Abstract
Background
Asthma is a chronic inflammatory condition of the airways, leading to excessive production of mucus and an inflammatory condition of the lungs. The primary clinical manifestations of asthma comprise thoracic constriction, coughing, wheezing, and dyspnea.
Objectives
The present work was designed to examine the therapeutic effects of aucubin in treating ovalbumin (OVA)-induced asthma in mice via suppressing inflammatory responses.
Materials and Methods
The aucubin-induced RAW 264.7 cell viability was assessed using the MTT test. The inflammatory cytokine levels in the lipopolysaccharide (LPS)-treated RAW 264.7 cells were analyzed using kits. The inflammatory cell levels in the bronchoalveolar lavage fluid (BALF) of the asthma-induced mice were assessed microscopically. The Th2 cytokines, tumor necrosis factor-alpha (TNF-α), immunoglobulin E (IgE), and interferon-gamma (IFN-γ) were assessed using assay kits. The amounts of the oxidative stress parameters were examined using kits. The lung tissues of the experimental mice underwent histological examination.
Results
The present work reveals that the aucubin treatment has no cytotoxic effect on RAW 264.7 cell growth. Furthermore, it decreased the nitric oxide (NO), TNF-α, and IL-6 levels in the LPS-induced cells. In addition, the treatment of aucubin in mice with OVA-induced asthma led to decreased T helper (Th)2 cytokine levels and inflammatory cell counts. The aucubin treatment was observed to reduce IgE and inflammatory markers, leading to the relief of airway inflammation. Furthermore, the aucubin treatment resulted in a decrease in the malondialdehyde (MDA), myeloperoxidase (MPO), and NO levels, while simultaneously elevating the antioxidant levels in the OVA-induced mice. The histopathological study revealed the therapeutic effects of aucubin on asthmatic mice.
Conclusion
The present results demonstrate the efficacy of aucubin in mitigating the asthmatic condition in OVA-induced mice, making it a promising therapeutic candidate to treat asthma.
Introduction
Asthma is a condition that is marked by excessive inflammation of the airways, resulting in mucus overproduction and inflammatory conditions of the pulmonary tissues (Heijink et al., 2020). It is characterized by the malfunctioning of airway epithelial cells, which are significant factors in the condition (Murphy & Hallstrand, 2021). The primary clinical manifestations of asthma comprise thoracic constriction, coughing, wheezing, and dyspnea. Additionally, individuals may have chest tightness, difficulty breathing, and other symptoms (McCracken et al., 2017). The worldwide occurrence of asthma ranges from 1% to 30%, and research indicates that there are over 300 million individuals suffering from asthma globally. The Global Initiative for Asthma (GINA) predicts that the number of asthma cases could reach 400 million by 2025 (Barcik et al., 2020; Hoch et al., 2019). Currently, the precise mechanism by which asthma develops is not completely reported. Nevertheless, airway inflammation is thought to be the primary pathophysiological alteration. The modules that participated in lung inflammation during asthma development include neutrophils, mast cells, eosinophils, T helper (Th) cells, dendritic cells, and other cytokines (Haj-Salem et al., 2015).
An association has been shown between the development of asthma and a disproportion in Th1/Th2 immunological reactions (Zhu et al., 2020). An exaggerated Th2 reaction results in the heightened secretion of Th2 cytokines. These inflammatory markers activate the synthesis of immunoglobulin E (IgE), excessive secretion of mucus, and the buildup of reactive oxygen radicals that infiltrate the airways (Antunes et al., 2022). Allergic asthma is commonly linked to the elevated formation of reactive oxygen species (ROS) and dysregulated antioxidant systems, resulting in oxidative damage to the pulmonary tissues. This oxidative stress disturbs the antioxidant mechanism, promotes inflammatory marker secretion, and leads to hyperplasia of goblet cells (Malaquias et al., 2018).
Asthma is often treated based on the severity of symptoms. Corticosteroids and antileukotrienes are the primary pharmacological agents used to treat asthma by reducing inflammation. Corticosteroids, when combined with bronchodilators, are considered preferred therapy alternatives (Holgate et al., 2015). Although these drugs are effective, they have various unwanted side effects (Sharma et al., 2022). Hence, considering its significance in terms of economic impact and life quality, there is a requirement for alternative therapies that have the same effectiveness, utilize innovative targeted methods, and result in fewer negative effects. It is of utmost importance to find pharmaceuticals that can effectively treat and prevent severe cases of asthma (Eger & Bel, 2019). Aucubin is a naturally occurring iridoid glucosidic compound found in several medicinal plants like Aucuba japonica, Rehmannia glutinosa, and Plantago asiatica (Zeng et al., 2020). Aucubin has been found to possess many salutary effects, including anti-inflammatory (Gao et al., 2021), antioxidant (Shen et al., 2019), antiapoptotic (Wang et al., 2019), hepatoprotective (Zhang et al., 2020), nephroprotective (Potočnjak et al., 2020), and neuroprotective (Park et al., 2024) activities. Nevertheless, the therapeutic potential of aucubin in treating animal models of asthma has not been investigated. Consequently, the goal of this work is to examine the therapeutic properties of aucubin in treating ovalbumin (OVA)-induced allergic asthma in mice.
Materials and Methods
Chemicals
The chemicals and other reagents utilized in this experiment, such as aucubin, aluminum hydroxide, OVA, and so on, were acquired from Sigma–Aldrich, USA. The biochemical study kits were acquired from Elabscience, USA, and Abcam, USA, respectively.
In Vitro Assays
Collection of Cell Line
The RAW 264.7 cells (ATCC, USA) were cultivated in a Dulbecco’s Modified Eagle Medium (DMEM) medium with 10% fetal bovine serum (FBS) and a 1% antibiotic mixture. The cells were cultivated in an incubator with a CO2 (5%) supply at 37°C. After attaining 80% confluency, they were collected by trypsinization and then utilized in further experimental methods.
Cell Viability Assay
Aucubin-treated RAW 264.7 cells were subjected to the MTT assay to study their cell viability. The cells were cultivated in a 96-well plate using DMEM for 24 h and then treated with the diverse dosages of aucubin (1, 2.5, 5, 10, 25, and 50 µM) for 24 h a 37°C. Afterward, a volume of MTT (20 µL) and DMEM (100 µL) was added to all wells. Subsequently, a solution of dimethyl sulfoxide (DMSO) (100 µL) was used to dissolve the formazan deposits that formed in each well. The measurements of absorbance were subsequently acquired at the 570 nm wavelength.
Analysis of Inflammatory Marker Levels
The inflammatory biomarkers, including IL-6, IL-1β, and tumor necrosis factor-alpha (TNF-α) concentrations, were examined in the treated RAW 264.7 cells. The tests were conducted three times using commercially acquired kits as per the manufacturer’s specifications (Elabscience, USA).
Experimental Animals
3–4-week-aged BALB/c mice, weighing around 22 ± 5 g on average, were purchased from the institutional animal house and utilized for the present study. All mice were caged in regulated laboratory situations with a 25 ± 1°C temperature, air humidity ranging from 50% to 60%, and a 12-h cycle of light and dark.
Treatment Groups
The mice were grouped into four groups, each having six mice (n = 6). Group I includes untreated control. Group II was assigned as the asthma-induced mice, which were sensitized to OVA to induce asthma. The mice were administered an intraperitoneal injection of aluminum hydroxide (100 µL) with OVA (20 µg) on the 1st and 14th days of the study. The mice were sequentially given 1% OVA through their nasal passages from the 21st to the 23rd day to initiate the asthma. The Group III mice were given aucubin at 10 mg/kg concentration through oral gavage 1 h prior to the OVA treatment. The Group IV mice were given a standard drug, dexamethasone (DEX), at 3 mg/kg concentration. On the 24th day of the study, all mice were euthanized using CO2 asphyxiation. Subsequently, the right lung tissues were surgically excised, and their wet and subsequent dry weights were assessed to determine the lung weight index.
Differential Cell Count in the Bronchoalveolar Lavage Fluid (BALF)
The BALF samples were obtained from the experimental mice using intubation and lavage using a 1 mL saline solution. The obtained samples underwent centrifugation at 5,000 rpm for 5 min. The BALF was stained using the Diff-Quik® reagent (Sigma–Aldrich, USA), and then differential cell counts were performed.
Analysis of Inflammatory and Biochemical Marker Levels
The concentrations of Th-2 cytokines, IgE, eotaxin, interferon-gamma (IFN-γ), and TNF-α in the BALF samples from the treated mice were measured using assay kits. The assays were performed in triplicate as per the manufacturer’s recommended guidelines (Elabscience, USA). The lung tissues were surgically excised and then blended using cold saline. The suspension was thereafter centrifuged at 6,000 rpm for 5 min. The supernatant was utilized to examine the concentrations of nitric oxide (NO) and myeloperoxidase (MPO) using kits (Abcam, USA).
Analysis of Oxidative Stress Marker Levels
The concentrations of the oxidative biomarker malondialdehyde (MDA), as well as the antioxidants catalase (CAT), superoxide dismutase (SOD), and GSH, were measured in the lung tissue homogenates. The kits were utilized to examine the concentrations of MDA, SOD, GSH, and CAT levels. The tests were conducted three times using the recommended procedures provided by the manufacturer (Elabscience, USA).
Histopathological Analysis
Lung tissues excised from the experimental mice were subjected to histopathological examination. The obtained lung tissues were treated with 10% neutral formalin, followed by paraffinization. The tissues were processed, sliced into sections with 5 µm thickness, and then stained using eosin and hematoxylin. The stained tissue slides were investigated using a microscope at a magnification of 40× to identify any histological changes.
Statistical Analysis
The obtained values were examined using SPSS software. The values are shown as the mean ± SD of triplicates. A one-way analysis of variance (ANOVA) and Dunnett’s post hoc assay were conducted to determine the significance among the treatment groups.
Results
Effect of Aucubin on the Viability of RAW 264.7 Cells
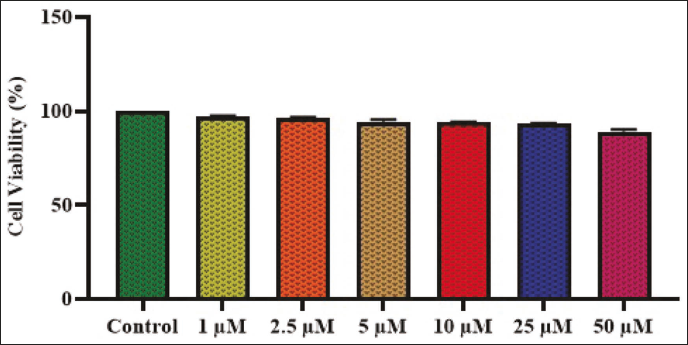
Figure 1 depicts the activity of aucubin on the growth of lipopolysaccharide (LPS)-induced RAW 264.7 cells. The findings revealed that the treatment with aucubin did not have any major negative impacts on the RAW 264.7 cell growth. There were no notable alterations in cellular viability when cells were subjected to aucubin treatment at concentrations ranging from 1 to 50 µM, as illustrated in Figure 1. Hence, it is evident that the treatment with aucubin had no toxic effects on the growth of RAW 264.7 cells. The dosages of aucubin at 2.5, 5, 10, and 25 µM were chosen for further assays.
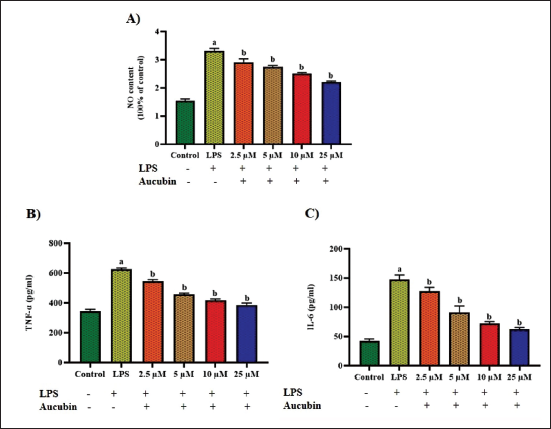
Effect of Aucubin on the NO and Inflammatory Cytokines Level in the LPS-induced RAW 264.7 Cells
Figure 2 displays the levels of NO, IL-6, and TNF-α in both the control and treated cells. The cells treated with LPS showed elevated NO, IL-6, and TNF-α levels relative to the control. Nevertheless, cells treated with dosages of 2.5, 5, 10, and 25 µM of aucubin demonstrated notable decreases in NO, IL-6, and TNF-α levels, as depicted in Figure 2. These outcomes proved the anti-inflammatory effectiveness of aucubin.
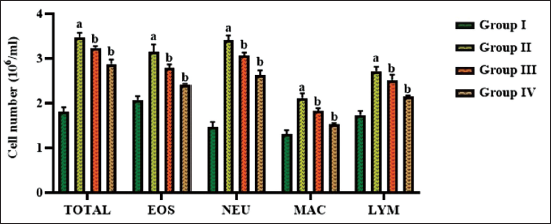
Effect of Aucubin on the Inflammatory Cell Numbers in the BALF of the Experimental Mice
Figure 3 demonstrates the levels of inflammatory cell numbers, including neutrophils, macrophages, eosinophils, and lymphocytes, in the BALF of the OVA-induced asthma mice. The BALF examination of asthma-induced mice revealed an elevation in the total cell count, with elevated levels of eosinophils, macrophages, neutrophils, and lymphocytes. However, the treatment of aucubin (10 mg/kg) showed a remarkable reduction in the total number of inflammatory cells in the OVA-induced mice (Figure 3), which is similar to the results of DEX treatment.
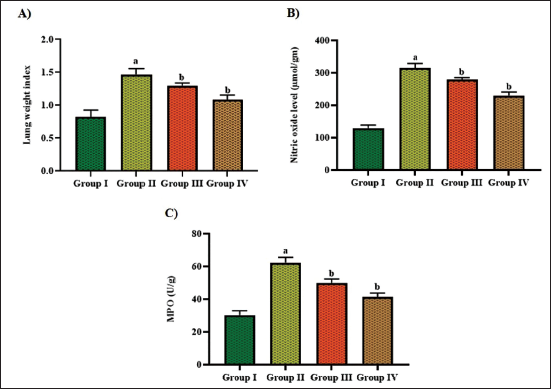
Effect of Aucubin on the Lung Weight and Biochemical Marker Levels
Figures 4 and 5 reveal the impact of aucubin treatment on the levels of lung weight and biochemical marker levels, including MPO, NO, eotaxin, and IgE, in the asthma-induced asthma mice. The mice with asthma exhibited a significant increase in lung weight, NO, MPO, IgE, and eotaxin levels in comparison to the control. Contrastingly, when aucubin was treated with 10 mg/kg concentration in the OVA-induced mice, there was a notable decrease in lung weight and other biochemical markers, as shown in Figures 4 and 5.
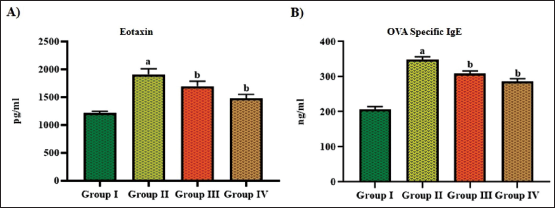
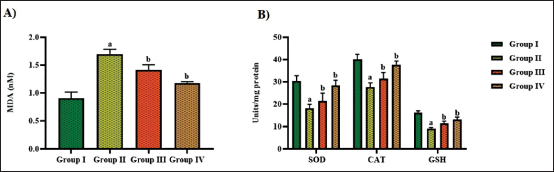
Effect of Aucubin on the Oxidative Stress Marker Levels
Figure 6 exhibits the concentrations of MDA and the antioxidant levels in the experimental mice. The mice exposed to the OVA challenge exhibited a notable upsurge in MDA and a subsequent diminution in GSH, CAT, and SOD activities, compared to the control. The aucubin treatment at a dosage of 10 mg/kg revealed a remarkable reduction in their MDA levels. Furthermore, there was an upsurge in the GSH, SOD, and CAT activities (Figure 6). These findings were also noted in the DEX-treated mice, which supports the antioxidant activities of aucubin.
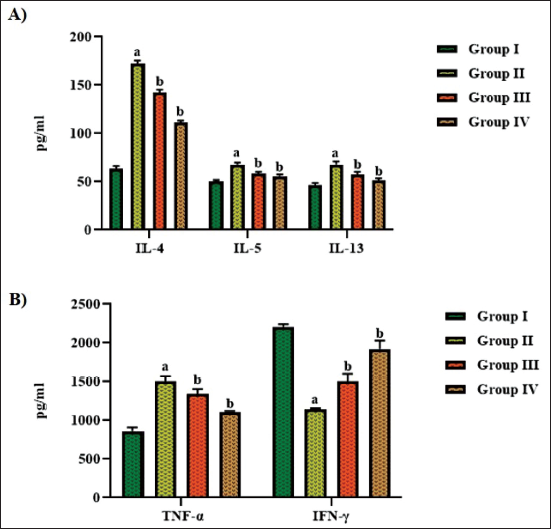
Effect of Aucubin on the Proinflammatory Cytokine Levels in the BALF
The Th2 cytokines (IL-4, IL-5, and IL-13), TNF-α, and IFN-γ levels in the BALF were assessed, and the outcomes are depicted in Figure 7. The mice with asthma exhibited a remarkable upsurge in the Th2 cytokines and TNF-α levels, whereas the levels of IFN-γ were decreased. Interestingly, the treatment of aucubin at a dosage of 10 mg/kg considerably reduced the Th2 cytokine levels and TNF-α in OVA-induced asthma mice. Conversely, there was an elevation in the IFN-γ levels in the mice with asthma. The treatment of DEX also demonstrated a drastic reduction in Th2 cytokines and TNF-α levels while simultaneously boosting the IFN-γ level in mice with asthma. These findings witnessed the anti-inflammatory potential of aucubin.
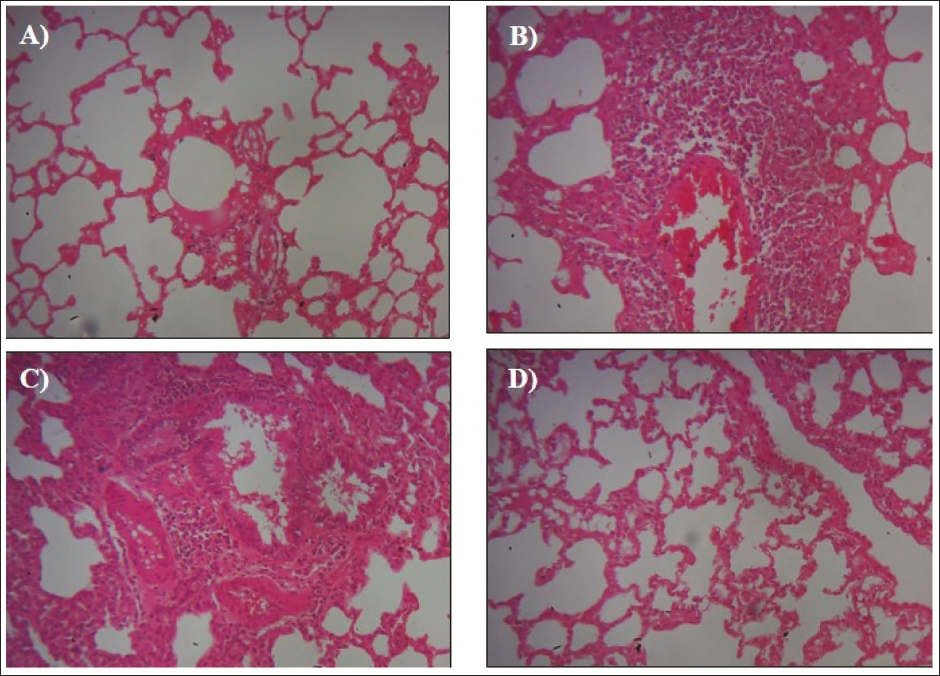
Effect of Aucubin on the Lung Tissue Histopathology
The salutary properties of aucubin treatment on the histological alterations in the lung tissues of the OVA-induced mice were investigated, and the results are shown in Figure 8. The control mice exhibited normal tissue architecture. In contrast, the mice with OVA-induced asthma showed an increased level of inflammatory cell infiltration in the peribronchial areas, along with thickening of the mucous membrane and airway epithelium. It is important to mention that the aucubin (10 mg/kg) treatment considerably decreased the inflammatory cell infiltrations, reduced damage to the airway epithelium, and reduced goblet cell hyperplasia in the OVA-induced mice (Figure 8). The DEX treatment also effectively reduced the OVA-induced damage to the lung tissues, which supports the therapeutic effects of aucubin.
Discussion
Allergic asthma is defined by inflammation and hyperresponsiveness of the airways (Hall & Agrawal, 2014). Allergens that trigger immunological and inflammatory cell infiltration in the airways can lead to allergic asthma development. The infiltration of inflammatory cells, including neutrophils, eosinophils, and lymphocytes, into the bronchial mucosa following exposure to allergens is facilitated by the targeted release of inflammatory cytokines (Papi et al., 2018). The presence of these cells can lead to pathological alterations such as increased sensitivity of the airways (airway hyperresponsiveness), inflammation of the airways, and airway remodeling (Humbert et al., 2019). Eosinophils contribute to the progression of asthmatic inflammation, and an elevation in eosinophil levels has been associated with the manifestation of asthmatic symptoms. Additionally, the occurrence of eosinophils in the airways is a crucial marker of inflammation and heightened sensitivity, which can be caused by an elevation in eosinophil cationic proteins (Lee et al., 2019). The present findings showed increased levels of inflammatory cell levels in the asthma-induced mice. Interestingly, the aucubin treatment successfully reduced these cells in the BALF of asthma-induced mice, which supports the anti-inflammatory properties of aucubin. The persistent inflammation of the airways caused by allergic sensitization is a significant pathological feature of asthma (Lambrecht & Hammad, 2015). It has been observed that the elevation of inducible nitric oxide synthase (iNOS) can impact mucus secretion in individuals with asthma by stimulating the synthesis of NO (Takezawa et al., 2016). The present results also found that the OVA-induced mice demonstrated increased NO levels, which were effectively reduced by the aucubin treatment.
Cytokines facilitate the progression of chronic inflammation in the airways. Th2 cells are specific types of cells that generate IL-4, IL-5, and IL-13, which contribute to the inflammatory process (Chung, 2015). Th2 cytokines, specifically IL-4, IL-5, and IL-13, have a significant role in the asthma development. The occurrence of these cytokines is closely related to inflammatory cell infiltration, the production of IgE antibodies, the activation of eosinophils, and the increased sensitivity of the airways (Ray & Cohn, 1999). IL-4 is a vital cytokine in regulating Th2 inflammation. It promotes the B-cell development and the transition to IgE. IL-13 serves pivotal function as an immunoregulatory cytokine in the asthma development. IL-13 holds greater significance than IL-4 when it comes to bronchial hyperresponsiveness and mucus secretion (Rincon & Irvin, 2012). Due to this, there have been experiments conducted on advanced biological anti-interleukin treatments for asthma, specifically targeting IL-4, IL-5, and IL-13 (Dunn & Wechsler, 2015). The outcomes of this work revealed that the OVA challenge drastically elevated the Th2 cytokine levels in the mice to the control. However, the aucubin treatment successfully reduced the Th2 cytokines in the asthma-induced mice, which proves the anti-inflammatory properties of the aucubin.
The TNF family, known for its inflammatory and immunosuppressive properties, has a notable effect on the development of asthma and inflammation in several tissues (Zdrenghea et al., 2015). TNF-α, a cytokine that promotes inflammation, has a significant effect on airway hyperresponsiveness (Mukhopadhyay et al., 2006). TNF-α plays a role in the initiation of airway hyperreactivity, inflammation, and the activation of profibrotic mechanisms in asthma. TNF-α induces inflammatory cell recruitment in asthma (Yang et al., 2010). The present findings demonstrated that aucubin remarkably decreased the TNF-α levels in the asthma-induced mice.
Oxidative stress has a pivotal role in the development of asthma. The generation of ROS and the presence of antioxidants, such as GSH and SOD, are kept in equilibrium to preserve regular physiological processes (Gwozdzinski et al., 2021). During oxidative stress, there is an elevation in the accumulation of ROS and a diminution in the capacity of the body to defend against them. This disruption in equilibrium leads to oxidative damage and worsened inflammation. The current investigation found the onset of oxidative stress in the asthma-induced mice, which aligns with previously reported findings (Grasemann & Holguin, 2021). Prior research has demonstrated that animals exposed to OVA exhibit elevated levels of ROS in their airways (Adam-Bonci et al., 2021). The buildup of these ROS leads to increased sensitivity and reactivity of the airways, known as airway hyperresponsiveness. The production of MDA is a key marker in determining the level of oxidative stress (Nishida et al., 2002). CAT, SOD, and GSH are crucial antioxidants that can effectively mitigate pulmonary damage and fibrosis in individuals suffering from asthma (Rogers & Cismowski, 2018). GSH is an essential antioxidant that hinders the production of harmful toxic radicals. In general, a decrease in GSH levels indicates an increase in ROS levels. CAT can mitigate cellular damage by turning hydrogen peroxide (H2O2) into oxygen and water (Sahiner et al., 2018). The results of this work demonstrated increased MDA levels while reducing levels of antioxidants such as CAT, SOD, and GSH in the pulmonary tissues of the OVA-induced mice. Interestingly, the aucubin treatment successfully diminished the MDA and subsequently increased the antioxidants in the OVA-induced mice. These findings prove that aucuba has potential antioxidant properties.
Conclusion
The present study demonstrated that aucubin treatment successfully mitigated the asthmatic condition in the OVA-induced mice. The aucubin treatment successfully reduced the Th2 cytokines and inhibited the IgE and other proinflammatory markers, resulting in a reduction of the inflammatory response in the airways. The aucubin treatment also reduced oxidative stress by increasing the antioxidant mechanisms. These results demonstrate the efficacy of aucubin in mitigating the asthmatic condition in OVA-induced mice, making it a talented salutary candidate to treat asthma. Furthermore, further study is necessary to clarify the precise therapeutic functions of aucubin to treat allergic asthma.
Abbreviations
BALF: Bronchoalveolar lavage fluid; CAT: Catalase; GINA: Global Initiative for Asthma; GSH: Reduced glutathione; IgE: Immunoglobulin E; LPS: Lipopolysaccharide; MDA: Malondialdehyde; MPO: Myeloperoxidase; NO: Nitric oxide; OVA: Ovalbumin; SOD: Superoxide dismutase.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
This work was approved by the Institutional Ethical Committee of Qingdao University, Qingdao, China (Ethical number: 2023/1-47).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
