Abstract
Phytochemical analysis of 3 members of the Urticaceae family (eg Pilea pumila, Laportea canadensis, and Boehmeria cylindrica) was conducted using GC-MS. P pumila and L canadensis have several recorded historical medicinal uses by Native American tribes, whereas B cylindrica does not. This study was conducted to understand the historical uses in terms of chemical composition. Plant material, separated into aerial or root portions, was dried and extracted using either ethanol or heptane at room temperature. Sterols, fatty acids and esters, terpenes and terpenoids, saturated and monounsaturated hydrocarbons, small oxygen-functionalized organic molecules, and tocopherols were observed using these methods. Plant sterols composed 30.3% to 62.2% of the observed extracts, followed by fatty acids and esters ranging from 0% to 49% and terpenes and terpenoids ranging 4.56% to 33.9%. All 3 plants contained γ-sitosterol as the dominant plant sterol and palmitic, linoleic, and linolenic acids and esters. The 3 plants differed significantly in the observed terpene and terpenoid composition. Germacrene D-4-ol, known to have antifungal activity, was the major terpenoid identified in P pumila (7.42%-7.76%), phytol (4.13%), α-cadinene (3.96%), and cadina-1,4-diene (5.53%) were observed in L canadensis treatments, and squalene (2.20%-7.04%), phytol (4.17%), and cadina-1,4-diene (6.68%) were observed in B cylindrica treatments. Possible rationales for Native American medicinal uses were evaluated.
Keywords
Introduction
Pilea pumila (clearweed), Laportea canadensis (wood nettle), and Boehmeria cylindrica (false nettle) are 3 members of the Urticaceae family found in North America. Two of these species, P pumila and L canadensis, have recorded medicinal uses from Native Americans; 1 the third, B cylindrica, does not have a historical record of medicinal applications. To our knowledge, the phytochemical analyses of these plants have not been previously investigated; our goal with the present study was to understand the historical uses of these plants in the context of their chemical composition.
P pumila (clearweed) is an herbaceous annual that is native to North America. It tends to grow 6 to 12 inches tall in open, moist, temperate woods and typically appears in colony formation. Both the stem and the opposite leaves are translucent (and thus its common name, clearweed), which is especially apparent on a sunny day. 2 Medicinal applications have been coaxed from P pumila: The Cherokee rubbed the stems between their toes when itching occurred in that area, 3 while the Iroquois inhaled the squeezed juice from the stem to help alleviate sinus issues. 4 These Native American applications suggest that an antifungal effect might have been derived from the plant—the former because the site of application strongly suggests that the issue treated was athlete's foot and the latter because Mayo Clinic researchers demonstrated that a persistent fungal issue is at the heart of chronic sinus infections. 5 Later research found Aspergillus spp. to be the most common fungus involved. 6
L canadensis (wood nettle) is a large, conspicuous herb growing to four feet possessing a stem that is densely covered with stinging hairs. The plant can often be found growing in dense colonies on streambanks as well as on trailsides in wet woods. L canadensis was also utilized by a variety of Native American peoples for medicinal purposes. The Iroquois implemented a warm infusion of the smashed roots as an aid to childbirth. 4 The Houma found that a decoction of the plant served as a febrifuge. 9 Applications for urinary disorders were most widespread among the Native Americans; however, the Pillager Ojibwe used the roots to make a tea as a diuretic for “various urinary ailments”, 10 as did the Meskwaki, who also used it to treat urinary incontinence. 11 Native peoples in the Caribbean region have used the related species, L aestuans (West Indian nettle), for urinary stricture. 12
B cylindrica (false nettle) is a perennial that grows 2 to 3 ft tall and occasionally branches. The plant's habitat is typically near water—streambanks, swamps, marshes, bottomland areas, etc.—but it can also be found in wet woodlands. Unlike with its two relatives described above, there are no published Native American uses for this plant. However, medicinal applications of related species have been appreciated in the Eastern Hemisphere: The leaves of B glomerulifera have been used for sprue and the fruits for headache. 13 B platyphylla has been implemented as a stomachic, to treat dysentery, as a topical application for burns, and as a hemostat. 14 In one study, it showed a large degree of activity against Gram-negative bacteria. 14
In an attempt to find a chemical rationale for the traditional healing applications of P pumila and of L canadensis, we performed a phytochemical analysis using GC-MS of each of these plants after discovering that no chemical profiling of these plants has been previously published. We included B cylindrica in our analysis despite a relative lack of historical usage of this plant, as it is also a member of the Urticaceae family whose chemical composition has not been previously published, it was readily available, and we were curious as to how its profile would compare to the other two plants. Herein, we describe the utilization of GC-MS to identify the composition of each plant following solvent extraction in both ethanol and heptane. This approach was adopted to allow for the potential detection of highly volatile constituents often absent from essential oils due to the higher temperatures (> 280 °C) necessary to convert them into the gaseous state. 15
Results and Discussion
Results
Samples of P pumila (clearweed), L canadensis (wood nettle), and B cylindrica (false nettle) were separately added to both ethanol and heptane to extract organic-soluble components of the plant. This method of extraction has been cited to be more comprehensive in profiling small organic molecules within the plant than distillation of essential oils; distillation restricts the included components to those with an amenable boiling point and largely results in terpenes and other low-molecular-weight hydrocarbons as the major components. 15 These two solvents having different polarities were chosen to augment the diversity of compounds that would be observed with just one solvent. Plant samples of L canadensis (wood nettle) and B cylindrica (false nettle) were divided into aerial components and root components and analyzed separately, as the historical uses of L canadensis were particularly attributed to the aerial or roots of the plant.
Compounds observed were categorized by primary structure type to provide an overview of the compounds detected. The compound categories included terpenes and terpenoids, saturated hydrocarbons and other small organic compounds, fatty acids and esters, tocopherols, and plant sterols as shown in Table 1. Plant sterols, notably γ-sitosterol, accounted for the highest percentage of composition in almost all of the treatments, ranging from 30.3% to 62.2% of the total composition. Fatty acids and esters followed with a range of 0% to 49% of the sample's total composition, and terpenes and terpenoids had a range of 4.56% to 33.9%. Saturated and monounsaturated hydrocarbons, an example of which would be n-docosane, had a much lower percentage, ranging from 0% to 4.74%; alcohols, aldehydes, an example of which would be nonanal, ranged 0.22% to 5.14%. Tocopherols were observed in the range of 0% to 2.60%. The complete chemical profiling list for each plant is shown in Tables 2 to 4, listed in order of highest percentage observed to lowest. Compounds with lower than 0.05% were observed and identified but were not included here for table clarity.
Total Relative Composition of Compounds Identified in Pilea pumila, Laportea canadensis, and Boehmeria cylindrica.

Trace: Compounds having <0.05% are not tallied.
Other: Unresolved and/or unidentified components combined with components having low NIST library match factors (<750) are rejected.
Relative Percent Composition of Pilea pumila (Clearweed) Extracts.

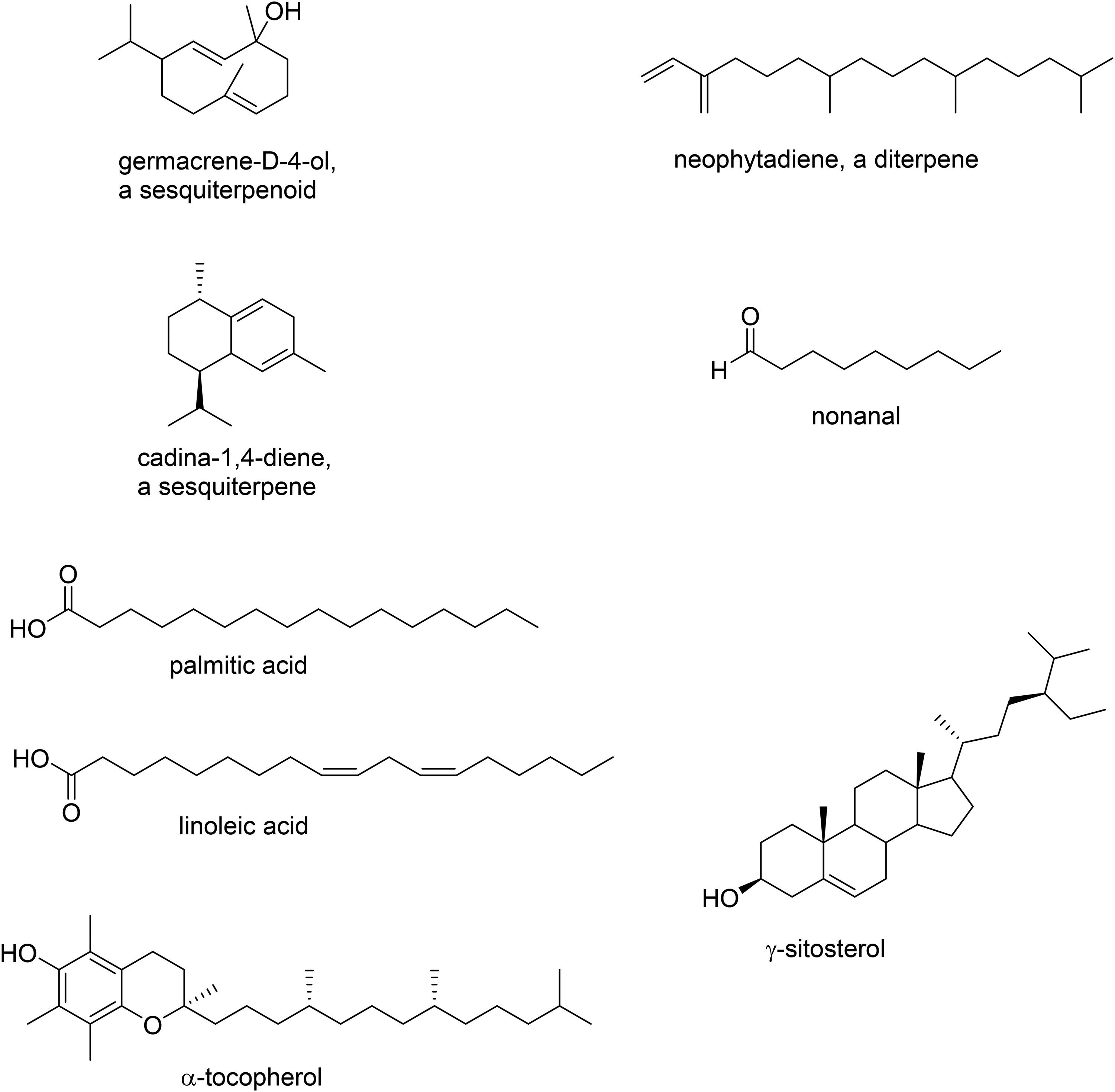
Representative structures of compounds observed.
Relative Percent Composition of Laportea canadensis (Wood Nettle) Extracts.
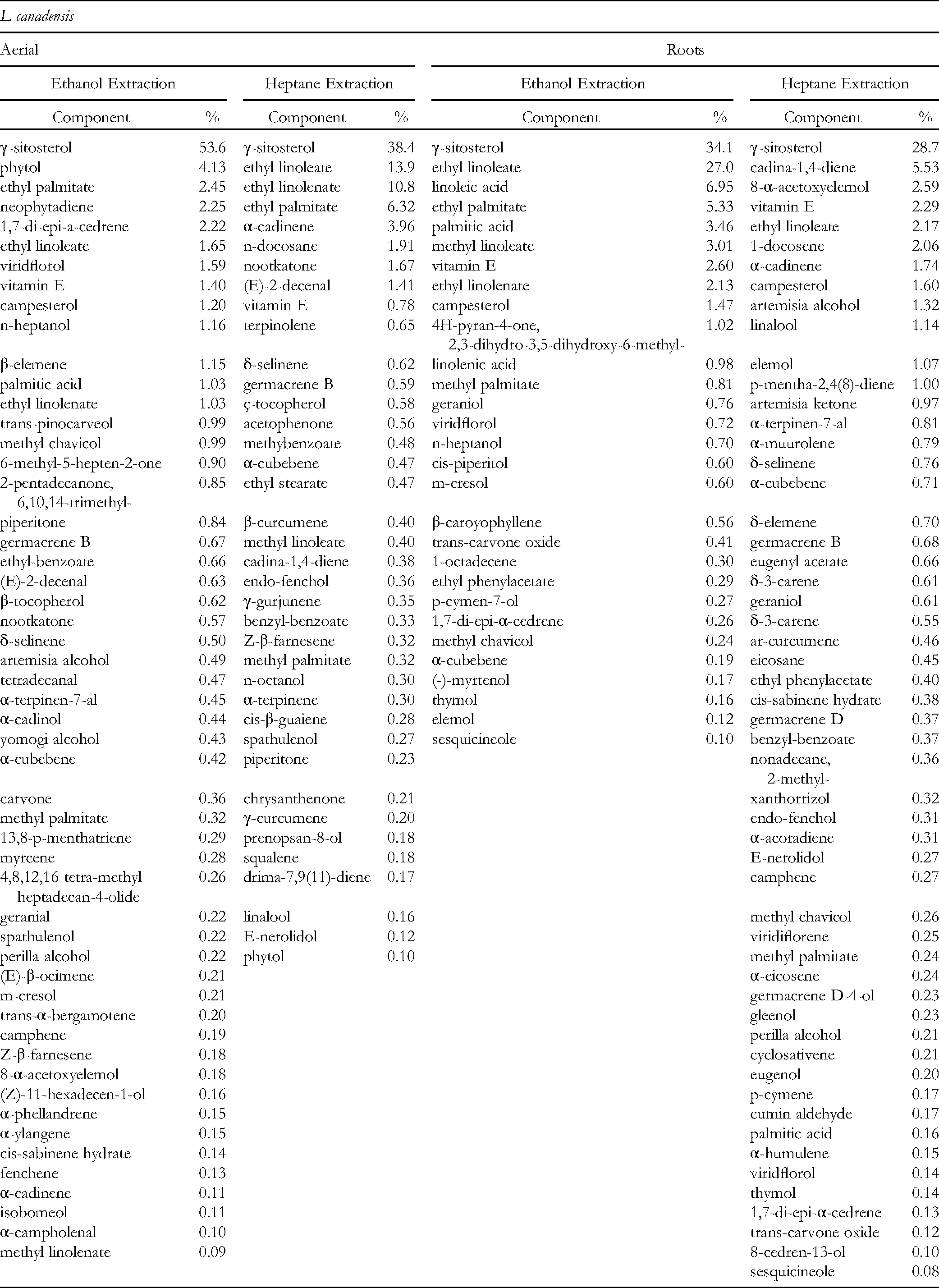
Relative Percent Composition of Boehmeria cylindrica (False Nettle) Extracts.

Discussion
Using ethanol and heptane as extracting solvents, followed by GC-MS analysis, several organic compound families were well characterized for these three members of the Urticaceae family: the sterol, fatty acid and esters, terpenes and terpenoids, saturated and monounsaturated hydrocarbons, small functionalized organic molecules such as alcohols, aldehydes, ketones, and esters, and tocopherols. Compared to distillation of essential oils, which often yields terpene and terpenoids as well as the saturated and monounsaturated hydrocarbons and small functionalized organic molecules, this method extracts a more comprehensive panel of plant constituents. For all treatment pairings of ethanol and heptane, fatty acids were observed as a higher percentage in ethanol samples, the more polar solvent, versus the more nonpolar solvent heptane, which is expected due to the polarity of the carboxylic acid functional group. By contrast, saturated and monounsaturated hydrocarbons were observed at a higher percentage in the heptane treatments.
All three members of the Urticaceae family contained γ-sitosterol as the major plant sterol observed, and the fatty acids and esters observed, principally palmitic, linoleic, and linolenic, are the same as were characterized for another member of the Urticaceae family, Urtica dioica L. 16 The plants differed in the terpene and terpenoid panels observed.
P pumila
The historical usages for P pumila by Native Americans were as a topical for the feet, presumably to treat fungal infections, and by way of inhalation of the stem juice for sinus issues. As noted above, the terpenoid compound isolated from P pumila as the highest percentage was germacrene-D-4-ol, a compound not observed in any of the L canadensis or Ba cylindrica treatments. This terpenoid is also a major component of the essential oil of Buddleja perfoliata, which showed potent antifungal activity against a variety of fungi in an analysis published in 2016. 17 The essential oil from Pinus nigra that included germacrene-D-4-ol as a major ingredient displayed antifungal activity against Aspergillus niger, a culprit in some ear and sinus infections and rarely in pulmonary infections, and where germacrene-D-4-ol specifically was highlighted as a key player in the oil's antimicrobial activity in binding with the FtsZ enzyme. 18 The presence of germacrene-D-4-ol in such a large quantity for P pumila supports the observed usage of this plant as an antifungal.
The biological activity of plants is largely due to the combination of chemical entities produced, however, rather than to a single component. In this regard, other compounds in P pumila have also demonstrated antifungal activity. β-elemene showed activity against Magnaporthe oryzae, a plant-pathogenic fungus that causes several diseases in rice. 19 Members of the cadinene family have also demonstrated antifungal activity in a study evaluating the extracts of Eupatorium adenophorum. 20 Plant essential oils with high neophytadiene content have been shown to have activity against Candida spp. and Aspergillus spp. in a study examining Apium graveolens L; 21 this terpene was identified not only in the P pumila, but also L canadensis and Boehmeria cylindrica. As can be seen, our study also unveiled methyl chavicol (alternately known as estragole) as a component of this plant. In 2005, Oxenham et al studied the antifungal and fungicidal effects of two chemotypes of basil (Ocimum basilicum) oil and its major individual components in a series of in vitro and in vivo experiments, with the result that the methyl chavicol component significantly reduced the mycelial growth of the plant pathogenic fungus Botrytis fabae. 22
Dodecanol, also identified in this study, was found to be fungistatic against Saccharomyces cerevisiae. 23 It has also been identified as having activity against the bacterium Salmonella choleraesuis; the mechanism of action is thought to be as a nonionic surfactant similar to the fatty acids. 24
Plant sterols and fatty acids, while fulfilling myriad roles in these plants, have been previously isolated and their biological activity determined. We identified a high amount of γ-sitosterol, an uncommon plant sterol, in P pumila. While this sterol does not seem to have been individually tested against fungal organisms in published studies, other sterols (the closely related β-sitosterol, stigmasterol, and ergosterol) have all demonstrated antifungal activity.25,26 A saturated fatty acid that we isolated, palmitic acid, has also shown antifungal activity.27,28 This was one of a spectrum of fatty acids (including also stearic acid, alpha-glyceryl linolenate, ethyl ester of linoleic acid, ethyl ester of linolenic acid, beta-glyceryl palmitate, and beta-glyceryl linoleate) that we found in this plant. Fatty acids tend to induce an increase in the cellular membrane fluidity of fungal organisms, causing their intracellular components to leak out and the cell to die. 29
In summary, there are a No. of compounds that support the historical usage of P pumila as an antifungal treatment.
L canadensis
L canadensis has historically been used by the Iroquois to assist during childbirth. As can be seen from our analysis, we isolated linalool from this plant's roots. This terpenoid has manifested a dose-dependent sedative effect on the central nervous system, while hypnotic, antinociceptive, and acute local analgesic effects have also been reported. 30 These are all effects that would have been appreciated by a woman experiencing childbirth pain. Linalool is one of the major components (some analyses have it as the major component) of lavender oil, which is used by way of aromatherapy in some maternity wards to ease the pain of childbirth. In a 2015 study, inhalation of this oil reduced the level of pain in a group of women during the first stage of labor as against a placebo inhalant. 31
The sesquiterpenoids/sesquiterpenes that we found (eg germacrene D, sesquicineole, δ-elemene, δ-selinene, α-cadinene, α-cubenene, etc.) could well explain the febrifuge effect elicited by the Houma, as these compounds stimulate diaphoresis, 32 which helps to resolve fever. They are present in a No. of plants commonly used in botanical medicine to achieve diaphoresis to assist in resolution of fever, with Achillea millefolium (yarrow) and boneset (Eupatorium perfoliatum) being perhaps the most famous.
With reference to the urinary disorders treated with L canadensis by Native American tribes, we found several demonstrated diuretic compounds, including germacrene D and δ-elemene. 32 γ-sitosterol, which we also isolated, is closely related to β-sitosterol (differing only in the spatial configuration of the C-17 side chain; the former is a C-24 isomer of the latter), a compound in wood nettle's relative, Urtica dioica (stinging nettle), and held to be the compound largely responsible for the benefits in urinary flow attributed to that plant's roots.33,34
L canadensis was the only one of the three plants with tocopherols contributing >1% to the total chemical composition. This plant was used for a No. of medicinal applications, and the antioxidants present, particularly vitamin E, in these leaves and roots may have contributed some additional healing.
B cylindrica
B cylindrica was the only plant of the 3 examined to have a second plant sterol, stigmasterol, present as a significant component in this analysis; additionally, the triterpene squalene was one of the highest percentage terpenes in all four of the B cylindrica treatments, a difference in the terpene panel from the other 2 Urticaceae species. B cylindrica lacks the historic Native American utilization of the other two plants, as we noted earlier. This is curious in that we found a No. of the same bioactive compounds in this plant as we did in L canadensis. One difference between B cylindrica and L canadensis in the chemical composition panel includes the lack of vitamin E (α-tocopherol) for B cylindrica roots, whereas it is a component of both the aerial and root portions of L canadensis.
Conclusions
Three different plants native to the Upper Midwest, two of which had historical medicinal uses, were analyzed using GC-MS. The two nettles were divided into aerial and root portions, and extracts for all plant portions were obtained using both ethanol and heptane. This type of extraction provided sterols, fatty acids and esters, panels of terpenes and terpenoids, saturated and monounsaturated hydrocarbons, small organic molecules, including some carbohydrates, and tocopherols. When looking to understand the observed medicinal uses of these plants, several components appear relevant. Our findings would seem to elucidate the traditional Native American (Cherokee and Iroquois) application of P pumila as something analogous to an antifungal, due most likely to germacrene-D-4-ol and to a No. of the monoterpenes and monoterpenoids.
With reference to L canadensis, we conclude that the sesquiterpenes we found most probably account for the febrifuge effect derived by the Houma as well as the diuretic effect elicited by the Pillager Ojibwe and the Meskwaki, with linalool most likely explaining the parturient effects utilized by the Iroquois. While B cylindrica does not have the historical utility of the L canadensis, some members of the terpene panel were similar. Future studies, using different extraction methods, would be needed to isolate possible alkaloids, additional glycosides, flavonoids, and coumarins.
Experimental
Sample Preparation
P pumila (clearweed), L canadensis (wood nettle), and B cylindrica (false nettle) were obtained from Locke Country Park, an open, moist, wooded area in Anoka County, Minnesota in late June and early July of 2021. The plants were fully grown and had not developed seeds. Voucher specimens of each plant were deposited with the Bell Museum of Natural History University of Minnesota Herbarium (MIN). The accession numbers are P pumila MIN965596, L canadensis MIN965595, and B cylindrica MIN965598. Plants were cleaned and dried at room temperature for three days in a high airflow velocity chemical fume hood. The dried plants were cut finely with a scalpel and ground with a mortar and pestle. The dried, ground samples (0.400 g) were added to either heptane (Sigma-Aldrich) or ethanol (EMD Millipore; 10 mL). The heterogeneous solutions were stirred at room temperature for four days. The solutions were then filtered. The filtrate solutions were centrifuged (8000 rpm, 3 min) and a portion of the supernatant was used without further refinement as the GC-MS sample.
GC-MS Analysis
All samples were processed on an Agilent 5977A gas chromatograph equipped with a quadrupole mass spectrometer. Separations were performed on an Agilent J&W DB-5ms™ fused silica column (30 m × 0.25 mm i.d. × 0.25 µm film thickness) using helium as a carrier gas at a flow rate of 0.8 mL/min. Upon optimization, 2 µL of each sample extract concentrated via rotary evaporation at ambient temperature was injected with a 1:10 split ratio with an inlet temperature of 200 °C. The oven temperature gradient employed for all separations was 60 °C to 170 °C at 15 °C/min, and from 170 °C to 285 °C at 3 °C/min, with a 15 min final hold. For mass analyses, samples were ionized via electron impact (EI) mode at 70 eV. The quadrupole frequencies were set to detect compounds ranging from 50 to 1000 Da. Solvent blanks (heptane and ethanol) were run before and after sample extracts to no ensure no contaminants were introduced during sample processing or previous separations. An initial solvent delay of 3 min was also employed.
The mass spectra were deconvoluted using the Automated Spectral Deconvolution and Identification System (AMDIS). The linear retention indices, RI, for compounds were determined by analysis of a standard solution of n-alkane C8-C40 series dissolved in n-hexane (Sigma-Aldrich). 35 Compounds were identified based on the comparison of their calculated retention indices and as well as by matching against reference compounds in the National Institute of Standards and Technology (NIST) mass spectral library.35,36 Compound concentrations (as normalized relative percent content) were subsequently quantified as a function of relative peak area percentage. The reported compounds shown in Supplementary Table S1 exceeded a NIST library database match factor threshold of >750 and were also within ± 10 RI values of previously reported reference values. 35
Supplemental Material
sj-doc-1-npx-10.1177_1934578X221080978 - Supplemental material for Phytochemical Investigation of Pilea pumila (Clearweed), Laportea canadensis (Wood Nettle), and Boehmeria cylindrica (False Nettle): Three Members of the Urticaceae Family
Supplemental material, sj-doc-1-npx-10.1177_1934578X221080978 for Phytochemical Investigation of Pilea pumila (Clearweed), Laportea canadensis (Wood Nettle), and Boehmeria cylindrica (False Nettle): Three Members of the Urticaceae Family by Rondedrick D. Sinville, Matthew G. Alfs and Sarah M. Dimick Gray in Natural Product Communications
Supplemental Material
sj-docx-2-npx-10.1177_1934578X221080978 - Supplemental material for Phytochemical Investigation of Pilea pumila (Clearweed), Laportea canadensis (Wood Nettle), and Boehmeria cylindrica (False Nettle): Three Members of the Urticaceae Family
Supplemental material, sj-docx-2-npx-10.1177_1934578X221080978 for Phytochemical Investigation of Pilea pumila (Clearweed), Laportea canadensis (Wood Nettle), and Boehmeria cylindrica (False Nettle): Three Members of the Urticaceae Family by Rondedrick D. Sinville, Matthew G. Alfs and Sarah M. Dimick Gray in Natural Product Communications
Supplemental Material
sj-docx-3-npx-10.1177_1934578X221080978 - Supplemental material for Phytochemical Investigation of Pilea pumila (Clearweed), Laportea canadensis (Wood Nettle), and Boehmeria cylindrica (False Nettle): Three Members of the Urticaceae Family
Supplemental material, sj-docx-3-npx-10.1177_1934578X221080978 for Phytochemical Investigation of Pilea pumila (Clearweed), Laportea canadensis (Wood Nettle), and Boehmeria cylindrica (False Nettle): Three Members of the Urticaceae Family by Rondedrick D. Sinville, Matthew G. Alfs and Sarah M. Dimick Gray in Natural Product Communications
Supplemental Material
sj-docx-4-npx-10.1177_1934578X221080978 - Supplemental material for Phytochemical Investigation of Pilea pumila (Clearweed), Laportea canadensis (Wood Nettle), and Boehmeria cylindrica (False Nettle): Three Members of the Urticaceae Family
Supplemental material, sj-docx-4-npx-10.1177_1934578X221080978 for Phytochemical Investigation of Pilea pumila (Clearweed), Laportea canadensis (Wood Nettle), and Boehmeria cylindrica (False Nettle): Three Members of the Urticaceae Family by Rondedrick D. Sinville, Matthew G. Alfs and Sarah M. Dimick Gray in Natural Product Communications
Supplemental Material
sj-docx-5-npx-10.1177_1934578X221080978 - Supplemental material for Phytochemical Investigation of Pilea pumila (Clearweed), Laportea canadensis (Wood Nettle), and Boehmeria cylindrica (False Nettle): Three Members of the Urticaceae Family
Supplemental material, sj-docx-5-npx-10.1177_1934578X221080978 for Phytochemical Investigation of Pilea pumila (Clearweed), Laportea canadensis (Wood Nettle), and Boehmeria cylindrica (False Nettle): Three Members of the Urticaceae Family by Rondedrick D. Sinville, Matthew G. Alfs and Sarah M. Dimick Gray in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
