Abstract
Background
Hepatocellular carcinoma (HCC) is the most common form of liver cancer, with a recurrence rate of 80–90% and a high mortality rate. It is an inflammation-related cancer where cytokines production plays a major role, resulting in a non-resolving inflammation in the tumor microenvironment, which promotes the disease. Therefore, targeting inflammation is a logical way to combat HCC. Natural products can be helpful in the co-treatment and prevention of HCC.
Hypothesis
This study aimed to evaluate the hepatoprotective properties of a methanolic extract of Apium graveolens L. (MAG) in diethylnitrosamine (DEN)-induced toxicity in BALB/c mice.
Materials and Methods
We checked the antioxidant, anti-inflammatory, and anti-cancer properties of MAG. DEN is known to induce oxidative stress by increasing reactive oxygen species (ROS) production. This can result in liver damage, increased SGOT, SGPT, and ALP activity in serum, increased expression of HCC biomarkers like AFP and GPC-3, and increased levels of the inflammatory biomarkers NF-κB, IL-6, IL-4, IL-1β, and TNF-α. The above factors can cause the activation of the inflammatory signaling pathways, triggering the development of HCC.
Results
MAG was able to lower the detrimental effects of DEN by restoring liver function; decrease oxidative stress by increasing superoxide dismutase (SOD), catalase (CAT), glutathione peroxide (GPx), and ϒ-L-glutamyl-L-Cysteinyl-glycine (GSH); decrease the inflammatory factors responsible for HCC; and increase caspase-3 activity. Molecular docking studies showed how phytoconstituents like luteolin, apigenin, and kaempferol present in MAG could potentially be responsible for lowering the effects of DEN in the mice’s liver.
Conclusion
Altogether, the present study showed that MAG was able to ameliorate inflammation in the DEN-induced liver carcinogenesis in BALB/c mice. To the best of the authors’ knowledge, this is the first report on the use of a whole plant (Apium graveolens) in an anti-cancer study in a mouse model.
Introduction
According to Cancer Statistics 2021, hepatocellular carcinoma (HCC) is one of the most aggressive cancers and is the third leading cause of death worldwide (Llovet et al., 2021). The etiological factors of HCC are mostly associated with inflammation (Li et al., 2020). Diethylnitrosamine (DEN) is a well-known hepatocarcinogen that forms DNA-carcinogen adducts in the liver. It can induce HCC without cirrhosis through the development of hepatocellular focal lesions (Carlessi et al., 2019; Rajewsky et al., 1966). Humans are vulnerable to DEN exposure since it has been reported to be present in foods such as smoked dried fish, smoked meat, salted and dried fish, soybean, cheese, tobacco smoke, and groundwater (Afé et al., 2021; Sarkar et al., 2020). About 70%–90% of HCC cases developed as a result of chronic inflammation. The immunological response triggered in liver cells plays an important role in the initiation and progression of HCC (Kumar & Le, 2016). Non-resolving inflammation, which is distinguished by the continuous production of pro-inflammatory cytokines, is a major driver of HCC (Yu et al., 2018). Nuclear factor-kappa B (NF-κB), a transcription factor, represents a molecular link between inflammation and cancer. Its activation stimulates the production of several pro-inflammatory cytokines, like tumor necrosis factor-alpha (TNF-α) and interleukin-1 β (IL-1β). High serum levels of TNF-α and IL-6 have been reported to contribute to the pathogenesis of HCC (Liu et al., 2017). IL-6 has been reported to induce the activation of different HCC signaling pathways like phosphatidylinositol 3-kinase- protein kinase (PI3K-AKT), Janus kinase/signal transducers and activators of transcription 3 (JAK-STAT3), NF-κB, and so on (Johnson et al., 2018; Lokau et al., 2019). Studies have also reported that non-resolving inflammation is considered to be a major contributor to cancer initiation and progression. Therefore, analysis of known biomarkers of HCC, such as alpha-fetoprotein (AFP), glypican-3 (GPC-3), and immune-related biomarkers might be useful in the diagnosis and prognosis of HCC (Abdel-Hamid et al., 2018; Amin et al., 2020).
Inflammation-induced recurrence of HCC has been reported in a lot of cases, and therefore, the downregulation of the inflammatory response is very critical in HCC. Lowered inflammation also has a desirable protective effect on the liver (Refolo et al., 2020). In the current study, the commonly consumed plant Apium graveolens L. (commonly known as celery) was evaluated for its ability to promote hepatoprotection by lowering the inflammatory response in the DEN-treated mice. A. graveolens L. belongs to the Apiaceae family and is a medicinal herb used as food and in traditional medicine (Aćimović & Milić, 2017). The presence of flavonoids such as apigenin, luteolin, and kaempferol in the plant has been reported to contribute to its healing properties (Minaiyan et al., 2021). Therefore, the present study was undertaken to check the anti-inflammatory and anti-cancer activity of A. graveolens against DEN-induced mice.
Materials and Methods
Chemicals
Diethylnitrosamine (DEN), aspartate aminotransferase (AST), alanine aminotransferase (ALT), and alkaline phosphatase (ALP) kits were purchased from Coral Clinical System, Goa, India. Xanthine oxidase, cytochrome c, glutathione reductase, reduced ϒ-L-glutamyl-L-cysteinyl-glycine (GSH), acetylthiocholine iodide, and 5-5′-dithiobis-(-2-nitro benzoic acid) (DTNB) were purchased from Sigma Chemical Co., USA.
Crude Extract Preparation
The preparation of plant extract was done according to Hadem et al. (2014). A. graveolens (celery) was bought from the Agriculture Department of the Government of Meghalaya, India, and identified and specified by an expert in the Agriculture Department of Meghalaya. A voucher specimen has been kept for future reference at the Department of Agriculture. Plant roots were removed and discarded, and the remaining stalks with the leaves were used after washing them in running water and finally in distilled water. It was then chopped into small pieces and dried in an oven at 37 °C–40 °C for 8–10 days. After that, the dried samples of celery were ground in a mortar and pestle to obtain a fine powder. The powdered form of the sample was used to prepare the methanolic extract by dissolving 100 g in 250 mL of solvent (methanol:water in the ratio of 4:1, v/v) and stirring overnight. The next day, the extract was then filtered, and the filtrate was evaporated to dryness using the rotary vacuum evaporator. Finally, the sample was obtained as lyophilized yellowish-brown powder of the extract. This powder was then dissolved in sterile normal saline at a concentration of 1 g powder/10 mL of saline and centrifuged at 2000g for 15 min to obtain a clear supernatant, which was used as methanolic extract of Apium graveolens (MAG) for all the experiments.
Experimental Animals
Swiss albino BALB/c male mice, aged 6–8 weeks (22 ± 13 g), procured from an inbred colony of Pasteur’s Institute, Shillong, Meghalaya, have been used for the experiment. The animals were housed in polypropylene cages (n = 5/cage) at room temperature (25°C–28°C) and maintained under a 12-h light/dark cycle. They were provided with standard mouse pellets and drinking water ad libitum, and these conditions were maintained constant throughout the experiments.
Preparation of DEN
DEN (density: 0.95 g/mL) was used to induce HCC in mice. It was prepared in Millipore water and administered intravenously at a dose of 20 mg/kg body weight (bw) (Hadem et al., 2014).
Toxicity of MAG
MAG toxicity was assessed in four groups of six mice each. MAG was administered by intraperitoneal (i.p.) injection at doses of 100, 250, 500, and 1000 mg/kg bw. The mice in the control group received only sterile saline injections. Toxicity and mortality were recorded for up to 7 days. Biochemical alterations induced by MAG were analyzed in serum after 24 h of exposure (Hadem et al., 2014).
Experimental Design
Experimental mice were randomly divided into eight groups, each group consisting of five mice, and their periodic induction with DEN and MAG is depicted in Table 1.
Experimental Design.
Biochemical Analysis
Aspartate and alanine transaminases (AST and ALT) and ALP activities were determined in the serum using the enzyme assay kit according to the manufacturer’s instructions using the Blood Analyzer 3000 (Evolution India R8C Laboratory). The protein content was estimated by the method of Bradford (1976), using bovine serum albumin as a control. The assays of superoxide dismutase (SOD) (Fridovich, 1986), catalase (CAT) (Aebi, 1984), glutathione peroxide (GPx) (Wendel, 1981), and GSH levels (Owens & Belcher, 1965) were carried out as per the respective standardized methods.
Immunoblotting
For immunoblotting experiments in the present study, the protocol of Baruah et al. (2018) was followed. Briefly, liver tissues from different treatment groups were homogenized in a 0.25% sucrose solution. After that, it was centrifuged at 20,000g for 45 min at 4°C, and the whole protein extract was quantified using the Bradford reagent. The protein sample was transferred onto a nitrocellulose membrane by electrophoresis. Subsequently, membranes were blocked for 1 h in 5% non-fat dried milk (NFDM) at room temperature (r.t.), and then they were washed with TTBS and TBS for 5 min (×3). It was then probed with the primary rabbit antibody against NF-κB, IL-2, IL-4, IL-10, IL-6, IL-1β, and GAPDH (ABCAM, UK) (1:1000 dilution) overnight at 4°C. The washing was repeated with TTBS and TBS, as stated earlier. After applying a secondary goat anti-rabbit antibody for 3 h, the immunoblot was detected using HRP substrate solution and quantified using Thermo My Image Analysis software.
Assay of AFP, GPC-3 Biomarker, TNF-α, and Caspase-3 in Serum
Assaying the effect of MAG on serum and tissue levels of different biomarkers was performed by a simple retro-orbital bleeding technique. The blood was separated from the serum by centrifugation at 2000 g for 10 min at 4°C. The level of TNF-α, AFP, GPC-3, and caspase-3 in serum was determined following the instructions provided in the assay kit (Boster Elabscience, USA).
Preparation of Ligand for Docking
A literature survey was carried out for the compounds present in the MAG. The 3D SDF structures of the MAGs were obtained from the PubChem database (Kim et al., 2016; Li et al., 2020). These structures were converted to .pdb files using the Open Babel software (O’Boyle et al., 2011), followed by their conversion into .pdbqt files using the AutoDock application of MGL tools (Forli et al., 2016).
Preparation of Protein for Docking
The 3D structures of the proteins kelch-like ECH-associated protein (KEAP) and signal transducers and activators of transcription (STAT3) were obtained from the Protein Data Bank (
Screening of Compounds Using AutoDock Vina
The compounds were screened for their ability to dock with the KEAP and STAT3 proteins using the AutoDock Vina (Trott & Olson, 2010). The PERL script was used for scripting. Briefly, the pdbqt files of compounds and proteins, along with the grid parameters of the proteins, were incorporated into AutoDock Vina. The AutoDock Vina program was run using the cmd prompt (Sharma et al., 2009). The bound structures were visualized using PyMol software, and the 2D images of the docked poses were generated using Discovery Studio.
Protein-Receptor Docking Study
Protein-receptor interaction studies of KEAP and STAT3 with their corresponding cellular receptors both in the absence and presence of selected MAG polyphenols were based on a previous study by Basu et al. (2020) with minor modifications. We used the HDOCK webserver for carrying out the study (Huang & Zou, 2008; Huang et al., 2014; Yan et al., 2017; Yan et al., 2020). In the current study, the net binding energy obtained after the docking study was analyzed.
Statistical Analysis Used
Results were expressed as the mean ± SD for at least three analyses for each sample. Statistical analysis was performed by GraphPad Prism 6 Software using a one-way ANOVA analysis of variance followed by Tukey’s multiple comparison tests. Significance was set at p < 0.05 and p < 0.01.
Results
Effect of MAG and DEN on Body and Liver Weights
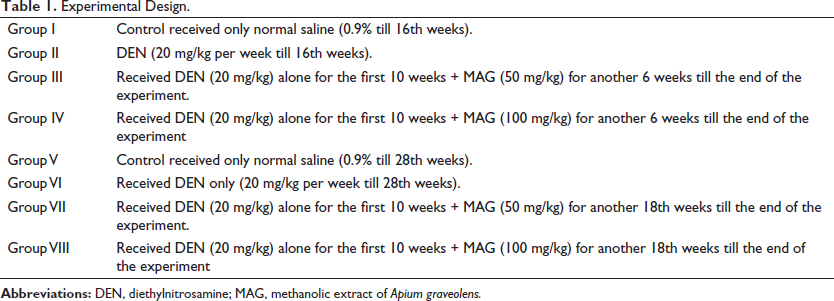
The liver weights (lws) and body weights (bws) are shown in Figure 1. When compared with the control groups, a significant increase **(p < 0.01) of lws and a remarkable decrease **(p < 0.01) in bws in the DEN-treated groups were observed. However, MAG-treated groups exhibited a trend of recovery ##(p < 0.01) toward the normal level in both lws and bws, in both treatment periods.
Toxicity Studies
Mice did not show any abnormalities/stress, such as circling, labored breeding, abdominal contractions, or mortality, after administration of MAG up to the dose of 800 mg/kg bw. AST activity exhibited a trend of increasing with higher doses of MAG, that is, 500 mg/kg bw onward. There was a significant **(p < 0.01) increase in its activity at 800 and 1000 mg/kg bw (Figure 1). The ALT activity of mice did not show any significant change up to a dose of 500 mg/kg bw. However, there was a significant **(p < 0.01) increase in its activity at 800 and 1000 mg/kg bw.
Effect of MAG Extract on Liver Morphology
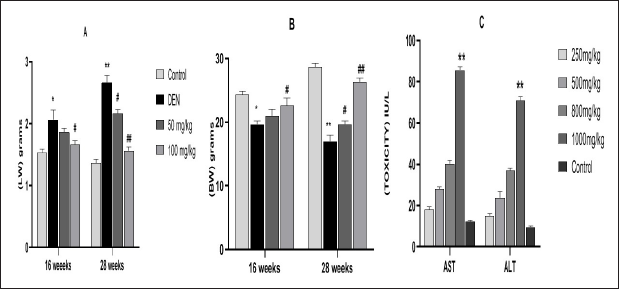
Figures 2A–F shows the morphology of liver tissues in different treatment groups. DEN treatment group showed the appearance of preneoplastic nodules and gray patch formation in Group II and Group IV mice. The nodules were significantly higher at 28 weeks **(p < 0.01) of DEN treatment alone compared to the DEN-exposed and MAG-treated groups. In this group, a decrease in nodule formation, which reverted close to control in Groups VII and VIII ##(p < 0.01), was observed.
Effect of MAG on Liver Marker Enzymes
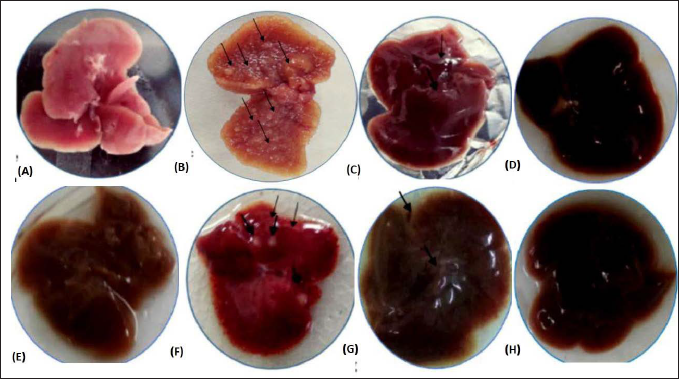
Liver enzyme levels are important indicators for hepatocyte damage. Figures 3A–C shows the level of liver enzyme activities such as AST, ALT, and ALP in the serum of the different treated groups compared with the control group. Mice exposed to DEN showed an increase in the level of these enzymes with a significant value of *(p < 0.05) in Group II and **(p < 0.01) in Group VI when compared to control mice. However, MAG treatment lowered the activity of the liver enzymes in a dose-dependent manner. More significant lowering was observed in the 28-week MAG-treated groups (VII and VIII) compared to the 16-week groups (III and IV). The level of enzymes in mice treated with MAG for 28 weeks (VIII) was close to a normal level, indicating amelioration of liver damage by the extract.
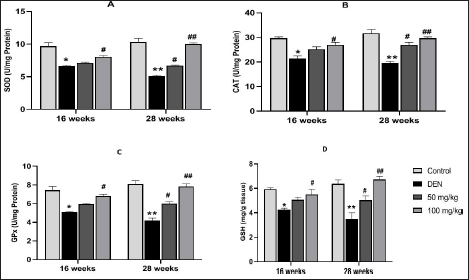
Effect of MAG on Antioxidant Enzymes Activities (SOD, CAT, and GPx) and Level of GSH
Figures 4A–D shows the antioxidant effect of MAG extract. Mice exposed to DEN exhibit significant inhibition of these enzymes in Group II with a p-value of
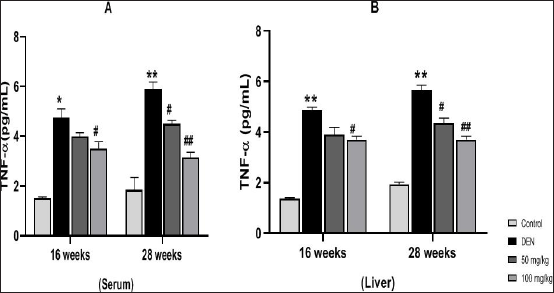
Effect of MAG on the Level of TNF-α
TNF-α is an important inflammatory mediator and an important serum prognostic marker for various types of diseases. Figures 5A and B showed that mice exposed to DEN resulted in a rapid accumulation of TNF-α in serum and liver tissue at 16 and 28 weeks of DEN exposure (Groups II and VI in A and B) in comparison to the control group. However, treatment with MAG extract significantly reduced the level of TNF-α in both liver and serum samples (Groups IV, VII (#p < 0.05) and Group VIII (##p < 0.01)).
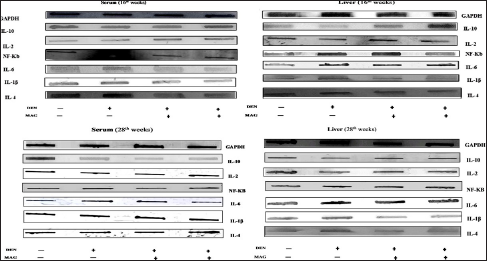
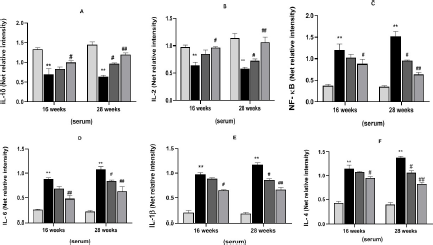
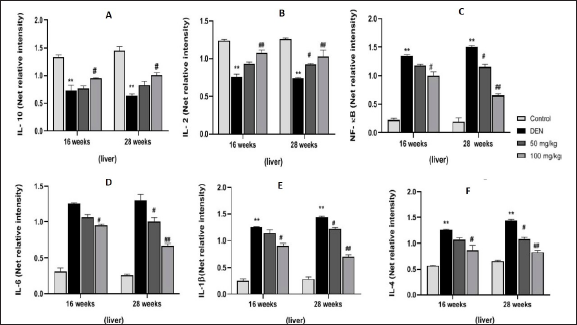
Effect of MAG on Levels of Inflammatory Cytokines
Figure 6 shows immunoblotted images, and Figures 7A–F and 8G–L show the level of expression of NF-κB, inflammatory mediators, IL-10, IL-2, IL-6, IL-1β, and IL-4 in the different treatment groups in both serum and liver samples. Intraperitoneal induction of DEN (20 mg/kg) leads to the activation of NF-κB in the nuclear extract of the liver, followed by transcription of pro-inflammatory cytokines. Thus, we observed a significant increase in NF-κB, IL-1β, IL-6, and IL-4 concentration levels in Group II (*p < 0.05) and Group VI (**p < 0.01) in serum samples, and the same trend was seen in liver samples at 16 and 28 weeks of the treatment period. In contrast, we observed a significant decrease in the levels of IL-10 and IL-2 in Group II (*p < 0.05) and Group VI (**p < 0.01) in both samples at both treatment periods. MAG extract significantly attenuated the levels of NF-κB and pro-inflammatory cytokines as shown in Groups IV and VIII (
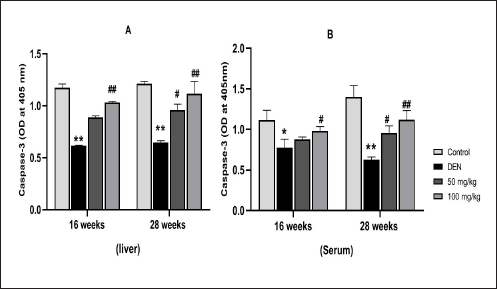
Effect of MAG and DEN on Apoptosis by Evaluating Caspase-3 Activity
Hepatoprotection by MAG was further confirmed by apoptotic studies. Figures 9A and B shows caspase-3 activity in liver and serum samples. MAG treatment resulted in the activation of caspase-3 in a dose-dependent manner, with the highest activity at 100 mg/kg bws at both 16 and 28 weeks of DEN-exposed and MAG-treated groups compared to DEN alone groups (Groups IV and VIII vs. Groups II and IV at 16 and 28 weeks of treatment).
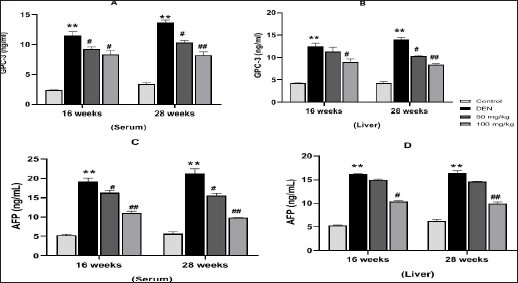
Effect of MAG on Levels of AFP and GPC-3 HCC Biomarkers
HCC biomarkers are important for HCC surveillance. Figures 10A and B shows levels of GPC-3 and Figures 10C and D shows levels of AFP in serum and liver. DEN exposure (20 mg/kg) significantly increased the levels of AFP and GPC-3 (Groups II and VI) with a significant value (**p < 0.01) in both samples. However, the levels of these biomarkers decrease in the MAG treatment groups (VIII), the highest being at a dose of 100 mg/kg bw in comparison to the DEN alone group. MAG was more effective in the 28th week’s treatment period as compared to the 16th week. The expression of these biomarkers in tumor cells was strikingly similar to that observed in the serum. These results indicated the anti-cancer activity of MAG against HCC, which can also be monitored in serum in a non-invasive manner.
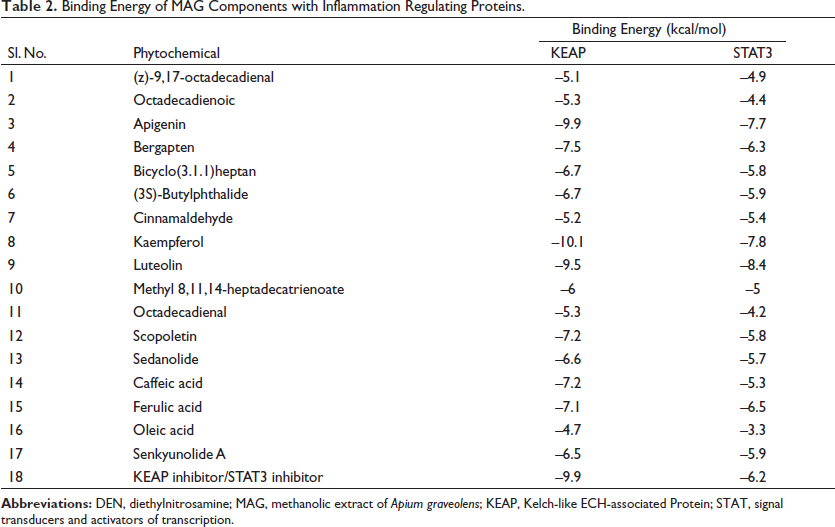
MAG Components Are Bound Favorably with Inflammation-regulating Proteins
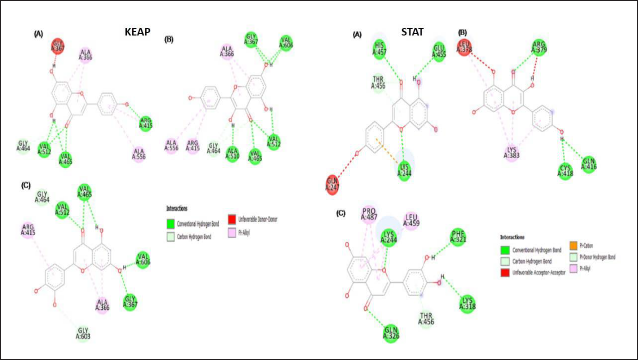
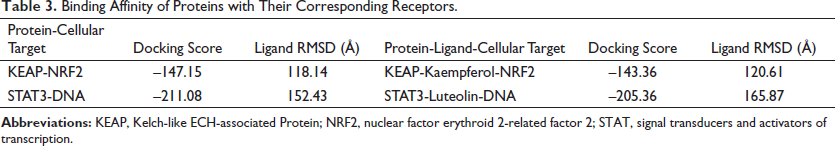
A literature search showed the presence of several compounds in the MAG (Kooti & Daraei, 2017) (Table 2). The AutoDock Vina-based screening revealed the favorable binding of most of the components of MAG with the KEAP and STAT3 proteins. The components apigenin, kaempferol, and luteolin showed the best binding with all three proteins. Their binding values were comparatively better than the binding energies of the commonly established inhibitors of these proteins, indicating the feasibility of their binding (Table 2). The 2D images of some of the best docking poses have been shown in Figure 11. Kaempferol showed the best binding with the KEAP protein, forming H-bonds with five amino acid residues. Luteolin formed the best docking with the STAT3 protein and formed H-bonds with four amino acid residues of the STAT3 protein, as shown in Figure 11. The protein-receptor binding analysis showed that the flavonoids kaempferol and luteolin interfered with the binding of KEAP and STAT3 to their respective cellular targets, which happen to be nuclear factor erythroid 2-related factor 2 (NRF2) and DNA, as evidenced by the increased binding energy (Table 3).
Binding Energy of MAG Components with Inflammation Regulating Proteins.
Binding Affinity of Proteins with Their Corresponding Receptors.
Discussion
The present study aimed to investigate the protective effect of a MAG against DEN-induced HCC in BALB/c male mice. As a vital organ of the body, the liver performs functions such as drug metabolism, digestion, and xenobiotic removal, as well as acting as a filter for toxins, making it susceptible to toxic overload that can cause inflammation and lead to cancer (Luo & Lin, 2021). An enlarged liver is a sign of an underlying problem, which could be inflammation. Increased liver weight and decreased body weight are two of the potential symptoms of HCC. DEN-induced hepatocellular damage can also be seen in the formation of nodules in the liver of DEN-treated mice, which is an indicative sign of a cirrhotic liver (Assy et al., 2009). The biometabolism of DEN by CYP2E1 generates highly reactive free radicals, resulting in oxidative stress and producing promutagenic products that are responsible for hepatocarcinogenesis. DEN administration resulted in an increment of the serum stress-specific enzymes (SGOT, SGPT, and ALP), indicating the toxic effect of DEN on the liver tissue, resulting in membrane permeability and leakage of these enzymes in the bloodstream (Pradeep et al., 2010). Also, it resulted in a significant elevation of oxidative stress, which is marked by a decreased level of antioxidant enzymes, thus disturbing redox homeostasis. On the contrary, treatment with MAG significantly reduced the level of liver enzymes and restored the content of antioxidant enzymes (SOD, CAT, GPx, and GSH) that are close to the normal range. Therefore, it can be concluded that the hepatoprotective activity of MAG against DEN-induced oxidative stress may be attributed to the presence of the phytoconstituents in MAG extract, which significantly repaired and reduced enzymatic leakage by ensuring membrane integrity and exerting as a powerful scavenger of reactive oxygen species (ROS) by protecting GSH reduction and potentiating of the enzymatic antioxidant defenses (Imran et al., 2019; Sukketsiri et al., 2016).
HCC is an inflammation-associated cancer (Todoric et al., 2016). As a consequence of DEN administration, pro-inflammatory cytokines such as TNF-α, IL-1β, IL-4, and IL-6 are significantly elevated in the liver. TNF-α and IL-6 induce NF-κB phosphorylation, thus increasing NF-κB protein expression, which further positively modulates the production of inflammatory mediators. High NF-κB expression in DEN-treated mice indicates the existence of chronic inflammation, tissue damage, tumor progression, and inhibition of apoptosis in cancer cells (Yu et al., 2020). A high level of expression of these inflammatory mediators and NF-κB indicates a continuous activation of transcription factor NF-κB, therefore, a continuous production of inflammatory factors, resulting in unresolved inflammation. Non-resolving inflammation is considered to be a hallmark of the development and progression of HCC. Indicating uncontrolled inflammation may be detrimental (Yu et al. 2018). In contrast to DEN, MAG administration significantly increases anti-inflammatory protein expression; IL-10 significantly suppresses tumor growth and inhibits NF-κB and STAT-3, which are the main links between inflammation and cancer (Cevey et al., 2019; Zhao et al., 2021). An increased level of anti-inflammatory cytokines indicates the ability of MAG to significantly attenuate the level of inflammatory mediators, subsequently lowering inflammation and thus playing a major role against DEN-induced tumorigenesis. Phytoconstituents’ presence in MAG is responsible for the anti-inflammatory effect as they could inhibit the transcription of inflammatory factors, lowering pro-inflammatory cytokines (Sivaramakrishnan et al., 2008). Our molecular docking studies have shown that STAT3 is inhibited by these flavonoids, which might explain the anti-inflammatory property of MAG.
The components of apigenin, kaempferol, and luteolin were reported to be present in A. graveolens and showed highly favorable binding with the KEAP protein. In our study, kaempferol binds to critical amino acids like ARG 415, which are directly involved in interaction with the NRF2 protein. Previous studies have shown that the binding of compounds with ARG 415 disrupts the interaction between KEAP and NRF2, leading to increased NRF2 activity (Davies et al., 2016). Flavonoids like resveratrol have previously been shown to interrupt the interaction between KEAP and NRF2, leading to increased NRF2 activity (Suraweera et al., 2020). NRF2 promotes the expression of antioxidant enzymes like SOD, CAT, and GPx (Wei et al., 2019). The increase in the activity of antioxidant enzymes like SOD, CAT, and GPx in the present study is indicative of relieving the KEAP-mediated suppression of NRF2 activity by the components in MAG.
The JAK-STAT3 pathway promotes the expression of pro-inflammatory cytokines (Salas et al., 2020). Luteolin, apigenin, and kaempferol demonstrated good binding with the STAT3 protein in its DNA binding region. Targeting the DNA binding region of STAT3 by small molecules has been shown to inhibit the activity of the STAT3 protein (Huang et al., 2014). The in vivo results of this study yielded further credibility to the docking study. There was a decrease in the levels of pro-inflammatory cytokines such as IL-6 and IL-4 in the MAG-treated mice. These further showed that the MAG components luteolin, apigenin, and kaempferol could carry out their anti-inflammatory effects by inhibiting the STAT3 protein.
Evading apoptosis is one of the hallmarks of cancer cells, as it allows the cell number to build up and increase tumor mass (Fouad & Aanei, 2017). The current study showed a low order of hepatic caspase-3 expression in the DEN-exposed mice group, indicating apoptosis evasion. Elevated NF-kB expression in DEN-treated mice promotes cell survival, therefore protecting cell death (Yu et al., 2020). MAG treatment increases apoptosis by inhibiting NF-kB, thus inducing cell death, and subsequently increases caspase-3 activity in a dose-dependent manner, thus showing its pro-apoptotic activity.
The current study revealed that the supplementation of DEN initiated the formation of HCC in DEN-exposed mice. This was proved by the elevated levels of AFP and GPC-3, an established biomarker for HCC. They are found to be elevated in HCC patients and adult animals when exposed to hepatocarcinogens (Zheng et al., 2018). A significant rise in AFP and GPC-3 levels was observed in the DEN group, indicating the manifestation of HCC (Zhang et al., 2018). MAG administration was able to significantly reduce the level of both of these biomarkers, indicating its inhibitory and anti-cancer potential against HCC (Hu et al., 2019).
Conclusion
In the present study, it was shown that the MAG exhibited potent antioxidant and anti-inflammatory activities, thus countering the detrimental effect of ROS, the toxic byproducts of aerobic metabolism. It appears to do so by preventing chronic inflammation, which is conducive to the development of cancer. Inflammatory biomarkers can act as potential predictive biomarkers for HCC, as non-resolving inflammation promotes HCC. It has been found that anti-inflammatory therapies are one of the most effective approaches to treat the disease. Molecular docking studies revealed that phytoconstituents such as apigenin, luteolin, and kaemferol are the ones responsible for the anti-oxidants, anti-inflammatory, and possibly anti-cancer properties in the MAG. A dose of 100 mg/kg showed better efficacy at countering DEN-induced HCC. The recurrence rate in HCC is known to be high. However, the 28th-week treatment resulted in better restoration, significantly closer to the control group. This suggests that the longer the intake of MAG, the better it is against inflammation, thereby preventing the recurrence of HCC. MAG is effective, safe, and cheap against the development of HCC with fewer side effects. Our findings in the present study demonstrated that a MAG does have the potential to inhibit inflammation triggered in HCC and perhaps can prevent its occurrence.
Footnotes
Summary
With more than 1 million cases expected by 2025, HCC, the most frequent kind of liver cancer, will continue to pose a health threat on a global scale. Despite being a fatal condition, early detection increases the likelihood of a successful course of therapy, such as the removal of a malignant tumor or a liver transplant. Patients are typically identified with HCC at advanced stages, which restricts their options for treatment. Other therapies work to lessen the symptom by trying to reduce or slow the tumor’s growth. According to studies, eating more fruits and vegetables can reduce your risk of developing cancer. By repressing the processes involved in cancer formation and boosting the mechanisms involved in cancer prevention, phytochemical substances found in plants may offer a therapeutic strategy for new cancer treatments. A variety of phytochemicals are present in the MAG, which is known to have anti-inflammatory and antioxidant properties. By scavenging free radicals generated during DEN administration, our results show that MAG can greatly reduce inflammation, a trait associated with cancer. It also significantly enhances endogenous antioxidant enzymes, thereby restoring redox balance. By suppressing the transcription factor NF-κB, which lowers the expression of pro-inflammatory mediators, MAG seems to considerably reduce inflammation. As a result, they play a critical role in the prevention of DEN-induced hepatocarcinogenesis. Our molecular docking experiments showed the mechanism of action of the constituents of MAG in the process of inhibiting inflammation and elevating antioxidant status. The administration of MAG demonstrated its pro-apoptotic action by raising caspase-3 activity, which causes cell death and reduces tumor mass. With DEN delivery, the HCC biomarkers AFP and GPC-3 were found to be raised, but they were considerably decreased after MAG treatment, demonstrating the anti-cancer potential of MAG. In conclusion, the phytoconstituents of MAG such as apigenin, luteolin, and kaempferol are perhaps responsible for their antioxidant, anti-inflammatory, and anti-cancer potential, indicating their therapeutic potential. Statistics show that the current cutting-edge therapeutic techniques and therapy regimen are partially effective, with a high recurrence rate of 70%–80% within 5 years following surgery in HCC. As a result, the therapeutic use of dietary supplements has drawn more attention due to its reduced risk of adverse effects and minimal toxicity to nearby cells. Perhaps, in the near future, plant-based therapeutics against HCC could become a reality and be universally accepted for maintaining human health.
Abbreviations
AFP: alpha-fetoprotein; AKT: protein kinase B; bws: body weights; CAT: catalase; CYP2E1: cytochrome-p450 family 2 subfamily E member 1; GPC-3: glypican-3; GPx: glutathione peroxide; GSH: ϒ-L-glutamyl-L-cysteinyl-glycine; HCC: hepatocellular carcinoma; IL: interleukin; JAK-STAT: Janus kinase/signal transducers and activators of transcription; LWS: liver weights; MAG: Apium graveolens methanolic extract; NF-κB: nuclear factor kappa B; NRF2: Nuclear factor erythroid 2-related factor 2; PI3K: phosphatidylinositol 3-kinase; ROS: reactive oxygen species; SOD: superoxide dismutase; TNF-α: tumor necrosis factor-alpha.
Acknowledgments
The authors would like to thank the support from the Department of Biochemistry, NEHU, Shillong, India, for all the necessary support to carry out the research work.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The work included in the submitted manuscript has been approved by the Institutional Ethics Committee (Animal Models) of North-Eastern Hill University, Shillong, Meghalaya, India.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
