Abstract
Objectives
Studies have shown that Momordin Ic (MI) has an antitumor effect on liver cancer, gastric cancer, and colorectal cancer. However, its effect on osteosarcoma has not yet been reported. The purpose of this study was to explore the effect of MI on several malignant phenotypes of osteosarcoma cells.
Methods
CCK-8 and EdU were used to evaluate the effect of MI on the proliferation of osteosarcoma cells. Apoptosis and cell cycle were observed by flow cytometry. Monodansycadaverine (MDC) staining was used to detect autophagy. Western blot was used to evaluate apoptosis, cell cycle, autophagy, and ferroptosis. Evaluation of osteosarcoma cell migration ability by wound healing assay. Colony formation assay was used to test the ability of cloning. Transwell invasion assay was used to detect the invasion level of osteosarcoma cells.
Results
The results showed that MI significantly inhibited the proliferative activity of osteosarcoma cells. Z-VAD-FMK, 3-MA, and Fer-1 were administered separately, and the results showed that except for Fer-1, the other two inhibitors could reverse cell activity to different degrees. Flow cytometry showed that MI induced G0/1 cell cycle arrest and increased apoptosis. MDC showed that MI induced autophagy in osteosarcoma cells. Western blot showed that autophagy and apoptosis proteins were significantly higher in MI group. Transwell invasion assay, wound healing assay, and colony formation assay confirmed that MI could inhibit the invasion, migration, and cloning ability of osteosarcoma cells.
Conclusion
In conclusion, our study confirmed that MI may exert antitumor effects by inducing apoptosis and autophagy in osteosarcoma cells.
Introduction
Osteosarcoma is the most common malignant bone tumor and originates from mesenchymal tissue; it often occurs in long bones, especially in the distal femur, proximal tibia, and proximal humerus metaphysis. 1 The 5-year overall survival rate of osteosarcoma patients remains stable at 60%-75%. Among newly diagnosed osteosarcoma patients, metastasis occurs in 10%-15% of patients, and lung metastasis is the most common metastasis among metastatic osteosarcoma patients, followed by bone metastasis. 2 Osteosarcoma with distant or skip metastasis in patients with a 5-year overall survival rate decreases by approximately 40%, predicting catastrophic results. 3 At present, the treatment of osteosarcoma mainly includes surgery, chemotherapy, and molecular targeted therapy. 4 However, the side effects and therapeutic effects of these agents are unsatisfactory, so new intervention drugs are urgently needed to improve the treatment efficacy.
Momordin Ic (MI), a natural small-molecule product, is the main active ingredient in saponins; it has a molecular weight of 764.94 and a chemical formula of C41H64O13 and is derived from the Chenopodiaceae plant Kochia. According to previous reports, MI has a variety of pharmacological activities, including anti-inflammatory effects, anti-rheumatoid effects, and blood sugar reduction, additionally, anti-pruritus.5–9 However, to date, there have been few reports on the antitumor activity of MI. In recent years, several scholars have begun to explore the antitumor effect of MI in liver cancer,10,11 prostate cancer, 12 cholangiocarcinoma, 13 colon cancer, 14 ovarian cancer, 15 and breast cancer, 16 and the results showed that this effect was significant. However, thus far, this phenomenon has not been studied in osteosarcoma.
Apoptosis, as a form of programed cell death, is a normal physiological process that mainly involves caspases, which are highly important for maintaining normal physiological activity. 17 Various lesions, such as neurodegeneration and autoimmune diseases, are common in apoptotic disorders. 18 Previous studies have shown that the occurrence of osteosarcoma is closely related to the inhibition of the apoptotic pathway. 19 The induction of apoptosis could be used as a new strategy for treating tumors.
Autophagy is a highly conserved metabolic process with a cell protection function. Autophagy is closely related to infection, immunity, and inflammation. Many studies have shown that autophagy is involved in the occurrence and development of cancer. Autophagy can be used as a tumor suppressor or tumor promoter to affect the progression of tumors. 20 The role of autophagy in different types of tumors has been widely studied, 21 and osteosarcoma proliferation, metastasis, chemotherapy, radiotherapy, and immunotherapy are also closely related. 22 In addition, relevant studies have shown that many apoptosis-inducing drugs often induce the activation of the autophagy pathway. For example, apoptosis and autophagy are upregulated at the same time in the treatment of breast cancer and colon cancer with ceramide, 23 and arsenic trioxide in the treatment of acute promyelocytic leukemia in a clinical trial also showed that both are activated at the same time. 24 In summary, autophagy and apoptosis work together and complement each other in combination in response to certain drugs, leading to cell death.
In this study, we found for the first time that MI could effectively inhibit the proliferation, migration, and colony formation of HOS and 143B osteosarcoma cells through apoptotic and autophagic pathways, which was consistent with the findings of relevant studies reporting that MIC has an antitumor effect but was not found to be related to the ferroptosis pathway. This study has enriched the potential drug development value of MI and provided new ideas for the development of new drugs for osteosarcoma in the future.
Materials and Methods
Reagents and Chemicals
MI (purity 99.71%) was purchased from MCE Company (Shanghai, China). DMEM and fetal bovine serum (FBS) were obtained from VivaCell (Shanghai, China). Penicillin streptomycin and trypsin were purchased from Invitrogen (USA). Z-VAD-FMK (Beyotime Biotechnology, Shanghai, China), 3-MA (MCE, Shanghai, China), and Fer-1 (MCE, Shanghai, China) were purchased. The following were used: CCK-8 kit (KeyGEN BioTECH, Nanjing, China); EdU proliferation detection kit (Servicebio, Wuhan, China); Cell Cycle Detection Kit (KeyGEN BioTECH, Nanjing, China); Annexin V-FITC/PI cell apoptosis detection kit (KeyGEN BioTECH, Nanjing, China); monodansycadaverine (MDC) staining kit (Beyotime, Shanghai, China), LC3B (Sangon Biotech, Shanghai, China); Bax (Cell Signaling Technology, USA); Bcl2 (Cell Signaling Technology, USA); PTGS2 (OriGene Technologies, Wuxi, China); Cyclin E (Cell Signaling Technology, USA); internal reference selection, GAPDH (Beyotime Biotechnology, Shanghai, China); secondary antibody selection, horseradish peroxidase-labeled goat anti-rabbit IgG (ZSGB-BIO, Beijing, China); and matrix gel (CORNING Company, USA).
Cell Culture
The human osteosarcoma cell line (HOS, 143B) was purchased from Procell (Wuhan, China), and the human osteoblast hFOB1.19 was obtained from Shanghai FuHeng Biology Co., Ltd The cells were cultured in DMEM containing 10% FBS (GIBCO), 100 U/mL penicillin, and 100 mg/mL streptomycin (Thermo Fisher Scientific, USA) at 37 °C and 5% CO2.
Cell Viability Assay
Cell viability was assessed by CCK-8. According to the instructions of CCK-8, after the cells were plated at the same density for 12 h, DMSO or MI was added for 8 h, and 10 μL of CCK-8 solution was added and incubated in the incubator for 2 h. The absorbance was detected at a wavelength of 450 nm by ELISA.
EdU Proliferation Assay
The cells were uniformly plated in 24-well plates at a certain density, and after the cells adhered to the wall, 20 μmol/L MI (IC50) was applied; 20 μM 2×EdU was prepared and placed in the incubator for preheating. In the semiliquid exchange mode, half of the primary cell culture medium on the culture plate was removed, and an equal volume of preheated 2×EdU incubation working solution was added for incubation for 4 h. Subsequently, 4% paraformaldehyde was used for fixation for 20 min, 0.4% Triton 100X was used for permeation for 15 min, and PBS was used three times for 5 min each. The prepared click reaction solution was added and incubated for 30 min in the dark at room temperature. After the cells were washed, Hoechst 33342 staining solution was added and incubated for 5 min again for fluorescence imaging and analysis of the proportion of proliferating cells.
Cell Colony Formation Assay
Single-cell suspensions were generated with complete medium, and the cells were inoculated in 6-well plates at a density of 1000 cells/well. The experimental group was treated with MI for 2 weeks, and the medium was changed every 4 days. After 2 weeks, the cells were washed twice with PBS, fixed in 4% paraformaldehyde, and stained with 0.5% crystal violet for 30 min. After gentle washing with PBS, images were captured for quantification.
Transwell Invasion Assay
The FBS was removed from the serum in DMEM 48 h in advance. The matrix gel was thawed, diluted at a ratio of 1:8, spread in the upper chamber, and gelatinized with 100 µL of serum-free medium. A cell suspension with a density of 2.5 × 105 cells/ml was prepared. Two hundred microliters of cell suspension (from which the experimental group was treated with MI in advance) was added to the Transwell chamber for culture for 24 h, after which the chamber was removed and gently dried. The cell suspension was fixed with 4% paraformaldehyde for 20 min, stained with 0.1% crystal violet for 10 min, air-dried naturally, and photographed for counting.
Wound Healing Assay
Wound healing assay was used to evaluate cell migration ability. Lines were drawn on the back of the 6-well plate in advance for positioning. The cells were inoculated into 6-well plates, and when the plates were filled, linear scratches were made in the cell monolayers using a 200 μL pipette tip. The experimental group was subjected to MI treatment, and the intersection position of the scratch and the line was clarified. Images were taken with an inverted phase difference optical microscope. At 0 h, 24 h and 48 h, the photos were taken at the corresponding marked positions. ImageJ software was used to determine the area of migration area.
Western Blot
Western blotting was conducted by adding 10 μL of phosphate inhibitor, 1 μL of protease inhibitor, and 5 μL of 100 mM PMSF to 1 mL of cold lysis buffer and mixing evenly for later use. After the cells were centrifuged and washed, 100 μL of lysate was added, and the mixture was placed on a shaking table at 4 °C for 30 s of vigorous shaking. After the cells were allowed to stand on ice for 4 min, this process was repeated five times. Finally, the cells were lysed by an ultrasonic shaker. Then, the samples were centrifuged at 12 000 rpm at 4 °C for 15 min. The supernatant was collected. The protein concentration was then detected by the BCA method according to the manufacturer's instructions. The same amount of protein extract was subjected to sodium lauryl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) (KeyGEN BioTECH, Nanjing, China) and transferred to PVDF membranes. The imprinted membrane was blocked with 5% skim milk at room temperature for 1 h, incubated with the primary antibody overnight at 4 °C and then incubated with the secondary antibody at room temperature for 1 h. Finally, immunoreactivity was detected via exposure to an ECL chemiluminescent kit (KeyGEN BioTECH, Nanjing, China).
Flow Cytometry Analysis
Cells (5 × 105 cells/well) were seeded in 6-well plates and treated with or without MI for 24 h. The cells were then harvested, washed, and placed at 4 °C overnight. The cells were then centrifuged, washed with cold PBS, and recentrifuged. The cells were then resuspended in 250 μL of PBS and stained with 10 mL of PI (1 mg/mL) and 10 mL of RNase A (10 mg/mL) in the dark for 30 min at room temperature. DNA was observed using flow cytometry (BD Biosciences, San Jose, USA) to calculate the percentage of cells in G0/G1, S, or G2/M.
The percentage of apoptotic cells was determined via flow cytometry using the Annexin V-FITC/PI Apoptosis Detection Kit. The cells were stained according to the manufacturer's instructions, and flow cytometry was performed.
Monodansycadaverine (MDC) Staining
MDC staining is used as a tracer of autophagy vesicles for autophagy detection. After MI(10 μmol/L) intervention, 1 ml MDC was added to the experimental hole and incubated in the dark at 37°C for 30 min. Then wash it with detection buffer for three times. Fix with 4% paraformaldehyde for 15 min, add DAPI, incubate for 5 min, and take photos under confocal microscope.
Statistical Analysis
The data are presented as the mean ± standard error (SEM) of at least three independent experiments. Differences between two groups were assessed using a two-tailed unpaired Student's t test. P < .05 was considered to indicate statistical significance. GraphPad Prism software was used for all the statistical analyses.
Results
Momordin Ic (MI) Inhibits the Proliferation of 143B and HOS Cells in Vitro
After intervention with 50 μmol/L MI for 24 h, optical microscopy showed that, compared with those in the DMSO group, more than 90% of the cells in the 143B and HOS MI groups had reduced activity and contraction, which changed from the adherent state to the suspended state, and many cell debris were observed. In the hFOB1.19 MI group, the number of adherent cells was reduced, and some cell debris was observed in the field of view. However, compared with that of the osteosarcoma cell line, the killing effect of MI on osteoblasts was significantly milder (Figure 1B and C). To determine the IC50 of MI on 143B and HOS cells, eight concentrations of DMSO, 1 μmol/L, 5 μmol/L, 10 μmol/L, 20 μmol/L, 30 μmol/L, 40 μmol/L, and 50 μmol/L were tested in this study. After 24 h of intervention, CCK-8 showed that OD of 143B and HOS cells were 50% of that of DMSO at 22.79 μmol/L and 19.48 μmol/L MI, respectively (Figure 1D). Owing to the similar IC50 values of the two cell lines, for the convenience of the experiment, 20 μmol/L MI was used for subsequent proliferation experiments. EdU proliferation assays revealed that the relative proportion of proliferating cells in the MI group was significantly lower than that in the DMSO group for both 143B and HOS cells after 20 μmol/L MI intervention (Figure 2A and B). The CCK-8 results showed that after 24 h of cell plating, the proliferation of the two kinds of cells gradually decreased, and the inhibition rate reached 50% after 36 h (20 μmol/L MI intervention for 24 h); Moreover, the inhibitory effect tended to stabilize after 48 h. (Figure 2C).

Momordin Ic (MI) inhibits osteosarcoma cells. (A) Chemical structure of MI. (B, C) Observation and quantitative analysis of the effect of MI on the proliferation of the osteosarcoma cell lines 143B and HOS and the human osteoblast line hFOB1.19 by optical microscopy. MI intervention concentration: 50 μmol/L; Intervention time: 24 h; (D) IC50 of MI in 143B and HOS cells. The data (n > 3) are presented as the means ± SEMs. The level of significance is indicated by ****P < 0.0001 compared with the absence of MI. MI, Momordin Ic.

MI affected the proliferation of 143B and HOS cells in a time-dependent manner. (A, B) EdU proliferation assay double staining and quantitative analysis. The inhibitory effects of MI on the proliferation of 143B and HOS cells were observed under a fluorescence microscope. The nuclei and duplicated DNA were stained with Hoechst and EdU-594, respectively. The MI concentration was 20 μmol/L, and the intervention time was 24 h. (C) MI inhibited 143B and HOS cells (CCK-8) in a time-dependent manner. The MI concentration was 20 μmol/L; the data (n > 3) are presented as the means ± SEMs. The level of significance is indicated by ns, ****P <0.0001 compared with the absence of MI. MI, Momordin Ic.
Proliferative Ability of Z-VAD-FMK and 3-MA to Reverse Partial MI Suppression
After pretreatment with 20 μmol/L MI in advance and the addition of 10 μmol/L Z-VAD-FMK (apoptosis inhibitor), 10 μmol/L 3-MA (autophagy inhibitor), or 10 μmol/L Fer-1 (ferroptosis inhibitor) for 24 h, the results showed that Z-VAD-FMK and 3-MA could partially rescue 143B and HOS cells, but Fer-1 had no significant effect, suggesting that MI might not induce ferroptosis but could kill tumor cells by inducing apoptosis and autophagy at the same time (Figure 3A-D). This conclusion was further validated by CCK-8 (
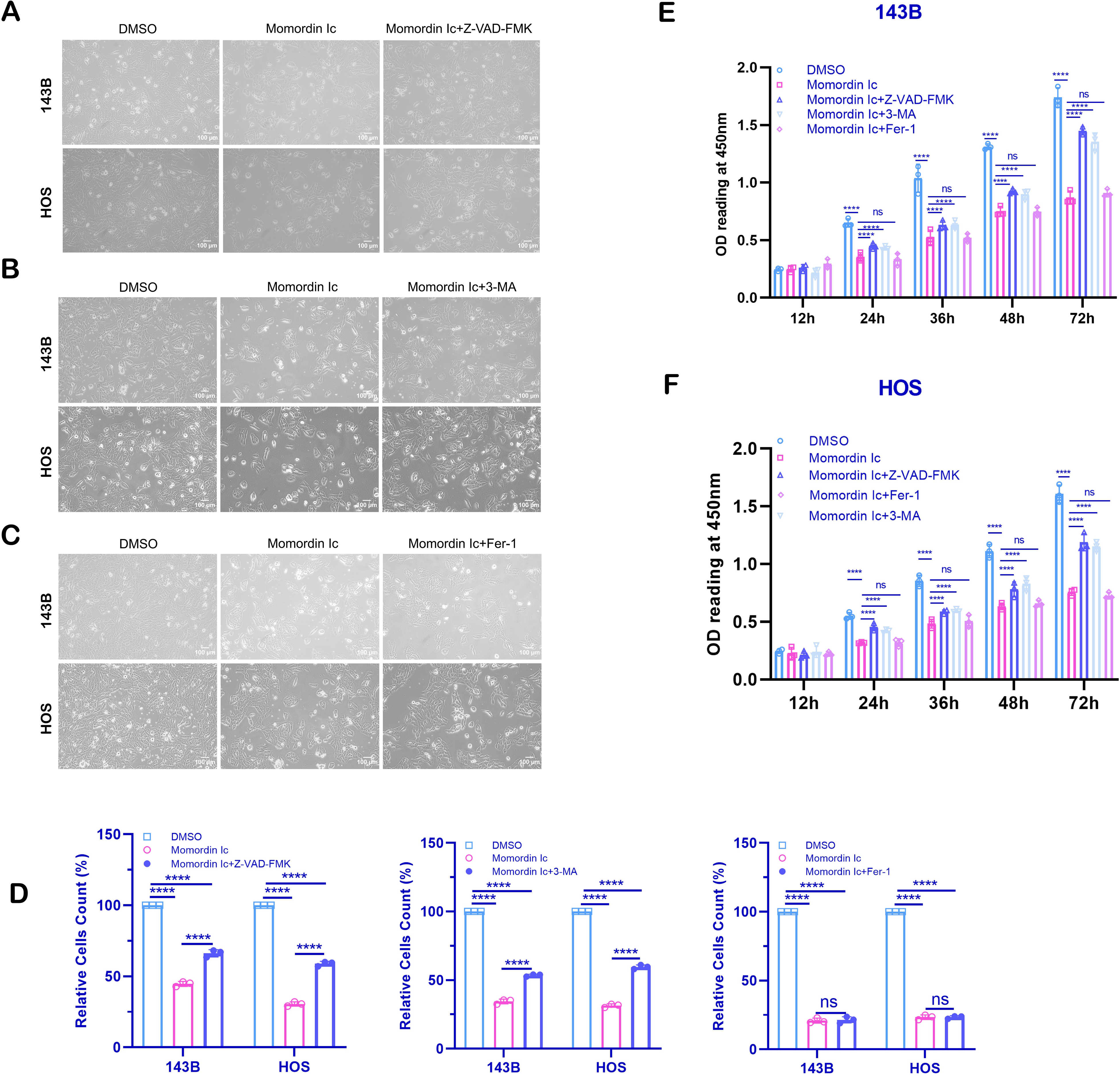
Z-VAD-FMK and 3-MA reversible partial MI killing of 143B and HOS cells. (A-D) The resistance of Z-VAD-FMK, 3-MA, and Fer-1 to MI inhibition was evaluated by optical microscopy, and quantitative analysis was also conducted. (E, F) CCK-8 was used to evaluate the resistance of Z-VAD-FMK, 3-MA, and Fer-1 cells to MI inhibition. The data (n > 3) are presented as the means ± SEMs. The level of significance is indicated by ns, ****P < 0.0001 compared with the absence of MI. MI (20 μmol/L, 24 h), Z-VAD-FMK (10 μmol/L), 3-MA, and Fer-1 (10 μmol/L). MI, Momordin Ic.
MI may Induce 143B and HOS Cell Cycle Arrest, Apoptosis, and Autophagy Instead of Ferroptosis
To test this possibility, we analyzed the cell cycle distribution and apoptosis via flow cytometry. The flow cytometry results showed that the GO/G1 ratios in the DMSO group of 143B and HOS cells were 32.32% and 31.02%, respectively, while those in the MI group were 39.35% and 39.50%, respectively. There was a significant difference, suggesting that MI mainly induced GO/G1 block (Figure 4A and B, E). The apoptotic assay results revealed that the apoptotic rates of 143B and HOS cells were 13.45%/3.34% and 12.64%/1.96%, respectively. Compared with that in the DMSO group, the apoptotic rate in the MI intervention group was significantly greater, and the difference was greater during early apoptosis (Figure 4C and D, F and G). Subsequently, in order to explore whether MI induced autophagy, we performed MDC staining. The results showed that MI treatment could increase the number of MDC-labeled vacuoles compared with DMSO treatment (Figure 4H

MI may induce cell cycle arrest and apoptosis in 143B and HOS cells. (A, B) Annexin V-FITC/PI double staining was performed on 143B and HOS cells by flow cytometry. (C, D) 143B and HOS cell RNase A/PI cell cycle detection was performed by flow cytometry. (E) Quantitative analysis of apoptosis. (F, G) Quantitative analysis of the cell cycle distribution. (H) The autophagy of osteosarcoma cells induced by MI was observed by MDC staining. The data (n > 3) are presented as the means ± SEMs. The level of significance is indicated by **P <0.01, ***P <0.001, and ****P <0.0001 compared with the absence of MI. MI intervention concentration: 20 μmol/L; intervention time: 24 h. MI, Momordin Ic; MDC, monodansycadaverine.
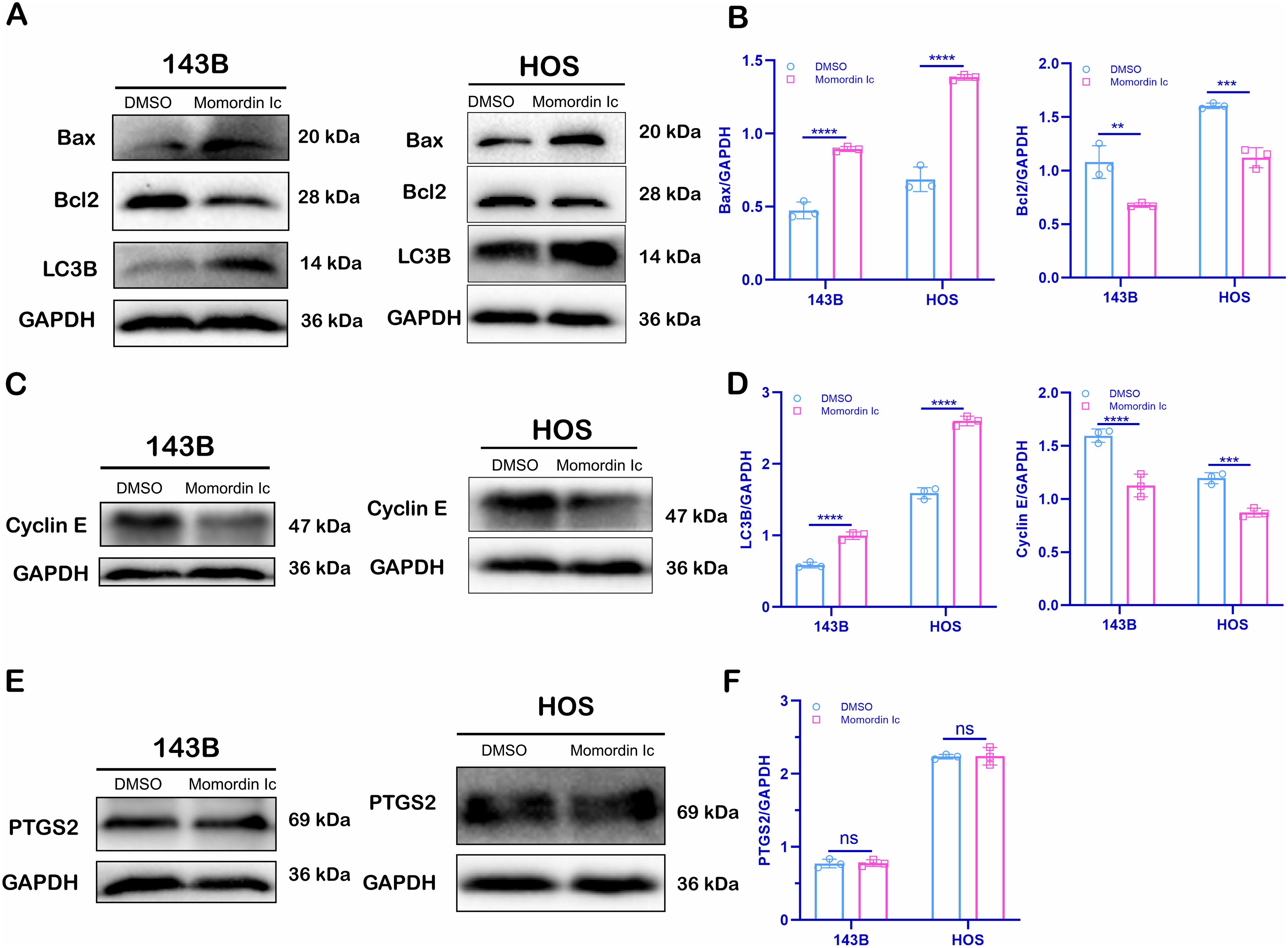
MI might not induce ferroptosis in 143B or HOS cells. (A, B) The expression of the apoptotic marker Bax/Bcl2 and the autophagy marker LC3B after 24 h of treatment with MI was detected by Western blot. (C, D) The expression of Cyclin E, a cell cycle marker protein, was detected by Western blot after 24 h of treatment with MI. (E, F) The expression of the ferroptosis-related protein PTGS2 was detected by Western blot after 24 h of treatment with MI. The data (n > 3) are presented as the means ± SEMs. The level of significance is indicated by ns, *P <0.05, **P <0.01 compared with the absence of MI. MI intervention concentration: 20 μmol/L. MI, Momordin Ic.
MI Inhibited Osteosarcoma Colony Formation Ability, Migration, and Invasion Ability in Vitro
In addition, we further explored the effects of MI on the colony formation, migration, and invasion ability of osteosarcoma cells. The results of wound healing assay showed that there was no significant difference between the 143B cell groups after 24 h of MI intervention, but a significant difference was observed between the two groups after 48 h (P <0.0001). In the case of HOS cells, significant differences were shown after 24 h of MI intervention (P <0.001). The above results indicated that MI could significantly inhibit the migration of osteosarcoma cells after treatment with 10 μmol/L MI (Figure 6A and B). The results of Transwell invasion assay showed that after 24 h of MI intervention (10 μmol/L), the number of cells penetrating the Matrigel in the MI group was significantly lower than that in the DMSO group (Figure 6C, F). Finally, the results of colony formation assay showed that after 10 days of MI intervention (10 μmol/L) in 143B cells, the colony formation rates of the MI group and DMSO group were 60.09% and 27.37%, respectively, and the difference was statistically significant (P <0.0001). After treatment with the same concentration, 63.79% and 21.05% of the HOS cells in the two groups were colonies, and the difference was statistically significant (P <0.0001) (Figure 6D and E). In conclusion, MI weakened the colony formation ability, migration, and invasion ability of osteosarcoma cells to different degrees.

MI's ability to inhibit the clone, migration, and invasion of 143B and HOS cells in vitro. (A, B) MI significantly slowed the migration of 143B and HOS cells after 48 h of treatment. (C) After 24 h of MI treatment, the invasion of 143B and HOS cells was significantly inhibited. (D) MI for 10 days significantly inhibited the colony formation ability of 143B and HOS cells. (E) Quantitative analysis of the plate colony formation ability. (F) Quantitative analysis of the Transwell invasion data. The data (n > 3) are presented as the means ± SEMs. The level of significance is indicated by ns, ***P <0.001, and ****P <0.0001 compared with the absence of MI. Intervention concentration of MI: 10 μmol/L; Intervention time: 24 h. MI, Momordin Ic.
Discussion
Apart from surgery and chemoradiotherapy, the current treatment for osteosarcoma is mainly focused on molecular targeted therapy and immunotherapy, but most of these treatments have unsatisfactory effects and strong side effects, which greatly limits their application in the treatment of osteosarcoma. 25 MI, a natural small molecular substance, was mainly used for its anti-inflammatory and anti-rheumatoid disease effects in the past. Later, several scholars explored the impact of MI in the field of cancer and found that MI could effectively kill tumor cells. However, its efficacy in osteosarcoma is still unknown. This study preliminarily explored the possible mechanisms involved in MI-mediated inhibition of osteosarcoma progression, as shown in Figure 7.

The possible mechanisms involved in MI inhibiting the progression of osteosarcoma. MI induces G0/G1 cycle arrest in osteosarcoma cells by inhibiting the expression of cyclin E. MI induces apoptosis in osteosarcoma cells by upregulating Bax and downregulating Bcl2. MI induces autophagy in osteosarcoma cells by promoting the expression of LC3B. MI may not induce ferroptosis. The diagram of the underlying mechanism is provided in Figdraw. MI, Momordin Ic.
In the beginning of this study, a high dose (50 μmol/L) of MI was used to explore its killing effects on osteosarcoma cell lines and osteoblasts. As expected, the killing effect of MI on osteosarcoma cell lines was much stronger than that on normal cells, which suggested the possibility of treating osteosarcoma without causing additional complications. After that, we studied the difference in effects of, DMSO and 7 various concentrations of MI, namely 1 μmol/L, 5 μmol/L, 10 μmol/L, 20 μmol/L, 30 μmol/L, 40 μmol/L and 50 μmol/L. The IC50 values of the two osteosarcoma cell lines were similar, indicating that MI has a stable and concentration-dependent killing effect on osteosarcoma cells. To explore whether MI was time dependent, we selected 20 μmol/L and found that MI began to exert antitumor effects after 12 h of intervention, and the inhibitory effect tended to be mild after 48 h; however, the change was not significant after 72 h, which was confirmed by the CCK-8 assay. These results indicated that there might be a peak of efficacy for the inhibition of osteosarcoma cells by MI, which is very similar to the effects of many drugs that induce killing via apoptotic autophagy.
To clarify how MI inhibits the proliferative activity of osteosarcoma cell lines, three phenotypic inhibitors were further selected in the present study to preliminarily determine what form of cell death MI induces. Studies have shown that Z-VAD-FMK and 3-MA can partially rescue the inhibition of proliferation of osteosarcoma cell lines by MI. These results indicated that the toxicity of MI on osteosarcoma cell lines might occur through the combined induction of apoptosis and autophagy. Subsequent apoptosis testing and analysis of the expression of Cyclin E and the apoptotic marker protein Bax/Bcl2 also confirmed that MI could induce apoptosis and G1/G1 cell cycle arrest. This result is consistent with those of previous studies. 12 Moreover, we further tested the expression of autophagy marker proteins and found that the expression of LC3B was significantly increased, indicating that MI induced autophagy. Our conclusions are consistent with those of Mi et al. 11 In conclusion, based on the studies of the authors and predecessors, we found that MI can inhibit the proliferation of osteosarcoma cells through both apoptosis induction and autophagy. However, the specific signaling pathway or mechanism involved is still unclear and requires further research.
As mentioned above, we initially treated cells with the ferroptosis inhibitor Fer-1, and no obvious reversal trend was observed via optical microscopy. To clarify the relationship between MI and ferroptosis, we tested the ferroptosis marker protein PTGS2 and detected no significant change in the expression level of the PTGS2 protein, suggesting that the inhibition of osteosarcoma cell proliferation by MI might not be able to trigger ferroptosis. However, this result is inconsistent with that of Wang et al, who concluded that MI downregulates PTGS2 expression in liver cancer cells. 10 It is speculated that this difference may be related to cancer species, cell type, and cell state because proteins may have different expression levels in different cancer species, different cell lines, or different cell culture states, and even, the mechanism of action of the same drug may differ among different cells.
In addition to exploring the proliferation of osteosarcoma cells induced by MI, we explored the effects of MI on colony formation, migration and invasion. These three experimental findings are consistent with our previous expectation that MI not only inhibits the proliferative activity of osteosarcoma cells but also may have significant effects on more malignant phenotypes of tumor cells. This study provides a research breakthrough point that may lead to more valuable findings in the future. However, notably, the inhibitory effects of MI on the migration and invasion of 143B and HOS cells were different at 24 h. MI obviously inhibited the migration of HOS cells after 24 h, but it had no obvious inhibitory effect on 143B cells. Our team speculated that differences in osteosarcoma cell subtypes may lead to differences in the inhibitory effects of MI. However, when exploring the inhibitory effect of MI on the activity of two kinds of cells, the results showed that MI had an obvious inhibitory effect after 24 h. In view of this inconsistency, we believe that different cell densities also affect the actual inhibitory effect of MI, including the onset time. The cell density (approximately 50%) in the previous experiments before MI intervention was not consistent with that in the migration experiment (close to 100%). The killing effect of the same amount of MI on cells at different densities is likely different. Overall, osteosarcoma treatment is a hot topic. Different types of osteosarcoma have different responses to the same drug, and different malignant phenotypes also have different resistances to the same dose of drug.
The limitation of this study lies in the lack of animal experiments to provide insight into the in vivo intervention effect on MI. In future studies, we will continue to explore the specific signaling mechanisms involved in MI to improve its early clinical application.
In conclusion, this study revealed the antitumor activity of MI in osteosarcoma cells. MI not only inhibits the proliferation of osteosarcoma cells by inducing apoptosis and autophagy but also significantly weakens the invasion and migration ability of osteosarcoma cells. This study not only enriches the therapeutic range of MI but also provides a new drug choice for the clinical treatment of osteosarcoma.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X241228966 - Supplemental material for Momordin Ic Inhibits the Partially Malignant Phenotype of Osteosarcoma Cells by Inducing Apoptosis and Autophagy
Supplemental material, sj-docx-1-npx-10.1177_1934578X241228966 for Momordin Ic Inhibits the Partially Malignant Phenotype of Osteosarcoma Cells by Inducing Apoptosis and Autophagy by Ruiqing Xu, Rui Huang, Jiandang Shi, Dawei Chu and Pengyu Yang in Natural Product Communications
Footnotes
Acknowledgements
Authors’ Contributions
Jiandang Shi was involved in the conception and design of this study. Ruiqing Xu and Rui Huang carried out the whole experimental process. Ruiqing Xu and Dawei Chu prepared the figures and interpreted the data. Ruiqing Xu and Rui Huang and Pengyu Yang drafted the manuscript. Jiandang Shi and Ruiqing Xu revised the manuscript. All authors read and approved the final manuscript.
Availability of Data and Materials
All basic data can be found in articles or supplementary documents or designated websites.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
Not applicable.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of Ningxia Province, (Grant Number: 2019AAC03193, 2022AAC03594).
Supplemental Material
Supplemental material for this article is available online.
Patient Consent for Publication
Not applicable.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
