Abstract
Objectives
Psoriasis vulgaris is an immune-mediated inflammatory skin disease that is associated with depression. In this study, we investigated the effect of Xiegan–Liangxue–Jiedu (XGLXJD) decoction, a traditional Chinese medicine formula, on psoriasis based on network pharmacology, molecular docking, and animal experiments.
Materials and Methods
A protein–protein interaction (PPI) network was constructed using the overlapping targets of XGLXJD decoction and psoriasis. Gene ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment analyses were performed using Metascape database. High-performance liquid chromatography (HPLC) was used to investigate the main compounds of XGLXJD decoction. Molecular docking was performed to predict the potential interaction between the main compounds and proteins of interest. A C57 mouse model of psoriasis was established via continuous exposure to imiquimod. Seven days later, the XGLXJD decoction was orally administered at increasing doses for 6 days. The psoriasis area and severity index were calculated. Hematoxylin and eosin staining was used to examine skin morphology. Enzyme-linked immunosorbent assay (ELISA) was used to determine the serum levels of adenylyl cyclase (AC), cyclic adenosine monophosphate (cAMP), protein kinase A (PKA), interleukin-17 (IL-17), and tumor necrosis factor-α (TNF-α). Sucrose preference test and forced swimming test were used to assess depression-like behavior. Immunohistochemical (IHC) staining and immunofluorescence were used to investigate the cAMP-response element binding protein (CREB) signaling pathway.
Results
Overall, 162 overlapping targets were generated. A total of 398 biological processes, 84 molecular functions, and 47 cellular components were identified via GO analysis, whereas 140 pathways were identified via KEGG pathway analysis. The most notable signaling pathways were cAMP as well as downstream IL-17 and TNF-α signaling pathways. HPLC analysis revealed that the main compounds of XGLXJD decoction were paeoniflorin, isorhamnetin, quercetin, luteolin, kaempferol, and baicalein. The molecular docking assay indicated that the docking energies of the main compounds of XGLXJD decoction to the top hub genes were less than −5 kcal/mol. ELISA revealed that the administration of XGLXJD decoction decreased the levels of pro-inflammatory cytokines (TNF-α and IL-17). Furthermore, the AC, cAMP, and PKA levels were enhanced after its administration. IHC staining demonstrated that the administration of XGLXJD decoction activated the AC–cAMP–PKA–CREB signaling pathway in skin. In addition, it enhanced sucrose preference and forced swimming time percentage. It also enhanced the expression of cAMP and PKA in the hippocampus.
Conclusion
XGLXJD decoction alleviated psoriasis and depression-like behavior. The AC–cAMP–PKA–CREB signaling pathway may play a crucial role in mediating this effect.
Introduction
Psoriasis vulgaris is an immune-mediated inflammatory disease that manifests mainly in the skin and joints (Noda et al., 2015). It severely impairs the quality of life of affected individuals. The estimated global prevalence of this disease ranges from 0.09% to 11.43% (Danielsen et al., 2013), whereas the estimated prevalence in the European Union, African Union, and China is 2.11%, 0.22%, and 0.56%, respectively (Liu et al., 2022). Psoriasis is characterized by hyperkeratosis, vascular regression, distorted morphology, excessive skin proliferation, aberrant differentiation of keratinocytes, and lymphocyte infiltration around superficial dermal vessels (Lowes et al., 2014; Perera et al., 2012). It is well-known that immune pathways are widely activated in psoriasis, leading to an imbalance between inflammatory response and skin turnover (Mudigonda et al., 2012). In addition to skin damage, psoriasis is closely linked to depression, and inflammation plays a crucial role in this linkage (González-Parra & Daudén, 2019). Therefore, treatment for psoriasis is not limited to repairing skin damage but improving the quality of life of affected individuals.
The Liangxue-Jiedu decoction is a famous Traditional Chinese medicine (TCM) formula used for treating blood-heat type disease (Li et al., 2021). According to TCM theory, blood-heat type disease is defined as a disorder caused by blood-heat factors, such as external invasion of wind-heat, inversion of external pathogenic factor wind-cold, and heat transformed from depressive mental injury (Tan et al., 2011).
Some modified decoctions based on the Liangxue-Jiedu decoction are widely used in clinical practice and preclinical research. For example, Liangxue-Jiedu-Huoxue decoction was shown to decrease the incidence of radiation pneumonitis (Xiao et al., 2010). The main application of Liangxue-Jiedu decoction is psoriasis. In a clinical trial, it was shown to reduce the psoriasis area and severity index in patients with progressive Psoriasis vulgaris (Sun et al., 2020). In a preclinical study, it was shown to improve psoriasis and comorbid dyslipidemia via regulation of the PI3K/Akt/mTOR pathway in the skin and liver (Xie et al., 2021). An artificial intelligence-based application predicted the multidimensional effect of Liangxue-Jiedu decoction in the treatment of psoriasis (Zhao et al., 2021).
Xiegan-Liangxue-Jiedu (XGLXJD) decoction is a modified version of Liangxue-Jiedu decoction used in clinical settings. It is composed of Buffalo horn (30 g), Rehmanniae Radix Praeparata (30 g), Radix Paeoniae Rubra (15 g), Cortex Moutan (12 g), Smilacis Glabrae Rhixoma (30 g), Radix Bupleuri (12 g), Sophora flavescens (12 g), Gardeniae Fructus (9 g), Cyperi Rhizoma (9 g), and licorice (6 g). Its raw medicine is decocted, and the filtrate is produced through modern technology, which transform into a commercial hospital preparation. Compared with Liangxue-Jiedu decoction, Buffalo horn, Rehmanniae Radix Praeparata add into the Liangxue-Jiedu decoction to become the XGLXJD decoction according to TCM theory, which aim to alleviate the brain function according to the TCM theory. In this study, we investigated the efficacy of XGLXJD decoction for psoriasis and its’ another symptom––depression via network pharmacology, molecular docking, and animal experiments.
In this study, we investigated the efficacy of XGLXJD decoction in psoriasis based on network pharmacology, molecular docking, and animal experiments.
Materials and Methods
Predicted Combination of Targets
TCM systems pharmacology database and analysis platform (TCMSP) (Ru et al., 2014) is a repository of 499 Chinese herbs registered in the Chinese pharmacopeia with 29,384 ingredients and 3,311 targets. Moreover, TCM Database@Taiwan (Sanderson, 2011) is the world’s largest TCM database for drug screening. In this study, these two databases were used to identify targets related to XGLXJD decoction. Further, online Mendelian Inheritance in Man (Amberger & Hamosh, 2017), Genecard (Rebhan et al., 1997), and DisGeNet (Piñero et al., 2017) databases were used to retrieve psoriasis-related targets. After removing duplicates, protein names were converted into gene abbreviations using UniProt (UniProt Consortium, 2018). A Venn diagram was constructed using Venny 2.1.0 website (
Network Construction and Analysis
A protein–protein interaction (PPI) network (Athanasios et al., 2017) of overlapping genes was constructed using the STRING database (Szklarczyk et al., 2019) with the species limited to “Homo sapiens” and the cutoff confidence score set at >0.9. To further explore the interactions between targets, a network was inputted into Cytoscape 3.8.2 (Shannon et al., 2003) and the cytohubba plugin (Chin et al., 2014) was employed to explore the top 10 hub genes based on the Maximal Clique Centrality (MCC) method (Cai et al., 2022).
Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment analysis.
GO analysis was employed for gene function classification into molecular functions (MFs), biological processes (BPs), and cellular components (CCs). DAVID database (Dennis et al., 2003) was employed for GO and KEGG pathway analyses. The predicted pathways were visualized using R software.
High-performance Liquid Chromatography (HPLC) Analysis
HPLC analysis was performed to identify the main compounds of XGLXJD decoction using 1260 Agilent high-pressure liquid chromatograph (HPLC, Kyoto, Japan). The run condition was set as follows: Agilent Eclipse XDB-C18 column (250 mm × 4.6 mm, 5 µm); mobile phase: methanol (A) and 0.2% phosphoric acid aqueous solution (B); gradient elution (0–20 min, 40% A ↓ 60% A; 20–25 min, 60% A; 25–29 min, 60% A ↓ 90% A; and 29–32 min, 90% A ↓ 40% A); flow rate: 1 mL/min−1; and detection wavelength: 279 (0–17 min) and 267 (17–25 min) nm. The standards of main ingredients of XGLXJD decoction and the XGLXJD decoction itself were dissolved in methanol and diluted to the desired concentrations (XGLXJD decoction 2.17 g/mL, quercetin 50 mg/mL, luteolin 50 mg/mL, kaempferol 50 mg/mL, isorhamnetin 50 mg/mL, baicalein 50 mg/mL and paeoniflorin 100 mg/mL, respectively). Each injection volume was 10 µL.
Reagents and Chemicals
XGLXJD decoction was provided by TCM Pharmacy of Longhua Hospital (lot number: 20220302). The biochemical kits for various compounds (adenylyl cyclase [AC], lot number: 20210103; cyclic adenosine monophosphate [cAMP]: 20210104; protein kinase A [PKA]: 20200624; interleukin-17 [IL-17]: 202106201, and tumor necrosis factor-α [TNF-α]: 20200911) were obtained from Nanjing Jiancheng Bioengineering Institute (Jiangsu, China). Primary antibodies (TNF-α catalog: ab1793, IL-17: ab91649, cAMP: ab235385, cAMP-response element binding protein (CREB): ab220798, PKA: ab32390, and phosphodiesterase (PDE): ab259945) were purchased from Abcam (MA, USA). Imiquimod cream was obtained from Mingxin Pharmaceutical Co., Ltd. (Sichuan, China, CAS No. 99011-02-6, lot number: 40201001). Vaseline was obtained from Jinlin Biotechnology Co., Ltd. (Shandong, China, CAS No.8009-03-8, lot number: 201905263).
Mouse Model Establishment and Drug Administration
Sixty male C57BL/6 mice (age: 7–8 weeks; weight: 18–20 g; SPF grade) were provided by Shanghai Jihui Experimental Animal Breeding Co., Ltd. Ethics approval was obtained from the Animal Ethics Committee of Shanghai University of Traditional Chinese Medicine (grant number: PZSHUTCM210423010, dated April 23, 2021). Mice were randomly divided into six groups (n = 10 in each group): (a) control, (b) model, (c) positive control, (d) low-dose XGLXJD decoction, (e) medium-dose XGLXJD decoction, and (f) high-dose XGLXJD decoction.
Except for the control group, mice in all groups were subjected to anesthesia treatment (pentobarbital sodium, intraperitoneal [i.p.], 30 mg/kg), and the hair over the center back (area = 2 × 3 cm) was shaved. Overall, 50 mg of 5% imiquimod cream was smeared onto the hairless area of the back of the mouse model using ophthalmic tweezers, once daily for 7 consecutive days (Zhang et al., 2016). Mice in the control group were smeared with 50 mg of medical vaseline using ophthalmic tweezers on the hairless area of the back.
Mice in the three XGLXJD decoction groups were orally administered with XGLXJD decoction granules dissolved in water at increasing doses of 10.85, 21.7, 43.4 g/kg once daily from days 8 to 13. Mice in the positive control group received Tripterygium wilfordii polyglycoside suspension (7.89 mg/kg once daily), according to the previously reported method (Chen et al., 2022).
Assessment of the Severity of Skin Damage
On day 13, the PASI was calculated based on the evaluation of mouse skin lesions for each of the following three symptoms: erythema, scaling, and thickening (Hung et al., 2018). Skin thickness was measured with a vernier caliper after anesthetizing the mice (pentobarbital sodium, i.p., 30 mg/kg).
Depression Behavior Test
Imiquimod-induced psoriasis-like inflammation model has been used to assess depression-like behavior (Guo et al., 2020). Sucrose preference test and forced swimming test were performed to investigate the effect of XGLXJD decoction on depression-like behavior.
Sucrose preference test was performed using a slightly modified version of the previously described method (Shen et al., 2021) before (day 7) and after XGLXJD decoction treatment (day 13). In brief, mice were pre-deprived of water and food for 1 day, and on day 2, two bottles were replaced with 2% sucrose solution and only water, respectively. After 12 h of ad libitum access, the two bottles were weighed and sucrose preference (%) was calculated using the formula: Sucrose solution intake/total intake (Sucrose + water solution) × 100% (Tu et al., 2018).
The forced swimming test was performed before (day 7) and after XGLXJD decoction treatment (day 13) (Hu et al., 2022). In brief, the day before the formal forced swimming test, mice were placed in a cylinder filled with water and forced to swim for 15 min. On day 2, the mice were forced to swim again and the immobile time was recorded for the last 4 min; the swimming time percentage was calculated as follows: (4 × 60-immobility time [s]) / (4 × 60) × 100%.
Histopathological Examination
Hematoxylin and eosin (HE) staining was performed to investigate skin damage. The skin over the central back was removed from the euthanized mice (pentobarbital sodium, i.p., 30 mg/kg), fixed, and embedded in paraffin. The paraffin-embedded sections were stained with HE solution and observed under a light microscope (MF31, Mshot, Guangzhou, China). The parameters of microscopy were as follows: numerical aperture, 1.25; brand, Mshot); the parameters of camera were as follows: brand, Msx2 (Mshot, Guangzhou, China); exposure time, 2 ms; gain setting: 1X.
ELISA for Cytokines
After skin harvesting, mouse serum was collected and the AC, cAMP, PKA, IL-17, and TNF-α levels were determined using commercial ELISA kits following the previously described method (Li et al., 2021). The data was obtained from optical density (OD) values with the help of a predetermined standard line.
Immunohistochemical (IHC) Assay in Skin
Briefly, skin tissues were collected, fixed, sliced into 4-µm sections, deparaffinized, and rehydrated. Then, these sections were blocked using a blocking solution for 1 h and incubated overnight at 4°C with primary antibodies (1:100–150 dilution; ab179800, Abcam), followed by incubation with secondary antibody for 20 min at 37°C. After washing, skin sections were photographed under a microscope (MF31, Mshot, Guangzhou, China). The integrated ODs and areas showing positive immunostaining for IL-17, TNF-α, cAMP, CREB, PKA, and PDE were assessed using NIH Image-Pro Plus 6.0 analysis software (Media Cybernetics Co., MD, USA).
Immunofluorescence (IF) Assay in the Brain Hippocampus
Brain hippocampus staining using IF was performed according to a previously described method (Jiang et al., 2022). Briefly, the whole mouse brain was perfused and fixed. Further, the brain was dehydrated, frozen, and sliced into 5-µm thick sections. After penetration using TritonX-100, the slides were blocked, stained, and photographed using an immunofluorescent camera (3DHistech Ltd., Budapest, Hungary). The expression levels of proteins were assessed using NIH Image-Pro Plus 6.0 analysis software (Media Cybernetics Co., MD, USA). The Cornu ammonis 1 (CA1) and Cornu ammonis 3 (CA3) regions were located in the hippocampus, consistent with a previous report (Gannon et al., 2022).
Statistical Analysis
The data was expressed as mean±SD. The statistical analysis was performed by SPSS using ANOVA analysis. A p<0.05 was considered as significant.
Results
Screening of active overlapping targets of “XGLXJD decoction” and “psoriasis”.
After removing duplicates, 251 and 4,452 potential targets of XGLXJD decoction and psoriasis, respectively, were obtained from the abovementioned databases. Finally, 162 overlapping targets were obtained; the Venn diagram is presented in Figure 1A. The networks formed between 138 compounds and 162 targets were visualized via Cytoscape (Figure 1B). Then, the 162 overlapping targets were imported into STRING website to construct the PPI network (Figure 1C). The network contained 161 nodes and 668 edges, and the average node degree was 8.22. The top 10 core target genes (hub genes) ranked by the MCC method using Cytohubba plugin were STAT3, JUN, MAPK3, RELA, ESR1, MAPK1, FOS, MYC, MAPK14, and HIF1A. The top five compounds ranked by the MCC method were MOL000098 (quercetin), MOL000006 (luteolin), MOL000422 (kaempferol), MOL004328 (naringenin), and MOL002714 (baicalein).
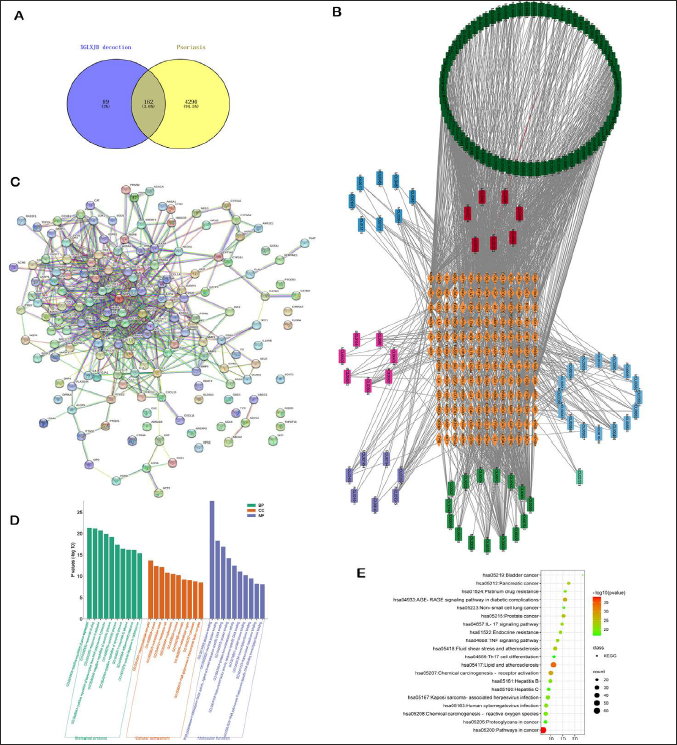
PPI, protein–protein interaction; BPs, biological processes; CCs, cellular components; MFs, molecular functions; GO, Gene Ontology; KEGG, Kyoto Encyclopedia of Genes and Genomes.
GO and KEGG Pathway Enrichment Analysis
The overlapping genes were imported into DAVID database and 398 BPs, 84 MFs, and 47 CCs were obtained using the threshold p value of <0.01. Each analysis ranked the top 10 genes according the −log10 (p values), as shown in Figure 1D. Four signaling pathways were obtained via KEGG pathway enrichment analysis using the threshold of p < .01. These were ranked by the P-value, as shown in Supplementary Table 1; the top 20 predicted signaling pathways are indicated in Figure 1E. Among them, TNF-α and IL-17 signaling pathways attracted our interest. Furthermore, the cAMP signaling pathway, which lies upstream of TNF-α and IL-17, also attracted our interest, although the cAMP signaling pathway ranked 106 among the 144 predicted signaling pathways (Supplementary 1).
Dominant Compounds in XGLXJD Decoction
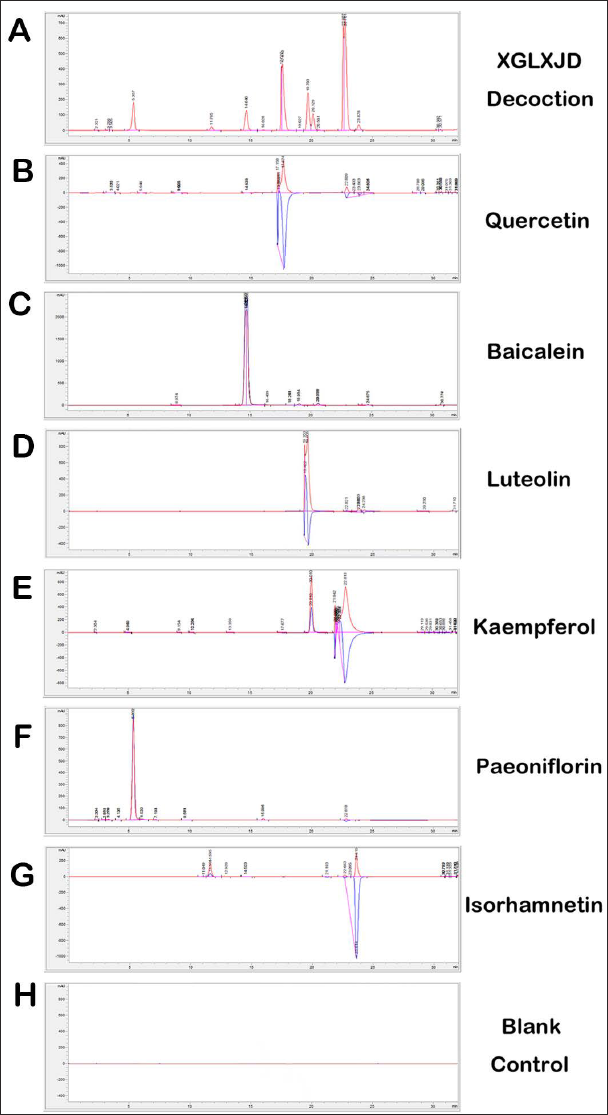
We performed HPLC to simultaneously determine the dominant compounds in network pharmacology analysis. Compounds detected in XGLXJD decoction under HPLC condition are shown in Figure 2A. The quercetin, baicalein, luteolin, and kaempferol standards appeared at the time points of 17.5, 14.8, 19.5, and 20.1 min, respectively (Figure 2B–2E). Hesperidin did not appear in the mobile phase. Two other compounds, namely paeoniflorin (5.3 min; Figure 2F) and isorhamnetin (22.8 min; Figure 2G) appeared under this HPLC condition and were also the main compounds of XGLXJD decoction. Thus, the main compounds included quercetin (9.97 ± 0.28 mg/g), luteolin (4.23 ± 0.21 mg/g), kaempferol (7.19 ± 0.04 mg/g), isorhamnetin (12.32 ± 0.26 mg/g), baicalein (6.74 ± 0.27mg/g), and paeoniflorin (0.21±0.01 mg/g).
HPLC Analysis of XGLXJD Decoction. (A) The representative HPLC chromatogram of XGLXJD decoction; (B) Quercetin; (C) Baicalein; (D) Luteolin; (E) Kaempferol; (F) Paeoniflorin; (G) Isorhamnetin; (I) The blank control contains only mobile phase.
Effect of XGLXJD Decoction on Skin Damage
As shown in Figure 3A, the control group showed no psoriasis-like lesions such as erythema, scales, infiltration, or thickening. Skin lesions in the low- and medium-dose XGLXJD decoction groups were similar to those in the psoriasis model group. The high-dose and positive control groups showed fewer skin lesions. In contrast, the model group showed the most obvious skin lesions, highest infiltration, deepest erythema color, and most numbers of scales.
XGLXJD Decoction Alleviated the Skin Damage, Regulated the Immune Progress and CAMP Signaling Pathway Activation in Serum of Mouse Model. (A) Representative skin among groups; (B) PASI index at day 3, 5, and 7; (C) Skin thickness; (D) HE staining of mouse skin, inner bar = 200 μm, magnification=200*. 1. Epidermal thickening. 2. Thickening of the spinous layers in a clubbed shape, extending towards the dermis. 3. Dermal superficial lymphocyte infiltration. 4. Dermal superficial capillary dilation; (E) AC levels; (F) Camp levels; (G) PKA levels; (H) TNF-α levels; (I) IL-17 levels. Statistical difference among model group and others were considered significant at the level of *p<0.05, **p<0.01 or ***p<0.001.
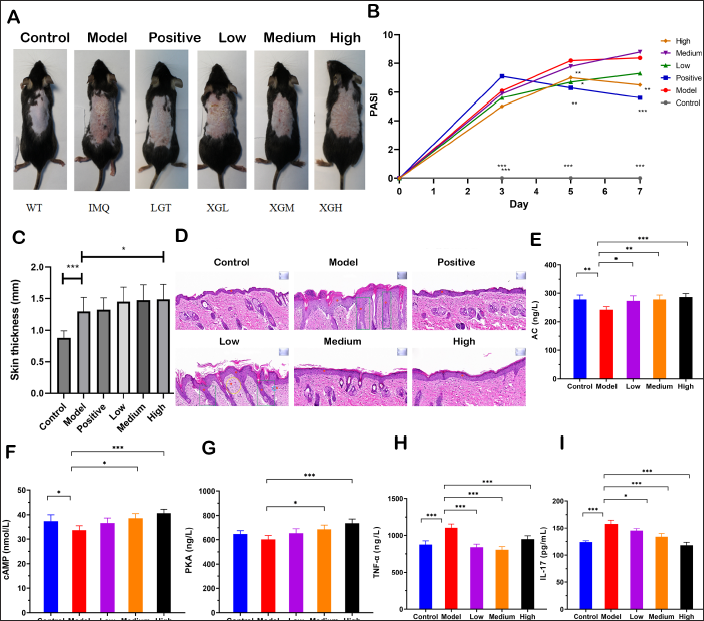
The PASI apparently increased in the model group compared with that in the control group (Figure 3B); however, medium- and high-dose XGLXJD decoction decreased the PASI on day 5, and high-dose XGLXJD decoction decreased the PASI on day 7. However, there were no significant differences in the low- and medium-dose decoction groups compared with the model group on day 7.
Skin thickness in the psoriasis model group increased significantly (Figure 3C) on day 7. High-dose XGLXJD decoction decreased the PASI. Moreover, there were no significant differences in the low- and medium-dose decoction groups compared with the model group.
A representative HE-stained skin section is shown in Figure 3D. The skin structure was normal in the control group. However, the model group showed obvious skin acanthosis, hyperkeratosis, hypokeratosis, and munro microabscesses in the epidermis. Telangiectasia and inflammatory cell infiltration were observed in the superficial dermis. The administration of XGLXJD decoction alleviated skin damage and inflammatory infiltration.
Effect of XGLXJD decoction on pro-inflammatory cytokines and AC–cAMP–PKA levels.
Compared with the control group, the serum levels of AC (Figure 3E), cAMP (Figure 3F), and PKA (Figure 3G) decreased in the model group. After the administration of XGLXJD decoction, a dose-dependent increase was observed in the levels of AC, cAMP, and PKA, indicating that XGLXJD decoction activated the AC–cAMP–PKA signaling pathway.
Further, the levels of pro-inflammatory cytokines TNF-α (Figure 3H) and IL-17 (Figure 3I) increased in the model group compared with those in the control group. However, compared with the model group, XGLXJD decoction decreased the levels of TNF-α and IL-17 in the three XGLXJD decoction groups.
Effect of XGLXJD Decoction on Protein Activity in Skin
Similar to the contents in serum, the IHC assay showed increased protein expressions of TNF-α (Figure 4A) and IL-17 (Figure 4B) in skin in the model group compared with the control group. However, compared with the model group, XGLXJD decoction decreased the protein expression of TNF-α and IL-17 in the three XGLXJD decoction groups.
XGLXJD Decoction Regulated the Immune Progress and CAMP Signaling Pathway Activation in the Skin of Psoriasis Mice Model, Inner Bar = 100 μm (left upper corner), magnification=200*. (A) TNF-α levels; (B) IL-17 levels; (C) cAMP levels; (D) CREB levels; (E) PKA levels; (F) PDEs levels. Scale inner bar= 200 μm. Statistical differences between the model group and others were considered significant at the level of *p<0.05, **p<0.01 or ***p<0.001.
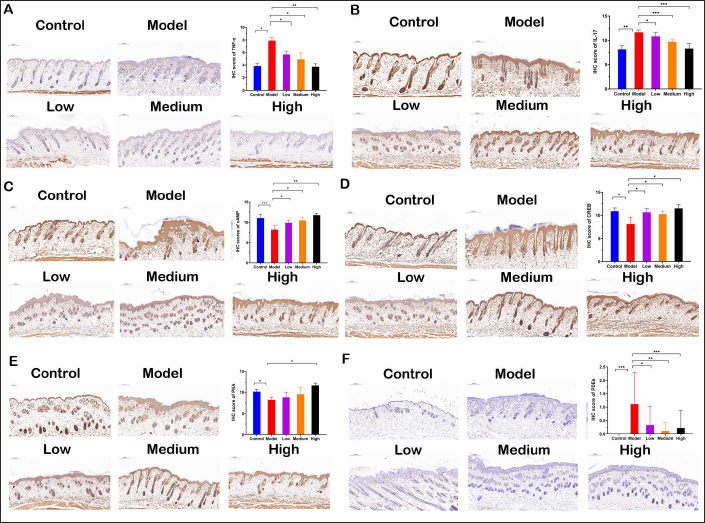
In contrast, the expression levels of cAMP (Figure 4C), CREB (Figure 4D), PKA (Figure 4E), and PDEs (Figure 4F) in skin were maintained at lower level in the model group compared with the control group. The XGLXJD decoction enhanced the expression of cAMP, CREB, PKA, and PDEs in the three XGLXJD decoction groups compared with the model group.
Effect of XGLXJD Decoction on Depression-Like Behavior and Protein Activity
As shown in Figure 5A, before treatment, there were no significant between-group differences in sucrose preference, except for the control group. However, the model group showed a significant decrease in sucrose preference compared with the control group. The administration of XGLXJD decoction enhanced the sucrose preference in the three XGLXJD decoction groups in a dose-dependent manner (Figure 5B). Furthermore, forced swimming time percentage over 240 s in the model group decreased markedly in the model group (Figure 5C), indicating that the mice reduced the struggling time activity, which is the most typical depression symptom. However, the administration of XGLXJD decoction increased the swimming time percentage in the three XGLXJD decoction groups in a dose-dependent manner (Figure 5D), indicating that XGLXJD decoction attenuated the depression symptom.
XGLXJD Decoction Alleviated the Depression Behavior in a Mice Model of Psoriasis. (A) The sucrose preference test among groups before treatment; (B) The sucrose preference test among groups after treatment; (C) The forced swimming test among groups before treatment; (D) The forced swimming test among groups after treatment; (E) cAMP expression in CA1 region and CA3 region; (F) CREB expression in CA1 region and CA3 region. Inner bar = 200 μm, magnification=200*. Statistical differences between the model group and other groups were considered significant at the level of *p<0.05, **p<0.01 or ***p<0.001.
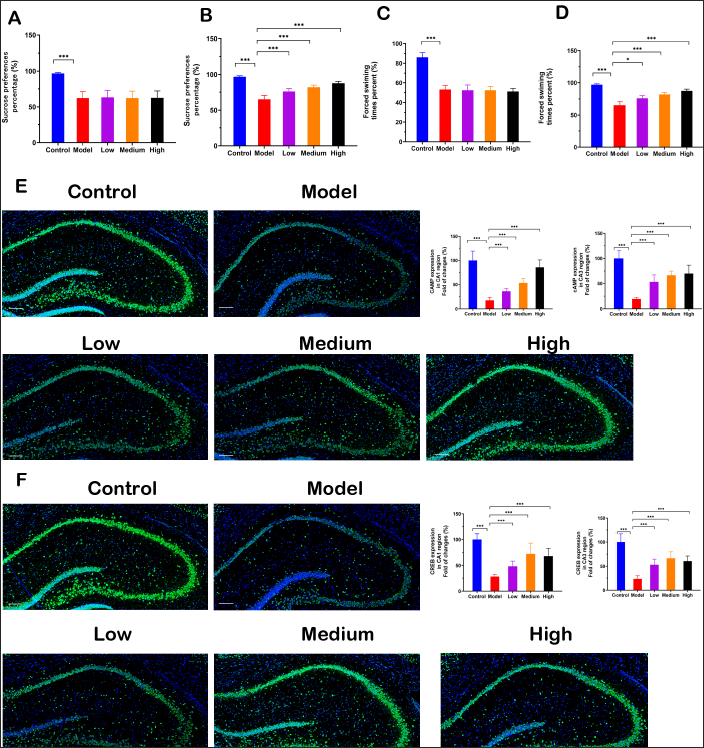
Consistent with the contents in serum, the cAMP and CREB protein expression were downregulated in the brain hippocampus in CA1 and CA3 regions (Figure 5E and 5F). However, administration of XGLXJD decoction increased the cAMP and CREB protein expressions in the hippocampus.
Discussion
This study demonstrated the efficacy of XGLXJD decoction in alleviating psoriasis-like inflammation via network pharmacology, molecular docking, HPLC analysis, and animal experiments. In this study, XGLXJD decoction decreased the PASI and alleviated skin damage, thereby ameliorating psoriasis-like inflammation in the mouse model. Moreover, XGLXJD decoction alleviated the depression-like behavior in the model.
Psoriasis frequently leads to multiple complications, and depression plays an important role in these complications (Nicholas & Gooderham, 2017). Depression impairs inflammatory responses, and inflammation promotes depression, thus activating a critical vicious cycle. In the TCM theory, human body is considered in its entirety, and TCM decoctions usually contain multiple herbs and ingredients that holistically target various organs of the body (Liu et al., 2021). TCM considers the human body as a complex dynamic system and focuses on maintaining balance in the body (Ma et al., 2016). Regarding psoriasis and other inflammatory disorders, the TCM theory considers the involvement of skin, brain, and other organs and aims to simultaneously treat whole body inflammation (Chan & Ng, 2020). XGLXJD decoction was prepared based on the TCM theory. The herbs in the decoction are used for cooling blood, which is the key mechanism of blood-heat type disease in the TCM theory. However, there is a need to explore the mechanism of action using modern techniques. Therefore, we performed network pharmacology analysis to predict the potential targets. In KEGG analysis, the AC–cAMP–PKA signaling pathway attracted our interest.
The AC–cAMP–PKA signaling system is the perfect post-receptor signal transduction system. Many ligands, such as hormones and other neurotransmitters, participate in this pathway. When these are transmitted into the brain and bind to specific receptors on the cell membrane, the G protein activates the AC to catalyze the hydrolysis of ATP to form cAMP, known as the second messenger (Zhang et al., 2020). Consequently, cAMP further activates PKA, which phosphorylates and regulates various target proteins. Increased levels of pro-inflammatory mediators, such as C-reactive protein and IL-6, are present in both psoriasis and depression, suggesting that inflammation represents a common fundamental link between depression and psoriasis (Hölsken et al., 2021). In this study, we predicted that the AC–cAMP–PKA signaling pathway and its regulation of inflammation may mediate the effect of XGLXJD decoction in alleviating psoriasis and inhibiting depression-like behavior.
cAMP binding abnormality is an important factor in psoriasis (Raynaud et al., 1989). A study demonstrated significant negative correlation between the severity of PASI and the binding of the cAMP analog to PKA (Raynaud et al., 1989). cAMP and CREB were also identified as therapeutic targets for psoriasis treatment in previous reports of arctigenin (Li et al., 2021) and salmeterol, a long-acting β2-adrenergic agonist (Xu et al., 2022). In this study, XGLXJD decoction was found to activate the AC–cAMP–PKA signaling pathway, which can inhibit the inflammatory response. The cAMP–PKA–CREB signaling axis modulates the downstream expression of pro-inflammatory cytokines, thereby inhibiting neuroinflammation (Jin et al., 2021; Park et al., 2016) and lung injury (Wang et al., 2022). The cAMP–PKA–CREB signaling axis modulates the downstream expression of IL-17 (Salinthone et al., 2010). PKA activation regulates cytokine profiles and modulates functional properties of both human naive and memory CD4(+) and CD8(+)T cells during immune and inflammatory processes (Shin et al., 1999). In this study, XGLXJD decoction activated the AC–cAMP–PKA signaling pathway in blood and skin, thereby inhibiting the downstream pro-inflammatory cytokines production.
The AC–cAMP–PKA signaling pathway is also closely related to the occurrence of depression. CREB, a transcription factor, is commonly activated by PKA and plays a crucial role in learning and memory in the brain (Lonze & Ginty, 2002). Changes in 133rd serine of CREB are closely related to the pathogenesis of depression (Peng et al., 2018). In this study, XGLXJD decoction activated the AC–cAMP–PKA signaling pathway, thereby alleviating depression-like behavior in mice.
Some aspects should be considered in future research. First, the effect of XGLXJD decoction on other manifestations of psoriasis (Rendon & Schäkel, 2019), such as psoriatic arthritis and kidney injury, should be investigated as the TCM theory considers human body in its entirety; this will be consistent with the trend of Evidence-based TCM: Beijing Declaration on ancient decoctions (Zhang et al., 2020). Second, as XGLXJD decoction is a TCM formula containing multiple compounds, its various active ingredients should be determined using better techniques, such as LC–mass spectrometry. Third, the pharmacokinetics of the standardized chemical components in the human body, including the absorption and metabolism patterns, should be further investigated.
Some limitations of this study should be considered. First, inhibition of the AC–cAMP–PKA signaling pathway was not performed as its inhibitor has not been largely applied in animal studies. Second, due to the relatively short duration of XGLXJD decoction treatment, we could not observe its long-term effects.
Conclusion
In this study, XGLXJD decoction was found to alleviate imiquimod-induced psoriasis-like inflammation in a mouse model. Furthermore, it alleviated depression-like behavior in mice. The AC–cAMP–PKA–CREB signaling pathway may play an important role in mediating the effect of XGLXJD decoction.
Abbreviations
XGLXJD: Xiegan-Liangxue-Jiedu decoction; PPI: Protein–protein interaction; GO: Gene ontology; KEGG: Kyoto encyclopedia of genes and genomes; HPLC: High-performance liquid chromatography; ELISA: Enzyme-linked immunosorbent assay; AC: Adenylyl cyclase; cAMP: Cyclic adenosine monophosphate; PKA: Protein kinase A; IL-17: Interleukin-17; TNF-α: Tumor necrosis factor-α; IHC: Immunohistochemical; CREB: cAMP-response element binding protein; HE: Hematoxylin and eosin; IF: Immunofluorescence; CA: Cornu ammonis.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This work was supported by Shanghai Artificial Intelligence Innovation and Development Project-Intelligent Dermatology Clinic Based on Modern TCM Diagnostic Technology [grant number 2020-RGZN-02038] and Shanghai Science and Technology Action Innovation Plan [Grant number 20Y21901300].
Supplementary Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
