Abstract
Objectives
Prolactinoma is a common intracranial tumor with a high incidence and serious harm to human health. At present, there is only one therapeutic drug in China, bromocriptine, and the Chinese herb
Materials and Methods
First, the extracts of
Results
Through the CCK-8 assay, the PVE inhibited the proliferation of MMQ cells. From the network pharmacology, the 21 targets, 9 signaling pathways, and 20 gene ontology (GO) projects were attained (
Conclusion
Our findings demonstrate that the PVE was verified against prolactinomas through the ESR1, MAPK3 targets, and the phosphoinositide 3-kinase/protein kinase B pathway.
Introduction
Prolactinoma is the most frequent pituitary neuroendocrine tumor, and recent epidemiological reviews have estimated that this tumor subtype accounts for 53% of pituitary neuroendocrine tumors (Inder & Jang, 2022). Prolactinoma causes various serious hazards to mankind’s health, for instance, amenorrhea, sexual dysfunction, galactorrhea, hyperprolactinemia, and infertility (Maiter, 2016; Zhou et al., 2018). In China, bromocriptine is a first-line drug against prolactinoma. Drug resistance frequently occurs, which brings clinical challenges (Wang et al., 2021; Xiao et al., 2020). Now that the pathogenesis of prolactinoma is unclear, drug development and therapeutic targets have occasion to be investigated imminently. So, seeking effective drugs with low side effects is essential. It has been reported that Traditional Chinese medicines have good effects on treating Prolactinoma, which has recently drawn the attention of scientists worldwide (Wei et al., 2017, 2018).
At present, although there are relevant reports of
As a representative research field, network pharmacology mainly utilizes bioinformatics to reveal the potentially complex relationships between multicomponent and multitargets, and its significant advantages can be directly verified through experiments based on the results displayed on the network (Li et al., 2021; Nogales et al., 2022). Molecular docking is a simulation method to predict the binding mode of receptor and ligand by analyzing the properties and interaction of receptor-ligand by the electric field force (Pinzi & Rastelli, 2019). It is mainly used for drug design and discovery based on the structure of receptors and ligands. The goal of this exploration is to use cyberpharmacology and molecular docking to forecast the possible targets and pathways of the anti-prolactinoma effect of the PV, combined with
Materials and Methods
Selection of Active Ingredients of the PV
The main active compounds of the PV were obtained by using the Traditional Chinese Medicine System pharmacology database and analysis platform (TCMSP;
Acquisition of Candidate Targets
The screened active compounds were imported into the PubChem database (
Construction of the PV–Target–Prolactinoma Network
In this research, the network of the PV–target–prolactinoma was established by Cytoscape 3.8.0 software. Subsequently, the mechanism of the PV against prolactinoma was further analyzed.
The GO and KEGG Pathway Enrichment
The 21 key targets were put into DAVID 6.8 database (
Construction of the PPI Network of Core Target
The 21 key targets of PV against prolactinoma were put into the STRING database (
Molecular Docking
In SYBYL-X 2.0 software, the binding ability of active substances to target proteins was assessed by Surflex-Dock module. Based on the PPI network and component–protein interactions, key components, and target protein molecules were selected for molecular docking. Download the construction of proteins directly from the PDB database, and modify it through SYBYL-X 2.0 software, including hydrogen addition, water removal, side chain repair, etc., to promote the reliability of molecular docking results (Feng et al., 2022). The 3D chemical structures of the active compositions were obtained from the PubChem database. A total score >7.0 indicates strong binding activity, a total score of 3.0–5.0 indicates moderate binding activity, and a total score of <3.0 indicates low binding activity (the lower the score, the lower the binding activity).
Drugs and Cell Culture
The PV(pharmaceutical batch number: 202208011) was supplied by Hubei Tianji Pharmaceutical Co. Ltd., and identified by Vice TCM Pharmacist Meng Junhua from Wuhan University Tongren Hospital. The PVE was extracted from 10 times the weight of the PV by refluxing water for 2 h and then condensed by evaporation. The following materials were occupied in this research: bromocriptine (Batch number: 35549; MedChem Express, New Jersey, USA). Rat prolactinoma tumor MMQ cell lines were suspended and cultured in DMEM (Gibco, Thermo Fisher Scientific, Inc.), medium containing 10% fetal bovine serum (FBS, Gibco, Thermo Fisher Scientific, Inc.) at 37℃, 5% CO2 atmosphere. The cells were treated with the PVE (0, 40, 80, 160 µg/mL), bromocriptine for 48 h.
Cell Viability Assay
Cell viability was tested by a CCK-8 assay. Depending on the maker’s guidance, log-phase MMQ cells were collected and adjusted to 1×105/mL. Different concentrations of the PVE (0, 40, 80, and 160 µg/mL) were individually added to each experimental group well. After being cultivated in an incubator for 48 h. The optical density values were tested at 450 nm. Then, calculate the cell survival rate and mortality rate.
Apoptosis Analysis
Apoptosis was detected by flow cytometry based on the manufacturer’s technical manuals. Cells were double-stained with PE Annexin V and 7-AAD for 15 min in the dark at room temperature and then analyzed by flow cytometry.
Western Blotting Analysis
Western blotting detected the expression of various proteins in the cells. At the end of the administration, the total protein of MMQ cells was extracted and its concentration was detected. The protein was separated and transferred to the polyvinylidene fluoride membrane by SDS-PAGE. The membrane is closed with 5% buttermilk at room temperature for 1 h, then incubated overnight in primary antibodies at 4°C. The antibodies used in this experiment were anti-BAX (1:2000); anti-ESR1 (1:2000); anti p-PI3K (1:2000); anti-mTOR (1:1000); anti p-AKT (1:1000); anti-MAPK3 (1:3000); anti p-AKT (1:2000); anti-PRL (1:6000); anti-Bcl-2 (1:2000), and cultured in secondary antibodies for 1h. The antibodies used were from Proteintech Co., Ltd. in China and Affinity in the USA. After elution, the TBST was exposed using electrochemical luminescence (ECL), and the gray values of the protein bands were determined by Image J.
Results
The PVE Repressed the Growth of MMQ Cells in vitro
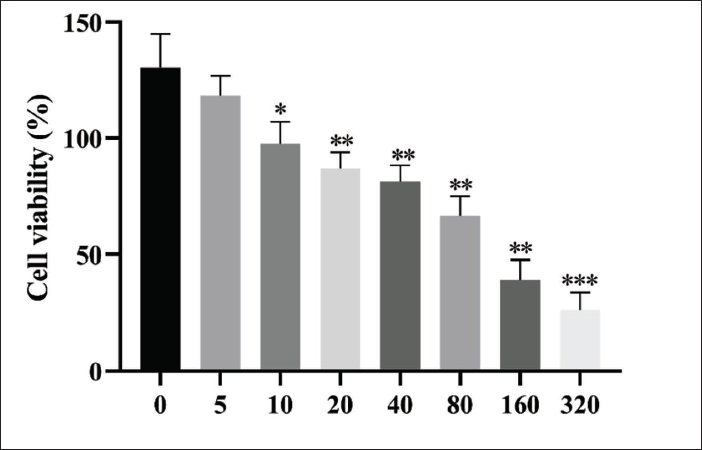
The CCK-8 results have demonstrated that MMQ cell viability decreased significantly after 5, 10, 20, 40, 80, 160, and 320 µg/mL PVE treatment (Figure 1). The viability of MMQ cells treated for 48h with the 320 µg/mL PVE was 46.32%. Thus, the 40, 80, and 160 µg/mL PVE were selected for the subsequent experiment.
Effects of the PVE on the Viability of MMQ Cells. After Treatment with the PVE for 48 h, the Viability of these Cells was Measured by CCK8 Assay. *p <0.05, **p < 0.01, and ***p <0.001 Vs. the Control Group.
Obtaining Active Compounds From the PV
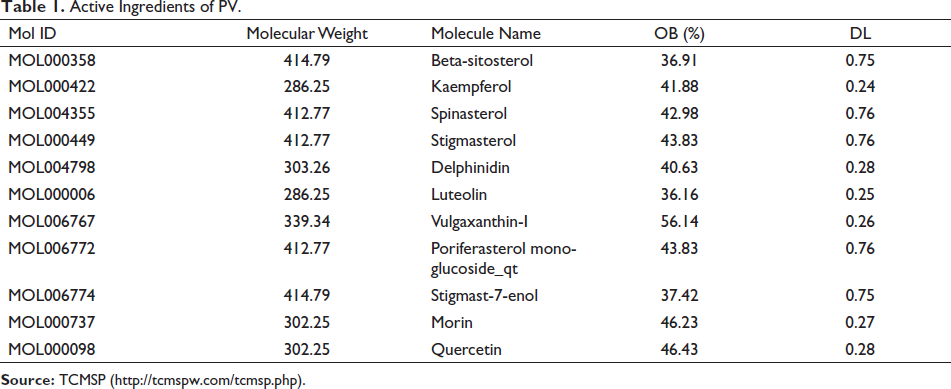
The chemical composition database of the PV was built by exercising the TCMSP and PubChem databases. Finally, the 60 main components of PV were screened, and 11 active ingredients were obtained according to the values of OB ≥ 30% and DL ≥ 0.18, as shown in Table 1.
Active Ingredients of PV.
Acquiring Intersection Targets
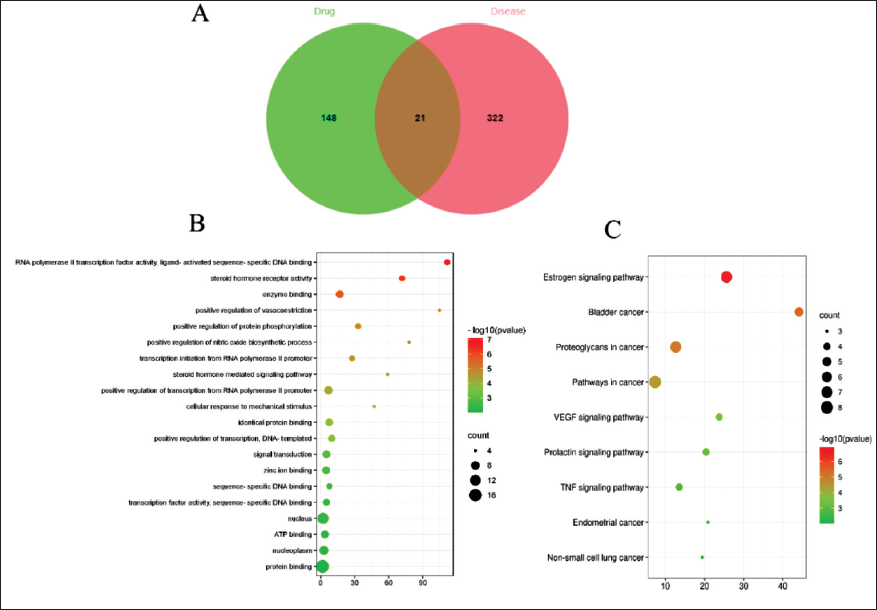
The 169 targets for the drug-active ingredients were gained through the PubChem database. Through the GeneCards database, 343 genes associated with prolactinoma were collected. The online mapping tool was used to import PV and prolactinoma-related targets, respectively, to draw the VENN diagram of drug-disease targets in Figure 2A, with a total of 21 intersection targets.
VENN Diagram and the Enrichment Analysis of Candidate Genes and Signal Pathways. (A) VENN Diagram of Intersection Targets Between the PV and Prolactinoma. (B) Top 20 GO Enrichment Bubble Chart. (C) Top Nine KEGG Pathways Bubble Chart. The Size of Bubbles Grows as “Counts” Increase.
Target Biological Function Analysis of the PV in the Treatment of Prolactinoma
The GO function and KEGG pathway of 21 key anti-prolactinoma targets were implemented by operating the David database. By the GO function enrichment analysis (
The Network of PV–target–prolactinoma and PPI Network Establishment and Analysis
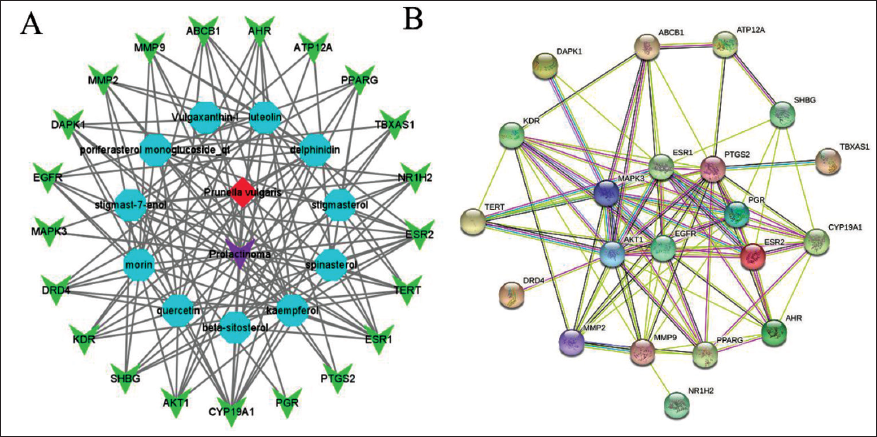
In Figure 3A, the 21 key targets were imported into the Cytoscape to visualize the “PV–target–prolactinoma” relationship network. In Figure 3A, the nodes have unlike shapes and colors where red represents the PV, purple represents prolactinoma, blue represents compounds and green represents the key targets. After the STRING database was integrated, input a PPI network into the Cytoscape 3.8.0 software for analysis. As shown in Figure 3B, the PTGS2 (degree = 16), AKT1 (degree = 15), ESR1 (degree = 15), MAPK3 (degree = 14), and EGFR (degree = 14)were all greater than the median degree (degree = 9) and ranked in the top 5. We initially believe that the PVE may mainly act on these targets to treat prolactinoma.
PPI Network and the Connection Between Active Ingredients and Targets. (A) Network of the Relationship Between the PV–target–prolactinoma. The Red, Purple, Blue, and Green Nodes Represent the PV, Prolactinoma, Active Ingredients, and Targets, Respectively. (B) PPI Network of the PV Targets to Treat Prolactinoma.
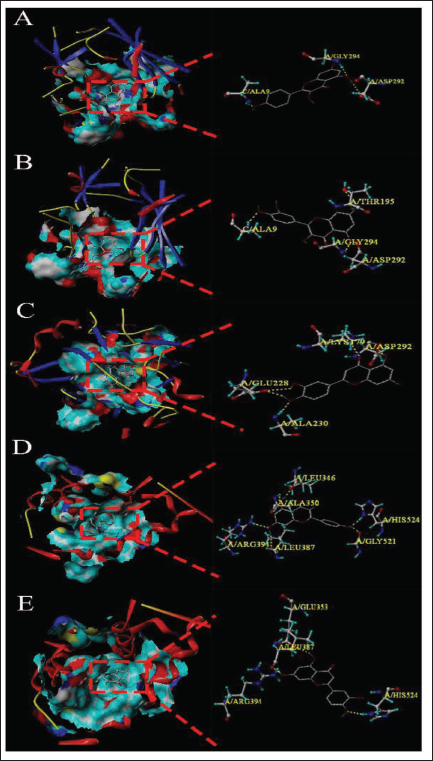
Molecular Docking Verification
The binding sites and binding scores can directly image the stability and interactivity of the docking model. Here, we used the SYBYL-X 2.0 software to conduct molecular docking of key components through the network of PV–target–prolactinoma and PPI network analysis and established the docking models of AKT1 and ESR1 with kaempferol, quercetin, and luteolin, respectively, as shown in Figure 4. It was indicated that multi-docking scores are greater than 4, showing that the binding between the ligand and the receptor was greatly firm, as shown in Table 2.
Molecular Docking Diagrams Between the Main Ingredients of the PV and the Core Targets. (A) Molecular Docking Between Kaempferol and AKT1. Kaempferol and Three Protein Residues (ALA9, ASP292, G LY294, and ASP292) in AKT1 Bonded to Each Other Through Hydrogen Bonds. (B) Molecular Docking Between Quercetin and AKT1. Quercetin and Four Protein Residues (THR195, ALA9, ASP292, and GLY294) in AKT1 Bonded to Each Other Through Hydrogen Bonds. (C) Molecular Docking Between Luteolin and AKT1. Luteolin and Four Protein Residues (GLU228, ALA230, LYS179, and ASP292) in AKT1 Bonded to Each Other Through Hydrogen Bonds. (D) Molecular Docking Between Kaempferol and ESR1. Kaempferol and Six Protein Residues (LEU346, ALA350, ARG394, LEU387, GLY521, and HIS524) in ESR1 Bonded to Each Other Through Hydrogen Bonds. (E) Molecular Docking Between Luteolin and ESR1. Luteolin and Four Protein Residues (GLU353, ARG394, LEU387, and HIS524) in ESR1 Bonded to Each Other Through Hydrogen Bonds.
Docking Score.
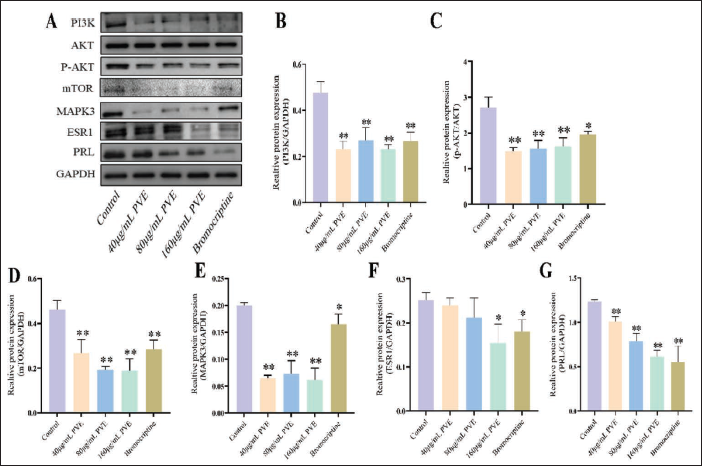
The PI3K/AKT Signaling Pathway Took Part in the Suppression of the PVE on PRL Secretion
In this study, bromocriptine was used as the positive control group, which is a common drug for treating prolactinoma. After treatment with 40, 80, and 160 µg/mL PVE, the protein expression of ESR1, MAPK3, mTOR, p-ATK/AKT and PI3K significantly reduced in MMQ cells (
Effects of the PVE on the PI3K, AKT, p-AKT, mTOR, MAPK3, ESR1, and PRL in MMQ Cells for 48 Hours. (A) Western Blotting Images. (B) Statistical Analysis of the Expression of the PI3K. (C) Statistical Analysis of the Expression of the p-AKT. (D) Statistical Analysis of the Expression of the mTOR. (E) Statistical Analysis of the Expression of the MAPK3. (F) Statistical Analysis of the Expression of ESR1. (G) Statistical Analysis of the Expression of the PRL. *p <0.05 and **p <0.01 Vs. the Control Group (n = 3).
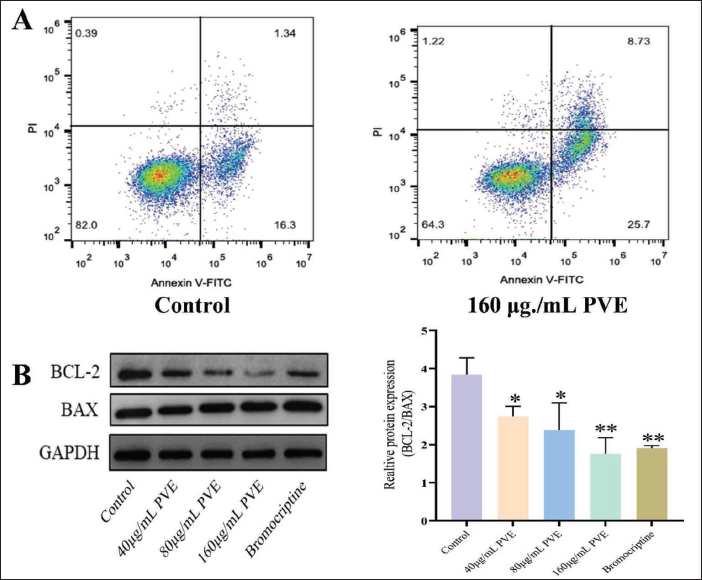
The Pro-apoptotic Mechanism Fluences the Inhibitory Effects of the PVE on MMQ Cells
After treatment with 40, 80, and 160 µg/mL PVE for 48h, the apoptosis rate was checked. As a result, the 160 µg/mL PVE could trigger apoptosis in MMQ cells (Figure 6A). We discovered that the PVE lowered the expression of Bcl-2, but made no difference to the expression of BAX (Figure 6B).
Effects of the PVE on the Bcl-2 and BAX in MMQ Cells for 48 Hours. (A) Apoptosis Rate of MMQ Cells Treated with the PVE (40, 80, and 160 µg/mL) was Detected by Annexin V-FITC and PI Double Staining. (B) Western Blotting Images and Analysis of the Bcl-2 and BAX Protein Expression After the PVE Treatment of MMQ Cells. *p <.05 and **p <0.01 Vs. the Control Group (n = 3).
Discussion
As one of the most effective choices of medicine, Chinese herbal medicine is also the most common medicine in China (Wang et al., 2018). The PVE has a good anti-cancer influence, which has further caught our attention to study (Wang et al., 2019; Zhang et al., 2020). Thus, the research plumbed the function and mechanism of the PVE on prolactinoma in MMQ cells. We utilized cyberpharmacology to explain the possible composition and action mechanism of the PVE. Finally, we successfully verified this through cell experiments. The results confirmed that the PVE may inhibit the proliferation of prolactinoma. The CCK-8 results suggested that the PVE had an anti-prolactinoma role. Interestingly, the ESR1, AKT1, and MAPK3 ranked among the top five in the analysis of cyberpharmacology. It was reported that the AKT and ESR1 inhibitors were often used to treat prolactinoma (Gao et al., 2017; Roof et al., 2018; Tang et al., 2019). This is consistent with our results and also validates the correctness of cyberpharmacology. Thus, the PVE might be an AKT or ESR1 inhibitor against prolactinoma. And we utilized the SYBYL-X 2.0 software to perform the molecular docking and described the probable binding sites of the PVE with relevant proteins. Therefore, elaborating on the ESR1 and AKT1 correlative pathways is consistent with our work objectives.
Estrogen plays a key role in the occurrence of prolactinoma, a possible mechanism is binding to the estrogen receptor, a nuclear steroid hormone receptor in the pituitary, consisting of Estrogen receptor α and Estrogen receptor β (Lv et al., 2012). ESR1, as one of the estrogen receptors, widely regulates various life activities. Estrogen could quickly and constantly activate membrane receptors through the PI3K/Akt and MAPK/ERK signaling pathways (Knowlton & Lee, 2012). Multiple reports have confirmed that the high expression of ESR1 could accelerate the occurrence of pituitary prolactinoma and kick start the progression of pituitary tumor (Delgrange et al., 2015). AKT1, as a member of AKT kinase, takes part in cellular progress, namely cell proliferation, apoptosis, and glucose metabolism. The PI3K/AKT pathway has formerly been reported to be connected with prolactinoma development (Dworakowska et al., 2009; Ferraris, 2022). It was shown that the PVE markedly suppressed the expression of AKT1 and Bcl-2/BAX protein. Thus, the PVE led to the apoptosis of MMQ cells by suppressing the PI3K/AKT1 pathway. Apoptosis, as one form of programmed cell death, refers to the orderly death of cells under the control of genes so as to hold a steady internal environment. A lot of natural drugs have been demonstrated to trigger apoptosis by intrinsic pathways (Xu et al., 2015). When the standards of oncogene signaling increase or DNA harm occurs, the inner pathway is induced, and the whole inner pathway is controlled by the Bcl-2 protein family. The Bcl-2 family contains both anti-apoptotic and pro-apoptotic proteins, which together adjust apoptosis. It was demonstrated that the PVE markedly decreased the expression of Bcl-2/BAX in MMQ cells. Consequently, the anti-prolactinoma effect of the PVE had a connection with apoptosis. Finally, it is not hard to show that PVE may be a potential candidate drug against prolactinoma. So, our work is meaningful.
Conclusion
In summary, utilizing cyberpharmacology, molecular docking, and cell experiments
Footnotes
Abbreviations
AKT: RAC-alpha serine/threonine-protein kinase; BAX: Apoptosis regulator BAX; Bcl: Apoptosis regulator Bcl-2; GO: Gene ontology; KEGG: Kyoto encyclopedia of genes and genomes; MAPK3: Mitogen-activated protein kinase 3; mTOR: Serine/threonine-protein kinase; PI3K: Phosphatidylinositol 3-kinase; PPI: Protein–protein interaction; PRL: Prolactin.
Acknowledgements
The authors thank Yu Zhang for excellent technical support, Jun-hua Meng and Yong-gang Chen for critically reviewing the manuscript.
Author’s Contribution
HZ and PN: designed the research and completed the experiment. HZ and Y-C: drafted the manuscript, PN and YZ: consulted literature and completed data statistics. XW: assisted in solving the experimental challenges. JM and Y-C: were major contributors in reviewing the manuscript. JM, PN, and YC: contributed equally to this study, and are both listed as first authors. HZ and Y-C: are rightfully corresponding authors. All authors have read the final manuscript and approved of the submission.
Data Availability Statement
The data that support the findings of this study are available from the corresponding authors upon reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This work was supported by Hubei Provincial Central Guidance Local Science and Technology Development Project [grant number 2020ZYYD026], Major Projects in Wuhan (WZ21M03) and Wuhan Municipal Health Commission scientific research project [Grant number WZ22A01].
Statement of Informed Consent and Ethical Approval
All experiments were approved by the Ethics Committee of Tongren Hospital Affiliated to Wuhan University.
