Abstract
Background
In this study, the ultrahigh performance liquid chromatography mass spectrometry/mass spectrometry (UPLC-MS/MS) method was established to test the contents of protocatechuic acid (C7H6O4), salvianolic acid B (C36H30O16), rosmarinic acid (C19H14O3), and cryptotanshinone (C19H20O3) in Qishen Yiqi dripping pills.
Materials and Methods
Methanol was chosen as the solvent according to a certain material–liquid ratio as a solution, and the ultrasonic extraction method was used to prepare the sample solution. UPLC-MS/MS was used to complete a quantitative analysis of the contents of Qishen Yiqi dripping pills. Then, the chromatographic conditions were shown as follows. The stationary phase was Agilent ZORBAX SB-C18 (2.1 mm × 100 mm, 1.8 µm), and the mobile phase was acetonitrile–0.1% formic acid water. The column temperature was 40℃, the injection volume was 3 µL, and the flow rate was 0.2 mL⋅min–1. Finally, the determination method was built to test four chemical constituents under the multiple reaction monitoring (MRM) analysis mode.
Results
The retention times of protocatechuic acid, salvianolic acid B, rosmarinic acid, and cryptotanshinone were 1.26, 1.32, 1.35, and 6.76 min, respectively. In addition, the concentration ranges of four compounds were 0.25–2.0 µg⋅mL−1, 10.0–50.0 µg⋅mL−1, 5.0–50.0 µg⋅mL−1, and 1.0–10.0 µg⋅mL−1, respectively. And the standard curve showed a good linear relationship in a reasonable range (r2 ≥ 0.9950). The average recovery rate was from 96% to 124%. The contents of protocatechuic acid, salvianolic acid B, rosmarinic acid, and cryptotanshinone were 0.12%–0.022%, 17%–8.4%, and 6.5%–8.6% in seven batches of Qishen Yiqi dripping pills, which were in the range of 23%–31%. The instrument ran stably, and the method had good accuracy and repeatability (relative standard deviation [RSD] ≤ 3%). Additionally, the test result was stable within 10 h.
Conclusion
The contents of the four constituents were different among different batches. The quantitative method was established to determine the four chemical components in Qishen Yiqi dripping pills, which could provide a reference for quality evaluation in vivo.
Introduction
Qishen Yiqi dripping pills are the dripping pills dosage form prepared by Modern Pharmaceutical Technology, including four Traditional Chinese Medicine (TCM), namely, Huangqi, Danshen, Sanqi, and Jiangxiang. Huang et al. (2023) found that the sovereign medicine is Huangqi, and the ministerial drug adjuvant drug is Danshen and Sanqi in the preparation. Besides, the adjuvant is Jiangxiang. Huang et al. (2023) also found that Qishen Yiqi dripping pills have an effect on smooth circulation and stopping pains, which could also promote blood stasis and stop bleeding. Based on modern pharmacological research, Qishen Yiqi dripping pills could adjust vitality and nourish the blood, stimulating pulse, and alleviating pain, which could be used to prevent myocardial injury and enhance the production of myocardial cells (Wu et al., 2023). Clinically, Zhang et al. (2022) proved that it mainly treats symptoms of qi deficiency and blood stasis, such as chest tightness and chest pain, shortness of breath, and spontaneous sweating. Chen L, et al. (2022) found that Qishen Yiqi dripping pills mainly play an auxiliary role in the treatment of patients with coronary heart disease, and Chen W, et al. (2022) demonstrated that Qishen Yiqi dripping pills could be combined with Western medicine to treat diseases such as hypertension and angina.
The quality of TCM is closely related to the stability of its active chemical components that especially affect the safety and effectiveness of the clinical application. In the development process of measuring the quality of TCM, the key focus has been on the control of active ingredients. The main objective of the study on active ingredients is to achieve overall control of the quality of TCM. Therefore, domestic and foreign scholars always use certain techniques and methods to quantify the active ingredients in the analysis of TCM, which aims to achieve the overall evaluation of TCM—Chinese herbal decoction pieces—intermediates—TCM preparations. In the study of Dai et al. (2021) and Yu et al. (2023), the ingredients of Qishen Yiqi dripping pills, tanshinone, catechin, salvianolic acid B, rosmarinic acid, and cryptotanshinone have various pharmacological activities, especially in reducing myocardial oxygen consumption and stabilizing heart rate. Generally, the HPLC method is used to analyze its content. With the development of mass spectrometry (MS) technology, the determination accuracy and sensitivity are greatly improved (Cheng et al., 2021; Guo et al., 2022; Rakotondrabe et al., 2023; Yan et al., 2022).
Ultrahigh performance liquid chromatography mass spectrometry/mass spectrometry (UPLC-MS/MS) technology has the characteristics of high resolution and high sensitivity, which is particularly suitable for the in vitro analysis of chemical components of Chinese Herbal Medicine. UPLC-MS/MS is widely used in the quality control of TCM preparations to provide basic research on effective substances (Qi et al., 2010; Wang & Li, 2022). The technology can be used as a benchmark for basic drug research to achieve complete and rapid analysis and identification of chemical components of conventional preparations. The advantages of good instrument stability, good analytical repeatability, and low detection limit in the quantitative analysis would provide technical support for the quality control of TCM (Belarbi et al., 2021; Bodalska et al., 2021; Li et al., 2022; Sands et al., 2021). Based on the principle of compatibility of TCM combined with UPLC-DAD-Q-MS/MS technology, this study has established the content determination method of four chemical components of Qishen Yiqi dripping pills to provide the basis for quality control. In this study, the basic research of effective substances of Qishen Yiqi dripping pills could provide the basic data for the scientific evaluation of the quality of the preparation.
Materials and Methods
Chemical Reagents
All standard substances, namely, protocatechuic acid (≥98%, Lot no. 200118), salvianolic acid B (≥98%, Lot no. 200122), and rosmarinic acid (≥98%, Lot no. 200812), cryptotanshinone (≥98%, Lot no. 201010), were purchased from Chengdu Phytochemical Pure Biotechnology Co., Ltd. Qishen Yiqi dripping pills (180711, 191007, 200510, 201003, 201108, 200906, and 200811, which were singed 1–7, and Tianshili Pharmaceutical Group Co., Ltd.). Methanol (Merck), Acetonitrile (Fisher Co., Ltd.), formic acid (Tianjin Kemiou Chemical Reagent Co., Ltd.), and purified water (Watsons Drinking Water Co., Ltd.).
Instrument
Agilent 6465A high-performance liquid chromatograph (Agilent Technology Co., Ltd.). Agilent Ultivo triple quad LC/MS system (Agilent Technology Co., Ltd.). Ultrasonic cleaner (JP-009, Shenzhen Alliance Cleaning Equipment Co., Ltd.). 1/100000 electronic balance (AL-240, Mettler-Toledo Instruments (Shanghai) Co., Ltd.). Ultralow temperature refrigerator (MDF-492, Sanyo, Japan). Chromatographic column: Agilent ZORBAX SB-C18 (2.1 mm × 100 mm, 1.8 µm).
Preparation of Test Solution
Preparation of Reference Solution
The standard substances were accurately weighed, namely, protocatechuic acid, salvianolic acid B, rosmarinic acid, and cryptotanshinone reference substances (weighing range was 0.960–1.040 mg). Then, we, respectively, put them into the 10-mL volumetric flask and added methanol to the scale. After blending, we got the reference solution with a concentration of 1.000 mg⋅mL−1.
Preparation of Sample Solution
A total of 20 drops of Qishen Yiqi dripping pills were accurately weighed to make a sample solution with two parallel samples prepared for each batch. Methanol was added to the volumetric flask to achieve the ratio of substance: liquid = 1: 10. Ultrasonic extraction was kept for 15 min at room temperature, and ultrasonic power was 40 Hz. The solution was cooled and stewed for centrifugation at 5,000 rpm for 5 min. The next step was that the supernatant was taken and stored at –20°C. During the determination, sample solutions were taken to filter by a 0.22-µm microporous membrane.
Chromatographic Condition and Mass Spectrum Condition
The stationary phase was the C18 column. The mobile phase was acetonitrile (A)–0.1% formic acid water (B). Gradient elution was as follows: 0–1.5 min, 50%–50% A, –2.5 min, –70% A, –4.0 min, –100% A, –5.5 min, –100% A. Flow rate was 0.2 mL⋅min−1. The column temperature was 40°C, and the injection volume was 3 µL. The autosampler temperature was 4°C.
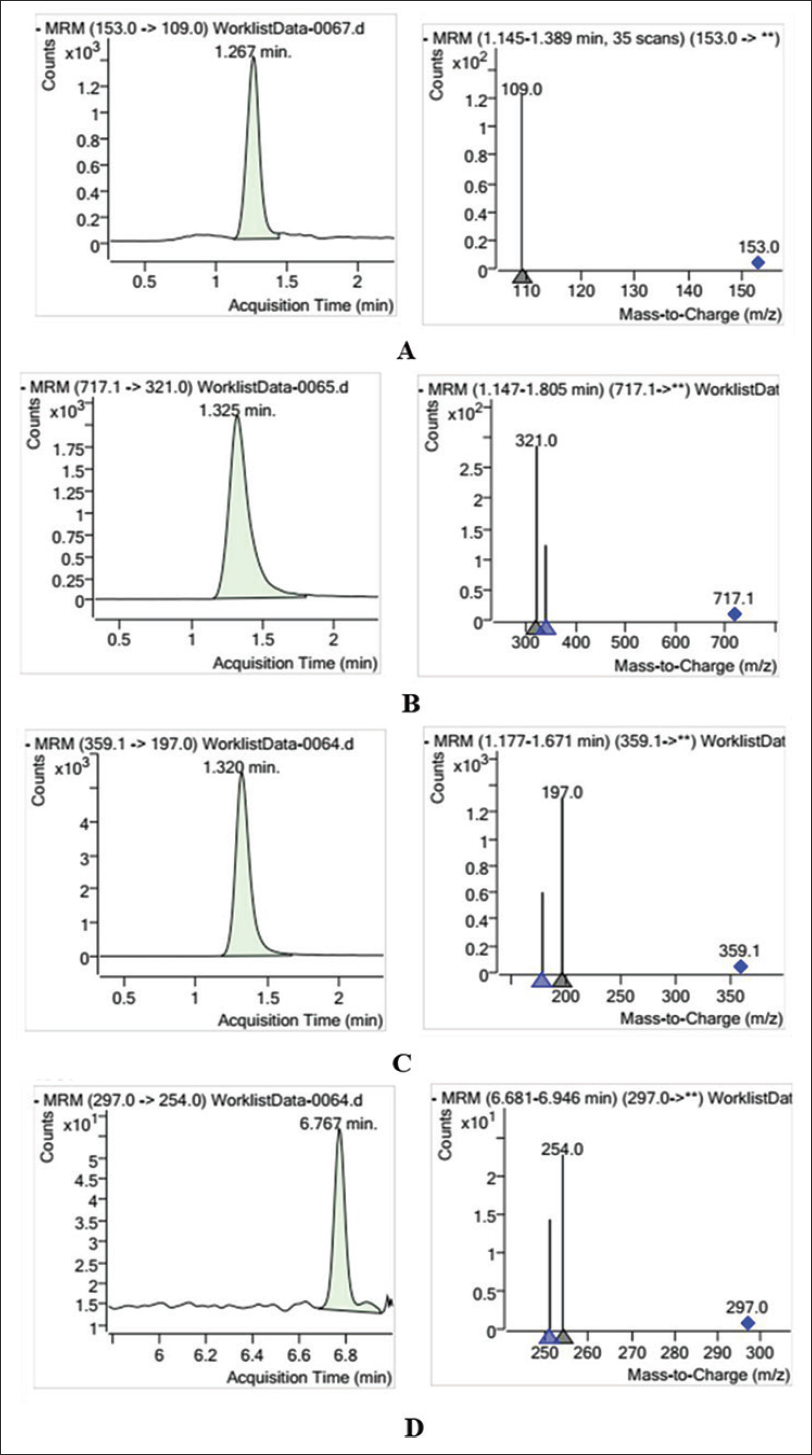
The electric spray ion (ESI) source adopts multiple reaction monitoring (MRM) scanning mode, sets parameters, and conducts measurement. The gas temperature was 340.0°C, and the sheath gas temperature was 380.0°C. Gas flow was set to 10.00 L⋅min−1, and sheath gas flow was 12.00 L⋅min−1. Clustering potential (DP) was 50.0–140 V. Collision energy (CE) was 0–50 V. Spray (PSI) was 40.0 V, and capillary voltage was –3,500 V. The mixed reference solution is injected into the mass spectrometer by the needle pump injection method, and the secondary mass spectrum scanning (sub-ion scanning) is carried out in the negative ion mode. According to the response intensity value, the two parameters of de-clustering voltage (DP/V) and collision energy (CE/V) are optimized (Table 1). The mass chromatogram of the four components is shown in Figure 1.
DP/V and CE/V of Four Components.
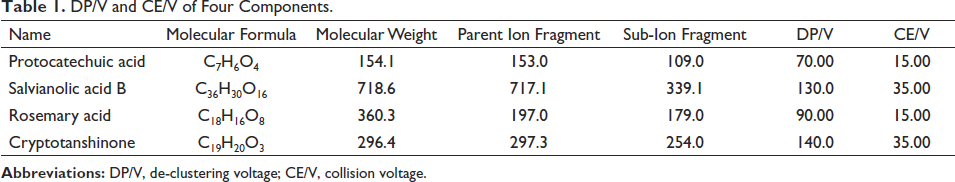
Results
Results of Repeatability Test
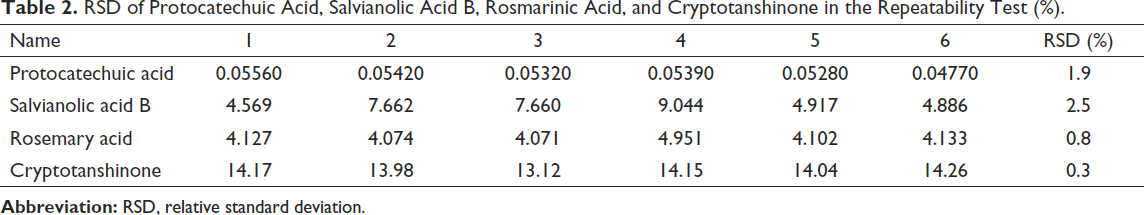
Take six samples of the same batch (Lot no. 180711) to prepare the test solution according to the method under the condition of preparation for the sample solution. The relative standard deviation (RSD) was calculated to be 1.9%, 2.5%, 0.8%, and 0.3%, respectively, which indicated that the method had good repeatability. The error was controllable in the preparation and dilution of the test solution (Table 2).
RSD of Protocatechuic Acid, Salvianolic Acid B, Rosmarinic Acid, and Cryptotanshinone in the Repeatability Test (%).
Results of Recovery Test
Take the process of protocatechuic acid’s operation as an example. Notably, 10 drops were taken from the same batch, which was accurately weighed 0.25 g. The total number of samples was nine and divided into three groups. Each group had three samples, which weighed 2.0 mg (the range of weight was between 1.800 and 2.200 mg), 4.0 mg (the range of weight was between 3.600 and 4.400 mg), and 6.0 mg (the range of weight was between 5.600 and 6.600 mg) of protocatechuic acid reference solution, respectively. The test solution was continuously injected with three lower gradient concentrations. Under the analytical conditions, the content of protocatechuic acid in the test solution that had been added to the reference substance was measured, and the recovery rate (%) was calculated. The average recoveries of protocatechuic acid, salvianolic acid B, rosmarinic acid, and cryptotanshinone were 98.0%, 101.2%, 122.0%, and 96.4%, respectively.
Results of Linearity
Under the established analytical conditions, the linear regression equation is obtained by taking the mass concentration of each reference substance as the abscissa (X) and the ion peak area as the ordinate (Y). Protocatechuic acid, salvianolic acid B, rosmarinic acid, and cryptotanshinone were 0.25–2.00 µg⋅mL−1, 10.0–50.0 µg⋅mL−1, 5.00–50.0 µg⋅mL−1, and 1.00–10.0 µg mL−1. Within the concentration range, there was a good linear relationship (r2 ≥ 0.9950). The values of r2 were 0.9971, 0.9980, 0.9984, and 0.9990. The results showed that there was a good linear relationship between the mass concentration of each compound and the response value (Table 3).
Linear Regression Equation of Protocatechuic Acid, Salvianolic Acid B, Rosmarinic Acid, and Cryptotanshinone.

Limit of Detection and Limit of Quantification
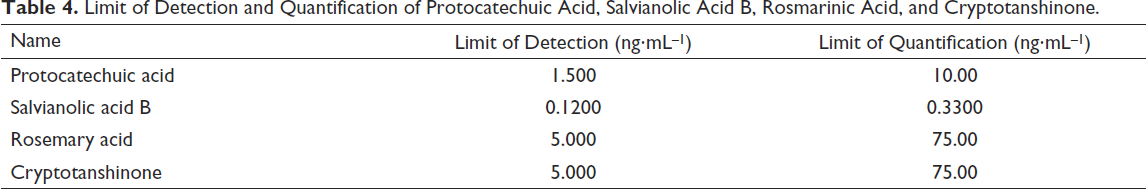
Gradually dilute the mixed reference solution to a certain multiple and measure according to S/N = 3 and S/N = 10 (Table 4).
Limit of Detection and Quantification of Protocatechuic Acid, Salvianolic Acid B, Rosmarinic Acid, and Cryptotanshinone.
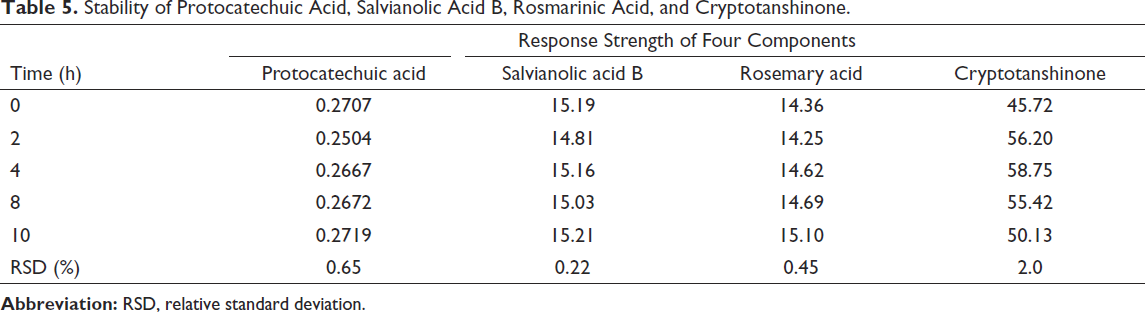
Results of Stability Test
Under the condition of preparation for sample solution, the samples were taken from Lot no. 180711 to do sample injection analysis at 0, 2, 6, 8, 10, 12, 24, and 48 h, respectively. Then, we calculated the RSD of the peak area in several different time spots. The results showed that the response values of four standard substances were stable within 0–10 h. It is indicated that the above four components should be determined within 10 h (Table 5).
Stability of Protocatechuic Acid, Salvianolic Acid B, Rosmarinic Acid, and Cryptotanshinone.
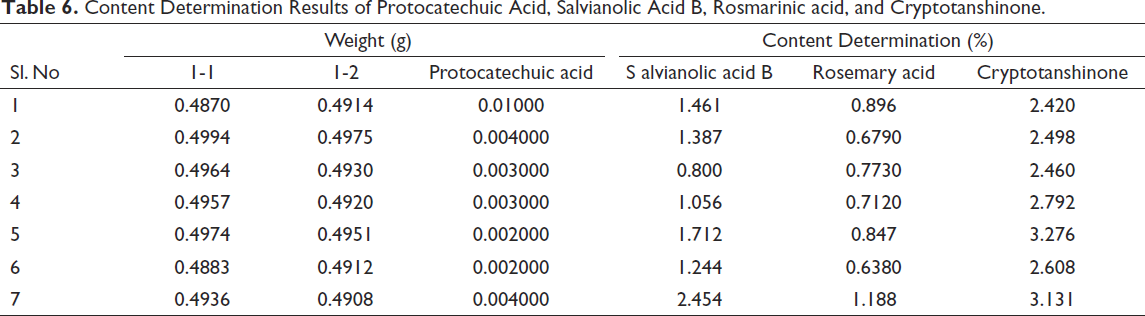
Content Determination
Take the solution to be tested under preparation for sample solution, and determine the content under the established analytical conditions. According to the standard curve, the contents of protocatechuic acid, salvianolic acid B, rosmarinic acid, and cryptotanshinone in the test solution 1–7 were obtained, respectively. The retention time of the above four components was 1.267, 1.320, 1.354, and 6.767 min, respectively (Table 6).
Content Determination Results of Protocatechuic Acid, Salvianolic Acid B, Rosmarinic acid, and Cryptotanshinone.
Discussion
Condition Screening
To establish a method to obtain stable quantitative detection, the effects of methanol–water, acetonitrile–0.1% formic acid water, and methanol–formic acid flow on the chromatographic peaks were compared in the experiment. Under the acetonitrile–water flow phase system, the chromatographic peaks appeared tailing phenomenon and the peak shape was unstable. Under the condition of methanol–water elution, the chromatogram peak of the control sample was significantly improved, while the chromatogram peak of the test sample was still trailing, affecting the quantification. The phase gradient change of acetonitrile–0.1% formic acid in the mobile phase showed a linear trend and increased well, which could meet the analysis requirements, and the obtained chromatographic peak had the best resolution and high ion response value. In the experiment, we explored the mass spectrum decomposition law of the four index components in both the positive and negative ion modes. The results showed that the response value of the compound ions was higher and more stable in the negative ion mode. Therefore, we chose to measure in the negative ion mode.
At present, LC-MS/MS technology has been widely used in the component identification of TCM. MSN technology provides structure information before and after the decomposition, which was conducive to the in-depth study of TCM components. Therefore, MS technology can ensure the analysis and characterization of the prototype components and their metabolic compounds of TCM. In the study, salvianolic acid B, as the representative component of many salvianolic acid compounds, is an easy-to-lose salvianolic acid residue during MS pyrolysis. The loss of –CO and –H2O will also occur in similar compounds. Salvianolic acid B contains substituents named –COOH and –OH. In the negative ion mode, it is easy to lose –H to get the molecular ion peak (m/z 717.1). Under the ionization condition, the parent ion is cracked and the Danshensu group is lost to obtain the fragment ion m/z 519.09 (C27H19O11), and a molecule of C9H10O5 is lost to obtain the fragment ion m/z 339.050 75 (C18H11O7).
Qishen Yiqi’s dripping pills prescription contains many chemical ingredients. Rosmarinic acid is often used as the indicator component for quality control of TCM. However, unstable substituent groups were in the structure, such as ester bond, phenolic hydroxyl, and so on; thus, it is easy to have large errors in the determination process. According to the results of Randjelović et al. (2023), the extraction rate of methanol for rosmarinic acid is higher than that of other organic solvents, and the stability is relatively high. Therefore, methanol is selected as the solvent for the preparation of the test solution. When the analysis is carried out by HPLC-MS, the selection of quantitative ion pair of this component and its key point. In combination with the literature, the parent ion peak (m/z 197.0) and the daughter ion fragment (m/z 179.0) with higher abundance are finally selected as the quantitative ion pair. The selection of quantitative ion pairs of protocatechuic acid and cryptotanshinone is also determined by the corresponding strength.
In the process of screening the linear range of each component, the concentration gradient of each reference solution is proposed by reference to the literature. Due to the difference between the instrument model and working conditions, we finally determined the concentration range used in the experiment and drew the regression equation after screening three standard curves. The method was established in the paper, which could rapidly and accurately determine the content of four chemical components in Qishen Yiqi dripping pills. Protocatechuic acid and rosmarinic acid have anti-inflammatory actions, antioxidant effects, and clinical significance. Besides, salvianolic acid B has the ability to scavenge oxygen free radicals and inhibit lipid peroxidation reactions, making it one of the known natural products with strong antioxidant effects. It has a protective effect on cardiac microvascular endothelial cells. Cryptotanshinone belongs to the terpenoid quinone class of compounds, which mostly have three- or four-membered ring adjacent or para-quinone structures on the skeleton, making it mostly active in anti-tumor, antioxidant, anti-inflammatory, antibacterial, and other activities. Therefore, the control of these ingredients would be very important for the whole evaluation of TCM preparations.
Selection of Measurement Technology
In this paper, the HPLC-MS/MS method was used to quickly determine the content of four chemical components in Qishen Yiqi dripping pills, including protocatechuic acid, salvianolic acid B, rosmarinic acid, and cryptotanshinone. The accuracy, sensitivity, and reproducibility were in line with the provisions of the Chinese Pharmacopoeia (2020), which could be used for the quality control of Qishen Yiqi dripping pills. The method could provide the basis for the quality control of TCM preparation and also provide the basis for the pharmacological mechanism pharmacokinetics, and other related studies provide a certain reference.
Analysis of Detection Results
In this paper, the determination results of different batches of Qishen Yiqi dripping pills showed that the contents of four components were significantly different. In the production process of preparations, there were many factors that influenced the production route, for example, different raw materials, improvement of the production process, parameters, and methods in the production route, etc.
The raw materials of decoction pieces, TCM decoction pieces—TCM extracts—TCM preparations. In addition to the growth environment affecting the quality, there are often conditions that affect the quality of the raw materials of the preparation, such as inappropriate harvest periods, different production years, and substandard products. Different processes would undergo different operations, which would also affect the quality of the preparation, for example, the processing, transportation, and storage of Chinese medicinal materials and prepared slices in the place of origin. Besides, the pre-treatment process of Chinese medicinal preparations is one of the important factors, including extraction, concentration, and drying.
The remarkable feature of TCM’s efficiency is “multi-component and multi-target.” TCM preparations contain diverse ingredients and complex chemical composition systems, which also show the same characteristics. Qishen Yiqi dripping pills are one of the common TCM preparations in cardiovascular clinics, but the material basis of action is still essentially unknown. The preparation is prepared by modern technology, having a more complex relationship between the process parameters and the quality connotation of the preparation.
The influence on the quality of Qishen Yiqi dripping pills can be summarized in research experiments and development processing. If the production process conditions are not optimized, the quality of the preparation will not be guaranteed. The quality of the same TCM preparation would be different produced by different manufacturers. Furthermore, the production process control of TCM preparations is another important factor affecting the quality of preparations, including equipment and facilities, personnel operation specifications, range of process parameters, deviation management scheme, and so on. According to the experimental results in this paper, it is suggested that the contents of the four components are different in different batches, which may be caused by process changes.
Conclusion
The results showed that the contents of the four components were different in different batches of Qishen Yiqi dripping pills. Among them, cryptotanshinone is the highest abundant, followed by salvianolic acid-B, which is the main chemical component of Salvia miltiorrhiza. The establishment of a multi-component UPLC-MS/MS method can be used as an important means for the study of the substance basis of drug efficacy. In this paper, a UPLC-MS/MS method was established to be suitable for the determination of four medicinal compositions of Qishen Yiqi dripping pills, including protocatechuic acid, salvianolic acid B, rosmarinic acid, and cryptotanshinone. The quantitative method had high sensitivity, good accuracy, and specificity, which could provide the basis for the production quality control of Qishen Yiqi dripping pills. In the future, we would combine in vivo ingredient determination and efficacy evaluation experiments to confirm the active ingredients, providing a scientific basis for objective evaluation of the quality of Qishen Yiqi dripping pills.
Summary
The established method rapidly identified the chemical ingredients of different products from Qishen Yiqi dripping pills. In particular, UPLC-Q-MS/MS was utilized to determine the contents of multiple components, which should be a powerful approach for achieving quality control of TCM.
Abbreviations
UPLC-MS/MS: Ultrahigh performance liquid chromatography mass spectrometry mass spectrometry; MRM: Multiple reaction monitoring; RSD: Relative standard deviation; TCM: Traditional Chinese medicine; HPLC: High-performance liquid chromatography; MS: Mass spectrometry; DAD: Diode array detector; DP: De-clustering potential; CE: ollision energy.
Footnotes
Acknowledgments
This work was supported by QMSI2020L-06. It was also supported by 202111230034.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Statement of Informed Consent and Ethical Approval
According to the review of the experimental animal ethics committee of Qiqihar Medical University, the animal experimental research content and experimental design meet the international ethical standards for the use of experimental animals and the provisions formulated by the experimental animal ethics committee of Qiqihar Medical University, so the animal experimental research is approved.
Funding
This work was supported by the fund named study on the therapeutic mechanism of Qishen Yiqi Dripping pills on statin-induced liver injury based on UPLC-Q-TOF-MS/MS (No. QMSI2020L-06).
