Abstract
MG132 is a potent, reversible, and cell-permeable 20S proteasome inhibitor and it is derived from a Chinese medicinal plant. The purpose of this study is to investigate the anticancer effects of MG132 against human osteosarcoma U2OS cells. We first performed MTT and colony formation assays to investigate the anti-proliferative effects of MG132. The results demonstrated that MG132 suppressed the proliferation of U2OS cells. Furthermore, we found that treatment with MG132 increased apoptosis and induced DNA damage in U2OS cells. Additionally, zymography, wound healing, and invasion assays showed that MG132 suppressed the enzymatic activity of matrix metalloproteinases, cell migration, and invasion, respectively of U2OS cells. Furthermore, western blotting assay was performed to investigate the apoptotic signaling pathways in MG132-treated U2OS cells. Our results showed that MG132 downregulated the expression of antiapoptotic proteins, including CDK2, CDK4, Bcl-xL, and Bcl-2, whereas it upregulated the expression of proapoptotic proteins, including p21, p27, p53, p-p53 (ser15, ser20, and ser46), cleaved forms of caspase-3, caspase-7, caspase-9, and PARP, and FOXO3 in U2OS cells. These results demonstrated that MG132 activated apoptotic signaling pathways in U2OS cells. Interestingly, MG132 downregulated the phosphorylation of Akt and Erk. Taken together, our results suggest that MG132 has anticancer effects in U2OS cells. Therefore, MG132 may be a potential therapeutic agent for the treatment of osteosarcoma.
Introduction
Osteosarcoma is a malignant bone tumor that occasionally metastasizes to the lungs. 1 It is the most usual type of bone tumor in adolescents and children, that occurs more frequently in males. 2 It is distinguished by high metastatic potential, rapid progression, and dismal clinical prediction. 3 Recently, several modern treatment strategies have been developed for osteosarcoma patients. 4 However, the adverse effects of radio- and chemotherapy cause significant morbidities, and therefore further studies are needed to eradicate these effects. 5 Chemotherapeutic reagents such as flavonoids family and cisplatin be extensively used for healing osteosarcoma patients. Hence, there is need for new and advanced strategies for the treatment of osteosarcoma patients. 6
MG132 is a potent, reversible, and cell-permeable 20S proteasome inhibitor. 7 It is a peptide aldehyde that inhibits the proteolytic activity of the 26S proteasome complex. It is a natural triterpene proteasome inhibitor derived from a Chinese medicinal plant. It was found to inhibit the proliferation of prostate cancer in nude mice. 8 Recently, proteasome inhibitors such as bortezomib, carfilzomib, and oprozomib have attracted much attention as new therapeutic agents for cancer. 9 In recent studies, MG132 was shown to induce anticancer effects and play an indispensable role in apoptotic signaling pathways in several kinds of cancer cells. MG132 induces apoptotic cell death through the ER stress-mediated apoptotic signaling pathway in Jurkat T cells. Furthermore, MG132 activates cisplatin-induced apoptosis in human osteosarcoma U2OS cells. 10
The p53 protein plays an important role in apoptosis, cell cycle progression, and repair of DNA damage. In particular, the role of apoptosis in the efficacy of DNA damaging agent-mediated tumor therapy was first suggested using
FOXO3 is a transcription factor that is generally distinguished by a stark fork head DNA-binding domain. 15 FOXO3 is regulated by a complex network of pathways. 16 It plays a significant role in cell cycle arrest via upregulation of p27. 17 Moreover, FOXO3 is known to induce arrest of cell cycle at the G1 and G2/M phases, and apoptosis in human cancer cells. 18 FOXO3 also acts as a tumor suppressor gene in many other cancer cells. 19 For instance, increase in FOXO3 expression correlates with the progression of apoptosis in human liver cancer HepG2 and SK-Hep1 cells. 20 In addition, FOXO3 is activated during cellular apoptosis in human ovarian cancer SKOV3 cells. 21 In our study, we demonstrated the function of FOXO3 in MG132-mediated apoptosis and focused on the anticancer effect of MG132 in human osteosarcoma U2OS cells. In brief, the purpose of this study was to confirm the anticancer effect of MG132 in human osteosarcoma U2OS cells.
Materials and methods
Reagents
MG132 was purchased from MedChemExpress (Monmouth Junction, NJ, USA) and it is dissolved in DMSO (Sigma-Aldrich, St. Louis, MO, USA). We referred to the structure of MG132 in previous research. 22 A 10 mM MG132 stock solution was prepared and stored at −80°C.
Cell culture
Human osteosarcoma U2OS cell line was purchased from the Korean Cell Line Bank (Seoul, Korea). The cells were incubated under standard conditions (5% CO2, 37°C, and 95% humidity). The cells were cultured in Dulbecco’s modified Eagle’s medium (DMEM; GIBCO, Grand Island, NY, USA) containing 10% heat-inactivated (56°C, 30 min) fetal bovine serum and 1% antibiotics (penicillin/streptomycin). At 80% confluence, the cells were harvested using 0.25% (w/v) trypsin-EDTA (Sigma-Aldrich).
Cell proliferation assay
U2OS cells were plated in 96-well plates (3 × 103 cells per well) and incubated under standard conditions (5% CO2, 37°C, and 95% humidity) overnight to attach. Subsequently, the cells were treated with various concentrations of MG132 (0, 1, 2.5, and 5 μM) and incubated at 37°C. After 24, 48, and 72 h of treatment, the media was suctioned out and replaced with fresh media containing 3-(4,5-dimethylthiazol-2yl)-2,5-diphenyl-2H-tetrazolium bromide (MTT) solution (5 mg/mL), and the cells were incubated 37°C for 1 h 40 min. Next, the supernatants were removed, and the cells were treated with DMSO to forming formazan. The plates were incubated at room temperature (25°C) for 20 min on a shaker (100 rpm). The absorbance level of formazan was measured at 570 nm using a microplate reader.
Colony formation assay
U2OS cells were plated in 6-well plates (1 × 103 cells per well) and incubated at 37°C for 18 h. The cells were then treated with MG132 (0, 1, and 2.5 μM) for 24 h. And then the media was replaced with fresh media containing 10% FBS and U2OS cells were incubated for 15 days, replacing the fresh media containing 10% FBS once every 3 days under standard conditions. The cells were rinsed with PBS twice and fixed using 4% paraformaldehyde for 30 min at room temperature (25°C). Subsequently the cells were rinsed with PBS twice and stained for 30 min with 1% crystal violet (Sigma-Aldrich) solution, and then the colonies were counted using chemi-doc systems.
Wound healing assay
This assay was performed to identify whether MG132 treatment suppressed cancer cell migration and invasion in cells. U2OS cells (2 × 105 cells/well) were plated in 6-well plates and incubated under standard conditions (5% CO2, 37°C, and 95% humidity) until the cells reached 90% confluence. Then, the center of the well was scratched with a P200 pipette tip and the cells were rinsed twice with PBS. The cells were treated with various concentrations of MG132 (0, 2.5, and 5 μM). After 24 and 48 h of treatment, the wound area closure was observed using a fluorescence microscope (CKX53; Olympus, Tokyo, Japan).
Boyden chamber assay
This assay was performed to detect the invasive ability of U2OS cells. Polycarbonate membrane with 8 µm pores (Neuro Probe, USA) was covered with gelatin using solution consisting of 0.1% gelation and 0.1 g/L acetic acid. U2OS cells were harvested using trypsin and resuspended in 0.1% FBS media. Media containing 1% FBS and MG132 (0, 1, and 2.5 μM) was used as chemoattractant (32 μL) in bottom chamber of a 48-well Micro Chemotaxis Chamber (Neuro Probe). Cells were seeded in the upper chamber (5 × cells per well in 50 µL media) and incubated for 24 h. The membrane was fixed with 4% paraformaldehyde for 20 min at 4°C and stained with a 1% crystal violet solution. Invaded cells were counted by a microscope (CKX53; Olympus, Tokyo, Japan).
Gelatin zymography assay
This assay was performed to investigate the activities of the matrix metalloproteinases (MMPs), particularly gelatinases MMP-2, and MMP-9, by gelatin zymography. U2OS cells (1.5 × 106) were plated in 100 mm cell culture dishes and incubated at 37°C overnight to adhere. The cells were treated with various concentrations of MG132 (0, 0.5, and 1 μM) in media containing 0.1% FBS, and incubated for 24 h. Then, the culture media was harvested and concentrated using an Amicon Ultra-15 centrifugal filter (Millipore, Billerica, MA, USA). Bradford assay was performed to detect protein concentration. Cell supernatants were mixed with SDS sample buffer and separated by 8% acrylamide gel containing 1.5% gelatin at 100 V for 3 h. The gel was incubated in 2.5% Triton X-100 buffer at 37°C in a shaking incubator for 30 min to remove SDS. Then, the gel was incubated 37°C in developing buffer (0.5 M Tris-HCl (pH 7.5), 50 mM calcium chloride) for 48 h. Subsequently, the gels were stained with Coomassie staining buffer.
Annexin V/propidium iodide (PI) staining assay
To investigate the rate of apoptosis of the cancer cells following treatment with various concentrations of MG132, this assay was performed. We used a Annexin V, PI apoptosis detection kit (BD Bio-science, Franklin Lakes, NJ, USA) to identify MG132-mediated apoptosis. U2OS cells (2 × 105 cells per well) were plated in 6-well plates and incubated at 37°C overnight to attach. The cells were treated with different concentrations of MG132 (0, 1, 2.5, and 5 μM) for 24 h. The cells were rinsed twice with PBS and resuspended in 1 × binding buffer diluted with PBS. Cells were stained with Annexin V and incubated for 20 min at RT (25°C) in the dark. After incubation, the cells were sorted by FACS (Beckman Coulter Brea, CA, USA).
Terminal deoxynucleotidyl transferase (TdT) deoxyuridine triphosphate (dUTP) nick end labeling (TUNEL) assay
This assay was performed to determine apoptotic cells using TUNEL staining (Promega, Madison, WI, USA). U2OS cells (2 × 105 cells per well) were plated in 6-well plates and incubated at 37°C for 24 h. The cells were treated with various concentrations of MG132 (0, 2.5, 5, and 10 μM) for 24 h. Then, U2OS cells were washed twice with PBS for 10 min each and fixed using 4% paraformaldehyde for 30 min at ice cold. After fixation, the cells were rinsed twice with PBS for 10 min and permeabilized with 1% Triton X-100 for 20 min at room temperature (25°C). Subsequently, the cells were treated with 50 µL rTdT enzyme buffer and incubated for 2 h at 37°C. Next, the nuclei of cells were stained with Hoechst 33342 (2 µL Hoechst 33342 solution was dissolved in 4 mL of PBS; Sigma-Aldrich) for 10 min. Finally, DNA fragmentation was observed using fluorescence microscopy (CKX53; Olympus, Shinjuku, Tokyo, Japan).
Western blotting
U2OS cells (1.5 × 106 cells per dish) were plated in 100 mm culture dishes and incubated 37°C to attach. The media was replaced with new media containing of MG132 (0, 2.5, and 5 μM) and cells were incubated for 24 h. Cell lysates were prepared using RIPA buffer (Sigma-Aldrich) containing PI and phenyl methane sulfonyl fluoride. After centrifugation, the supernatant was transferred to another tube. Proteins were quantified using Bradford assay. The protein samples were resolved by SDS-PAGE at 110 V for 1 h 40 min and transferred to methanol-activated polyvinylidene fluoride membrane at 50 V for 2 h. The membranes were then blocked using 3% skim milk for 1 h at RT (25 °C) and incubated with specific primary antibodies (mouse anti-ß-actin (1:5000 dilution), rabbit anti-p-AKT (1: 1000 dilution), rabbit anti-AKT (1: 1000 dilution), rabbit anti-p-ERK (1: 1000 dilution), rabbit anti-ERK (1: 1000 dilution), rabbit anti-p-JNK (1: 1000 dilution), and rabbit anti-JNK (1: 1000 dilution), rabbit anti-p-p38(1: 1000 dilution), and rabbit anti-p38 (1: 1000 dilution) (Santa Cruz, CA, USA), rabbit anti-p21 (1: 1000 dilution), rabbit anti-p27 (1: 1000 dilution), rabbit anti-p53 (1: 1000 dilution), rabbit anti-p-p53 (Ser15) (1: 1000 dilution) rabbit anti-p-p53 (Ser20) (1: 1000 dilution) rabbit anti-p-p53 (Ser46) (1: 1000 dilution), rabbit anti-MDM2 (1: 1000 dilution), rabbit anti-CDK2 (1: 1000 dilution), rabbit anti-CDK4 (1: 1000 dilution), rabbit anti-FOXO3 (1: 1000 dilution), rabbit anti-Bcl-2 (1: 1000 dilution), rabbit anti-Bcl-xL (1: 1000 dilution), rabbit anti-Bax (1: 1000 dilution), rabbit anti-cleaved caspase-3(1: 1000 dilution), rabbit anti-caspase-3 (1: 1000 dilution), rabbit anti-cleaved caspase-7 (1: 1000 dilution), rabbit anti-caspase-7 (1: 1000 dilution), rabbit anti-cleaved caspase-9 (1: 1000 dilution), rabbit anti-caspase-9 (1: 1000 dilution), rabbit anti-cleaved PARP (1: 1000 dilution), rabbit anti-PARP (1: 1000 dilution) (Cell Signaling, Danvers, MA, USA)) at 4°C overnight. Then the membranes were rinsed with TBS-T three times every 15 min and incubated with secondary antibodies (Santa Cruz). The membranes were rinsed with TBS-T for one more step and chemiluminescence was measured using detection reagents (GE healthcare, Little Chalfont, UK) and Chemi-doc detection system (Bio-rad, Hercules, CA, USA).
Cell cycle analysis
The cells were seeded at a density of 2 × 105 cells in 6-well cell culture dish to attach, and treated with different concentrations of MG132 (0, 1, 2.5, and 5 μM). Following that, cells were harvested using by 0.25% trypsin-EDTA and fixed using cold 70% EtOH for 2 h at -20°C. Subsequently, cells were centrifuged at 1500 rpm for 3 min and incubated with PI (Sigma-Aldrich) (50 μg/mL PI and 200 μg/mL RNase A) for 30 min at room temperature in dark room. The cell cycle distribution data were measured by FACS (Beckman Coulter).
Statistical analysis
The results are expressed as arithmetic mean ± SEM (the standard error of the mean). To compare the statistical meaning between the groups, two-sided unpaired Student’s t-test was used. Experiments were repeated three times. And the representative data were shown. Statistical analysis was conducted using one-way ANOVA, and a p-value of < 0.05 was considered statistically significant.
Results
MG132 inhibited the proliferation in U2OS cells
First, we determined the effect of treatment with different concentrations of MG132 (0, 1, 2.5, and 5 μM) on cell proliferation in U2OS cells treated for 24 h. Cell proliferation decreased significantly at 5 μM concentration of MG132 (Figure 1(b)). We observed a dose-dependent anti-proliferative effect at different concentrations of MG132 in U2OS cells. For this experiment, U2OS cells were treated with 0, 1, 2.5, and 5 μM MG132 for 24, 48, and 72 h, and then analyzed by the MTT assay. As shown in Figure 1(c), MG132 demonstrated an anti-proliferative effect on U2OS cells. We analyzed cell viability and found cell survival rate of nearly 25% at 5 μM concentration of MG132 after 72 h.
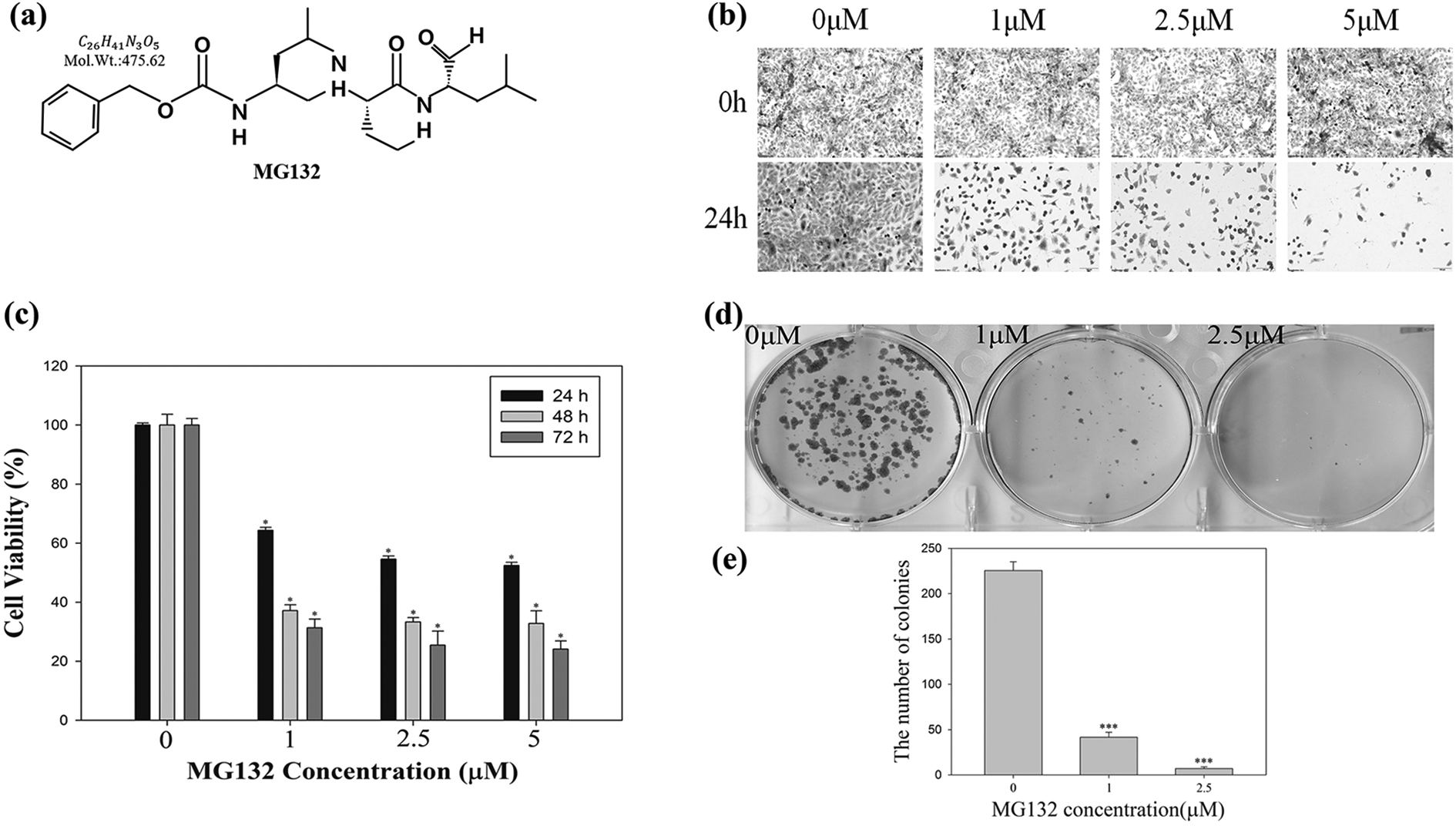
Effects of MG132 on proliferation, and colony formation in human osteosarcoma U2OS cells. (a) MG132 chemical structure (Harhouri et al.). (b) Morphological changes in MG132-treated U2OS cells. (c) Cytotoxicity of MG132 in U2OS cells. Cells were treated to increasing doses of MG132 (0, 1, 2.5, and 5 μM) for 24, 48, and 72 h. Cell viability was determined by MTT assay. (d) Colony formation assay of MG132-treated U2OS cells. Cells were treated with 0, 1, and 2.5 μμ of MG132 for 24 h, allowed to grow into colonies for 14 days, and colonies was calculated. (e) The results were analyzed using the Student’s t-test. *
We also performed colony formation assays to examine cell survival in culture media containing MG132. Treatment of U2OS cells with 0, 1, and 2.5 μM MG132 for 15 days (Figure 1(d)) decreased the number of colonies to about 41.6 and 7, respectively (Figure 1(e)). These results indicate that MG132 suppresses the growth rate and inhibits the proliferation of human osteosarcoma U2OS cells.
MG132 induced sub-G1 cell cycle arrest in U2OS cells
To verify the pattern of cell cycle distribution, a cell cycle arrest analysis was performed. U2OS cells were treated with various concentrations of MG132 (0, 1, 2.5, and 5 μμ) for 24 h and measured using flow cytometry (Figure 2(a)). After 24 h of treatment with 5 μμ of MG132, the cell cycle distribution of cells in the sub-G1 population mounted from 2.5% to 28.4%. As shown in the graph (Figure 2(b)), there was a significant increase in the proportion of cells in the sub-G1 phase compared to the control group. These results demonstrate MG132 induces sub-G1 cell cycle arrest in U2OS cells.
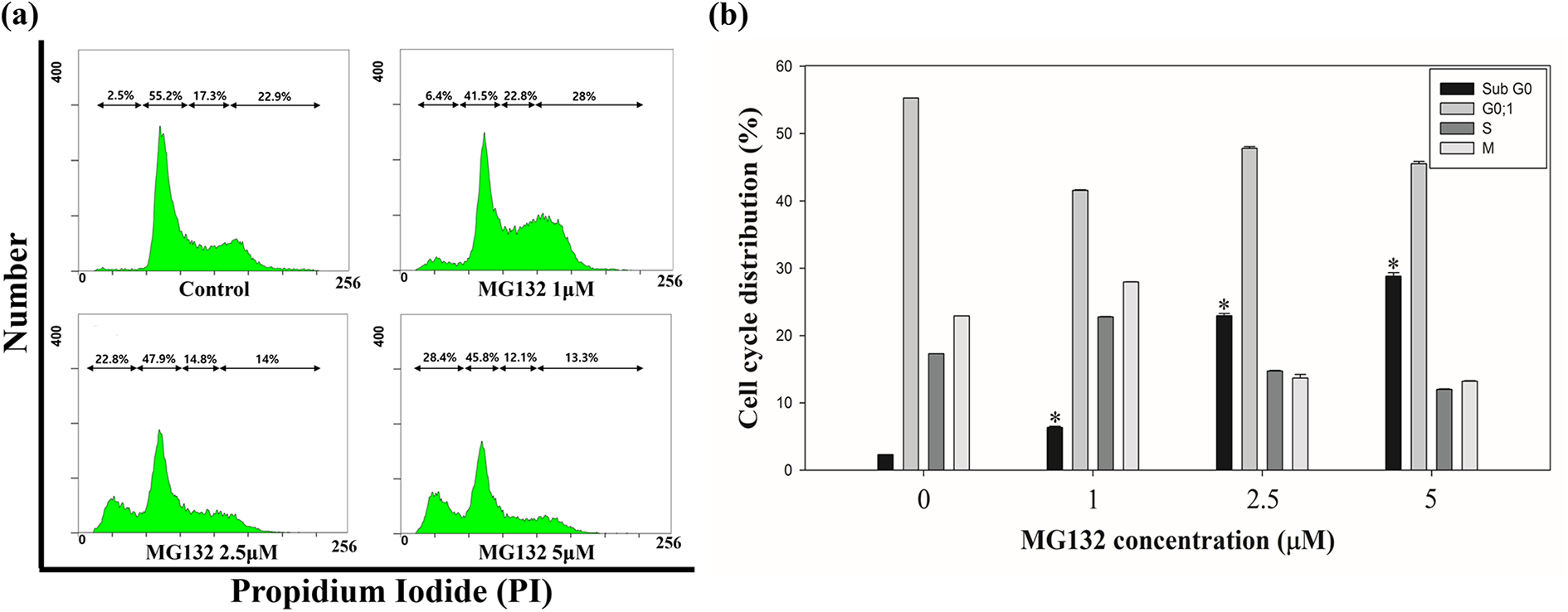
Cell cycle arrest assay of U2OS cells. Effectiveness of MG132 on the cell cycle distributions in osteosarcoma U2OS cells were analyzed by FACS. Cells were treated with 0, 1, 2.5, and 5 μμ of MG132 for 24 h and stained using propidium iodide (PI). Following that, the cells were determined using FACS. (a) Sub-G1 cell cycle arrest of U2OS cells. The part of cells in sub-G1 phase was found to be 2.5% in the control group, and 6.4, 22.8, and 28.4% in U2OS cells treated with 1, 2.5, and 5 μμ MG132, respectively. (b) Distribution of cells of each phase of the cell cycle was assessed following MG132 treatment. Our data was statistically analyzed using the Student’s
MG132 induced apoptotic cell death and DNA fragmentation in U2OS cells
Annexin V/propidium iodide (PI) staining was performed to measure MG132-induced apoptosis in U2OS cells. The viable cell population was 96.8% in the control group whereas it was 68.2%, 44.3%, and 38.6% in groups treated with 0, 1, 2.5, and 5 μM MG132, respectively (Figure 3(a)). In contrast to this, the rate of apoptotic cells percentage was 2.6% in the control group whereas it was 26.9%, 47.5%, and 53.8% in the 1, 2.5, and 5 μM MG132 treated groups, respectively (Figure 3(b)). Thus, we found MG132 induces apoptotic cell death in U2OS cells.
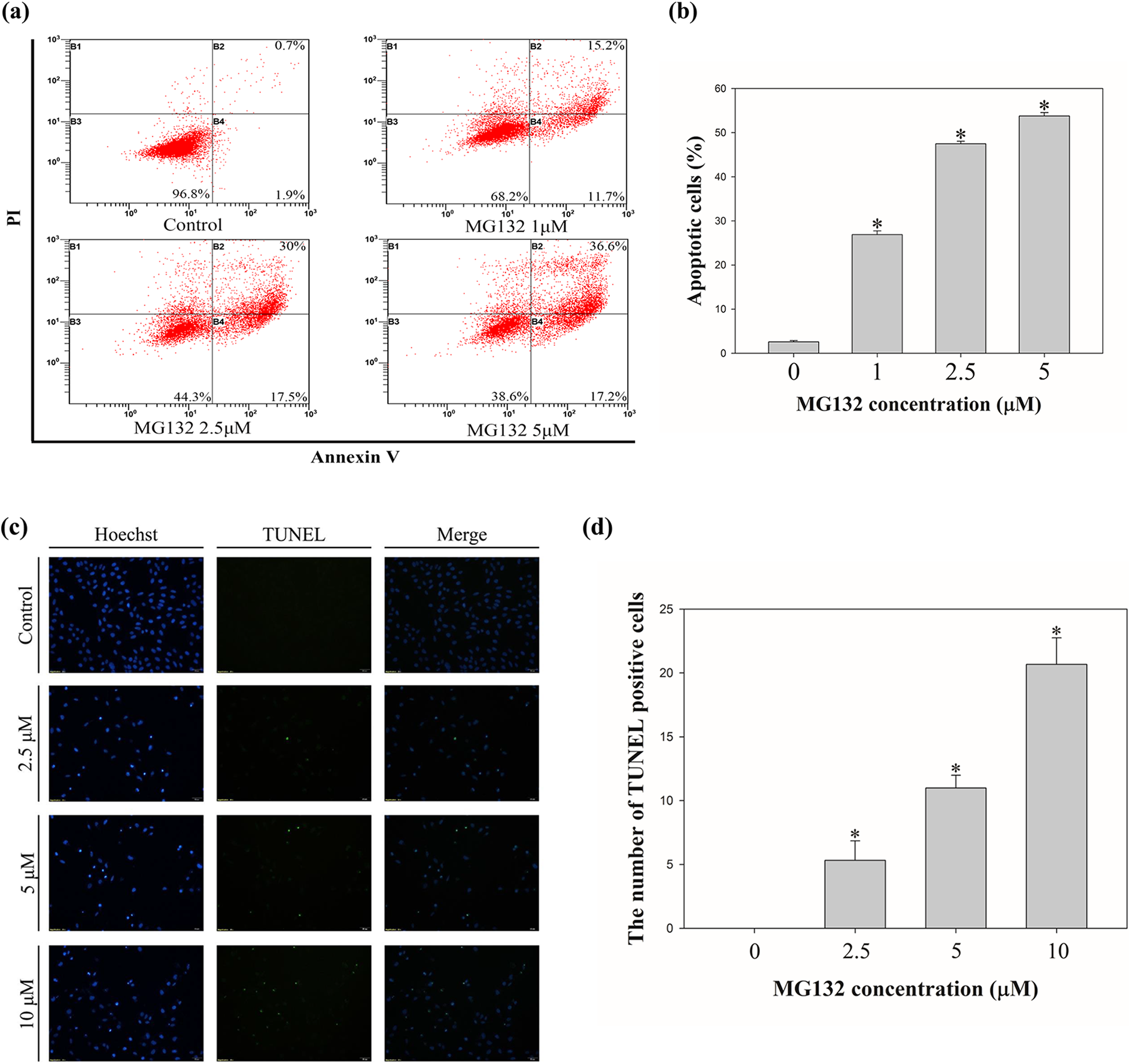
Induction of Apoptosis and DNA fragmentation in MG132-treated U2OS cells. (a) Increase of apoptotic rates in U2OS cells. MG132 induced apoptosis in dose- and time- dependent manner in osteosarcoma U2OS cells. U2OS cells were treated with 0, 1, 2.5, and 5 μμ of MG132 for 24 h. And the cells were analyzed by using FACS. The apoptotic cells rate is evaluated via Annexin V/PI double staining assay. (b) Quantitative analysis of apoptotic cells after treatment of MG132 for 24 h using flow cytometry. *p < 0.05 compared to control group. (c) Measurement of DNA fragmentation against apoptotic cells by TUNEL assay. U2OS cells were treated with 0, 2.5, 5, and 10 μμ MG132 for 24 h and induction of DNA fragmentation was displayed by fluorescence microscopy. Blue fluorescence indicates the nucleus stained using Hoechst 33342 and green fluorescence indicates the fragmented DNA stained by TUNEL. (d) The bar graph indicates that the number of TUNEL-positive cells following MG132 treatments (0, 2.5, 5, and 10 μμ).
Moreover, we performed TUNEL assay to further explored DNA fragmentation in the nuclei of U2OS cells, which is a hallmark of apoptotic cell death. Following incubation of U2OS cells with MG132 (0, 2.5, 5, and 10 μμ) for 24 h, the cells were analyzed using a fluorescence microscope. We detected that the proportion of TUNEL-positive cells was significantly increased in treatment group (Figure 3(c)). And as shown in the graph (Figure 3(b)), there was an upregulation of TUNEL-positive cells. Thus, these results demonstrate that MG132 induces apoptosis and DNA fragmentation in U2OS cells.
MG132 reduced invasion and migration of U2OS cells
We performed scratch assay to determine the migratory ability of U2OS cells following MG132 treatment (Figure 4(a)). In this experiment, cells were treated MG132 (0, 2.5, and 5μM) for 24 and 48 h. U2OS cell migration rate was remarkably decreased after MG132 treatment. Our result shows that the scratched area was covered after 48 h in the control group. The bar graph indicated that the area without cells (wound area) was markedly higher in the MG132 treated group by comparison with that in the control group (Figure 4(b)). These results suggest that MG132 treatment decreased the migration ability of U2OS cells.
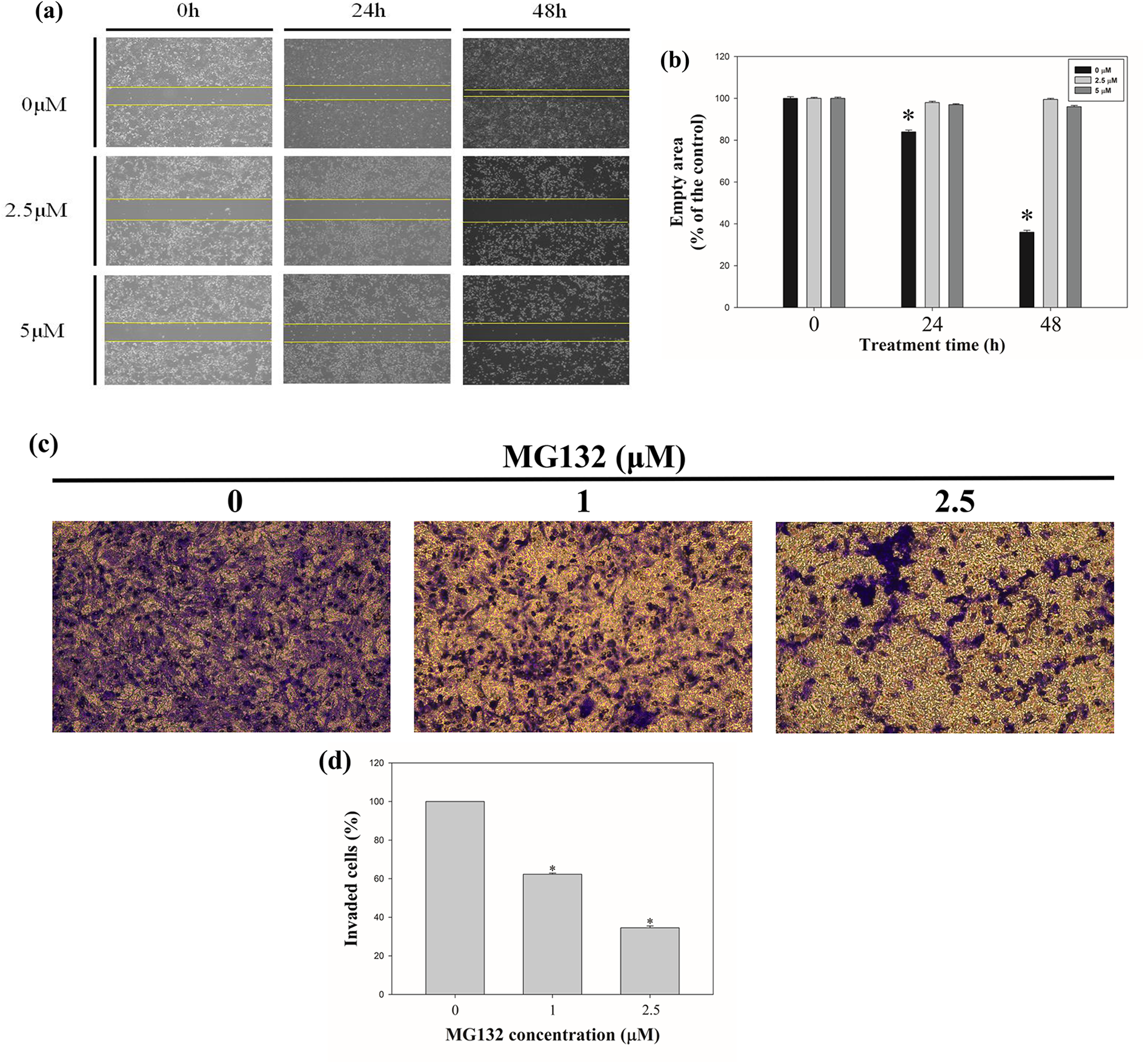
Inhibitory effects of MG132 on the invasion and migration of osteosarcoma U2OS cells. (a) Wound healing ability was assessed following MG132 treatment. And the empty area was measured after 24 and 48 h. (b) Quantitative analysis of the wound area (0, 2.5, and 5 μμ) after 24 and 48 h following treatment with MG132. The graph indicates the area without cells after given incubation time points (0, 24, and 48 h) (c) The invasive ability of U2OS cells was assessed following MG132 treatment. Invasion of U2OS cells treated with various concentrations of MG132 (0, 1, and 2.5 μμ) was detected using Boyden chamber assay. (d) Quantitative analysis of invaded cells after treatment of MG132 (0, 1, and 2.5 μμ). The graph indicates that the percentage of invaded cells were decreased as the MG132 concentration increased.
To determine the effects of MG132 on the invasive ability of U2OS cells, a Boyden chamber assay was performed. We found that the number of invading cells decreased following treatment with MG132 (Figure 4(c)). The bar graph showed that the number of invaded cells was considerably reduced in MG132 treatment group compared to that in the control (Figure 4(d)).
In addition, gelatin zymography assay was performed to measure the gelatinase activity of MMP-2 and MMP-9. The enzymatic activity level of MMP-2 and MMP-9 downregulated after treatment of U2OS cells with MG132 (Figure 5(a)). The expression level of MMP-2 in U2OS cells was considerably suppressed following treatment of MG132 (Figure 5(b)). The expression of MMP-9 also decreased after treatment with MG132 (Figure 5(c)). Our results suggest that migration and invasion ability of U2OS cells decreased following treatment with MG132.

Inhibitory effects of MG132 on the enzymatic activity osteosarcoma U2OS cells. (a) Expression levels of MMP-2 and MMP-9 were downregulated after treatment of MG132 (0, 0.5, and 1 μμ) for 24 h. (b) Downregulation of MMP-2 expression level as MG132 concentration increased. The bar graph shows that the quantified expression levels of MMP-2. (c) Downregulation of MMP-9 expression level as MG132 concentration increased. The bar graph shows that the quantified expression levels of MMP-9.
MG132 activated p53 signaling and caused the accumulation of FOXO3 in U2OS cells
We carried out western blot analysis to demonstrate the apoptotic mechanism by which MG132 induced apoptosis in U2OS cells. Treatment with MG132 altered the expression of mitogen-activated protein kinases (MAPKs). In Figure 6(a), the protein expression levels of Akt, Erk, Jnk, and p38 were maintained with increasing concentration of MG132. However, the levels of p-Akt, p-Erk, and p-p38 decreased as MG132 concentration increased. Furthermore, the level of p-Jnk was upregulated as MG132 concentration increased.
Next, we analyzed the level of proteins related to the cell cycle (Figure 6(b)). As MG132 concentration increased, the expression levels of p21, p27, p53, and p-p53 (ser15, ser20, ser46) were increased. However, the expression of MDM2, CDK2, and CDK4 was decreased.
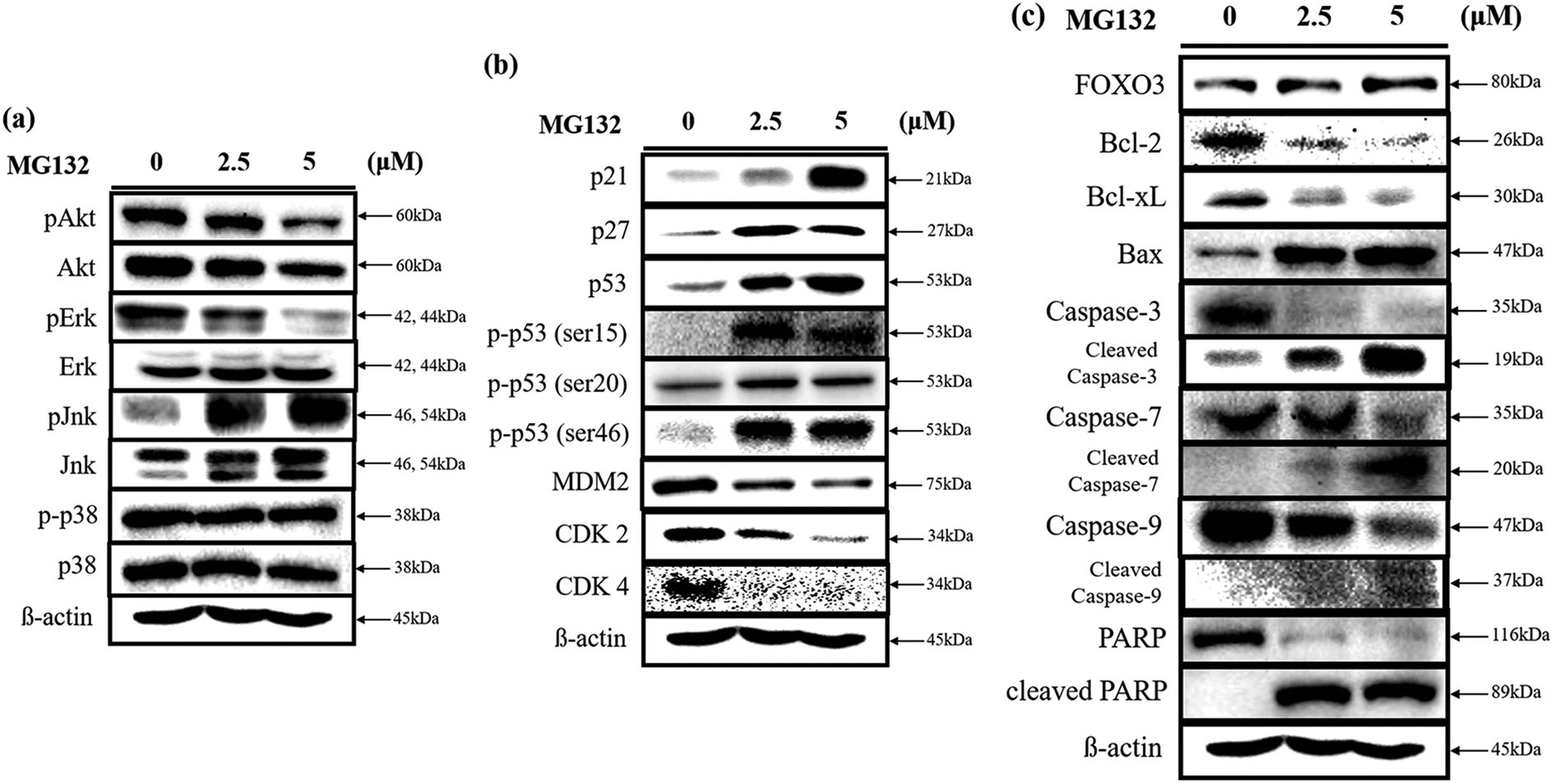
Analysis of various marker proteins in MG132-treated U2OS cells by western blot. MG132 (0, 2.5, and 5 μμ) was treated with U2OS cells. (a) Expression levels of MAP kinase proteins after treatment of MG132. The expression levels of total Akt, Erk, Jnk, and p38 were maintained, whereas that of phosphorylated Akt, Erk, Jnk, and p38 were decreased following MG132 treatment. (b) Expression levels of proteins that related to cell cycle after treatment of MG132. Expression levels of p21, p27, p53, p-p53 (ser15, ser20, and ser46) were increased, whereas that of MDM2, CDK2, and CDK4 were decreased following MG132 treatment. (c) Expression levels of proteins related to apoptosis signaling after treatment of MG132. Expression levels of Bax, FOXO3, cleaved caspase-3, cleaved caspase-7, cleaved caspase-9, and cleaved PARP were increased, whereas that of Bcl-2, Bcl-xL, caspase-3, caspase-7, caspase-9, and PARP were decreased following MG132 treatment. β-actin was used as a loading control.
We also analyzed the expression of proteins related to apoptosis including caspase-3, caspase-7, caspase-9, and poly (ADP-ribose) polymerase (PARP) (Figure 6(c)). The results demonstrated that the expression level of caspase-3, caspase-7, caspase-9, and PARP was remarkably decreased as MG132 concentration increased. In contrast, the cleaved form of these proteins increased substantially as MG132 concentration increased. In addition, the level of anti-apoptotic proteins such as Bcl-xL and Bcl-2 were decreased, whereas the level of the proapoptotic protein such as Bax was increased as the MG132 concentration increased. Furthermore, the expression level of FOXO3 protein increased as the MG132 concentration increased. Consequently, we identified a modern mechanism in which FOXO3 induces apoptosis in osteosarcoma cells in accordance with MG132 treatment via increase of Bax and decrease of Bcl-xL and Bcl-2. Taken together, our results suggest MG132 induces apoptotic cell death in U2OS cells. It also stimulates the p53 signaling pathway and activates the accumulation of FOXO3 protein in U2OS cells.
Discussion
Proteasomes participate in the regulation of proteins that control cell growth, proliferation, and death, and play a pivotal role in maintaining cell homeostasis, making them a potential target for tumor therapy. 23 To date, many proteasome inhibitors have been exploited to cure human cancers. 24 For example, proteasome inhibitor, bortezomib, exhibited discriminative cytotoxicity against tumor and malignant cells and displayed anticancer effects in a few pretreated patients. 25
MG132 is derived from a Chinese medicinal herb. 26 It is a vigorous proteasome and calpain suppressor, and a strong antitumor drug. MG132 was shown to have anticancer effects in human thyroid cancer BCPAP, IHH4, K1, and C643 cells by modulating the activation of transcription factor FOXO3a. 25 MG132 induced therapeutic effects and inhibited tumor progression in breast cancer MCF-7 cells by inactivating the nuclear factor (NF)-κB signaling pathway. 27 We investigated the overall anticancer effects of MG132, including inhibition of migration and apoptotic cell death in human osteosarcoma U2OS cells.
p53 is well-known tumor suppressor protein involved in several molecular mechanisms in the cell. 28 It plays a crucial role in mediating apoptosis induced by DNA tumor virus oncoproteins that promote apoptosis through numerous pathways. 8 Under normal conditions, p53 protein level is not high because it is controlled via negative feedback by MDM2, an E3 ubiquitin protein ligase that intercedes the downregulation of p53 through proteasomes. Cellular stress inhibits the binding of p53 and MDM2 by inhibiting the phosphorylation by p53 and MDM2 proteins, and causes p53 acetylation, leading to p53 accumulation and activation. 29 In this study, MDM2 decreased as the MG132 concentration increased. p53 activates the transcription of many genes that induce apoptosis, such as CD95/Fas, Bax, Puma, and caspase enzymes. 30 Furthermore, p53 interferes with the transcription of genes that inhibit cell death, such as Bcl-2 or directly binds to Bcl-2 or Bcl-xL to increase the permeability of the mitochondrial outer membrane, thereby activating the release of cytochrome c. 31 In this study, the proapoptotic protein, Bax, increased, whereas the antiapoptotic proteins, Bcl-2 and Bcl-xL, decreased with increasing concentration of MG132.
p53 also plays an important role in the cell cycle. When DNA damage is induced in the cell, p53 expression levels rapidly increase. As a result, the expression of p21Waf1/cip1, one of the target genes of p53, is activated. 32 In this study, the expression levels of p21 and p27 proteins increased as MG132 concentration increased. Cyclin-dependent kinases (CDKs), such as cdk2 and cdk4, also decreased as the MG132 concentration increased. These results indicate that MG132 may induce cell cycle arrest. The expression of p53 is upregulated by chemotherapy in many cancer cells. 8 For example, in human liver cancer HepG2 cells, the expression of p53 is upregulated as the dose of Broussochalcone A increases (20). In addition, the expression of p53 is enhanced as the concentration of esculetin increases in human colon cancer LoVo cells. 33 In another study, the expression of p53 in human cervical cancer HeLa cells was found to increase following treatment with Ursolic acid. 34 In this study, we found that the expression of p53 is upregulated in a concentration-dependent manner in human osteosarcoma U2OS cells following treatment of the cells with the proteasome inhibitor, MG132. We also found that phosphorylation of p53 such as ser15, ser20, and ser46, was increased by MG132 treatment in a dose-dependent manner. These proteins are related to apoptosis and cell cycle arrest in cancer cells.
MAPKs mediate several physiological progresses. Erks play a crucial role in cellular separation. 35 Erk activates extrinsic and intrinsic apoptotic pathways by distributing cytochrome c from the mitochondria into the cytoplasm or by activating caspase-8. 36 Nevertheless, no remarkable changes in Erk expression level were observed in the MG132-treated U2OS cells. C-Jun amino-terminal kinases (Jnks) and p38 proteins are also important mediators of the cell proliferation, migration, and differentiation. Jnk protein plays a crucial role in mediating mitochondrial apoptosis proteins such as Bax and Bid. 37 Jnks activate apoptotic signaling pathways by increasing proapoptotic proteins. 38 However, it also presents anti-apoptotic functions relying on the cell type and activation of another signaling pathways. 39 We analyzed p38 and Jnk MAPK expression in MG132-treated U2OS cells. The results showed phosphorated p38 MAPK expression were reduced in MG132-treated U2OS cells. Interestingly, p-Jnk expression was increased in MG132-treated U2OS cells. Furthermore, regulation of the Akt/Bad pathway was found to induce mitochondria-mediated apoptotic cell death in human liver cancer HepG2 cells. 40
Moreover, caspase-3 has an important role in cell morphology changes and fragmentation of DNA related to apoptotic cell death. 41 Caspase-7 and caspase-9 play a crucial function in apoptosis. Activation of caspase-7 produces morphological changes, including exposure to phosphatidylserine (PS) and DNA fragmentation. 42 Activation of caspase-3 and caspase-7 caused cell death in monocytic THP-1 cells. Vitalization of PARP induced programmed cell death in C6 glioma cells.43,44 Caspase-9 induces apoptosis via activation of caspase-3 in human cervical cancer cells, and Apaf-1 apoptosome mediates activation of caspase-9.45,46 We demonstrated that treatment with MG132 increased cleaved caspase-3, caspase-7, and caspase-9 levels in U2OS cells. Our results show MG132 induce caspase-3-, caspase-7-, and caspase-9-mediated apoptotic effects in U2OS cells. Poly (ADP-ribose) polymerase (PARP) is a crucial factor in genome stability, and caspase-mediated cleaved PARP is a biochemi-hallmark of programmed cell death. 47 In this study, that treatment of MG132 induced the cleavage of PARP in U2OS cells. Thus, these results indicated that MG132 may induce PARP-mediated apoptotic effects in U2OS cells.
FOXO3 plays a crucial role in cancer cells. FOXO3 is a transcriptional factor that is classified by specific fork head DNA-binding domain. The translocation of FOXO3 into the nucleus induces FOXO3-mediated cell death. 48 It is involved in many signaling pathways and plays an important role in many physiological processes. 49 Because FOXO3 is known for playing a crucial role in inducing tumor repression via arrest of cell cycle and apoptosis in many tumor cells, we verified that the cells treated with MG132 would go through apoptosis through the up-regulation of FOXO3 protein. 50 It has been reported that casticin induces apoptotic cell death in human hepatocellular carcinoma through the FOXO3a activation. 51 In this study, we demonstrated that FOXO3 levels in human osteosarcoma U2OS cells increased after treatment with MG132 in a dose-dependent manner. MG132 is also involved in the Akt/FOXO3 signaling pathway. In our study, we investigated Akt protein expression levels remained constant. However, pS473 Akt phosphorylation decreased protein expression. Therefore, our results indicated that anticancer effects of MG132 treatment promote the induction of FOXO3-mediated apoptosis signaling pathway. And its application may lead to an innovative therapeutic reagent for osteosarcoma (Figure 7). Taken together, MG132 induces apoptosis and inhibits migration of human osteosarcoma U2OS cells.

Schematic representation of MG132-induced Akt/Foxo3 signaling pathway. This diagram shows that MG132 inhibits MDM2 protein levels and upregulates p53 protein levels. In addition, MG132 treatment increases the expression level of proapoptotic protein such as Bax and decreases the expression levels of antiapoptotic proteins such as Bcl-xL and Bcl-2. As a result of this Akt/FOXO3 signaling pathway, MG132 induces apoptotic cell death and cell cycle arrest.
Footnotes
Author contributions
H.K.L. designed the experimental materials and methods, performed all assay, and wrote the manuscripts. S.H.P. served critical trouble shooting of experiments and contributed to the interpretation of the results. And M.J.N. contributed to writing the final manuscripts.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
