Abstract
Background
Osteosarcoma (OS) is a highly metastatic primary bone malignancy, and the treatment options remain inadequate. Hence, exploring innovative natural medications is required. Prunetin (PRU) is an isoflavone that has been a proven anticancer agent in numerous cancer cell lines. However, the activity of PRU against OS remains uncertain.
Materials and Methods
Here, we studied the anticancer activity of PRU (20 and 25 µM) on human OS cells MG-63 and investigated its latent mechanism. The PRU activity of MG-63 cells cytotoxicity, intracellular ROS, metastasis, apoptosis, anti-apoptotic proteins, MAPK/STAT-3, and AKT signaling pathways was assessed by MTT assay, DCFH-DA, DAPI, PI, AO/EB, cell adhesion, and RT-PCR analysis. Findings unveiled that PRU could constrain MG-63 cell viability and adhesion through elevated intracellular ROS and elicited apoptosis.
Results
Likewise, PRU (20 and 25 µM) avert the MG-63 cell proliferation, which stimulates apoptosis by the enhancement of Bax and caspases, while it diminishes Bcl-2 in a dose-dependent way. Furthermore, PRU could reduce Pin-1, and anti-apoptotic elements, as well as trigger apoptotic signaling pathways. Our data established that PRU alleviates MG-63 cell proliferation and metastasis via ROS-mediated apoptosis, which triggers MAPKs/STAT3 and AKT pathways, suggesting that PRU is a promising natural remedy for OS. In order to comprehend the therapeutic target for cancer, we assessed the effect of PRU on the expression of Pin1, which is thought to be over-expressed in many human malignancies. According to our findings, PRU specifically suppressed Pin1 expression to reduce the expression of Akt, STAT3, P38, JNK, P65, and IL-6. We evaluated the impact of PRU on the expression of Pin1, which is allegedly over-expressed in many human malignancies, to better understand the therapeutic target for cancer. Researchers state that PRU inhibited the expression of Akt, STAT3, P38, JNK, P65, and IL-6 in particular, by suppressing Pin1 expression.
Conclusion
Together, these results suggest that PRU may be an effective treatment for bone cancer in people by preventing Pin1 expression.
Introduction
Osteosarcoma (OS) is the supreme primary bone cancer malignancy, which demonstrates great potential for metastasis and is the most common pediatric bone malignancy (Fan et al., 2014; Wang et al., 2014). Children and adolescents show a high incidence of OS; this accounts for ~5% of total pediatric cancers (Errani et al., 2021). The prevalence of OS is 8.7 per million among people younger than 20 years, and the risk is higher in men (Mirabello et al., 2009). It mostly initiates from long bones with rapid growth, comprising the tibia, femur, and humerus (Li et al., 2015). A low survival rate of OS has been observed in early pulmonary metastasis (Picci, 2006). While the survival ratio of OS patients is progressively improving after chemotherapy and surgery, the five-year survival rate is 65%–70%, whereas the metastatic patients are only 20% (Botter et al., 2014). However, clinical use of chemotherapy is limited owing to its resistance. Therefore, all the developments in novel natural remedies are crucial for the enlightening prognosis of patients with OS.
Several signal transduction pathways that determine cell fate are regulated by the enzyme peptidyl-prolyl cis-trans isomerase NIMA-interacting 1 (Pin1) (Zhou & Lu, 2016). Most human malignancies, including ovarian, breast, colorectal, and cervical cancers, among others, exhibit abnormally high Pin1 expression (Angelucci & Hort, 2017). A poor clinical prognosis has been associated with Pin1 expression levels. By turning on more than 40 oncogenes and deactivating more than 20 tumor suppressors, overexpression of Pin1 speeds up tumor development. Pin1-null mice exhibit normal development, reduced tumor growth, and strong tumorigenic resistance. Pin1 inhibition disrupts the collaboration of the carcinogenic pathways in both CSC and non-CSC tumor cells, which justifies the creation of targeted therapeutics. These results imply that Pin1 may serve as a novel target for cancer treatment and a prognostic cancer marker (Moore & Potter, 2013; Zhou & Lu, 2016).
OS is the second leading cause of cancer-related death among children and adolescents due to the development of lethal metastases (Ottaviani & Jaffe, 2009). Common primary OS locations include the distal proximal tibia, femur, head, shoulder, jaw, and pelvis. Local discomfort, localized edemas, and sporadic joint dysfunction are the common symptoms. Almost 90% of cases are identified as high-grade OS at the time of diagnosis, which is normally done with X-rays (Fine et al., 2009). Currently, pre- and post-operative chemotherapy, as well as surgical procedures, serve as the comprehensive therapeutic options for OS (Carrle & Bielack, 2006). Despite recent advancements, there are still problems with OS treatment, especially for patients with metastases and chemotherapy resistance. Therefore, the development of novel and unconventional medicines continues to garner interest (Gaspar et al., 2020).
Prunetin (PRU) is a set of isoflavone, a characteristic O-methylated flavonoid that is isolated from the prunus family and belongs to the Rosaceae plants (Hu & Li, 2018). PRU has been revealed to display several pharmacological effects, including anti-cancer, anti-inflammatory, stress response, anti-obesity, and controlling proteolytic action (Ahn et al., 2013; Piegholdt et al., 2016). PRU has been established to exert an anti-obesity effect which has facilitated aldehyde dehydrogenase enzyme inhibition of hepatocytes in human (Nam et al., 2016; Sheikh & Weiner, 1997). PRU is too exposed to control the proteolytic action in osteoarthritis. Few reports established that PRU repressed gastric cancer cell growth (Vetrivel et al., 2020). Thus far, to the finest of our awareness, there is no described information on the anti-cancer perspective of PRU on bone tumor cells. In this present research, we examined the anti-proliferative, anti-metastatic, and apoptotic action of PRU on human OS cells, in an in vitro model. We illuminated the cytotoxic perspective and action of apoptosis instigated by PRU on MG-63, an OS human cell.
Materials and Methods
Chemicals
PRU, antibiotics, Phosphate-buffered saline (PBS), Dulbecco’s Modified Eagle Medium (DMEM), Fetal bovine serum (FBS),3-(4, 5-Dimethylthiazol-2yl)-2,5-Diphenyltetrazolium Bromide (MTT), Dichloro-dihydro-fluorescein diacetate (DCFH-DA), Acridine orange/Ethidium bromide (AO/EB), 4’,6-diamidino-2-phenylindole (DAPI), Propidium Iodide (PI), trypan blue, and Dimethyl Sulfoxides (DMSO) were procured from Gibco (CA, USA). Analytical-grade biochemicals and solvents were also used.
Cell Culture
MG-63, a human OS cell line, was obtained from Shanghai Aiyan Biotechnology Co. Ltd., (Shanghai, China). These cells were cultivated in DMEM media, together with FBS (10%), and 1% antibiotics (penicillin/streptomycin) at 37°C in a 5% CO2 atmosphere, and less than 95% humidified air.
MTT Cell Viability Assay
Human OS cell proliferation was performed by consuming the MTT experiment. Concisely, MG-63 cells were sowed into 96-wells (1×105 cells/well) and cultivated at 37°C in a wet incubator, ensuring 5% CO2. After overnight incubation, the cells were preserved with different amounts of PRU (0–35 µM) for 1 day. Later, the treated cells were added to the MTT (10 µL) solution and taken for an additional 4 hr to allow the transformation of MTT into formazan insoluble crystals by the mitochondrial dehydrogenase. The successive formazan was liquefied by adding DMSO (150 µL). The optical density was determined at 490 nm by employing a multifunctional plate reader (BD Biosciences, NJ, USA). Cell proliferation was measured as a percentage of viability against MG-63 control cells (100%). The IC50 value has determined the formula as shown below:
Cell viability inhibition (%) = (Optical density of Control-Optical density of Test) × 100
Determination of Intracellular ROS
Human OS cells MG-63 were sowed in six-well plates each and kept for one day, then diverse quantities of PRU (Control, 20, and 25 µM) were added. Then, all groups were stained with DCFH-DA (10 µM) and afterwards preserved at 37°C for 30 min. The stained cells were cleaned with ice-cold PBS twice to eliminate extra dye. The fluorescence was measured by a multimode reader as described previously (Velu et al., 2021).
Apoptosis Evaluation by AO/EB Staining
The apoptotic morphology in human OS cells MG-63 exposed to PRU at a concentration of control, 20, and 25 µM was evaluated by AO/EB staining (Kasibhatla et al., 2006). MG-63 cells were added with 20 and 25 µM PRU and kept for one day. The control and treated cells were added to the dye mixture including AO/EB (100 µg/ml of each dye). All groups were sustained for 20 min under the dark, ensuring that the unbinding dye was detached by PBS and detected over a fluorescence microscope (Olympus, Tokyo, Japan).
Apoptosis Assessed by DAPI Staining
Human OS cells MG-63 were sowed 1×105 cells in 96 well plates and added to PRU (control, 20, and 25 µM) fixed with paraformaldehyde (4%) at 37°C for 10 min. These conserved MG-63 cells were stained with DAPI to evaluate the nucleus alterations related to apoptosis by the method described earlier (Zheng et al., 2015). Then, all samples were fixed on a glass slip and viewed through a fluorescence microscope BX51.
Evaluation of PI Staining for Apoptosis
The apoptosis nuclei were examined by PI staining performed. Human OS cells MG-63 were preserved with diverse dosages of PRU (control, 20, and 25 µM) and conserved for 48 hr. Then, treated cells were assembled, and staining with PI was allowed to the protocol (Zheng et al., 2015). The fluorescent red emission from the nuclei was evaluated under a microscope (Olympus).
Cell Adhesion Assay
Human OS cells MG-63 were put on the media of 96-well plates and pretreated with PRU (Control, 20, and 25 µM), then sowed on a fibronectin-coated (500 µL/well). All treated groups were kept at 37°C for a variation between 60 and 100 min and PBS was eroded two-fold for the purpose of eradicating the non-adherent cells. Then, stationary with paraformaldehyde (4%), all wells were kept for 5 min at 4°C. Afterwards, .4% trypan blue staining to the adherent cells and calculated by using microscopy (Olympus).
Estimation of mRNA Expression Levels
Total RNA was sequestered from human OS cells MG-63 according to the company’s protocols by consuming TRIzol® reagent (Abcam, USA). The sequestered RNA was reverse transcribed into cDNA, accomplished by a high-capacity cDNA Reverse Transcription kit (Abcam) permitting the company’s procedures. Then, the Fast Start SYBR Green master mix (Abcam), was employed to examine the cDNAs adapted to the manufacturer’s techniques. The band intensity was studied by employing 1.5% agarose gels, which were exposed to electrophoresis; at last, the band intensity was determined in ImageJ 1.48 software.
Western Blot Investigation
SDS-PAGE was used to separate the proteins from equal amounts of extract (25 g of total protein), and the proteins were then transferred to Polyvinylidene Fluoride (PVDF) membranes. After blocking, primary and secondary antibodies diluted in 5% bovine serum albumin were successively incubated with the membranes. The secondary antibodies for detection were Immunoglobulins (IgG) that had been tagged with Horseradish Peroxidase (HRP). Cell Signaling Technology provided these antibodies (Beverly, MA, USA). Using the AlphaImager 2200, the proteins’ expression was investigated (Bio-Techne, Minneapolis, MN, USA).
Statistical Analysis
All tests were independently performed thrice and the results were stated as mean ± SD. A statistical study was accomplished by GraphPad Prism software, where version 8.0.1 was employed to perform an Analysis of Variance (ANOVA) and Duncan’s examination in subsequence. Statistics showed a significant reflection of p < 0.05.
Results
Anti-proliferative and Cytotoxicity Properties of PRU on MG-63 Cells
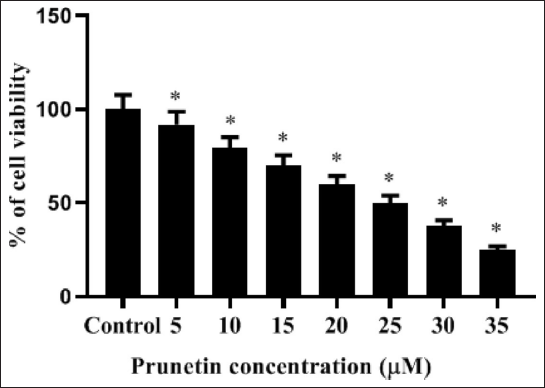
The human MG-63 cell viability is assessed by MTT test with different quantities (0–35 µM) of PRU. Data exposed that PRU had reduced viability through its cytotoxic and antiproliferative actions on MG-63 cells in a quantity-dependent way. PRU treatment at a concentration of less than 10 µM could not expressively alter the viability of MG-63 cells. However, the higher amount of PRU (15, 20, 25, 30, and 35 µM) substantially reduced (p < 0.05) the proliferation of MG-63 cells against an untreated control. From the MTT test, we detected the IC50 value of PRU as 25 µM for MG-63 cells. Based on the figure of the inhibitory concentration data of 20 and 25 µM, PRU has been taken for further trials (Figure 1).
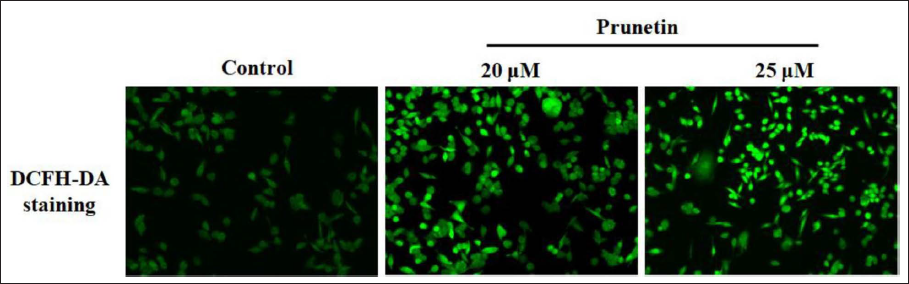
Influence of PRU on the Intracellular ROS Accumulation in MG-63cells
The formation of intracellular ROS is associated with assorted stimuli and can activate cell death. ROS intensities were improved in MG-63 cells after being added with 20 and 25 µM PRU for one day. The gathering of ROS, DCFH-DA-branded cells was noticed beneath a fluorescence microscope. The intensity of ROS was greatly augmented (p < 0.05) with 25 µM of PRU in contrast to untreated control cells (Figure 2).
PRU-induced Apoptosis on MG-63 Cells Demonstrated by Dual Staining
Apoptotic cells imagined the distinct morphological variations accomplished by the AO/EB dual staining. MG-63 control cells presented uniformly stained green live cells. PRU (20 and 25µM) treatment exposed increased cell apoptosis in a quantity-dependent mode. Initial apoptotic cells unveiled membrane blebbing and chromatin condensation in treatment with PRU (20µM), which displayed yellow dots in the light greenish cells. It has exhibited orange color in the late apoptotic cells that have wasted their membrane integrity owing to ethidium bromide co-stain in PRU (25 µM) treatments (Figure 3A).
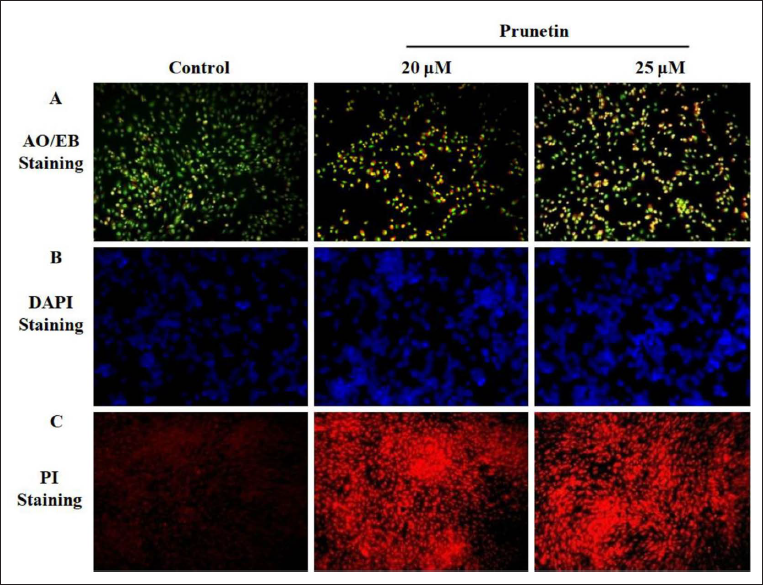
PRU-triggered Apoptosis on MG-63 Cells Showed by DAPI Staining
Human MG-63 cells added with DAPI staining revealed the survival of typical viable cells with usual nuclei. Treatment with PRU in the MG-63 cancer cells elicited apoptosis that heightened the nuclear morphology and nuclear bodies’ fragmentation compared to MG-63 control cells. PRU (20 and 25 µM) exposed to MG-63 cells viewed chromatin reduction, membrane blebbing, the destruction of the nuclear envelope, and cellular collapse. These effects emphasized that PRU-triggered apoptosis in a dose-dependent way (Figure 3B).
PRU-inspired Apoptosis on MG-63 Cells Evidenced by PI
PI was an effective Stain to distinguish the apoptotic nuclei of MG-63. PI stain travels into the cells after losing the integrity of the membrane, which is associated with the weakening of membrane polarization, which indicates cell death. The staining with PI exposed that PRU inclined apoptotic actions on MG-63 in a concentration-needy manner. Treatment with PRU (20 and 25 µM) increased the apoptosis effect in contrast to MG-63 control cells. Thus, PRU-driven apoptosis could be one of the mechanisms for reducing MG-63 cell proliferation (Figure 3C).
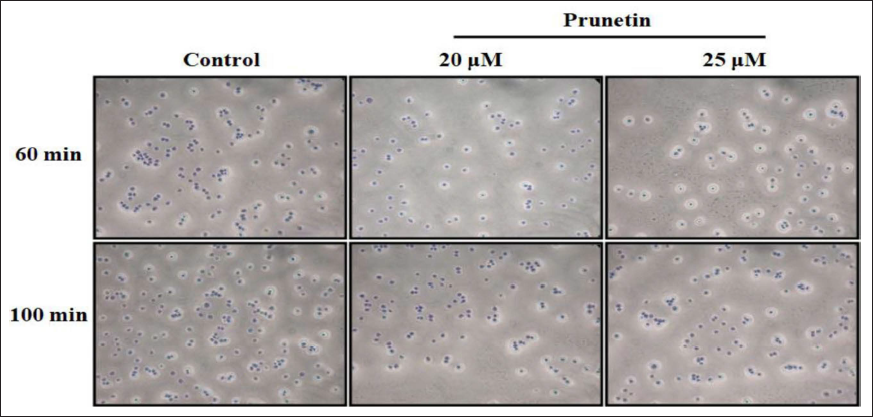
Influence of PRU on MG-63 Cells Adhesion
Human OS cells MG-63 adhesion on the extracellular matrix has a considerable action in generating metastasis of the malignant cells. The addition of PRU in the MG-63 cells exposed silencing on extracellular matrix proteins adhesion. The number of MG-63 adherent cells was remarkably diminished on treatment with PRU and then reduced after 60 and 100 min against control cells (Figure 4). Treatment using PRU (20 and 25 µm) with MG-63 cells significantly declined (p < 0.05) the number of cell adhesions in a quantity-reliant manner when compared to the control.
Influence of PRU on Apoptotic Proteins mRNA Expression Levels
Administration of PRU (20 and 25 µM) to MG-63 cells for measuring the levels of mRNA expression of apoptotic proteins. The untreated MG-63 cells demonstrated that Bcl-2 was elevated, while caspase-3, Bax, caspase-9, and -8 levels were lessened. Treatment with PRU expressively reduced Bcl-2, while Bax, caspase-3, -8, and -9 the mRNA levels were elevated in a dose-dependent manner against untreated control (Figure 5).
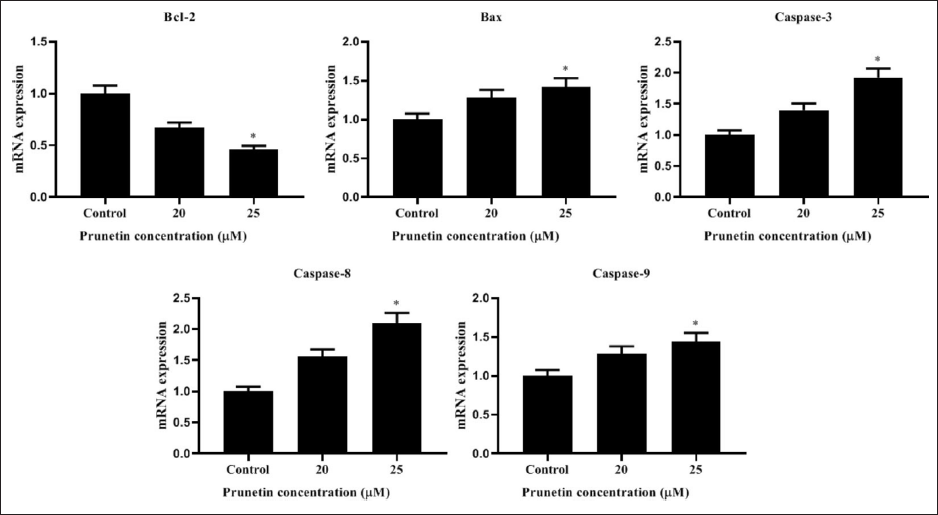
PRU Inhibits the mRNA Levels of Human MG-63 cells
The mRNA levels of Pin-1, STAT-3, p38, JNK, and p65 were reduced on PRU-treated MG-63 cells versus control cells. Administration of PRU (20 and 25 µM) on MG-63 cells was attenuated in the mRNA expression levels of apoptotic proteins in a quantity-needy way (Figure 6).
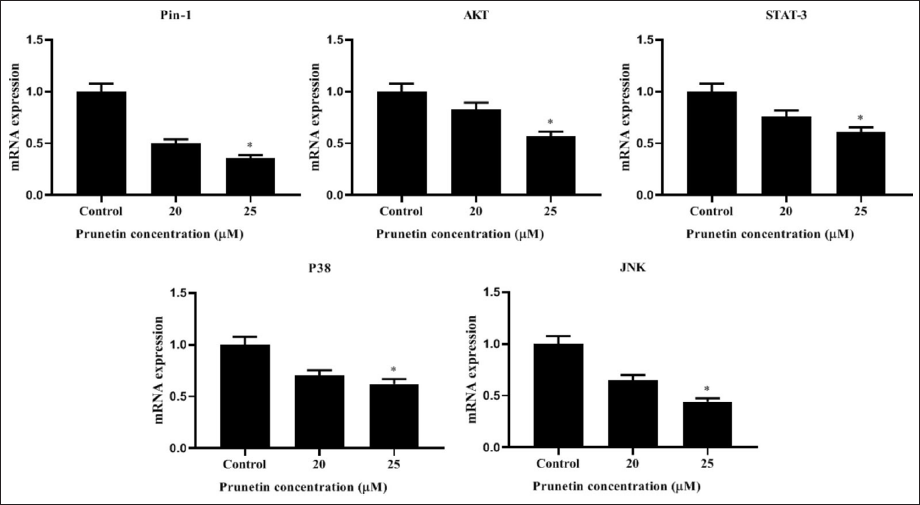
PRU Inhibits the Protein Levels of Human MG-63 cells
Analysis of Pin1 expression helped clarify the molecular processes by which PRU impacts bone cancer cells. The protein expression of Pin1 was dramatically down-regulated in a dose-dependent manner after 24 hr of treatment with different PRU dosages (Figure 7). PRU was tested on a group of oncoproteins that are Pin1 substrates and whose protein stability is maintained by Pin1 to support the theory that PRU shows powerful anticancer efficacy against bone cancer by inhibiting Pin1. According to our research, PRU therapy significantly decreased the expression of pin1, p-Akt/Akt, p-STAT3/STAT3, p-P38/P38, and p-JNK/JNK (Figure 7).
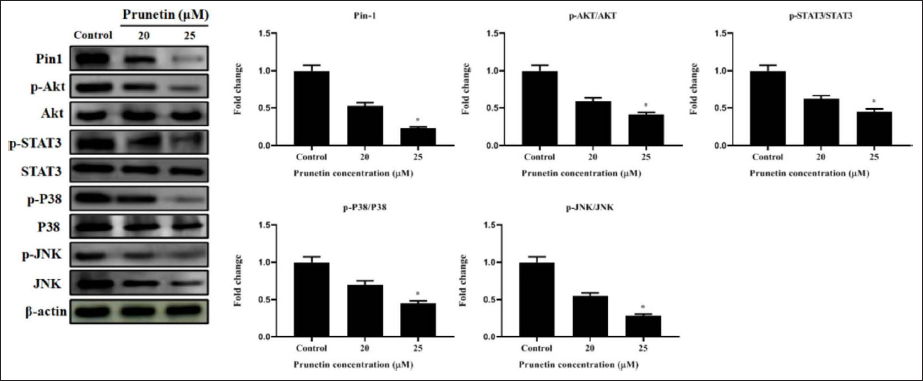
Discussion
OS is the utmost prime bone cancer that mostly happens in adolescents and children (Fan et al., 2014; Wang et al., 2018). It shows high morbidity and mortality owing to poor prognosis and dismal therapeutic outcomes (Errani et al., 2021; Picci, 2007). Metastasis is the prominent source of death in OS patients, and the remedial alternatives remain poor(Fan et al., 2014). Despite recent advances in diagnostic technology and treatment, it is incompetent to invent an effective drug to treat OS, due to drug resistance and metastasis. Scientists have explored novel agents or molecules that may be used to treat numerous tumors including OS. PRU has been revealed to display several valuable activities, including anti-inflammatory, anti-cancer, anti-obesity, and also controlling proteolytic action (Ahn et al., 2013; Carrle & Bielack, 2006; Fine et al., 2022). However, the anticancer efficacy of PRU on human OS cells MG-63 has not yet been reported so far. Thus, this research is intended to identify an effective isoflavone, PRU against human OS cells MG-63 and to evaluate its latent molecular actions. The current research established that PRU treatment on OS cells considerably reduced tumor cell proliferation, cell adhesion, and induced cell apoptosis in a quantity-dependent way.
Atypical proliferation plays a vital action in carcinogenesis and expansion. Hence, the regulation of a typical propagation signal was an active technique to employ antitumor properties (Wang et al., 2014). We established that PRU repressed MG-63 cell viability and cell adhesion in a quantity-dependent manner by the stimulation of apoptosis. These results point out that PRU may not only function as an inhibitor of tumor cell proliferation, migration, and metastasis but can also act as an apoptosis promoter in OS. Li et al. (2022), have reported similar results of the anti-cancer activity of PRU against hepatic carcinoma induced by a diethylnitrosamine in the rat model. Uncontrolled growth is one key feature of tumor cells (Wang et al., 2017). Suppression of tumor growth can be accomplished by the inhibition of cancer cell proliferation. Thus, PRU may be a possible pharmacological action to control OS.
Apoptosis is considered by biological and morphological alterations comprising chromatin condensation and subsequently DNA degradation (Khan et al., 2014), and it can be a crucial target for anticancer treatment. Induction or the restoration of apoptotic signaling pathways leads to apoptosis in cancer cells so numerous anti-cancer approaches target apoptosis (Wong, 2011). Our research confirmed that PRU had a cytotoxic effect on MG-63 cells through the induction of ROS-mediated cell death. ROS and the mitochondrial oxidative metabolic process play a vital action in the induction of apoptosis under both physiological and pathological conditions. ROS can activate diverse pathologies, including triggering apoptotic signaling pathways, the distraction of intracellular redox homeostasis, and oxidative modifications of biomolecules (Circu & Aw, 2010; Trachootham et al., 2009). ROS is a key mediator of apoptosis in OS cells. It has been documented that nimbolide prompts ROS generation and apoptosis in human OS cells U2OS and MG-63 (Liu et al., 2015). Another report indicated that Plumbagin stimulates apoptosis through intracellular ROS formation and mitochondrial apoptotic signaling pathways in U2OS and MG-63 cells (Chao et al., 2017). Here, we established the action of PRU on ROS-prompted cell death.
One of the hallmarks of the cancer cell is apoptosis resistance (Zhang et al., 2017). So, apoptosis stimulation in malignant cells is a central feature of cancer remedies. This finding indicates that the triggering of cell apoptosis is one of the mechanisms involved in PRU on cells. Bcl-2 family proteins that consist of anti-apoptotic and pro-apoptotic members show a vital part in the intrinsic pathway by controlling the discharge of pro-apoptotic elements from the mitochondria (Tsujimoto, 1998). Bcl-2 could alleviate the mitochondrial membrane permeability and averts apoptosis, whereas Bax could enhance the mitochondrial membrane permeability and stimulate apoptosis (Besbes et al., 2015). Bcl-2 and Bax have a synergistic influence on cell apoptosis control with diverse Bcl-2/Bax ratios (Golestani Eimaniet al., 2014). Our research emphasized that PRU usage added to reduced Bcl-2 mRNA levels, but enhanced Bax levels in OS cells. The caspase family is an alternative significant facilitator of apoptosis, and caspase-3 is a prominent enzyme that triggers death protease, which in turn catalyzes numerous cellular proteins (Porter & Jänicke, 1999). Caspase-3 is one of the vital genes in apoptotic elevation, which directly prompts cell death in cancer cells (Yang et al., 2017). Our research exposed that PRU could elevate Bax/Bcl-2 ratio, caspase-8, -9, and -3 mRNA levels. These findings indicate that cell apoptosis regulation is another significant mechanism of PRU against OS cells MG-63.
Apoptosis is measured as the chief cytotoxic activity by which anti-tumor remedies employ their anti-cancer effects (Donovan & Cotter, 2004). In this study, MG-63 cells added with PRU reduced the mRNA expression levels of Pin-1, Akt, STAT-3, Akt, p38 MAPK, and JNK in an amount-dependent way. MAPKs signaling have been revealed to show an exceptional role in malignant cell propagation and cell death (Wu et al., 2019). It not only averts apoptosis by constraining caspase stimulation (Yang et al., 2004), but also activates the anti-apoptotic factors expression (Konopleva et al., 2012). The signaling of JNK has been demonstrated to be connected to cell death by controlling pro-apoptotic elements expression, and can also stimulate the caspase activity (Su et al., 2013). The role of Pin1 is protein isomerization and swapping the protein possessions. Pin1 also promoted Akt-facilitated GSK-3β deactivation (Kim et al., 2009). Akt protein controls the function of numerous substrates including cell multiplication, cell cycle progression, and cell development (Fresno Vara et al., 2004). Inactivation of Akt signaling triggered cancer cell death. The proteins in the STAT family are present in the cytoplasm and able to translocate in the nucleus and binding to DNA resulting in activation (Banerjee et al., 2017). Among the STAT family members, STAT-3 is prominently linked to tumors and contributes to the progression of signal transduction intricate in several cytokines. Inflammation is an ultimate component that has been associated with tumor invasion, metastasis, and cancer mortality (Singh et al., 2017). NF-κB-p65 is a substantial nuclear transcription factor in cells and triggers apoptosis by directing the cell cycle in the cancer cells (Zhu et al., 2015).
According to Siegel et al. (2023), bone cancer is an untreatable malignant tumor with a high fatality rate. The existing therapeutic approaches, such as surgical resection, radiation, and chemotherapy, have consistently improved recently, although bone cancer survival rates have only marginally increased. The fundamental cause of this is that bone cancer is a very heterogeneous illness, and blocking one pathway results in feedback and the simultaneous activation of numerous interacting and/or redundant pathways. Thus, the need for new treatment targets is urgent. In earlier research, Pin1 was discovered to be highly overexpressed and inversely linked with the development of the disease in bone cancer (Russo Spena et al., 2018). Targeting Pin1 as cancer therapeutic is desirable since it induces the simultaneous collapse of many carcinogenic pathways when Pin1 is inhibited (Luo et al., 2009).
Our findings showed that PRU inhibited Pin1 expression in a dose-dependent manner and that Pin1 inhibitors present a unique strategy to increase PRU therapeutic effectiveness against bone cancer. Our findings indicated that PRU controls the MAPK/STAT-3/NF-κB-p65 pathways, and these outcomes showed the latent mechanism of PRU-facilitated anti-cancer activities on MG-63 cells.
Conclusion
In conclusion, our results demonstrated that PRU subdued proliferation and inflammation, as well as triggered apoptosis over the ROS-facilitated MAPK/STAT3/NF-κB and AKT signaling in MG-63 cells. This may be the prime report to show the latent molecular actions of the anti-cancer properties of PRU on MG-63 cells and according to the model, suppresses Pin1 in bone cancer cells, which limits cancer stem cell characteristics. PRU may be used as a model molecule to create inhibitors that specifically target Pin1, which may be a novel target for bone cancer. Thus, PRU has the possibility to be employed as an anti-cancer agent against OS. Further research has warranted.
Abbreviations
PRU: Prunetin; ROS: Reactive oxygen species; MTT: 3-[4,5-dimethylthiazol-2-yl]-2,5 diphenyl tetrazolium bromide; DCFH-DA: Dichloro-dihydro-fluorescein diacetate; DAPI: 4′,6-diamidino-2-phenylindole; PI: Propidium iodide; AO/EB: Acridine orange/ethidium bromide; Bax: Bcl-2-associated X protein; Bcl-2: B-cell lymphoma 2; MAPK: Mitogen-activated protein kinase; STAT3: Signal transducer and activator of transcription 3; AKT: Protein kinase B; OS: Osteosarcoma; DMSO: Dimethyl sulfoxides; ANOVA: Analysis of variance.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Summary
Prunetin suppressed cell growth of bone cancer cells.
Prunetin induced apoptosis via inhibited MAPK/AKT signaling pathway.
