. To test this assumption, we build a transient middle cerebral artery occlusion (tMCAO) model.
Materials and methods
Animals
In our design, 100 adult male SD rats with weight of 300–400g were applied and purchased from Jinan Pengyue Experimental Animal Breeding Co., Ltd (Permission Number: SYXK(Lu)20,180,030). The animal studies were performed in accordance to the institutional ethics guidelines for animal experiments and the National Research Council’s Guide for the Care and Use of Laboratory Animals, approved by the animal management committee of Laizhou People’s Hospital. Prior to model establishment, the rats were housed adaptively for 1 week in an SPF animal room with 12 h-light/dark cycle. During this period, rats have free access to diet and drinking water, and the temperature and humidity were 22 ± 2°C and 50 ± 10%, respectively.
Establishment of tMCAO models and grouping
tMCAO models were constructed by reference to experimental method described by Sapkota et al. 33 with a slight modification. Briefly, rats were anesthetized with 3% pentobarbital sodium (45 mg/kg) i.p. and then placed on the surgical operating plate and kept in a supine position. Next, the isolation of right common carotid artery was performed carefully. A nylon thread was used to occlude the middle cerebral artery for 2h. Then, the thread was removed, restoring blood flow (reperfusion). And reperfusion time was carried out according to the designed time (4 h, 8 h, 12 h, and 24 h) in our study. During period of modeling, rats were kept in heated surgical platform with the temperature of 37 ± 0.5°C. After MCAO, rats were allowed free access to food and water. After the indicated time of reperfusion, the rats were anesthetized through intraperitoneal injection of 3% pentobarbital sodium (45 mg/kg) and sacrificed via cervical dislocation. Then, some brain tissues were extracted immediately and stored at −80°C freezer and 4% paraformaldehyde for following analysis. And other tissues were kept in −20°C freezer for TTC staining.
To test the effect of different times of reperfusion on expression of LPA5 and NLRC4 in ischemic brain tissues, in Experiment 1 (Figure 1(a)), we set a time gradient of reperfusion (4 h, 8 h, 12 h, and 24 h). 8 rats were involved in each time point. And immunofluorescence experiments and western blot assays were employed to measure LPA5 and NLRC4. In Experiment 2 (Figure 1(b)), we continued to analyze the regulation of LPA5 on NLRC4 in ischemia area. Rats were randomly divided into five groups (n = 8): sham group, CI/R group, negative control group (Scrambled siRNA), LPA5-siRNA1# group, and LPA5-siRNA2#. Sequence of LPA5 siRNA1# is CCGGCGTGCTGGATCCACTGGTTTACTCGAGTAAACCAGTGGATCCAGCACGTTTTTG, and sequence of LPA5-siRNA2# is TGCTGTT GACAGTGAGCGAGCGAGATACACAT Graphical abstract of this design. There were two experimental designs, named as Experiment 1 (A) and Experiment 2 (B). Firstly, tMACO rat was performed at ischemia of 2h and reperfusion for different time points, and we selected the optimal time point of reperfusion for the following study according to the peak value of NLRC4 at 4h. Secondly, LPA5 siRNA and its negative control vectors were icv. injected after reperfusion of 4h, and after 24h, the brains were collected for the further experiments.
CGTTTGCATGTAGTGAAGCCACAGATGTACATGCA-AACGATGTGTATCTCGCGTGCCTACTGCCTCGGA, and scrambled siRNA sequence is TGCTGTTGACAG-TGAGCGAGCTTACG
CTGAGTACTTCGAGTAGTGAAGCCACAGATGTACTCGAAGTACTCAGCGTAAGCGTGCCTACTGCCTCGGA. LPA5-siRNA1#, LPA5-siRNA2#, and scrambled siRNA were provided from Shanghai GenePharma. Each rat received 3 μL solution of LPA5-siRNA1#, LPA5-siRNA2#, and scrambled siRNA through immediate intracerebroventricular (icv.) injection after reperfusion of 4h. The liquid was injected into lateral ventricles at a speed of 0.2 μL/min using a mini-pump (RWD, Shenzhen, China).
Triphenyltetrazolium chloride staining
2,3,5-triphenyltetrazolium chloride (TTC) staining kit (T8170, Beijing Solarbio Science & Technology Co., Ltd) was used to visualize the ischemic infarction. The frozen brain tissues were taken out and cut into five slices (about 2 mm for each section). And the slices were stained in 2% TTC solution for 30min at 37°C in the dark environment. Finally, images of sections were obtained and the infarct size was analyzed by Image J 1.49p (National Institute of Health, USA).
Hematoxylin-eosin (H&E) staining
After fixation by 4% paraformaldehyde for 2 days, the slices (5 μm) were dewaxed and rehydrated in gradient alcohol. Next, the sections were placed into water for 5 min and stained successively with hematoxylin and eosin. After dehydration and transparency, the tissues sections were sealed using neutral resin. The images were observed by using DM1000 LED microscope (LEICA, German).
Immunofluorescence staining assay
Briefly, the paraffin sections (5 μm) of brain tissues were de-paraffinized, rehydrated, and antigen-retrieved. Then 0.3% hydrogen peroxide was added in order to quench endogenous peroxidase activity, followed by 3% BSA in PBS solution to block non-specific expression in sections. Afterward, the slices were washed by PBS, and incubated overnight at 4°C with the following primary antibodies: rabbit anti-rat monoclonal antibody LPA5/GPR92 (1:200, bs-15,366R, Bioss) and NLRC4/CARD12(1:300, bs-20,016R, Bioss). After that, the slices were incubated for 2 h at room temperature with secondary antibodies goat anti-rabbit IgG H&L (Cy3) antibody (1:500, A0516, Beyotime). Then, the sections were stained with DAPI (C1005, Beyotime) for 20 min at room temperature. Finally, tissue immunofluorescence images were photographed by using LSM800 laser confocal microscopy microscope (Zeiss, German), and the mean fluorescence intensity of LPA5 and NLRC4 was analyzed by using Image J 1.49p software (National Institute of Health, USA).
Western blot
Ipsilateral brain hemispheres were collected and homogenized. Total protein was extracted using RIPA lysis (P0013 B, Beyotime) and quantified through BCA protein assay kit (P0009, Beyotime). 50 μg protein sample in each group was loaded onto 10% SDS-PAGE and transferred into PVDF membranes. After blocked by 5% skimmed milk, the membranes were cultured with primary antibodies overnight at 4°C. The antibodies were diluted according to the corresponding dilution ratio in instruction. Rabbit anti-rat monoclonal antibody: LPA5/GPR92 (1:1000, orb157371, Biorbyt), NLRC4 (1:1000, orb412596, Biorbyt), AIM2 (1:800, orb330613, Biorbyt), caspase-1 (1:900, orb213641, Biorbyt), cleaved caspase-1 p20 (1:1000, WL02996a, Wanleibio), mature IL-1β (1:500, WL00891, Wanleibio), precursor IL-1β (1:1000, obr500658, Biorbyt), and β-actin (1:2000, ab8227, Abcam). Next day, the membranes were incubated with secondary antibody HRP-conjugated goat anti-rabbit IgG (H&L) (1:2500, ab6721, Abcam) for 1h at room temperature. Protein bands were visualized with enhanced chemiluminescence (ECL) solution. Finally, the protein bands were obtained under ChemiDoc XRS+ gel imaging system (Bio-Rad, USA), which were analyzed by using Image J 14.9p (National Institute of Health, USA).
Statistical analysis
All data are represented as mean ± standard deviation (SD). One‐way analysis of variance (ANOVA) followed by LSD post-tests was used to analyze significant difference between multiple groups through IBM SPSS statistics 19.0. When p-value was less than 0.5, statistical difference was considered significant.
Result
The expression of LPA5, NLRC4, and NLRC4-related proteins in brain tissues of ischemia-reperfusion rats
As shown in Figure 2(a), we found that LPA5 protein in CI/R group exhibited a significant increase than sham group (p < 0.01). And LPA5 expression gradually increased with prolonged reperfusion time. At 24 h, LPA5 expression was highest than other time points of reperfusion (p < 0.05 or p < 0.01). And we also found the cytoplasmic localization of LPA5. In addition, we also analyzed NLRC4 expression in ischemia-reperfusion brain tissues. However, NLRC4 expression is quite different from LPA5 expression. The immunofluorescence results (Figure 2(b)) indicated that NLRC4 expression in CI/R at different time points was higher than sham group (p < 0.05 or p < 0.01). But at 4 h, NLRC4 expression was highest than the other three CI/R groups (p < 0.01). And NLRC4 expression gradually decreased with prolonged reperfusion time. When reperfusion time was 24 h, there was the lowest expression of NLRC4. Nevertheless, NLRC4 expression in CI/R-24 h group was still higher than in the sham surgery group with a significant difference (p < 0.05). Consistent with immunofluorescence assay, western blot results (Figure 3(a)) of LPA5 and NLRC4 showed a similar tendency. After different reperfusion (4, 8, 12, and 24h), immunofluorescence experiment was implemented to detect expression of LPA5 and NLRC4 in brain tissues of tMCAO model. All experiments were repeated at least 4 times. The scale bar is 20 μm. Compared with sham group, *p < 0.05, **p < 0.01; compared with CI/R-4h group, &&p < 0.01; compared with CI/R-8h, ^p < 0.05, ^^p < 0.01; compared with CI/R-12, %p < 0.05. Western blot was used to detect the expression of LPA5, NLRC4, AIM2, Cleaved caspase-1, and Mature IL-1β protein. Image J 1.49p software was used to analyze the grey values of each protein band. All experiments were repeated at least 4 times. Compared with Sham group, **p < 0.01; compared with CI/R-4h group, &p < 0.05, &&p < 0.01; compared with CI/R-8h, ^p < 0.05, ^^p < 0.01; compared with CI/R-12, %%p < 0.01.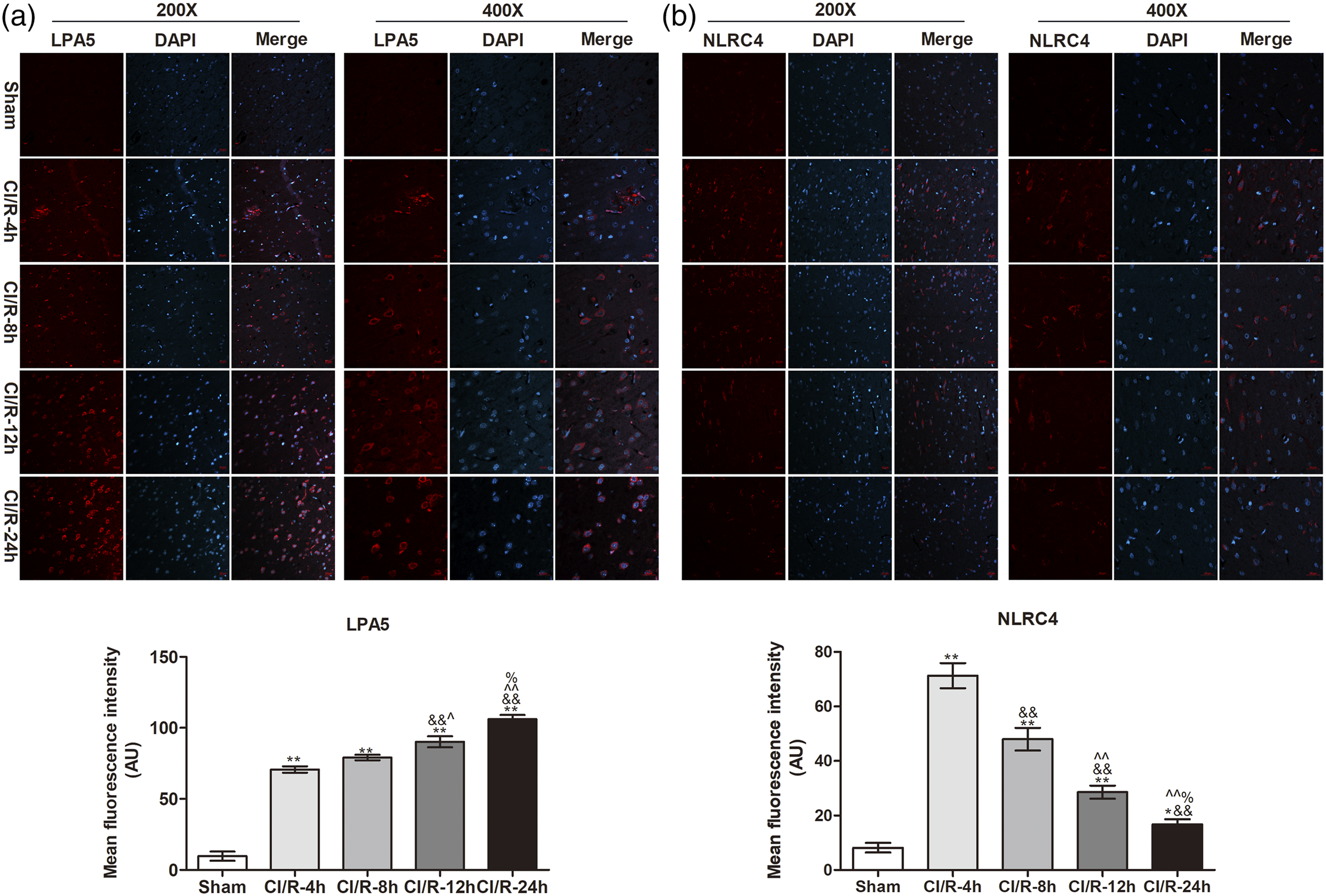

Meanwhile, we detected the NLRC4-related proteins via western blot (Figure 3(b)). The results showed that ischemia-reperfusion raised the expression of AIM2, and boosted the conversion of caspase-1 and IL-1β from inactive form to active form. And From Figure 3(b), we found that AIM2, the ratio of cleaved caspase-1/caspase-1, and mature IL-1β/precursor IL-1β in CI/R-4 h were higher than other time points (p < 0.01). As the reperfusion time increased, the above protein was gradually decreased. At 24 h of reperfusion, those proteins reached lower level than other time points (p < 0.01), and was still higher than sham group (p < 0.01). We chose the reperfusion of 4h for the following analysis.
Low-expressed LPA5 improved the region of brain injury induced by ischemia-reperfusion
Intracerebroventricular injection of two LPA5 siRNAs was carried out and HE staining and TTC staining were used to analyze the pathological changes and cerebral infarction in the ischemic area. From the overall view in Figure 4(a), the ischemia zone was reduced after injection of LPA5-siRNA. And we also provided the images at different magnification to observe the pathology in the ischemic and non-ischemic areas, indicating that there was no neuronal damage, shrinking, and vacuolus in sham group. However, a severe injury of ischemic brain in CI/R-4 h group and si-control group was existing compared with its non-ischemic region. There were a large number of empty bubbles, nuclear wrinkle shrinkage, and disturbed tissues in ischemic zone. While LPA5 siRNA was treated, the improved appearance was observed in LPA5-siRNA1# group and LPA5-siRNA2# group. Then, the brain tissues were stained to analyze the infarct volume via TTC staining. As shown in Figure 4(b), the infarct size of other groups was significantly higher than sham group (p < 0.01). There was no significant difference between CI/R-4 h and si-control groups (p > 0.05). After LPA5-siRNA injection, the infarct volume was significantly decreased (p < 0.01), especially LPA5-siRNA1#. Transfection of LPA5 siRNA decreased the infarct volume and improved the pathological status. (A) HE staining; (B) TTC staining. All experiments were repeated at least 4 times. The scale bar was 100 μm and 200 μm. Compared with Sham group, *p < 0.05, **p < 0.01; compared with CI/R-4h group, &&p < 0.01; compared with si-control, %%p < 0.01.
LPA5 silence inhibited NLRC4 expression and its associated proteins
We performed immunofluorescence staining (Figure 5) and western blot (Figure 6(a)) to analyze the NLRC4 expression after LPA5 siRNA injection. As shown in Figure 5, mean fluorescence intensity of LPA5 and NLRC4 was markedly decreased compared with CI/R-4 h group and si-control group after LPA5-siRNA through icv. injection (p < 0.01), indicating that LPA5 siRNA could inhibit NLRC4 expression, particularly in LPA5-siRNA1#. And there was a similar result between CI/R-4h and si-control groups without significant difference (p > 0.05). Consistent with immunofluorescence results, western blot (Figure 6(a)) also showed a reduced relative expression of NLRC4 protein after LPA5-siRNA treatment. Furthermore, the relative expression of AMI2, the ratio of cleaved caspase-1/caspase-1, and the ratio of mature IL-1β/precursor IL-1β (Figure 6(b)) were also inhibited by LPA5-siRNA. There was significant difference between CI/R-4h and two LPA5-siRNA groups, si-control and two LPA5-siRNA groups (p < 0.01). Given all that, LPA5 silence could inhibit NLRC4 in the brain tissues of CI/R rat model, thus reducing brain damage in the ischemic area and the area of cerebral infarction. Immunofluorescence assay was performed to detect the LAP5 and NLRC4 expression in brain tissues of CI/R rats after silencing LPA5. All experiments were repeated at least 4 times. The scale bar is 20 μm. Compared with Sham group, *p < 0.05, **p < 0.01; compared with CI/R-4h group, &&p < 0.01; compared with si-control, %%p < 0.01. Western blot assay was carried out to determine LPA5, NLRC4, AIM2, Cleaved caspase-1, and Mature IL-1β protein after LPA5 siRNA treatment (n = 4). (A) Western blot was employed to detect LPA5 and NLRC4 expression. (B) Western blot was used to analyze AIM2, Cleaved caspase-1, and Mature IL-1β protein. Compared with Sham group, *p < 0.05, **p < 0.01; compared with CI/R-4h group, &&p < 0.01; compared with si-control, %%p < 0.01.

Discussion
In our draft, we found a high expression of LPA5 and NLRC4 after CI/R. Meanwhile, the LPA5 expression exhibited gradually rising trend with a time-dependent manner, and NLRC4 content reached to the peak value at 4h of reperfusion, followed by a gradual decline, but still higher than sham group. Consistent with the reporting results, 32 LPA5 was upregulated in post-ischemic brain. LPA5 is expressed throughout the whole body,34,35 playing an important role in diverse pathological conditions.36–40 To date, LPA5 was a critical regulator to control microglia activation and production of pro-inflammatory cytokines. 32 Gaire BP et al. reported that LPA5 could regulate NLRP3 inflammasome activation to participate in Imiquimod-Induced Psoriasis-Like Lesions. Nevertheless, whether LPA5 modulates NLRC4 to be involved in cerebral ischemia reperfusion is unknown, and this is the focus of this study in our manuscript.
In our study, we found the close relationship between LPA5 and NLRC4. Low-expressed LPA5 caused the downregulation of NLRC4. It is well known that activated NLRC4 could lead to cascade in many different signaling pathways. According to the previous reports, NLRC4 could directly interact with pro-caspase-1 through CARD–CARD interactions, which converted pro-caspase-1 to active, mature caspase-1.41,42 And active caspase-1 could interact with 70 substrates and cleave these substrates, including IL-1β.42,43 For ischemia-reperfusion, oxidative stress induced an inflammatory response through proteins including IL-1β production. 44 Recent study demonstrated that a unified mechanism for downstream caspase-1 recruitment through CARD–CARD interactions by NLRC4 and ASC. Structure modeling was further used to verify that full-length NLRC4 assembles via two separate symmetries at its CARD and its nucleotide-binding domain (NBD), respectively. 45 Consistent with the findings mentioned above, LPA5 siRNA-induced downregulation of NLRC4 could inhibit activation of pro-caspase-1 and precursor IL-1β.
In this paper, we established tMCAO rat model to verify whether LPA5 regulates NLRC4 to affect histological change of ischemic region. Through immunofluorescence staining of LPA5 and NLRC4, we found abundant expression of LPA5 and NLRC4 in brain tissues, indicating the enhanced inflammatory responses after ischemia-reperfusion. Meanwhile, we also measured the downstream proteins (caspase-1 and IL-1β) of NLRC4, and found that caspase-1 and IL-1β were activated and changed into active forms: cleaved caspase-1 and mature IL-1β, respectively. Further, we used immunofluorescence assay and western blot to analyze the effect of LPA5 on NLRC4, and the results showed that LPA5 silence led to the low-expressed NLRC4. Inhibiting LPA5 could reduce the infarct size to improve the injury induced by ischemia-reperfusion. We also analyzed the downstream proteins (caspase-1 and IL-1β) of NLRC4, and found that LPA5-siRNA could inhibit NLRC4 expression further to decrease activation of caspase-1 and IL-1β. In brief, we concluded that ischemia reperfusion led to enhanced inflammatory responses with the upregulation of LPA5 and NLRC4. Furthermore, LPA5 controls NLRC4 further to affect aggravated brain damage. In the future, we discuss deeply the relevant signaling pathway involved in the interaction with LPA5 and NLRC4 during ischemia-reperfusion.
Footnotes
Authors’ contributions
All authors were involved in the design of this study. Material preparation and purchase, data acquisition, and analysis were performed by Xuling Zhang and Tao Huang. Experiment operation was carried out by Xuling Zhang, Tao Huang, and Lubo Lang. The first draft of the manuscript was written by Xuling Zhang. And all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work to take public responsibility for appropriate portions of the content.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Animal experiments were carried out in accordance with the institutional ethics guidelines for animal experiments and the National Research Council’s Guide for the Care and Use of Laboratory Animals, which was approved by Laizhou People’s Hospital.
Data availability statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
