Abstract
Objectives
The aim of this study is to analyze the chemical composition of Aloe officinalis Forssk and to investigate its potential medicinal use, especially as a natural antibacterial agent.
Methods
The red flowers of Aloe officinalis Forssk were collected from Al Kadimi, Jazan province, Saudi Arabia, and processed into a methanolic extract (MEAOF) by maceration. The extract was comprehensively analyzed by GC-MS, LC-MS, FT-IR, and NMR spectroscopy. The antioxidant activity of MEAOF was evaluated using the DPPH assay, while its antibacterial potential against various human pathogenic bacteria was investigated using the agar well diffusion method.
Results
The analyzes identified several bioactive compounds in MEAOF, including stearic acid, palmitic acid, 9,12-octadecadienoic acid (Z, Z) and dasycarpidan-1-methanol acetate. MEAOF exhibited significant antibacterial activity against both Gram-positive and Gram-negative bacteria. In addition, MEAOF demonstrated an antioxidant scavenging capacity of 87.96%.
Conclusion
The study confirms the significant therapeutic potential of the flowers of Aloe officinalis Forssk and emphasizes their strong antibacterial and antioxidant properties. These results support the use of extracts from the flowers of Aloe officinalis Forssk in the development of new drugs to combat antibiotic resistance and reduce oxidative stress and underline their value for medical applications.
Introduction
For thousands of years, nature has served as a cradle of medicinal agents, and a significant proportion of contemporary medications have been derived from natural origins. Many of these medications have roots in traditional or phytomedicinal remedies.1,2 Aloe officinalis Forssk, commonly known as Aloe vera, is a perennial succulent plant of the Asphodelaceae family. Aloe vera originated in the Arabian Peninsula but is now grown in many parts of the world for medicinal and agricultural purposes. The plant has thick, fleshy leaves that grow in a rosette shape. The leaves are green and contain a gel-like substance known for its many health benefits. This gel is commonly used in skincare products, cosmetics, and natural remedies.3,4
Aloe officinalis Forssk has gained attention for its antimicrobial properties, particularly its ability to inhibit the growth of certain bacteria. Antibiotic resistance is a major and growing global health problem that occurs when bacteria develop ways to resist the effects of antibiotics that would normally eliminate them or inhibit their growth. 5 Infected wounds have been effectively treated using topical antimicrobial medications. Antibiotic resistance in wound management can lead to treatment failure and consequences. There is an increasing demand for the creation of new antimicrobial agents that have a broader range of effectiveness and can be applied topically. Nevertheless, there is no guarantee that more recent topical treatments will not acquire resistance to germs. 6 In the regions of Jazan and Asir in Saudi Arabia, a large variety of indigenous flowering plants are widespread. 1 In contemporary therapeutic practice, these flowers are extensively utilized, with their essential oils, infusions, juices, petals, and aromatherapy applications used for holistic healing purposes that affect both mental and physical health.7,8 The current study aimed to analyze the chemical composition of Aloe officinalis Forssk flower using GC-MS, LC-MS, FT-IR, and NMR spectroscopy. The study also shown its potential as an antioxidant and antibacterial agent.
Materials and Methods
Materials
Scharlau, Spain provided the bacteriological media that was utilized in this research. Other materials employed in this study were supplied by Ejadah Medical Supplies Est in Riyadh, Saudi Arabia.
Study Area, Plant Collection, and Identification
Aloe officinalis Forssk is a desert plant commonly found in Al Kadimi, Jazan province, Saudi Arabia. Therefore, Al Kadimi was the study area, which is in Jazan province, southern Saudi Arabia, approximately 30 km East of Jazan city (Figure 1). Aloe officinalis Forssk was harvested in September 2023 from Al Kadimi. The herbarium curator identified the plant and deposited a voucher specimen in the herbarium of Jazan University (reference number = JAZUH 1308).

The plant collection (A) Al Kadimi, Jazan province (B & C) growth of plant in the desert region of, Saudi Arabia (D) development of a flower bud originating from the aerial part of the stem (E) an overview of flower arrangement and organization (F) drying process of harvested flowers.
Processing of Flowers
The red flowers of Aloe officinalis Forssk were carefully plucked out and thoroughly rinsed with both tap water and Millipore water. The flowers were then air-dried in the laboratory at room temperature for one week, as shown in Figure 1. After drying, the flowers were blended, ground into a powder using a grinder, and stored at room temperature in a sealed container.
Maceration 9
A 25-gram sample of powdered dried flower material was soaked in 100 mL of methanol for one week to allow for thorough maceration. After this period, the mixture was spun at a force of 2000×g for 10 min in a Sigma brand tabletop centrifuge to separate the debris. The clear liquid layer obtained was filtered using Whatman no. 1 filter paper to eliminate any residual particles, and the collected liquid, or filtrate, was left to evaporate at room temperature (25 °C), resulting in a methanolic extract named MEAOF. The MEAOF was subsequently evaluated for its antibacterial efficacy in further experiments.
GC-MS Analysis
The MEAOF was put through an additional purification step by dissolving it in HPLC-grade methanol. The solution was then passed through a Millex-GV Syringe Filter Unit with a pore size of 0.2 µm, composed of polyvinylidene fluoride (PVDF) from Merck KGaA, Darmstadt, Germany. To ascertain phytochemical composition of the resulting filtered MEAOF, it was subjected to Gas Chromatography-Mass Spectrometry (GC-MS) analysis. The GC-MS analysis utilized Thermo Scientific equipment with an AS 3000 autosampler and an ISQ detector. A 2 µL sample of the extract was introduced into a TR 5MS capillary column for partial component separation. Helium served as the carrier gas, maintaining a flow rate of 1.2 mL/min. Mass spectrometry was conducted, with spectral data analyzed via Xcalibur software. Interpretation of the mass spectra relied on the NIST and MAINLIB software libraries to identify the bioactive compounds present and recorded.
LC-MS Analysis
MEAOF was subjected to detailed analysis through Liquid Chromatography-Mass Spectrometry (LC-MS) employing a Waters Xevo G2-S system, Milford, USA. This comprehensive characterization process was guided by the protocols outlined by Khojali et al (2023), ensuring the accuracy and reliability of the results obtained.
10
The technique facilitated the precise identification and quantification of the molecular constituents within MEAOF
FT-IR Spectroscopy
FT-IR spectroscopy was employed to analyze the functional groups within MEAOF. The analysis was conducted using a Nicolet iS10 FT-IR spectrophotometer from Thermo Scientific in the USA. Samples were prepared using the KBr pellet technique, and the resulting spectra were recorded with the FT-IR spectrophotometer. These spectra were then compared with those of a reference KBr pellet. The spectral analysis covered a range from 400 to 4000 cm−1, with the resolution set at 4 cm−1.
Nuclear Magnetic Resonance (NMR) Spectroscopy
The air-dried MEAOF underwent analysis through nuclear magnetic resonance spectroscopy (NMR) after sample preparation in deuterated water (D2O). The NMR spectra, including 1H NMR and 13C NMR, were recorded using a Bruker 400 Ultra Shield NMR spectrometer. This instrument operated at a frequency of 400 MHz for 1H NMR and 100 MHz for 13C NMR.
Evaluation of the Antibacterial Potential of MEAOF1,6
Bacterial Strains Utilization and Culture Standardization
The study employed various bacterial strains, including Staphylococcus aureus, Streptococcus pyogenes, Bacillus subtilis, Proteus vulgaris, Klebsiella pneumoniae, Escherichia coli, and Pseudomonas aeruginosa. A 24-h culture was initiated and adjusted to a standardized concentration by diluting it in a gradient ranging from 10−1 to 10−7 using nutrient broth. The bacterial culture viability was determined by quantifying the colony-forming units per milliliter (CFU/mL).
Assessment of Antibacterial Susceptibility
The antibacterial activity was assessed following the method outlined by Sivakumar et al. 6 The bacterial cultures for antibacterial testing were prepared using Muller-Hinton agar plates and were ready after a 24-h subculture from stock culture. Both the MEAOF samples and a conventional antibiotic, ciprofloxacin, were tested using the agar well diffusion method. A sterile cotton swab soaked with a culture containing a specific concentration of CFU/mL for each bacterial strain selected in the study was used for inoculation. The cotton swab was swabbed over the Muller-Hinton agar plates and allowed to dry for about 10 min before applying the samples. The agar plates were inoculated and then drilled with a sterile stainless-steel drill to create wells for the agar well diffusion procedure. Antibacterial activity was assessed by measuring the zones of inhibition around the wells following 24 h of incubation at 37 °C. The results, showing the antibacterial range based on the size of these zones, were methodically documented in Table 2, demonstrating the relationship between the diameter of inhibitory zones and the antibacterial activity spectrum.
Antibacterial Study.

Each value is the mean of 3 batches with standard deviation. The statistical analyses were done using the Prism 9, Graph Pad Instat software system, USA. #CFU- Colony Forming Unit; MEAOF: Methanolic extract of Aloe officinalis Forssk flower. All the values of MEAOF were lesser than Ciprofloxacin significantly at P < .05 level.
Antioxidant Activity by 2,2-Diphenyl-1-Picrylhydrazyl (DPPH) Assay
The 1,1-diphenyl-2-picrylhydrazyl (DPPH) stable radical method, as outlined by Brand-Williams in 1995, was employed to measure the antioxidant capacity of MEAOF.
11
The DPPH control plays a crucial role in antioxidant testing as it provides a baseline for measuring the natural absorption of the radical. By comparing the absorbance of the DPPH solution alone (without sample) with the absorbance when a sample is added, the ability of the sample to scavenge free radicals can be determined. This control ensures that any observed absorbance changes are due to the antioxidant activity of the sample and no other factors, allowing accurate quantification of the radical scavenging potential of the sample. For the preparation of the DPPH solution, 2.4 mg of DPPH was dissolved in 100 mL of methanol. Then 3.995 mL of this methanolic DPPH solution was combined with 5 µL of the MEAOF solution for the assay (At). 3 mL of solution containing DPPH in 100 µL of methanol was used as a control (Ac). The absorbance at 30 min of the resulting mixture was measured at 517 nm employing a spectrophotometer. Each measurement was performed in triplicate. The percentage of antioxidant capacity was calculated using a specific formula.
Statistical Analysis
The statistical analysis was done using the Prism 9, Graph Pad Instat software system, USA. Statistical analyses were performed using one-way ANOVA, followed by Tukey's test (post hoc test). Values of P < .05 was considered statistically significant for all analyses.
Results
The analysis of MEAOF using various techniques like GC-MS, LC-MS, FT-IR, and NMR revealed a range of bioactive compounds. The GC-MS and LCMS analyses of MEAOF show distinct bands, as shown in Figures 2 and 3. Several bioactive MEAOF were identified and are listed in Table 1. Stearic acid, also known as octadecanoic acid, is a type of saturated fatty acid. It was detected in MEAOF at a retention time (RT) of 40.89 min. The identification carried a probability index of 20.41% and represented 22.53% of the total peak area in the GC-MS chromatogram. Palmitic acid (n-Hexadecanoic acid), identified with a RT of 37.58 min, showed a probability index of 38.36% and occupied 17.48% of the chromatogram. Additionally, palmitic acid was detected in the LC-MS analysis under negative ionization mode. The compound 9,12-octadecadienoic acid (Z, Z) was identified in MEAOF with a RT of 40.94 min and a probability index of 27.32%, accounting for approximately 12.53% of the total peaks in the GC-MS chromatogram. Furthermore, the LC-MS analysis also exhibits 9,12- octadecadienoic acid (Z, Z) in negative ionization mode. Dasycarpidan-1-methanol, acetate (ester) an alkaloidal compound has been identified in the MEAOF with a RT of 13.51 min and a probability index of 33.79%, accounting for approximately 7.7% of the total peaks in the GC-MS chromatogram. Remarkably, the compound was also identified in the positive ionization mode in the LC-MS analysis. Methyl benzoate, commonly referred to as benzoic acid methyl ester, was identified in MEAOF with a RT of approximately 21.51 min, representing around 14.81% of the chromatogram. MEAOF sample analysis exhibited stigmasterol at RT of 4.59 min, a probability index of 13.73%, and represented 2.12% of the chromatogram peak. Additionally, stigmasterol was detected in MEAOF using the positive ionization mode in the LC-MS analysis. Methyl stearate was identified at RT 16.10 min with a 21.59% probability index and occupied very less area of 0.21% in of the chromatogram peak. Additionally, several compounds were determined at specific RT, accounting for distinct percentages of the chromatogram area, as detailed in Table 1. Ethyl iso-allocholate, identified as a steroidal compound, was analyzed, and determined using the positive ionization mode of LC-MS in 2.34 RT with 98% of probability index. LC-MS analysis, utilizing positive ionization, identified 17-pentatriacontene, diisooctyl phthalate, hexyl-heptadecane (9-hexyl-), and 2-cyclohexyl-eicosane at specific RT. Conversely, employing negative ionization, LC-MS analysis determined the presence of heptadecane, norethandrolone, betulin, and phytol at designated RTs.
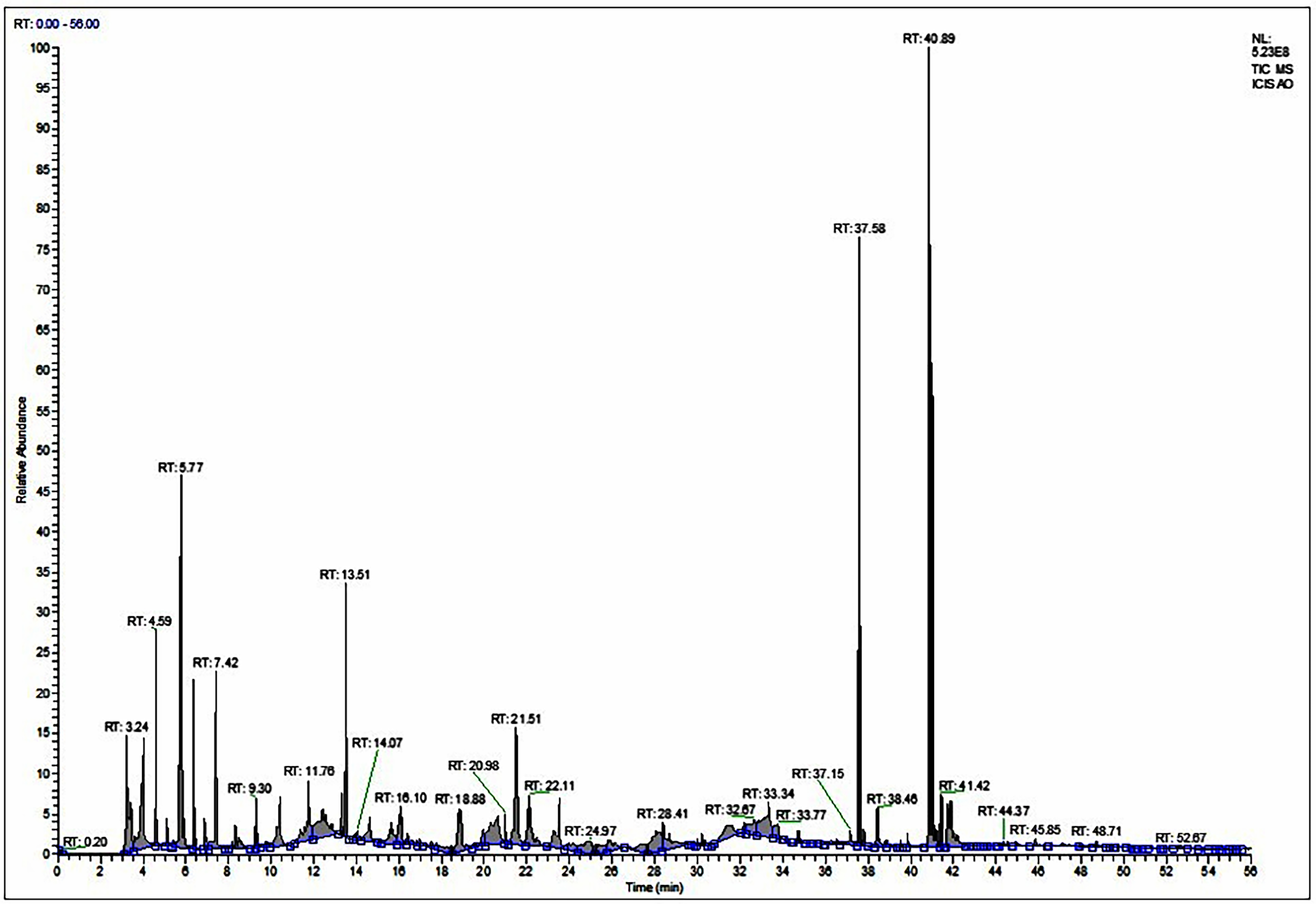
GC-MS chromatogram of MEAOF.

The mass spectra of MEAOF through LC-MS analysis (A) positive ionization mode spectrum (B) negative ionization mode spectrum.
GC-MS Detection of Possible Bioactive Compounds of MEAOF.

The FT-IR analysis revealed specific band patterns corresponding to significant functional groups, as depicted in Figure 4. The presence of the phenolic O-H group is suggested by a broad, parabola-shaped signal at 3396 cm−1, indicating stretching vibrations. The analysis identified bands at 2928.30, 1631.37, 1480.80, 1261.13, and 1057.64 cm−1. The bands suggest the presence of various functional groups, indicating the existence of alkanes, carboxylic acids, alkenes, aliphatic amines, sulfoxides, and sulfides. The bands at 866.69, 814.25, and 595.08 cm−1 correspond to alkyne C-H bending, alcohol OH out-of-plane bending vibrations, and methylene groups, respectively.

FT-IR analysis of MEAOF.
The 1H NMR analysis gave the fingerprint region of MEAOF with the most deshielded proton 3.368 and 4.915 ppm in the proton dimension (Figure 5A). The 13C NMR spectrum showed unique peaks at 37.74, 42.18 to 49.15, 51.37 to 56.11, 60.56 to 68.61, 70.45 to 77.30, 82.33, 99.15 to 98.45 and 102.33 ppm (Figure 5B). 37.74 ppm indicates carbon that is part of a methylene (-CH2-) group. The peaks at 42.18 to 49.15 ppm are the range of chemical shifts is also indicative of alkyl carbons with slightly more deshielded environment compared to the peak at 37.74 ppm. the peaks at 47.114 to 48.135 ppm correspond to C–N group. The chemical shift range of 51.37 to 56.11 ppm is indicative of certain types of carbon environments within a molecule that includes carbons in alcohol, ether, amine, or halide groups. The study initially conducted a qualitative assessment of antioxidant properties, which indicated promising antioxidant effectiveness. This led to a subsequent quantitative evaluation of antioxidant activity, revealing that the MEAOF possesses an antioxidant scavenging capacity of 87.96%. Interestingly, MEAOF exhibited a good spectrum of antibacterial activity against the screened human pathogenic organisms. The antibacterial efficacy was good in both Gram positive and Gram-negative bacteria (Table 2). The spectrum of activity was sequenced as Escherichia coli (22 ± 1.7 mm) > Bacillus subtilis (21.3 ± 1.5 mm) > Enterococcus faecalis (20.6 ± 2.5 mm) > Staphylococcus aureus (20.3 ± 2.5 mm) > Streptococcus pyogenes (19 ± 1.7 mm) > Pseudomonas aeruginosa (17.3 ± 1.5 mm) > Proteus vulgaris (17.3 ± 0.5 mm) > Salmonella choleraesuis (16.6 ± 3 mm) > Klebsiella pneumoniae (16.6 ± 1.5 mm).
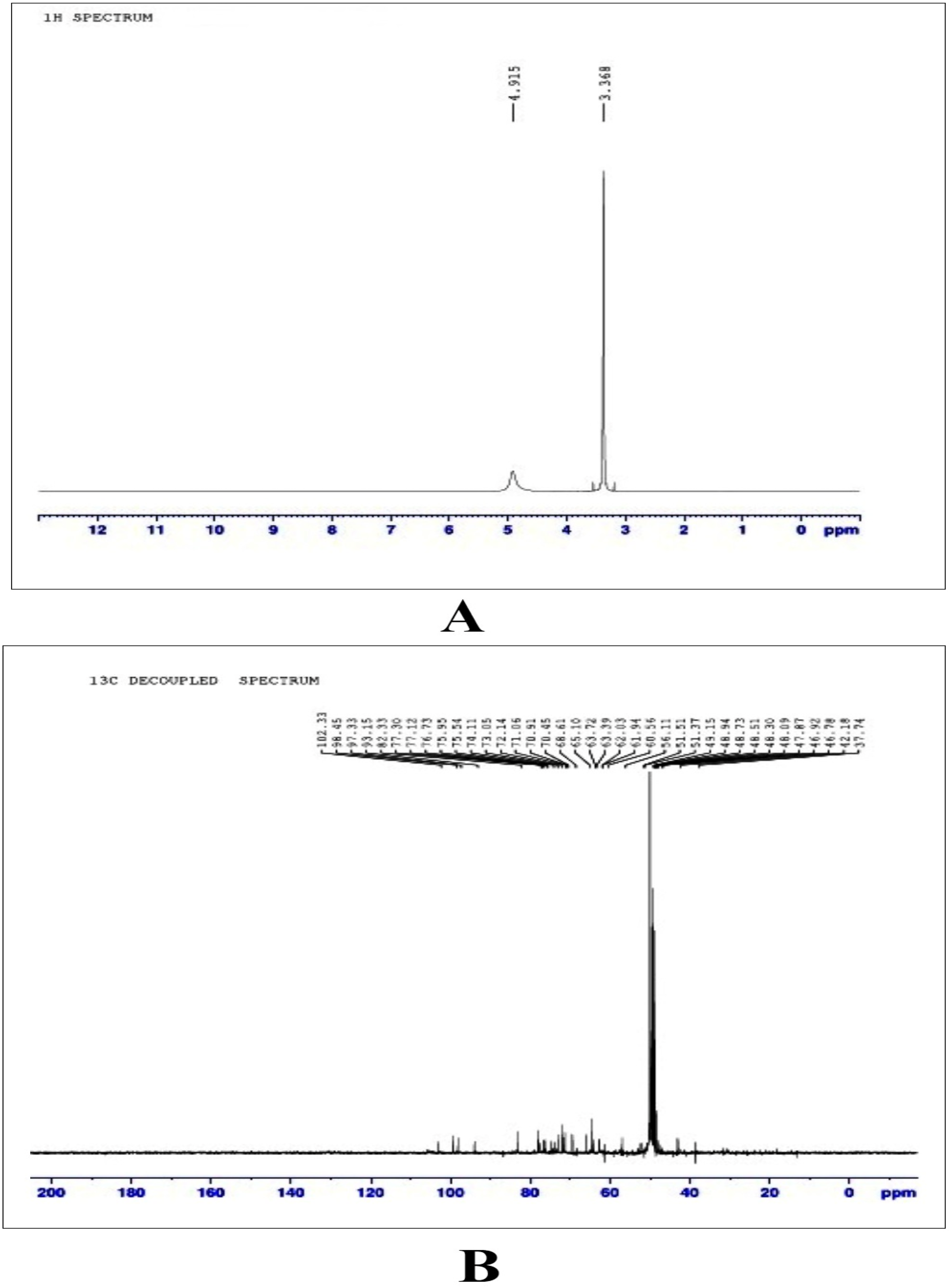
NMR study. (A) 1H NMR analysis of MEAOF (B) 13C NMR analysis of MEAOF.
Discussion and Conclusion
Bioactive compounds with diverse molecular structures and potential therapeutic applications are abundant in nature and natural products. Throughout history, plants have been harnessed for treating various illnesses, attributed to their rich reservoir of biologically active molecules possessing medicinal properties. 6 A variety of plants are used for their medicinal properties in folk remedies throughout Saudi Arabia, with a focus on the southern areas of the country. 12 The present research aimed to explore the bioactive components of MEAOF using GC-MS, LC-MS, FT-IR, and NMR analyses.
Despite its limitations, GC-MS was chosen for this study because it can analyze volatile compounds with high sensitivity and specificity. Although less effective for non-volatile or polar compounds, the technique provided valuable insights that significantly improved our understanding of the bioactive compounds in MEAOF. This study identified several bioactive compounds, including stearic acid, palmitic acid, 9,12- octadecadienoic acid (Z, Z), dasycarpidan-1-methanol, acetate, methyl benzoate, and stigmasterol. For instance, Palmitic acid (n-Hexadecanoic acid) was noted for its significant role in inhibiting prostate cancer cell proliferation and its antibacterial. Recent investigations underscore its significant role in impeding the proliferation of prostate cancer cells, evident in both laboratory experiments and animal models. This inhibitory mechanism involves cell cycle arrest at the G1 phase, marked by diminished levels of cyclin D1 and phosphorylated Rb protein, coupled with heightened expression of p27 protein, as elucidated in the research conducted by Zhu et al in 2021. 13
Furthermore, previous studies by Syed et al in 2022 and Maysa et al in 2020 have recognized palmitic acid for its broad-spectrum antibacterial and antifungal properties.14,15 It notably exhibits potent antibacterial activity against biofilm-forming bacteria.14,16 In previous research, we identified the presence of hexadecanoic acid, methyl ester, within the exudate gel of Aloe fleurentiniorum, a plant traditionally used for medicinal purposes by the people of Aridah and Fayfa mountains of Saudi Arabia. 6
The compound 9,12-octadecadienoic acid (Z, Z), also known as linoleic acid, was identified in MEAOF. Dietary intake of linoleic acid is associated with a lower risk of mortality from all causes, cardiovascular disease, and coronary heart disease. However, it may slightly increase the risk of cancer mortality. Conversely, higher blood levels of α-linoleic acid are associated with a lower risk of mortality from all causes and coronary heart disease. In addition, α-linoleic acid may have the added benefit of reducing blood clot formation.17,18 Dasycarpidan-1-methanol, acetate (ester) an alkaloidal compound has been identified in the MEAOF and the compound has been reported to exhibit anti-inflammatory, antibacterial, antifungal, and anticancer properties. 9 Methyl benzoate, commonly referred to as benzoic acid methyl ester, was identified in MEAOF. Methyl benzoate has remarkable insecticidal properties and acts both as a contact poison and as a fumigant. It also acts as an egg-laying deterrent and attracts certain insect species due to its natural odor. These properties make methyl benzoate a promising plant insecticide for integrated pest management strategies, providing effective pest control without negatively impacting human health or the environment. In the study, stigmasterol, a compound recognized for its potent anti-osteoarthritic properties by inhibiting pro-inflammatory mediators, was identified within the same MEAOF sample. The anti-inflammatory efficacy of stigmasterol has been supported by research findings.19,20
Octadecanoic acid, methyl ester, commonly known as methyl stearate, is an organic compound that belongs to the class of fatty acid methyl esters. In our previous research, we identified Octadecanoic acid, methyl ester in the exudate gel of Aloe fleurentiniorum. 6 Ethyl iso-allocholate, a steroidal compound, was determined MEAOF. An earlier study suggested that ethyl iso-allocholate, obtained from Trigonella foenum graecum L., induces caspase-dependent apoptosis in cancer cells. 21 Furthermore, it was found to decrease tumor growth, inhibit metastasis and angiogenesis in vivo, while also exhibiting safety for normal tissues. Significant interactions were observed between ethyl iso-allocholate and all protein targets, with a notable emphasis on caspase-1, indicating its therapeutic potential in terms of anti-inflammatory and anti-tumor effects. 22 Betulins are a naturally occurring triterpene that is found extensively distributed throughout the plant kingdom. Betulin displays enormous pharmacological potential, such as anti-inflammatory activity, anti-HIV, anticancer, and antibacterial properties.23–26
The FT-IR analysis identified specific peak patterns 3396, 2928.30, 1631.37, 1480.80, 1261.13, 1057.64, 866.69, 814.25, and 595.08 cm−1. The specific groups identified are associated with tannins, fatty acids, carbohydrates, steroids, phytosterols, and saponins. These patterns also suggest the presence of compounds like terpenoids and glycosides. Previously, the leaf extract of Aloe officinalis Forssk was found to display characteristic absorption peaks at 3454.3, 1635.52, 2351.1, 2065.6, 1041, 1251.7, 1402.2, and 572.8 cm−1. 27
Similarly, research has shown that the gel exudate from Aloe fleurentiniorum possesses a range of bioactive compounds, as evidenced by its distinct spectral fingerprint in the regions of 3354, 2945, 2832, 2523, 2046, 1707, 1451, 1108, 1031, 880, 737, and 610 cm−1, according to findings by Sivakumar et al, 2021. 6 Furthermore, a study by Saad et al, 2020, highlighted that the exudate gel from the flower of Caralluma retrospiciens (Ehrenb.) showcased numerous specific absorption bands at 3287.45, 2850.32, 2595.91, 2524.69, 2216.53, 2041.07, 1651.70, 1450.28, 1309.57, 1111.25, and 534.93 cm−1.
NMR spectroscopy was used in this study despite its limitations in analyzing mixtures due to signal overlap. Despite these challenges, NMR provided valuable insight into the composition of the mixture and helped to identify and quantify the organic compounds. This analysis revealed the presence of numerous bioactive substances in the MEAOF. The deshielded peak at 3.368 ppm signifies the existence of a methoxy group, while alkoxy proton presence implies the existence of aromatic amines. The 1H NMR shifts from 3.57 to 4.91 ppm indicate the presence of fatty acids with a terminal methyl group. Based on the results of a prior study, a shift in ppm concentration between 3.70 and 5.10 suggested the presence of protons associated with the glycerol component. 28 In the same investigation, it was noted that the presence of olefinic protons of unsaturated fatty acids was detected in the region of 5.2-5.5 ppm. However, the present study demonstrated the deshielded proton was able to determine up to 4.91 ppm in proton NMR.
The study initially conducted a qualitative assessment of antioxidant properties, which indicated promising antioxidant effectiveness. This led to a subsequent quantitative evaluation of antioxidant activity, revealing that the MEAOF possesses an antioxidant scavenging capacity of 87.96%. The presence of compounds such as palmitic acid, phytol, and 9,12- octadecadienoic acid (Z, Z) in MEAOF could be responsible for its antioxidant properties. This finding aligns with previous research, such as the study by Bouchra et al (2019), which highlighted the antioxidant potential of tannin extracts from the green rind of Aloe vera. 29
Similarly, research by Hu et al (2003) demonstrated a significant antioxidant effect of the extract of Aloe vera, with a free radical scavenging activity of 72.19%. 30 However, the current study surpasses these findings by showing a higher antioxidant level in the red flowers of Aloe officinalis, indicating its superior efficacy in combating oxidative stress. Interestingly, MEAOF exhibited a good spectrum of antibacterial activity against the screened human pathogenic organisms. The antibacterial efficacy was equal in both Gram positive and Gram-negative bacteria. The observed antibacterial effect may be due to the presence of bioactive compounds such as palmitic acid, dasycarpidan-1-methanol, acetate etc Similarly, an earlier study also suggested the antibacterial effect of these compounds present in Aloe vera leaves. 29 This study highlights the considerable therapeutic potential of Aloe officinalis Forssk and many indigenous plants from the Jazan and Asir regions of Saudi Arabia. In this investigation, detailed analytical techniques such as GC-MS, LC-MS, FT-IR, and NMR were used to identify several bioactive chemicals in MEAOF and highlight their significant therapeutic capabilities such as antibacterial, antioxidant, anticancer, and anti-inflammatory activities. Compounds such as palmitic acid, 9,12- octadecadienoic acid (Z, Z) and dasycarpidan-1-methanol acetate have strong antibacterial properties and are therefore promising for the development of new treatments to combat antibiotic resistance. Reactive oxygen species (ROS) in bacterial cells can interact directly with bacterial cell membranes, disrupting their integrity and causing cell lysis, thereby increasing the permeability of bacterial cells to MEAOF. MEAOF has attracted considerable attention due to its dual antioxidant and antibacterial properties. These extracts are rich in bioactive compounds that can neutralize ROS and attenuate oxidative stress, protecting cells from damage. In addition, MEAOF can interfere with bacterial signaling pathways and enzyme activities, inhibiting bacterial growth.31,32 This study highlights the significant therapeutic potential of Aloe officinalis Forssk flower, particularly its antibacterial and antioxidant properties, which are related to the presence of various bioactive compounds identified using advanced analytical techniques. Despite the limitations of GC-MS in analyzing non-volatile or polar substances, the results underline the importance of natural products in the development of new medical compounds, particularly in addressing the growing challenges of antibiotic resistance and oxidative stress-related diseases. This study emphasizes the crucial role of natural products and knowledge in promoting pharmaceutical innovation. Through scientific validation, this research adds to the global body of studies on desert plants and opens avenues for the development of effective natural-based therapeutics that may lead to breakthrough health strategies in the future.
Footnotes
Acknowledgements
The authors gratefully acknowledge the funding of the Deanship of Graduate Studies and Scientific Research, Jazan University, Saudi Arabia, through Project Number: RG24-S091.
CRediT Authorship Contribution Statement
Santhosh Joseph Menachery: Conceptualization, Funding acquisition; Sivakumar S. Moni: Conceptualization, Methodology, Investigation, Data curation, Formal analysis, Writing – original draft, Amani Khardali, Ali Ahmed Ali Albarqi, Hana F. Abdallah: Supervision, Resources Supervision, Resources, Project administration, Software; Mohamed Eltaib Elmobark, Mohammad Firoz Alam Dalia Daws, Razan Willie Areshyi: Validation, Hissah Ali Sofyani: Visualization, Supervision,Validation, Software.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This article does not involve any experiments or studies involving human or animal participants were conducted as part of this research. Therefore, ethical approval is not applicable for this article.
Funding
The authors disclosed receipt of the financial support from the Deanship of Graduate Studies and Scientific Research, Jazan University for the research, authorship, and/or publication of this article: This work was supported by the Deputyship for Research & Innovation, Ministry of Education in Saudi Arabia for funding this research work through the project number RG24-S091.
Statement of Human and Animal Rights
This article does not include any research involving human or animal participants and therefore, not applicable.
Statement of Informed Consent
Not applicable as this research did not involve experiments or studies with human or animal subjects.
