Abstract
Aim/Background
Oxidative and inflammatory damage induced by bacterial infections has been attributed as a more concerning health issue raised by health professionals and researchers due to large antibiotic drug resistance. Natural products and their derived medicines play an immense role in fighting pathogenic micro-organisms. Considering these factors, the study is aimed at exploring the biological effect of andrographolide against gram-positive and negative bacterial strains that induce oxidative and inflammatory onsets using in silico approaches.
Materials and Methods
In silico docking analysis was conducted using AutoDock Vina to determine the biological interactivity of andrographolide with proteins (catalase (CAT), tumor necrosis factor (TNF-α), and cytochrome P450 VDH mutant T107A with bound Vitamin D3) involved in pathogenesis caused by bacterial infection. Furthermore, absorption, distribution, metabolism and excretion (ADME) analysis was performed to evaluate the pharmacokinetic behavior of andrographolide by assessing several parameters such as lipophilicity, skin permeability, GI absorption, blood-brain barrier (BBB) permeability.
Results
The results of the study showed that andrographolide exhibited significant interaction with TNF-α with amino acids such as GLN B: 27, ILE B: 136, and ASP B: 45 for site 1 and ASN D: 30, ARG C: 31, and ARG C: 82 for site 2. However, the effect of andrographolide was found to be comparable with that of other proteins. In ADME analysis, the boiled egg plot represents the andrographolide that exists in the white region, which represents a good distribution of andrographolide, while lipophilicity in the form of the consensus logP was found to be 2.3 with high GI-absorption and no BBB permeability.
Conclusion
The study concludes that andrographolide can be a promising agent against oxidative and inflammatory stress induced by bacterial onsets and may reduce the vitality of several gram-positive and gram-negative bacterial strains.
Introduction
Infectious diseases remain a serious health issue, accounting for around 41% of the world’s disease burden and causing severe morbidity and mortality as reported by disability-adjusted life years (DALYs). Micro-Organisms are the smallest living organisms that cannot be seen with the naked eye and often exist around us in water, soil (with the highest proportion), and air. Many different types of microbes are categorized as pathogenic and non-pathogenic. Additionally, it is known that the human body has millions of various kinds of bacteria (Cavicchioli et al., 2019; Venkatachalam et al., 2021).
The World Health Organization (WHO) is a global organization tasked with upholding the highest human health and well-being standards. According to the WHO, micro-organisms have been acknowledged as sobering dangers that are alarming the world’s health. Notably, less than 5% and maybe as little as 1% of all bacteria can be grown in a lab (Aboudharam et al., 2010; Mourya et al., 2019). Most of the microbial communities grown in laboratories are utilized to investigate the determining impact of organic or synthetic antibacterial drugs. Additionally, the demand for medications with anti-bacterial resistance was challenged in the pharmaceutical industry (Clebak & Malone, 2018; Piddock, 2012). Due to the excessive or improper use of such treatments, antimicrobial resistance in modern medicine is continuously increasing and is now considered an emergency (Kiran et al., 2021; Naseer et al., 2021).
In addition, regulatory compliance and economic incentives are the primary deficits that complicate the expansion of novel antibiotics by the pharmaceutical sector. Since bacterial resistance to current medications has been continuously increasing for decades, it is crucial to uncover the mechanism of resistance using a different bio-molecular approach and to discount the effectiveness of the therapeutic drug to reduce resistance and optimize the biological effectiveness of antimicrobial agents (Alshibl et al., 2020; Breijyeh et al., 2020).
Natural-derived active medicines are essential in treating a variety of pathogenic onsets, including those caused by microbes, pharmacological poisons, or even physical illnesses (Alhozaimy et al., 2017). Most medications used in modern therapies, perhaps even more than 50%, are made from naturally derived medicines (Rana et al., 2021). Additionally, several naturally occurring substances are being investigated for their potential as the most effective medications for treating microbial infections. The Andrographis paniculata plant yields andrographolide, a naturally occurring component characterized as a labdane diterpenoid. It has been reported that A. paniculata exhibits many pharmacological effects, including anti-bacterial, antiviral, anti-platelet, anti-tumor, and immunomodulatory activity. Andrographolide is a potential anti-inflammatory agent (Dai et al., 2019; Hanh et al., 2020; Worakunphanich et al., 2021).
Although it is reported that the exact mechanism of andrographolide is considered an anti-inflammatory effect coordinated by its antioxidant characteristics, reactive oxygen species (ROS) generated inside the body are a normal cellular process; excessive production of ROS leads to cellular damage (Gautam et al., 2021). Together with nitric oxide, the most prevalent ROS, such as hydrogen peroxide (H2O2) or superoxide anion (O2), cause the intracellular vitality extortion of micro-organisms (Chandra et al., 2018; Sharma et al., 2018). Extracellular DNA trap is regulated by the interleukins (ILs) as well as the mitogen-activated protein kinase (MAPK) pathway that involves the extrusion of DNA developed with biomolecules. With antimicrobial and proteolytic activity, neutrophils and other leukocytes defend themselves from infectious processes or pro-inflammatory agents (Gautam, 2022). Based on the observations, the study is related to examining the impact of andrographolide using in silico methods on bacterial resistance as well as bacterial-induced oxidative and inflammatory onsets.
Materials and Methods
Software Information
The AutoDock tool (Version 1.5.7) was used to prepare the protein and ligand, while Discovery Studio was used to visualize the protein and ligand interaction. Cytoscape and Metascape software were used to assess the network pharmacology analysis, while the SWISS absorption, distribution, metabolism, and excretion (ADME) online tool was used for the determination of the pharmacokinetic analysis of andrographolide.
In silico Docking Analysis
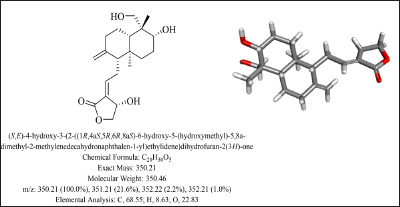
This study aimed to explore the anti-bacterial effect, and bacterial strains induced oxidative and inflammatory insults, reducing the activity of andrographolide through in silico methods. Andrographolide is one of the most prominent constituents found in Andrographis paniculata. The chemical structure and complete elemental analysis are depicted in Figure 1. Different proteins involved in oxidative stress, inflammatory stress, and bacterial resistance were assessed for their biological interactions with andrographolide. The binding mechanism and affinity of andrographolide can be simulated using molecular docking.[20,18]
Chemical Structure, Elemental Analysis and 3D Structure of Andrographolide.
Accession of the Target Protein
The three-dimensional and high-resolution structure of the targeted proteins was obtained from the RCSB Protein Data Bank (
Ligand Preparation
The ligands were downloaded in SDF format from the PubChem database (pubchem.ncbi.nlm.nih.gov) and formatted in PDB and PDBQT using software (BIOVIA Discovery Studio Visualizer 2021). The software was then used to process the ligands for molecular docking analysis using AutoDock by adjusting ionization, torsion, degree of freedom, and stereochemical variation (Wangthong et al., 2007).
Protein Structure for Docking
Targeted proteins such as CAT, TNF, and cytochrome P450 VDH mutant T107A with bound Vitamin D3 were prepared and refined using AutoDock. The RCSB protein database was used to download the protein structures in PDB format with resolutions of 2.75, 2.65, and 2.57 and R-value free values of 0.272, 0.253, and 0.231 for each protein. Docking simulations were used to perform molecular docking using the AutoDock Vina and BIOVIA Discovery Studio Visualizer programs. Additionally, the creation of the ligand and enzyme used in the docking analysis was carried out using the Discovery Studio program, as was the execution of the docking analysis utilizing the command prompt procedure (Ali et al., 2022).
ADME Analysis
A Swiss ADME analysis of andrographolide was performed to determine its pharmacokinetic behavior. The SwissADME (
Network Pharmacology Analysis
Network pharmacology analysis of andrographolide was performed using Cytoscape software against various genes involved in inflammation and oxidative damage pathophysiology. This analysis was performed as per standard protocol with some modifications (Gautam, 2022). Each gene was obtained from a gene card and evaluated for its biological interaction with andrographolide. Protein–protein interaction (PPI) and compound protein interaction networks were generated to follow interaction information and its significance. A gene ontology analysis was performed to determine the pathophysiological pathways involved in reducing oxidative and inflammatory stress.
Results
In silico docking and ADME studies for andrographolide were successfully performed to determine the bacterial-induced antioxidant and anti-inflammatory effects.
In silico Docking Analysis
In in silico docking analysis, the biological interaction of CAT, TNF, and cytochrome P450 VDH mutant T107A with bound Vitamin D3 with andrographolide was evaluated successfully. The spacing angstrom central grid box dimensions were set for two different sites, such as site 1 and site 2, to determine the variability in the biological interaction of andrographolide. The central grid box dimensions for each protein at two different sites were also evaluated successfully. The results of the in silico studies revealed that andrographolide has a strong interaction with the TNF-α protein, which has been considered by the generation of conventional hydrogen bonds as well as binding energy (affinity kcal/mol). The interaction profile of andrographolide with the targeted genes is summarized in Table 1.
Interaction Profile of Andrographolide.
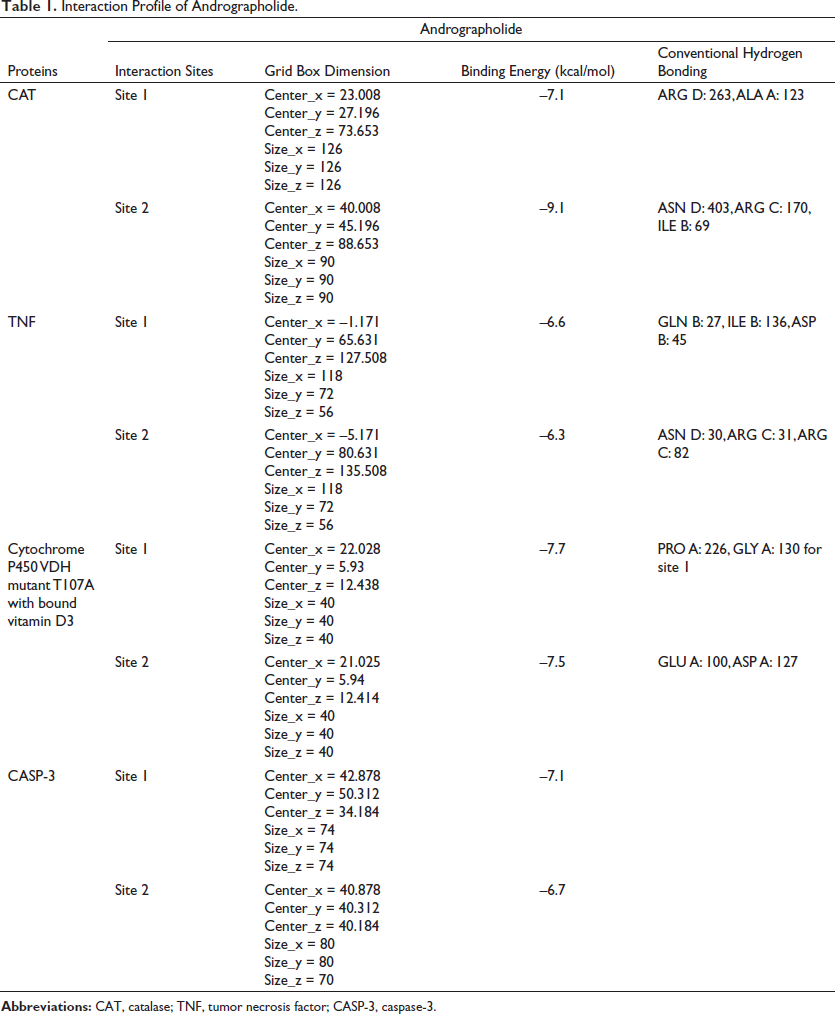
Docking Interaction of Andrographolide with CAT
The interaction of docking analysis of andrographolide with CAT was determined at two different sites, including site 1 as well as site 2. The results of the study showed that andrographolide produces significant binding energy with CAT, which was found for different sites at –7.1 and –9.1 kcal/mol, respectively. The conventional hydrogen bond that was produced during analysis showed that andrographolide exhibited ARG D: 263 and ALA A: 123 for site 1 and ASN D: 403, ARG C: 170, and ILE B: 69 for site 2. The results of the study are represented in Table 1 and Figure 2.
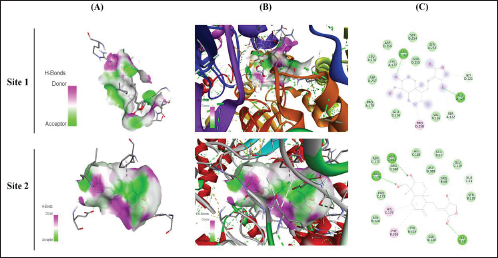
Docking Interaction of Andrographolide with TNF
The interaction of docking analysis of andrographolide with TNF-α was determined at two different sites (site 1 and site 2). The study showed that andrographolide produces significant binding energy with TNF-α, which was found for different sites at –6.6 and –6.3 kcal/mol. The conventional hydrogen bond that was produced during analysis showed that andrographolide exhibited GLN B: 27, ILE B: 136, and ASP B: 45 for site 1 and ASN D: 30, ARG C: 31, and ARG C: 82 for site 2. The results of the study are represented in Table 1 and Figure 3.
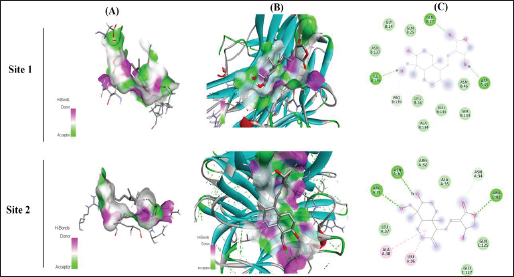
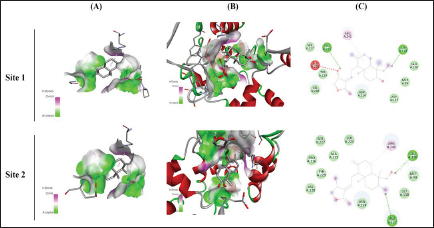
Docking Interaction of Andrographolide with Cytochrome P450 VDH Mutant T107A with Bound Vitamin D3
The interaction of docking analysis of andrographolide with Cytochrome P450 VDH mutant T107A with bound Vitamin D3 was determined at two different sites, which include site 1 and site 2. The results of the study showed that andrographolide produces significant binding energy with TNF-α, which was found for different sites at –7.3 and –7.5 kcal/mol. The conventional hydrogen bond that was produced during analysis showed that andrographolide exhibited PRO A: 226 and GLY A: 130 for site 1 and GLU A: 100 and ASP A: 127 for site 2. The results of the study are represented in Table 1 and Figure 4.
In silico Docking Analysis of Andrographolide with Cytochrome P450 VDH Mutant T107A with Bound Vitamin D3 Protein on Two Different Sites. Column A Represents Ligand Interaction and Bonding Pattern, Column B Represents the 3D Structure of Protein and Ligand Interaction, and Column C Represents the 2D Structure of the Ligand with Specified Amino Acids and Residual Energy.
Docking Interaction of Andrographolide with CASP-3
The interaction of docking analysis of andrographolide with Cytochrome P450 VDH mutant T107A with bound Vitamin D3 was determined at two different sites, which include site 1 and site 2. The results of the study showed that andrographolide produces significant binding energy with TNF-α, which was found for different sites at –7.1 and –6.7 kcal/mol. The conventional hydrogen bond that was produced during analysis showed that andrographolide exhibited ASP C: 135 and TRY D: 197 for site 1 and TRY D: 197 for site 2. The results of the study are represented in Table 1 and Figure 5.
In Silico Docking Analysis of Andrographolide with CASP-3 Protein on Two Different Sites. Column A Represents Ligand Interaction and Bonding Pattern, Column B Represents 3D Structure of Protein and Ligand Interaction while Column C Represents 2D Structure of the Ligand with Specified Amino Acids and Residual Energy.
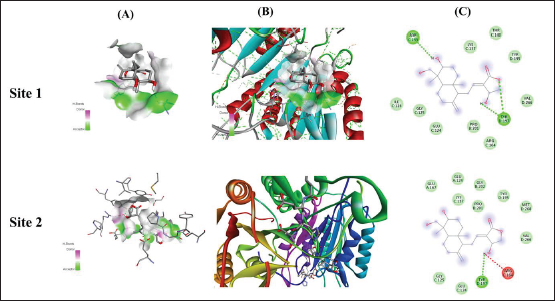
In silico docking analysis of andrographolide with the CASP-3 protein on two different sites. Column A represents ligand interaction and bonding pattern, column B represents the 3D structure of protein and ligand interaction, and column C represents the 2D structure of the ligand with specified amino acids and residual energy.
ADME Analysis
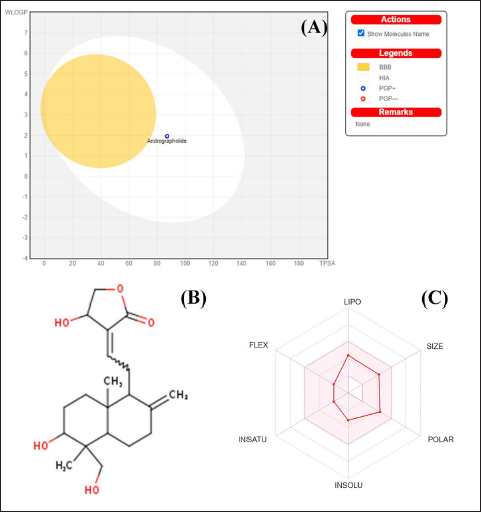
ADME analysis of andrographolide was conducted using the Swiss ADME tool successfully. The study aimed to determine the pharmacokinetic behavior or bioavailability response of andrographolide. The study was performed as per the reference protocol stated in the referenced procedures of SWISS. Several parameters, such as lipophilicity, and drug-like response consensus logPo/w, TPSA, ESOL Log S values, BBB permeant, GI absorption, and log Kp (cm/s) (skin permeation), were determined for the assessment of the pharmacokinetic behavior of andrographolide. TPSA serves as an appropriate marker for the evaluation of ADME aspects, especially those related to absorption and cerebral availability. The TPSA value of andrographolide was determined to be 86.99. The consensus logPo/w was found to be 2.3, which represents the accuracy of the forecast for the physico-chemical parameters, and lipophilicity, which reflects the lipophilicity of anticipated molecules, is used to predict the consensus logPo/w. A hydrophilic substance has a low logP value, whereas a lipophilic substance has a high logP value (Ali et al., 2022). This is comparable to a hypothesis put forth by Potts and Guy that links the size and lipophilicity of the molecule to the skin permeability coefficient (Kp). The less permeant the medication is to the skin, the more negatively skewed the value of log Kp. The log Kp value of –6.9 supports our findings that andrographolide considerably enhances skin permeability. In the consensus log, the plain polarity or lipophilicity of two different criteria, Po/w and TPSA, has a considerable influence on a molecule’s BBB permeant affinity. If the molecule completely encloses the egg’s yolk, it has a high BBB penetration rate and remains within the white-zone range of HIA absorption. Since andrographolide was found outside in the boiled egg plot (white region) of the ADME analysis (Gautam, 2022). The results of the study suggest that it has a low affinity for permeation. The ADME outcomes are depicted in Figure 6.
Network Pharmacology Analysis
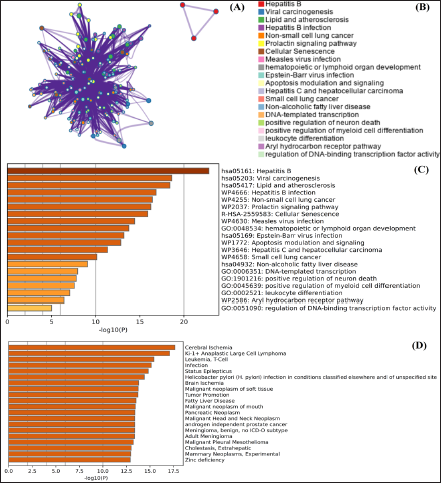
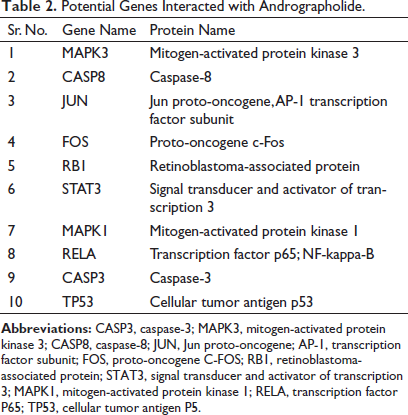
Network pharmacology analysis was conducted to investigate the multi-mechanistic and therapeutic approach of andrographolide as well as to identify the pattern of substantial and partial gene interaction with the targeted components. The compound and protein interaction (CPI) and PPI interaction networks were generated to validate compounds and gene interactions. During data interpretation and analysis, the genes that did not interact with the drug at all or just partially interacted with it were eliminated. The maximum number of additional interactors was set to 10, and the confidence score of the network between genes and chemicals was set to 0.004. The study’s findings demonstrated that PPI interaction exhibited the following: number of edges: 44, average node degree: 8.8, average local clustering coefficient: 0.978, expected number of edges: 17, and PPI enrichment p-value: 1.33e–08. The findings showed that each gene exhibited partial and significant interaction with each other, while the genes that were not found to have interaction were excluded during interpretation analysis. In CPI, the interaction of genes and andrographolide considerably contributes to the biological control of pathogenic and pathophysiological onsets. Out of the 100 genes examined in this study, 10 targeted genes (Table 1), such as mitogen-activated protein kinase 1 (MAPK1), caspase-8 (CASP8), caspase-3 (CASP3), signal transducer and activator of transcription 3 (STAT3), and retinoblastoma-associated protein (RB1), were found to interact significantly with andrographolide. The CPI network of genes and compounds is depicted in Table 2 and Figures 7 and 8.
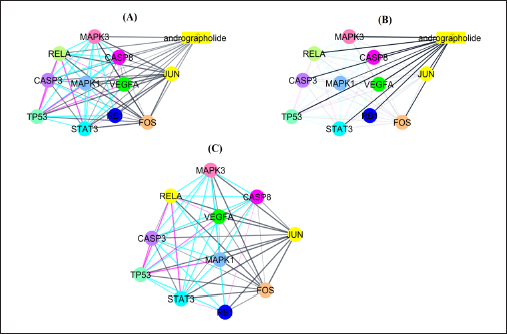
Gene Ontology Analysis to Investigate Multi-Mechanistic and Therapeutic Action of Andrographolide Based on Interaction with the Genes.
Potential Genes Interacted with Andrographolide.
Discussion
Microbial infection remains a concerning issue due to the anti-bacterial resistance of a wide variety of drugs (Ali et al., 2022). Throughout history, medicinal plants or phytochemical constituents have played an immense role in alleviating several acute and chronic diseases (Khan et al., 2022). Medicinal plants are also a good source for mitigating bacteria-induced diseases (Kiran et al., 2021). There have been several studies conducted to explore and validate the scientific facts concerning the anti-bacterial effect of natural products/medicinal plants, or phytoconstituents due to a large number of anti-bacterial drug resistances (Mehrotra, 2020). Based on the observations, the study is associated with exploring the therapeutic effect of andrographolide against bacterial infection-induced diseases using several in silico approaches (Dhama et al., 2022).
In silico studies have been well explored for the assessment of new drug discovery and development as well as to determine the multi-targeted effect of active constituents (Gautam, 2022). In silico docking study was conducted to find the biological interaction of andrographolide with several proteins (CAT, TNF, cytochrome P450 VDH mutant T107A with bound Vitamin D3, and CASP-3) involved in bacterial infection, their induced oxidative stress, and inflammatory stress. The findings of the in silico study showed that andrographolide exhibited the highest interaction with TNF-α with the development of 3 and 3 conventional hydrogen bonds in each site, while binding energy was found to be –6.6 and –6.3, respectively. Andrographolide produced a conventional hydrogen bond via the interaction of the –OH group with the amino acids TRY D:197 and TRY B:195. It has been noted that the stability of protein-ligand complexes with higher binding affinities than ordinary medicines is greatly influenced by the formation of conventional hydrogen bonds. Other van der Waals bonds include the carbon-hydrogen bond, the unfavorable donor-donor bond, pi-cation, pi-sulfur, alkyl, pi-donor hydrogen bond, and the pi-alkyl bonds. However, the effect of andrographolide was also found to be comparable with that of other proteins. According to numerous types of research, andrographolide improves SOD as well as CAT activity in samples transfected with agents that cause oxidative stress. For instance, the administration of andrographolide (5, 7, and 10 mg/kg, orally) reversed the effects of hexachlorocyclohexane exposure on SOD and CAT activity (Trivedi et al., 2007). Exposure to carbon tetrachloride decreased SOD and CAT activity in rat erythrocytes (CCl4). After being stimulated by CCl4, rats were treated with A. paniculata (1 g/kg) methanolic extract to recover SOD and CAT activity (Akowuah et al., 2009). (Wang et al., 2021) evaluated the andrographolide effect against synovial inflammation in osteoarthritis by expressing and regulating the signaling pathway of TNF2 trafficking. It was demonstrated that treatment with andrographolide slowed the development of OA and synovial inflammation. Additionally, a reduction in dorsal horn neuron activity and pain hypersensitivity was seen after treatment. Additionally, androgen significantly decreased TNFR2 or inflammatory cytokines. The NF-B signaling route needs TNFR2, and androgen-induced dilapidation of TNFR2 was linked to the function of the lysosome, which in turn decreased the NF-B signaling pathway’s cascade phosphorylation of p65 (Wang et al., 2021). CASP-3 is one of the most prominently expressed proteins and regulates the apoptotic function of the irregular cellular development process. It has been reported that it plays a vital role in inflammatory cascades and regulates pathogenesis induced by inflammation (Khan et al., 2022).
The assessment of the pharmacokinetic behavior of medications obtained from natural or synthetic sources has been done using ADME analysis. The primary parameters to determine the outcomes of the drug’s pharmacokinetic behavior include skin permeability, logP-value, GI absorption, and brain permeability (Ali et al., 2022). According to our research, andrographolide has no brain permeability, the least skin permeability, and lower lipophilicity. Furthermore, it can be suggested that andrographolide has good GI absorption and skin permeability and may lead to a better therapeutic site.
Gene ontology analysis was performed to determine the multi-targeted and therapeutic effects of andrographolide against pathogenesis induced by bacterial infection through the induction of oxidative stress and inflammatory stress (Gautam, 2022). The findings showed that andrographolides play a multi-mechanistic role in bacterial and viral infections through the regulation of several pathophysiological pathways. The outcome of the analysis is depicted in Figure 8.
A study conducted by Tan et al. (2010) evaluated the effect of andrographolide on the regulation of epidermal growth factor receptor (EGFR) and transferrin receptor trafficking in epidermoid carcinoma (A-431) cells. The outcome of the study showed that andrographolide treatment inhibited cell growth, down-regulated EGFRs on the cell surface, and affected the degradation of EGFRs and TFRs. The EGFR was internalized into the cell at an increased rate and accumulated in a compartment that co-localizes with the lysosomal-associated membrane protein in the late endosomes. Hence, it was shown that andrographolide might affect receptor trafficking by inhibiting receptor movement from the late endosomes to the lysosomes. The downregulation of EGFR on the cell surface also indicates a new mechanism by which andrographolide may induce cancer cell death (Tan et al., 2010).
Furthermore, it is also reported that andrographolide induces DNA damage in prostate cancer cells. A study conducted by Forestier-Román et al. (2019) evaluated the effect of andrographolide-inducing DNA damage on the prostate tumor. Tumor growth was evaluated using an orthotopic xenograft model in which the prostates of SCID mice were injected with 22RV1, and mice were treated three times per week with Andrographolide 10 mg/kg. andrographolide decreased tumor volume, MMP11 expression, and blood vessel formation in vivo. Gene expression analysis identified cellular compromise, cell cycle, and “DNA recombination, replication, and repair” as the major molecular and cellular functions altered in tumors treated with andrographolide. Within DNA repair genes, we confirmed increased expression of genes involved in DNA double-strand break repair. Consistent with this observation, we detected increased γH2AX in andrographolide-treated tumors and in cells in culture. Hence, it was demonstrated that andrographolide inhibits PCa by promoting DNA damage (Forestier-Román et al., 2019).
Zi et al. (1998) reported the biomolecular activity of EGFR and TGF (Zi et al., 1998). It is reported that andrographolide inhibits transforming growth factor (TGF)-α-mediated tyrosine phosphorylation of EGFR in advanced human prostate cancer DU145 cells. It down-regulates EGFR signaling via significant inhibition in the expression and secretion of different growth factors, binding to and activating EGFR. Andrographolide subsequently impaired downstream mitogenic events and acted as an anticancer agent in tumor cell lines (Deep & Agarwal, 2007).
Conclusion
The present study concludes that andrographolide can be a promising agent against bacterial infection as well as oxidative and inflammatory stress induced by bacterial strains, as it shows significant interaction with CAT and TNF as well as acts as a potential agent against CASP-3-induced cellular degradation or inflammation. Network pharmacology analysis revealed the multi-therapeutic action of andrographolide in bacterial-induced inflammation and oxidative stress via positive and negative regulation of several gene expressions, especially for proteins belonging to MAPKs and CASPs. Furthermore, the study would support the further observation made on the oxidative and inflammatory damage due to bacterial infection of andrographolide to explore the mechanism of action for alleviating oxidative and inflammatory damages in bacterial infection.
Summary
Natural products and their derived medicines have been used exponentially to treat acute and chronic diseases globally. Microbial infections and their induced oxidative and inflammatory damage have been concerning in the era of antibiotic drug resistance. Natural constituents have been used as an alternative and provide potent therapeutic effects. Andrographolide is one of the prominent constituents of A. paniculata and has been used for treating several acute and chronic ailments. Due to a lack of evidence and molecular approaches, in this study, the multi-mechanistic and therapeutic effect of andrographolide against bacterial infection-induced oxidative and inflammatory damage has been depicted using various in silico approaches. The results showed that andrographolide has a prominent effect against various biomolecular proteins such as CASPs, CAT, MAPK, and TNF-α, which are responsible for regulating oxidative and inflammatory stress. Furthermore, it can be demonstrated that andrographolide can be a promising agent for treating bacterial infections and their induced oxidative and inflammatory damage and might act as a prominent therapeutic agent that can superimpose those anti-bacterial agents that are obviated due to anti-bacterial resistance.
Footnotes
Abbreviations
ADME: absorption, distribution, metabolism and excretion; BBB: blood–brain barrier; CPI: compound and protein interaction; CAT: catalase; CASP3: caspase-3; DALYs: disability-adjusted life years; PPI: protein–protein interaction; MAPK3: mitogen-activated protein kinase 3; CASP8: caspase-8; JUN: Jun proto-oncogene; Ap-1: transcription factor subunit; FOS: proto-oncogene C-FOS, IL: interleukins; RB1: retinoblastoma-associated protein; STAT3: signal transducer and activator of transcription 3; MAPK1: mitogen-activated protein kinase 1; RELA: transcription factor P65; nuclear factor kappa-B, NF-κ-B; TP53: cellular tumor antigen P5; TNF: tumor necrosis factor; TPSA: topological polar surface area; WHO: World Health Organization; EGFR: epidermal growth factor receptor.
Acknowledgments
The author would like to thank the Department of Biology, Faculty of Science, Taibah University, Al-Sharm, Yanbu El-Bahr 46429, Saudi Arabia, for providing facilities for conducting research.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Statement of Informed Consent and Ethical Approval
Not applicable.
