Abstract
Introduction
Primary Sjögren’s syndrome (pSS) is a chronic inflammatory disease primarily affects exocrine glands dysfunction. Oxidative stress (OS) is a phenomenon occurring as a result of an imbalance between the generation of free radicals and antioxidant defense system. Hence, we aimed to establish the status of OS and inflammatory response according to the pSS disease activity index. In this context, we investigated malondialdehyde (MDA), and antioxidant enzymes during pSS. The possible association between MDA and nitric oxide (NO) levels and between MDA and some pro-inflammatory cytokines (IL-1β, IL-6, TNF-α, and IL-33).
Methods
The study has been conducted on 53 pSS patients. The antioxidant enzymes, represented by glutathione peroxidase (GSH-Px), catalase (CAT) and superoxide dismutase (SOD), were estimated by a colorimetric activity kit. Whereas, MDA value was assessed by measuring thiobarbituric acid reactive substances. Moreover, pro-inflammatory cytokines (IL-1β, IL-6, TNF-α, and IL-33) and NO were respectively quantified by enzyme-linked immunosorbent assays (ELISA) and the modified Griess.
Results
Interestingly, we report a notable reduction in our pSS patients’ antioxidant enzyme activity, while NO, MDA and proinflammatory cytokines values were significantly increased. pSS patients with higher disease activity had much stronger increases in NO and MDA levels. No significant difference was assessed in CRP level. Additionally, substantial significant correlations between plasmatic NO and MDA levels and between MDA, NO and IL-1β, IL-6, TNF-α cytokines were reported. However, no significant association was found between NO, MDA and IL-33 concentrations.
Conclusion
Collectively, our data showed altered oxidant-antioxidant balance in pSS patients. MDA, NO, IL-1β, IL-6, TNF-α seem to be good indicators in monitoring disease activity. Oxidative stress was closely related to inflammation in pSS. Exploiting this relationship might provide valuable indicators in the follow-up and prognosis of pSS with a potential therapeutic value.
Introduction
Sjögren’s syndrome (SS) is a chronic systemic autoimmune condition, essentially defined by an inflammatory infiltration of the exocrine glands (primary SS [pSS]). This condition leads to xerostomia and xerophtalmia symptoms.1,2 Other systemic connective tissue diseases may coexist with SS. In this situation, secondary SS (sSS) is then diagnosed.
The clinical spectrum of SS expands from sicca symptoms in the affected tissues such as eyes and mouth to extra-glandular systemic symptoms, which are associated with either immunocomplexes deposition (cutaneous vasculitis, hypocomplementemia, and peripheral neuropathy) or lymphoepithelial infiltrates in parenchymal organs. The most significant concern and the main factor impacting the mortality rate for SS patients is the development of B-cell non-Hodgkin lymphoma.3,4
As many other complex disorders, SS seems to occur from a confluence of genetic and environmental factors along with aberrant autoimmune responses, leading to B and T cell hyperactivity, autoantibody production causing damages target organs.5,6 In fact, SS is induced by intricate relationships between various immune cell types. In these conditions, cytokines are considered as essential components of this communication network. Abundant evidence now supports that cytokines imbalance contributes to immune dysfunction, triggers inflammation, and induce tissues damage during pSS.
Moreover, a number of strong lines of evidence highlight the involvement of a crucial interplay between changes induced by the immune-inflammatory responses and oxidative stress (OS). Chronic inflammation and disturbance in immune homeostasis might lead to more marked OS and more severe cell injury. 7
Oxidative stress represents the imbalance between pro- and anti-oxidant systems, occurring with an increased secretion of reactive nitrogen which includes essentially nitric oxide (NO), and peroxynitrite (ONOO−); or oxygen species (ROS and RNS) or a decline in the ability of antioxidative defense system, leading to alteration in the function of different components and signaling pathways.
The relationship between oxidative injury, which is mainly characterized by malondialdehyde (MDA), and inflammation is tight because the stress could be originated by the inflammasome and the TLRs signaling. 8 Growing evidence indicates that C-reactive protein (CRP) levels augment significantly in response to inflammation, injury and, infection, and exhibit elevated expression during inflammatory disease. 9 Similarly, it has been reported that OS is elevated in patients with autoimmune disease, owing to overproduction of free radicals.10,11 The antioxidant system includes antioxidant components like glutathione (GSH) and diverse antioxidant enzymes, like catalase (CAT), superoxide dismutase (SOD), peroxiredoxins (PRXs), and glutathione peroxidases (GSH-Px).12,13 SOD constitutes an antioxidant enzyme converting superoxide anion (O2−) into O2 and H2O2. CAT and GSH-Px are also antioxidant enzymes, catalyzes the dismutation of H2O2. Myeloperoxidase (MPO) converts H2O2 into hypochlorous acid (HOCl), a potent cytotoxic oxidizing agent. 14
In this context, less is known about the implication of OS in pSS. Studies of oxidative injury during pSS are limited. In this sense, the purpose of our study was to situate the status of OS characterized by MDA, and antioxidant enzymes in relation to the pSS disease activity index. In the same way, we performed correlation studies to assess the potential interconnection between OS markers, nitric oxide, and pro-inflammatory cytokines.
Patients and methods
Study subjects
Clinical and laboratory characteristics of patients with primary Sjögren’s according to the ESSDAI score.

Abbreviation: SD, standard deviation. (pSS-I:Low; pSS-II:moderate; and pSS-III: high disease activity); ESSDAI: EULAR Sjögren’s Syndrome Disease Activity Index
Plasma analysis
CRP levels have been quantified using nephelometric assay, while, anti-SSA, anti-SSB and antinuclear (ANA) Abs have been performed according to a standard protocol.
Assessment of antioxidant enzymes
After an overnight fast, blood samples were aseptically taken in the morning and centrifuged for 10 minutes at 3000 r/min. Samples were than kept at −80°C until analysis. The levels of antioxidant enzymes were measured in accordance to the instructions of manufacturer. In a spectrophotometer, the absorbance was measured at 340 nm for serum glutathione peroxidase (MAK437-1 KT, Sigma-Aldrich, St Louis, MO, USA), at 540 nm for catalase (219,265-1KIT, Sigma-Aldrich, St Louis, MO, USA), and at 450 nm for superoxide dismutase (19,160-1KT-F, Sigma-Aldrich, St Louis, MO, USA).
Lipid peroxidation analysis
Lipid peroxidation was quantified using the determination of thiobarbituric acid reactive compounds (TBARS) in plasma. 18 Following the MDA reaction with thiobarbituric acid, the absorbance at 532 nm was detected using a spectrophotometer.
Nitric oxide levels assessment
Plasma nitrites NO2− as an index of plasma NO concentrations, were evaluated by the modified Griess method as described previously. 19 The absorbance was assessed in a spectrophotometer at 543 nm. The amounts of nitrites were calculated relative to standard curve.
Cytokines assay
Plasma IL-6, TNF-α, IL-1β, and IL-33 levels (sensitivity: 1.6, 1.7, one and 0.2 pg/mL, respectively) were assessed with commercial ELISA kit following the instructions provided by the manufacturer (Thermofisher scientific®). The concentrations were evaluated relative to standard curves.
Statistical analysis
All results were reported as mean ± standard deviation (SD). Statistical analyses were performed using Graph-Pad PrismR software (GraphPad Inc., La Jolla, CA, USA). Statistical significance was evaluated by Student’s t-test for normal distribution and Mann–Whitney U test for abnormal distribution of unpaired variables. Correlations between variables were performed by Pearson’s correlation analysis for normally distributed data. Otherwise, Spearman’s rank correlation test was used. A
Results
Lipid peroxidation, CRP and nitric oxide analysis
As displayed in Figure 1, our data revealed that plasma NO and MDA in pSS patients were considerably higher when compared to HC ( Plasma NO2-, MDA and CRP levels in healthy controls (HC, 
Antioxidant enzyme assays
As reported in Figure 2. Our data indicated that SOD (EC 1.15.1.1), GSH-Px (EC 1.11.9) and CAT (EC 1.11.1.6) activities were significantly decreased in patients with pSS, when compared to HC ( CAT, SOD and GSH-Px activities from healthy controls (HC, 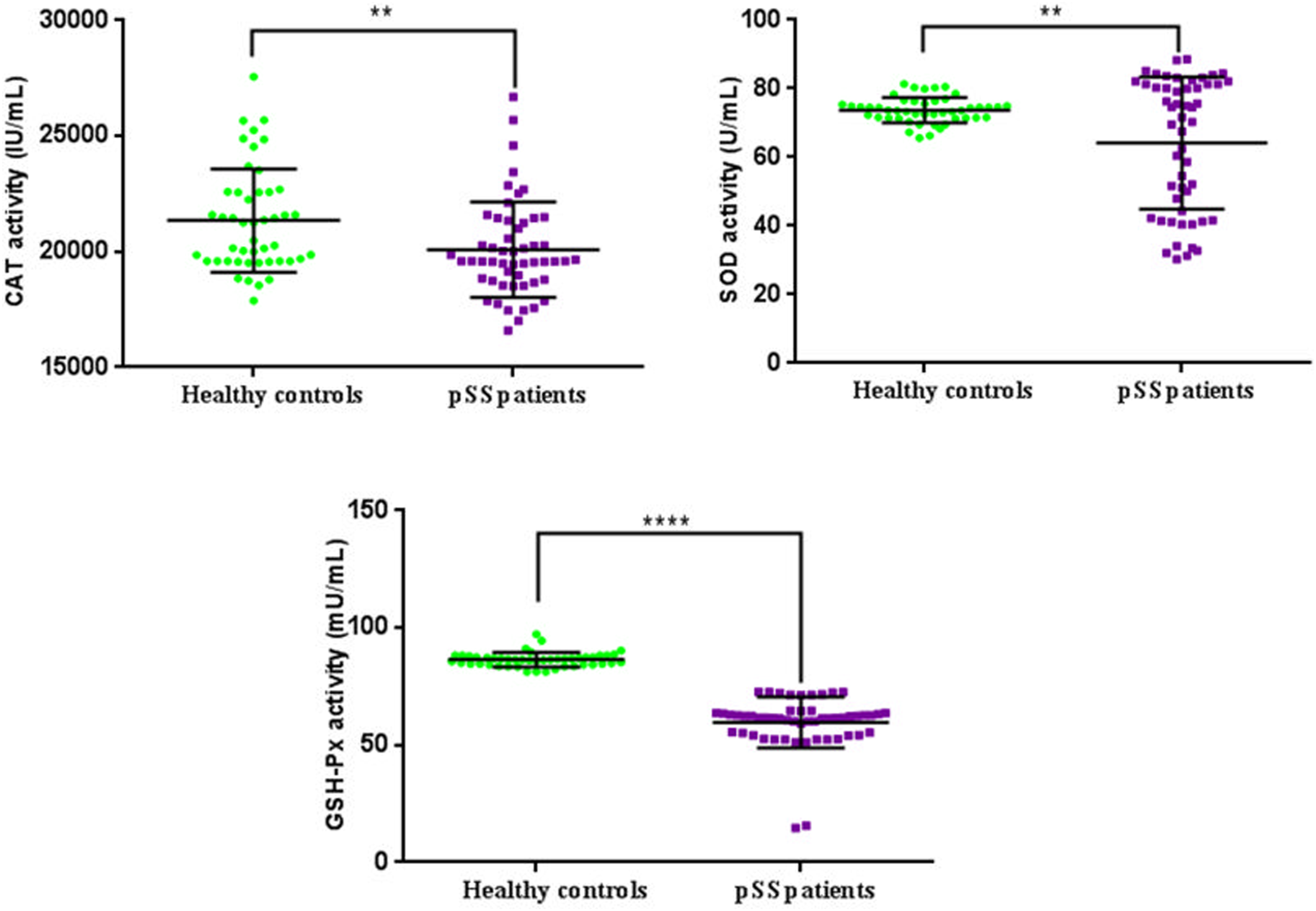
Plasma cytokines levels
As seen in Figure 3, IL-1β, IL-6, TNF-α, and IL-33 plasma values were significantly elevated in pSS patients in comparison to HC ( IL1-β, IL-6, TNF-α and IL-33 plasmatic levels in healthy controls (HC, 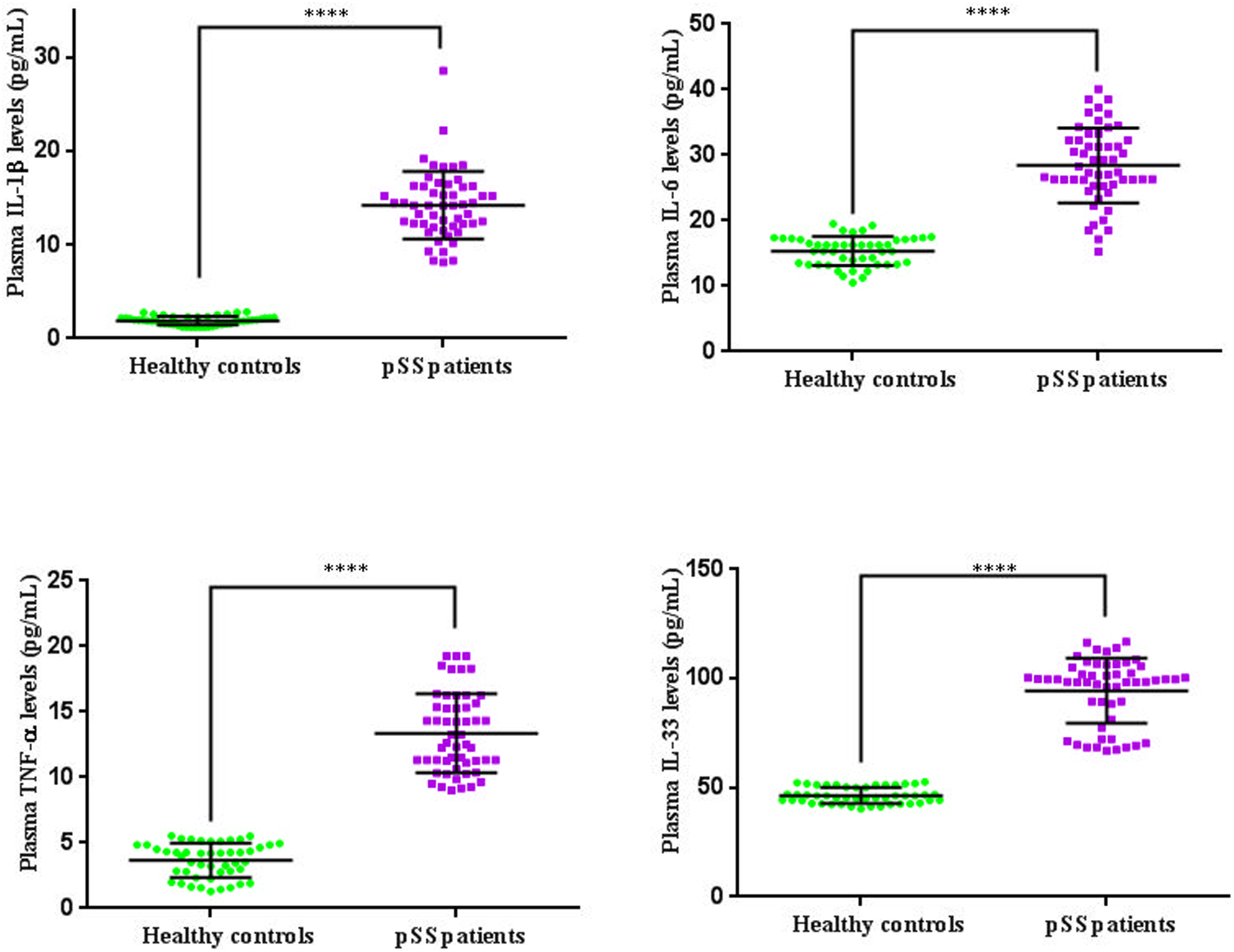
Cytokines and oxidative stress variables measurement according to the pSS disease activity state
Cytokines and oxidative stress variables measurement according to the ESSDAI score.

Abbreviation: SD, standard deviation. (pSS-I:Low; pSS-II:moderate; and pSS-III: high disease activity)
Correlation studies
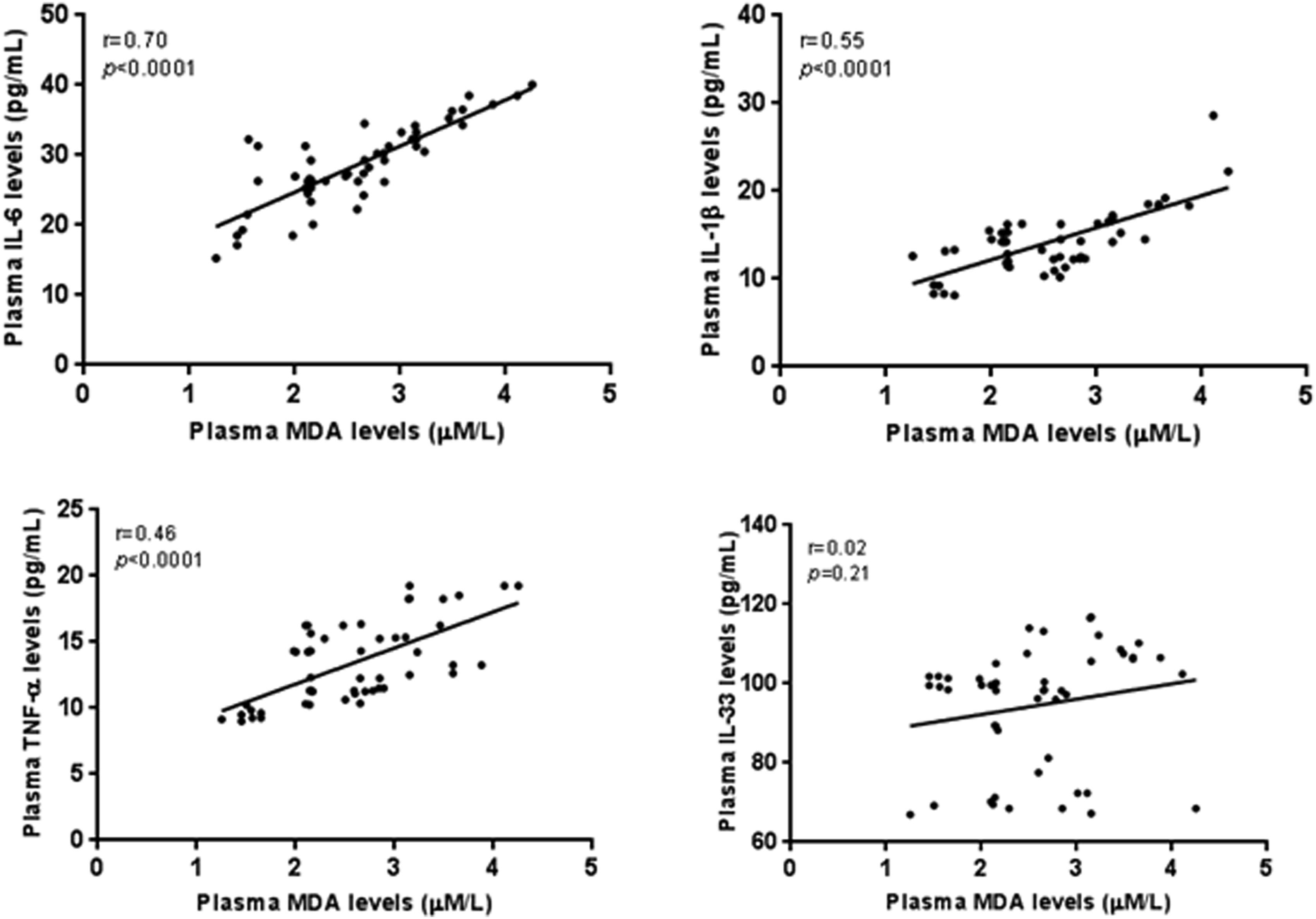
Our data indicated, a potent positive correlation between NO and MDA concentrations among either pSS patients ( (a) Correlation between NO2- and MDA levels. Plasma production of NO2- strongly correlated with MDA levels (r = 0.66, Correlation between IL-6, IL1β, TNF-α, IL-33 and MDA levels. Plasma production of IL-6, IL1-β, TNF-α and IL-33 positively correlated with MDA levels (r = 0.70, Correlation between IL-6, IL1β, TNF-α, IL-33 and NO2- plasma levels. Plasma production of IL-6, IL1β, TNF-α and IL-33 positively correlated with NO2- levels (r = 0.59, 

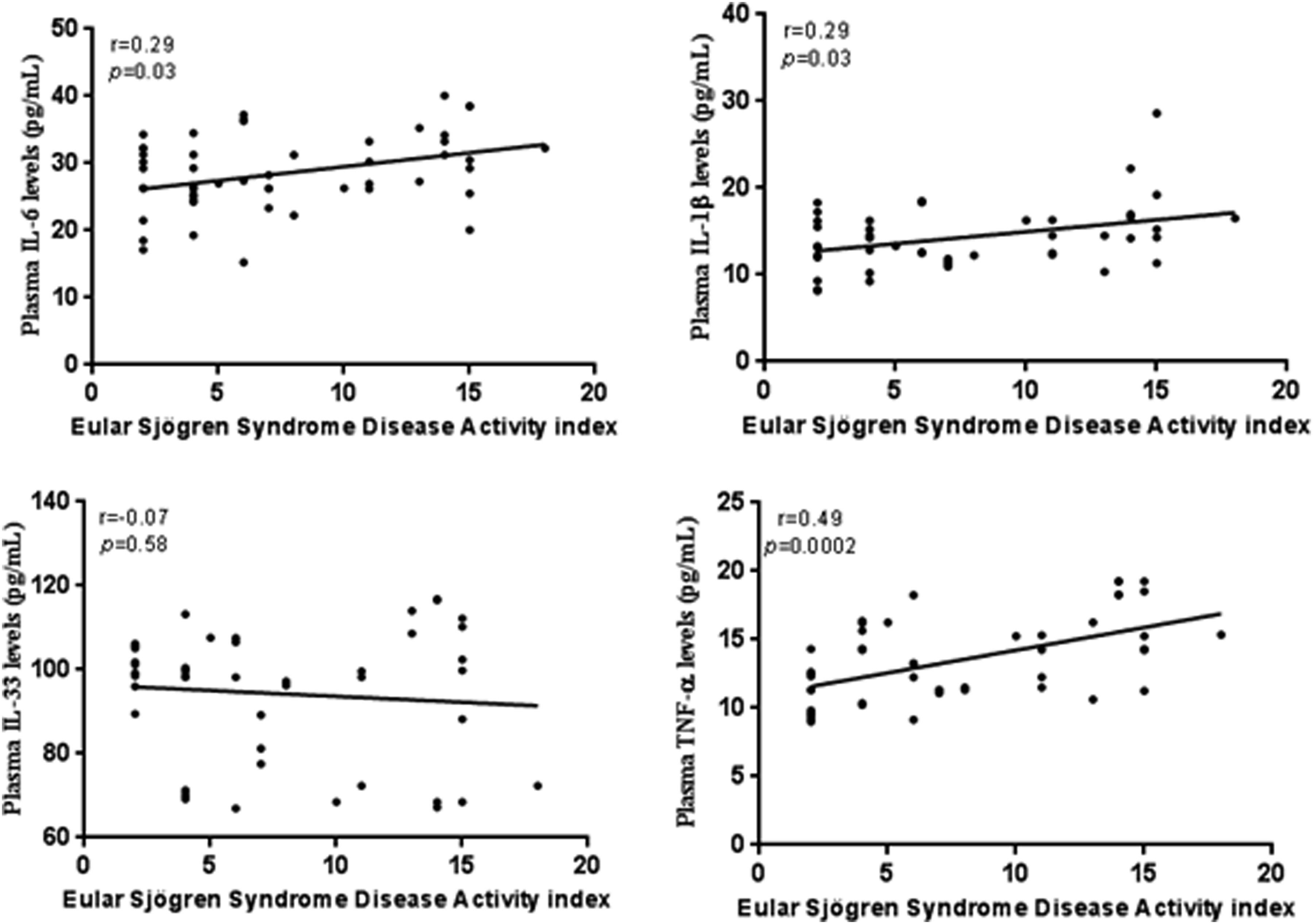
Moreover, we performed correlation between some oxidative stress variables, pro-inflammatory cytokines and ESSDAI score. A strong positive correlation was recorded between the NO, MDA levels and disease activity ( Correlation between oxidative stress variables and the pSS disease activity index. Correlation between CAT, SOD and GSH-Px activities, NO2-, MDA levels and the pSS disease activity index. A significant positive correlation was shown between plasmatic production of NO2-, MDA and ESSDAI score (r = 0.69, Correlation between proinflammatory cytokines and the PSS disease activity index. Correlation between IL-6, IL1β, TNF-α, IL-33 and the pSS disease activity. Plasma production of IL-6 IL-1β and TNF-α positively correlated with ESSDAI score (r = 0.29, 
Discussion
Several factors having direct or indirect impact upon oxidant/antioxidant systems have been involved in pSS etiology. 20 Oxidative stress is considered as the off-balance of free radicals and antioxidants. At low or moderate concentrations, these molecules are essential for some biological processes such as intracellular cell signaling and homeostasis, induction of mitogenic response, immune defense against pathogens, and cell death.21,22 In this context, accumulative literature evidence has shown that autoimmune phenomena characterizing pSS process results in an increase in OS. In turn, imbalance of oxidative stress exacerbates autoimmune diseases. It is acknowledged that infections, UV irradiation, ionizing radiation stress, smoking and others exogenous sources can induce and aggravate the pathobiology of autoimmune diseases.13,9 These factors seem to have a tight relationship with oxidative stress. Indeed, ROS and RNS can be induced by environmental factors. 23
Despite the fact that NO is considered as a key signaling molecule in several physiological activities, however, like most other inflammatory mediators, excessive production of NO in response to inflammatory stimuli mediate cellular/tissue injury.24–27 Importantly, NO is a crucial marker for oxidative stress and inflammation. 28 Our report revealed a high rise in NO levels, which can result in the production of peroxynitrite. This last potent oxidant molecule induces oxidative cell injury by altering DNA, protein, and lipids, and in turn amplify the pathologic processes of diseases, leading to proinflammatory, deleterious and destructive effects.29–31 Indeed, reaction between O2- and NO forms H2O2 which is far more deleterious and reactive than its precursors and it is able to oxidize and nitrate nucleotides, proteins, and lipids.32,33 Interestingly, a similar pattern of increase was observed for MDA production was observed in our study. In the same line, prior research concluded that lipid peroxides play a role in the etiology of a number of autoimmune disorders and allows us to support the role of OS during pSS.34,35 Increased levels of MDA indicate an important generation of ROS. 36 Free radicals are byproducts of normal cellular metabolism. However, if they are not properly scavenged, these reactive species may initiate lipid peroxidation, which may damage cell structure and function.37,38 MDA has been extensively considered in LPO studies and suggested as a biomarker of lipid peroxidation. 39 In this context, increased MDA concentrations have been linked with divers clinical characteristics and tissue damage during systemic lupus erythematosus.40,41
Under physiological conditions, the endogenous ROS can be scavenged by enzymatic antioxidants like: SOD, glutathione peroxidase and CAT. 42 However, under some pathological situations, the dynamic balance between the production and degradation of ROS may be defective and consequently could leads to a marked augmentation in the levels of ROS. 42 Hence, we also aimed to measure activities of antioxidant enzymes. In this context, we report a strong reduction in the enzymes activities of CAT, GSH-Px, and SOD during pSS. Meanwhile, lowered activity of antioxidant enzymes could explain the increase of MDA level described in our report. The depletion of the antioxidant defense system following the strong formation of free radicals may provide an explanation for the decline in antioxidant enzymes. Indeed, the antioxidant system is not able to efficiently detoxify free radicals, leading consequently to OS status.
Enzymatic antioxidants preserve the oxide/reduction in cell equilibrium. This is why, antioxidant system should maintain the balance of redox status. Anti-oxidant enzymes have been intensively studied during autoimmunity but the results were controversies. Our result agrees with previous works supporting a significant diminution in SOD activity in patients with autoimmune diseases.43,44 Reduction in SOD activity is probably attributable to the SOD antibodies which cause lead to the production of immune complexes production. 45
In the current study, low amounts of GSH-Px and catalase activities were also detected. The major peroxisomal antioxidant responsible for catalyzing the conversion of H2O2 into O2 and H2O is catalase, without free radicals production. 46 Whereas, Glutathione peroxidase is considered as an intracellular antioxidant enzyme which reduces H2O2 to H2O in the presence of reduced glutathione as a hydrogen donor. Glutatione peroxidase plays a pivotal role during lipid peroxidation damage. 47 Intriguingly, the reduced activities of CAT and GSH-Px could be caused to the inactivation of the enzymes by hydrogen peroxide. 48
In our current study, CRP levels did not show any significant association with outcome in our category of patients. Our findings are consistent with a prior research reporting that pSS is characterized by a relatively low CRP response. 49
Hence, we report that plasma of TNF-α, IL-1β, IL-6, and IL-33 increased markedly in pSS patients relative to controls. This finding provides additional support to the notion that inflammatory mediators are crucial for the pathophysiology of SS. In fact, there are several compelling lines of evidence delineating the destructive function of inflammation in pSS50,51 Next, we investigated whether proinflammatory cytokines and OS factors varied based on the stage of pSS disease activity. Remarkably, our data revealed that the increases of NO and MDA levels were significantly greater in pSS patients with higher disease activity. When we combine our two observations concerning high concentrations of NO and MDA described here and the decrease in antioxidant enzymes, we can suggest the existence of a compromised antioxidant balance, which may result in elevated ROS levels or decreased antioxidant levels, ultimately leading to a state of oxidative stress. Indeed, In fact, OS originates when the antioxidant defense is surpassed by the production of ROS. In a comparable manner, it has already been shown that increased OS is correlated with the activity of autoimmune diseases.52,53
Furthermore, we observed a noteworthy elevation in the amounts of IL1β, IL-6, and TNF-α, particularly among patients with pSS expressing mild and high activity disease. Greater response in samples with more active pSS conditions implies that these markers might be helpful in assessing how the disease is progressing as well as clarifying the pathophysiological causes. However, CRP levels̍ does not seem very useful in assessing the disease progression in this condition.
Therefore, and consistent with our present result revealing the eminent importance of NO, MDA related oxidative stress, and proinflammatory cytokines in SS, we then, sought to evaluate the relationship between serum oxidative stress marker and inflammation in SS by performing correlation study. As far as we know, no earlier report had examined the relationship between proinflammatory cytokines, and oxidative stress during pSS. In this way, our data indicated a highly positive association between plasma NO and MDA measurements during pSS. To our knowledge, this is the first reporting an interconnection between NO and MDA measurements during pSS, suggesting that the NO status is associated with lipid peroxidation in this condition. Accumulative literature evidence has supported that oxidative stress induces oxidation of lipids. In this context, it has been postulated that interaction of ROS with lipids, especially the hydroxyl radical, reacts with lipid membranes and leads to MDA production.41,54
Here, we also reported a lack of correlation between MDA, CRP and MDA. This data could be related to the fact that levels of CRP have been found within normal limits in our category of patients. In the same line, no correlation between CRP and MDA was also described in previous works.55,56
Remarkably, our result revealed a positive significant correlation between NO, MDA and the proinflammatory cytokines (IL1β, IL-6 and TNF-α), supporting consequently a crucial relationship between enhanced OS, inflammation and pSS disease and that both OS and inflammation are intimately related in pSS. This observation has not been established for IL-33. Subsequently, IL1β, IL-6 and TNF-α, are considered as potent inflammatory cytokines and consequently may have a crucial role and higher association with OS, contrary to IL-33. In the same way, it has been previously reported free radical generation has been shown to be triggered by a rise in inflammatory mediators and likewise, excessive ROS production may trigger pro-inflammatory signaling pathways and activate the release of numerous inflammatory mediators, including IL-1β and TNFα, via several different mechanisms. 57 Indeed, disproportionate OS could play a significant function during the inflammatory response of gene expression promoting inflammation, notably via activation of the nuclear transcription factor NF-kβ and activator protein-1 which in turn up-regulates proinflammatory cytokines. 41 When summarizing the results of some studies, a strong correlation has been established between OS and inflammation in a number of diseases, including type 2 diabetes and irritable bowel syndrome.58,59
In our report, the highest correlations were noted between IL-6 and MDA, and between TNF-α and NO. According to this result, these potent proinflammatory cytokines appears to be associated more closely to OS than other cytokines. This positive correlation between oxidative stress marker and these proinflammatory cytokines suggests that reducing inflammation and OS could alleviate pSS disease severity. Moreover, According to some research, excessive OS may contribute significantly to the pathophysiology of autoimmune diseases by increasing inflammation. 60 Inflammatory processes have been shown to reduce cellular antioxidant capacity and induce oxidative stress. 61 Both OS and inflammation processes feed off each other, creating a vicious cycle that can sustain perpetuate the inflammatory response. 62
Moreover, to assess if a link between some markers of OS, proinflammatory cytokines and pSS exists, we examined their relationship with pSS disease activity index. Interestingly, a positive significant correlation between NO, MDA, the proinflammatory cytokines (IL-6, TNF-α, and IL-1β) and pSS disease activity was revealed. These markers seem usefulness for monitoring pSS disease evolution. Nevertheless, the amount of IL-33 does not seem to have an effect on the disease’s activity.
However, it is important to take into account some limitations of our work when evaluating the findings. Firstly, our study was performed without sample size calculation. Large-scale study is required to validate these results. The second limitation of this study concerns the failure to predict the responses of patients using different therapeutic compounds.
Conclusion
The outcome of our study increasingly supports a potential interaction between inflammation and OS in pSS disease. Indeed, MDA, proinflammatory cytokines, and NO showed significant association, supporting consequently a crucial interconnection between inflammation and OS process during pSS. Therefore, we propose to focus on the impact of antioxidants administration, which may be beneficial against disease-related increases in oxidative stress and may positively affect the clinical damage induced by oxidative stress, since it has been established that oxidative stress was clearly implicated in the pathogenesis of pSS.
Footnotes
Acknowledgments
All of the study participants are acknowledged and thanked by the authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partially funded by the Algerian Thematic Agency for Research in Health Sciences (ATRSS). Code number: 04/06/01/14/080.
