Abstract
Objective:
Although several theories have been proposed including developmental/neurodegenerative processes, neurotransmitter abnormalities, viral infection, and immune dysfunction, the exact causative factor of schizophrenia is unclear. A relationship between inflammation and schizophrenia has been supported by abnormal cytokine production and altered antioxidant status. This study was aimed to examine the alterations in serum oxidative–antioxidative status and cytokine levels of schizophrenic patients.
Methods:
A total of 91 schizophrenic patients from Saudi Arabia and 50 age- and sex-matched healthy controls were enrolled in the present study. Fresh blood samples were collected to measure the levels of cytokines and markers of oxidative stress by spectrophotometric assays simultaneously.
Results:
We observed that there was a significant increase in the levels of tumor necrosis factor-α, interleukin (IL)-1β, and IL-6 and a decrease in the levels of interferon-γ. Lipid peroxides are elevated in serum, while total-sulfhydryl levels were decreased. Also, the activities of superoxide dismutase and glutathione peroxidase were decreased, while the activities of catalase, glutathione reductase, and myeloperoxidase were found to be elevated in serum.
Conclusion:
We conclude that inflammation resulting from dysregulation of cytokines and altered antioxidant systems may play a critical role in the etiology of schizophrenia.
Introduction
Schizophrenia is a disorder that is characterized by distorted thinking, hallucinations, and reduced ability to feel normal emotions. It has long been associated with immunity, environment, and hereditary factors. 1 –3 Activation of the inflammatory response system in acute schizophrenia has been suggested recently by several studies. 4, 5 Many investigators have reported the involvement of immunological dysfunction in schizophrenic patients, 6 –8 although the results are conflicting, the primary focus of most of the studies has been plasma levels of cytokines or the T-helper 1 and 2 (Th1/Th2) imbalance. 9 –14 However, more and more converging evidence indicates that oxidative stress mechanisms may also play a role in the pathophysiology of schizophrenia. 15,16
Over the past decade, immunologic, neurochemical, and neuroendocrine activities of the cytokines involving the brain and their behavioral effects have been demonstrated. Therefore, research concerning the role of cytokines in schizophrenia has also been expanded. 17 Schizophrenic patients have aberrant proportions of immunocompetent cells and varied levels of cytokines, especially proinflammatory interleukins (ILs). 18 Theoretically, alterations in the levels of cytokines can simply be a consequence of mental stress or sleep deprivation associated with the onset or exacerbation of the disease. On the contrary, they can modify metabolism of neurotransmitters and neuroendocrine hormones and influence neural development and neurodegeneration, as supported by findings in fundamental neuroscience. 17 –19
If the balance between rate of formation of free radicals and the rate of their neutralization is not maintained, an oxidative damage, known as oxidative stress, occurs. 20 There is abundant evidence that free radicals are involved in membrane pathophysiology in the central nervous system (CNS) and may play a role in neuropsychiatric disorders including schizophrenia. 21, 22 The brain and nervous system are particularly prone to the free radical damage, since the membrane lipids are very rich in polyunsaturated fatty acids and areas of human brain are very rich in iron, which plays an essential role in generating free radicals. 23 –25 The radical-induced damage may be important in schizophrenia, as there is increasing evidence that oxidative injury contributes to the pathophysiology of schizophrenia. 23 –25 Free radicals, primarily the reactive oxygen species, superoxide radicals, and hydroxyl radicals, which are highly reactive, having an unpaired electron in an atomic or molecular orbit, are generated under physiological conditions during aerobic metabolism. 21 As free radicals are potentially toxic, they are usually inactivated or scavenged by antioxidants before they can inflict damage to lipids, proteins, or nucleic acids. The human body has a complex antioxidant defense system that includes the antioxidant enzymes superoxide dismutase (SOD), glutathione peroxidase (GPX), catalase (CAT), and glutathione reductase (GR). These block the initiation of free radical chain reactions. 21
There have been many studies investigating independently the role of cytokine imbalance and alteration in the antioxidant status in schizophrenia but to our knowledge, till date, there is no study that correlates these two fundamental aspects of inflammation in one study.
Aims of the study
In this context, the present study was aimed to assess the levels of pro-/anti-inflammatory cytokines such as tumor necrosis factor (TNF)-α, IL-1β, interferon (INF)-γ, IL-6, and IL-10 in serum and also to elucidate the oxidant–antioxidant status in schizophrenic patients. Serum malondialdehyde (MDA) levels were measured as thiobarbituric acid reactive substances, which serve as an index of extent to lipid peroxidation (LPO). The antioxidant enzymes SOD, CAT, GPX, and GR in serum were estimated. Myeloperoxidase (MPO) that is produced by activated neutrophils/macrophages is a known marker of inflammation assayed in serum.
Materials and methods
Patients and controls
A total of 140 subjects visiting Riyadh Military Hospital (RMH), Riyadh, Saudi Arabia, were involved in this study. Of the total, 91 Saudi unrelated schizophrenic patients (males) from the Outpatients Psychiatric Clinic of RMH were recruited. The diagnosis of schizophrenia was made according to the Diagnostic and Statistical Manual of Mental Disorder (Fourth Edition) criteria administered for the lifetime prevalence of mental disorders. 22, 23 To ensure the diagnostic reliability, a systemic search into the case notes of the patients was made. Subjects with any other Axis I disorders as well as those with alcohol and substance-related disorders, such as major depression and bipolar disorders, and earlier hospitalization were excluded. Of the total 140 subjects, the remaining 50 healthy unrelated male subjects were recruited as normal controls. Each subject was interviewed by a psychiatrist so as to rule out the subjects with a family history of mental illness. None of the control subjects had a first or second degree relative with any mental illness.
Neither the schizophrenic patients nor the control subjects had smoking habit (less than four cigarettes per day) or suffered from substance abuse/dependence, and all recruited subjects were free of immunosuppressive medication. This study was approved by the Ethical Committee of RMH and informed consent was obtained from each subject and/or his family member recruited in this study.
Reagents and apparatus
All reagents used in this study were of the highest purity grade (Sigma Chem Co., St Louis, Missouri, USA). For spectrophotometric analysis, we used Perkin Elmer Lamda 35 ultraviolet–visible spectrophotometer (Perkin Elmer, Waltham, MA, USA). For enzyme-linked immunosorbent assay (ELISA), we used the Thermo Scientific Corp (Thermo Fischer Scientific, Waltham, MA, USA). Multiskan MS instrument.
Collection of blood samples and separation of serum
Blood samples were collected using standard venipuncture technique between 9:30 and 11:00 a.m. after a 12-h fasting. Serum samples were separated immediately after centrifugation at 2000 g for 10 min at +4°C and stored at −20°C until analysis, which was performed in the same run to avoid inter run analytical variation.
Cytokine measurements
Human cytokines TNF-α, IL-1β, INF-γ, IL-6, and IL-10 were assayed using Quantikine Immunoassay ELISA Development System (R&D Systems, UK). All assays were carried out in duplicate by the same operator using the recommended buffers, diluents, and substrates. The concentrations of the samples in each plate were calculated according to each standard curve and the dilution factor.
Assay of enzymatic and nonenzymatic biomarkers of oxidative stress
SOD was assayed by the method of Marklund and Marklund. 26 One unit of SOD inhibitory activity is that the amount of enzyme that inhibits autoxidation of pyrogallol by 50% per minute at pH 8.2 at 25°C.
CAT was assayed according to the method of Claiborne as described by Giri et al. 27, 28 One unit of CAT will decompose 1 μmol of hydrogen peroxide (H2O2) to oxygen (O2) and water (H2O) per minute at pH 7.0 at 25°C. Results are calculated as mean ± SEM for five different preparations.
GPX activity was determined by the method of Flohe and Gunzler. 29 One unit of GPX is the amount of enzyme that causes the formation of 1 μmol of nicotinamide adenine dinucleotide phosphate (NADP+) from NADPH per minute at pH 8.0 at 25°C. Results are calculated as mean ± SEM for five different preparations.
Total-sulfhydryl (SH) groups were determined by the method of Sedlak and Lindsay. 30 LPO in serum was determined spectrophotometrically by the method of Ohkawa et al. 31 MPO was assayed by previously described method of Bradley et al. 32
Statistical analysis
Study groups were compared for variance by Student’s t test using SPSS 14.0 (SPSS Inc., Chicago, Illinois, USA). The data were expressed as mean ± SEM; p < 0.05 was considered as significant.
Results
Among the 90 male schizophrenic patients enrolled in this study, the mean age was 38 ± 7.54 years and the mean disease duration was 9 ± 4.5 years. Age of onset of the disease varied from 19 to 64 years. The mean age of controls was 30.81 ± 7.54 (Table 1).
Characteristics of the study participants.a

aData are presented as mean ± SEM.
Cytokine levels in schizophrenic patients and normal controls
The baseline characteristics of the study participants are presented in Table 1. Because of matching criteria, no statistically significant difference was noted in age.
The levels of IL-1β, IL-6, and TNF-α of schizophrenic patients were significantly higher than those of normal controls (Table 2). On the other hand, INF-γ values for schizophrenic patients were significantly lower than those of normal controls (Table 2). However, IL-10 levels of the patients showed no significant difference from controls (Table 2). There were no significant correlations between the cytokines and clinical variables including age, duration of illness, age of onset, number of admissions, numbers of episodes, or psychopathology scores (data not shown).
The mean ± SD values of pro-/anti-inflammatory cytokines in normal controls and schizophrenic patients.a

IL: interleukin; IFN: interferon; TNF: tumor necrosis factor; NC: no change.
aValues in parentheses represent percentage change from respective controls.
b p < 0.01: significantly different from control by Student’s t test.
c p < 0.05: significantly different from control by Student’s t test.
Levels of enzymatic and nonenzymatic biomarkers of oxidative stress
The mean ± SEM of serum total-SH, MDA, SOD, CAT, GPX, GR, and MPO is indicated in Table 3. There was a statistically significant increase in the serum MDA levels in schizophrenic patients when compared with the controls. The activities of serum antioxidant enzymes CAT, GR, and MPO were significantly increased in schizophrenic patients compared with controls. On the other hand, the activities of SOD and GPX were found to be decreased in schizophrenic patients as compared to controls. The levels of serum total-SH was significantly decreased in patients with schizophrenia as compared to controls.
The mean ± SD values of nonenzymatic and enzymatic biomarkers of oxidative stress in normal controls and schizophrenic patients.a
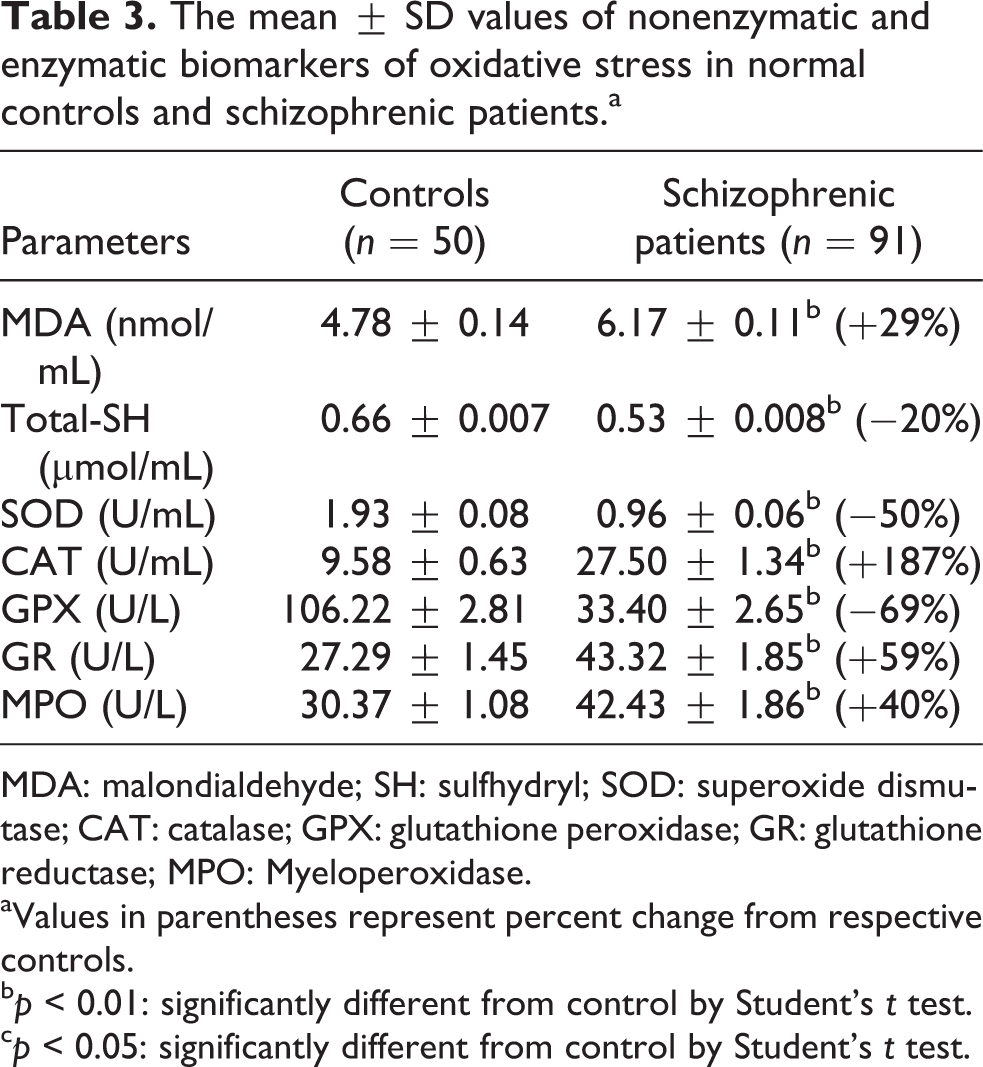
MDA: malondialdehyde; SH: sulfhydryl; SOD: superoxide dismutase; CAT: catalase; GPX: glutathione peroxidase; GR: glutathione reductase; MPO: Myeloperoxidase.
aValues in parentheses represent percent change from respective controls.
b p < 0.01: significantly different from control by Student’s t test.
c p < 0.05: significantly different from control by Student’s t test.
Discussion
It has been suggested over and over again by different authors that assessment of serum/plasma cytokines levels is the most appropriate way to investigate if they play significant role in the pathophysiology of schizophrenia. Serum/plasma cytokine levels may be produced by blood cells, endothelium, or may originate from the brain. Abnormal regulation of cytokine activity and alterations in the antioxidant defense mechanisms are important hallmarks of inflammation. Thus, this study provides a comparative evaluation of cytokine–antioxidant status from schizophrenic patients and from healthy controls.
We demonstrated that proinflammatory cytokines IL-1β, IL-6, and TNF-α were significantly elevated in schizophrenic patients as compared to healthy controls. These results are consistent with some previous studies that showed higher production of IL-1β, IL-6, and TNF-α levels in schizophrenic patients. 33 –37 However, in our study, other inflammatory cytokine, IFN-γ, was found to be decreased from normal controls in consistent with a previous study. 37 These results are in agreement with earlier studies that demonstrated Th1 hyperactivity in bipolar disorder. 38 –41 However, our studies contradict other studies that found no alteration in IL-1β, IL-6, and TNF-α and increased or no significant change in IFN-γ levels in schizophrenic patients. 42 –48 In our study, the different activities of these inflammatory cytokines in schizophrenic patients suggest that specific subtypes of cytokines, monocytic proinflammatory cytokines, may be associated with the immunopathogenesis of schizophrenia.
Our study also examined a Th2 anti-inflammatory cytokine, IL-10, and found that its concentration in the schizophrenic patients was not significantly different than in controls. Our results are in accordance with but contradicts those studies that have indicated an increase in the levels of IL-10 in schizophrenic patients. 44 –50 These results suggest that an overproduction of monocytic Th1 cytokines may be associated with the etiology of schizophrenia.
We postulate that an imbalance between proinflammatory and anti-inflammatory cytokines may be involved in the pathophysiology of schizophrenia through various mechanisms. Cytokines are known to interact with neuroendocrine system, for example, the hypothalamic–pituitary–adrenocortical system, the autonomic system (epinephrine and norepinephrine), and the neurotransmitter system (dopamine, serotonin, and glutamine). 51, 52 In addition, cytokines might cross the blood–brain barrier (BBB) either through leaky areas or by active transport. 53 Increased permeability of the BBB may enable activated immune or neurotoxic cytokines to enter the CNS and trigger psychopathological changes. 54
For more than a decade, inflammation has been implicated in chronic psychiatric disorders. The etiology of schizophrenia remains unexplained, but recently a vascular–inflammatory–genetic theory has been proposed, bringing together environmental and genetic factors that influence the inflammatory response and potentially contribute to the disease. 55 Taking all findings together, it seems reasonable to hypothesize that the changes in cytokines in schizophrenic patients could lead to disruption of the homeostatic mechanisms among the immune, endocrine, and neurotransmitter systems and induce cell loss and decrease neurogenesis.
Our results also indicate that there is increase in free radical generation and antioxidant defense is impaired in schizophrenic patients. Impaired antioxidant defense and increased LPO have been reported in chronic schizophrenic patients. 56, 57 In the present study, LPO product, that is, MDA levels was found to be elevated significantly in serum of schizophrenic patients respective to controls. This confirms the involvement of increased oxidative stress. Rise in MDA could be due to the excessive oxidative damage to lipid membranes and compartments in these patients. 56, 57
We observed a significant decrease in the levels of total-SH (free and protein-bound SH groups; nonenzymatic antioxidant defense system) in schizophrenic patients when compared with the controls. SH groups are important chain-breaking antioxidants, responsible for scavenging the free radicals and suppression of peroxidation in aqueous and lipid region of the cell. The decrease in the level of this nonenzymatic antioxidant parameter may be due to the increased turnover, for preventing oxidative damage in these patients, suggesting an increased defense against oxidant damage in schizophrenic patients. Similar reports of decreased GR levels in schizophrenic patients were reported by various studies. 58 –60
SOD is the important antioxidant enzyme having antioxidative effects against superoxide anion. Our results confirm previous studies that reported a decrease in SOD activity, but contradict others who have found no change or elevated levels of SOD in schizophrenic patients. 56, 57, 60, 61
GPX, an oxidative stress-inducible enzyme, plays a significant role in the peroxyl scavenging mechanism and in maintaining functional integration of the cell membranes. 62 We found decreased levels of GPX in schizophrenic patients as compared with the controls in accordance with previous studies, but our results contradict with some studies that found an increase in GPX levels. 56– 61
We also observed a significant increase in the activity of CAT in patients with schizophrenia compared with the controls. CAT is the enzyme that protects the cells from the accumulation of H2O2 by dismutating it to form H2O and O2 or using it as an oxidant in which it works as a peroxidase. 63 We observed an increase in CAT activity in schizophrenic patients, as compared with normal healthy subjects as previously documented, although some studies have reported a decrease in CAT levels in schizophrenic patients. 56, 57, 60, 64
GR plays an important role in GPX reactions as it is required for the conversion of oxidized glutathione (GSH) to reduced GSH. Thus, it is essential in maintaining the reducing capability of the cell. 65, 66 In our study, we observed an increase in the activity of GR as compared with the controls. MPO, considered to be a biomarker of inflammation, is an enzyme stored in azurophilic granules of polymorphonuclear neutrophils and macrophages and released into extracellular fluid in the setting of inflammatory process. 67, 68 We observed a significant increase in the levels of MPO in schizophrenic patients as in controls. Both GR and MPO have not yet been studied in the context of schizophrenia.
We conclude that there is a growing evidence base supporting the role of inflammation in the etiology of schizophrenia. The results of our study provide support to the notion that pro-/anti-inflammatory cytokines and dysregulation of the oxidant–antioxidant balance play important roles in the pathophysiology of schizophrenia. It is suggested that interventions that reduce oxidative stress and augment the antioxidant system may be helpful in the management of schizophrenia patients. However, further research studies are warranted to understand the mechanisms and pathways underlying cytokine imbalance and oxidative stress in schizophrenia.
Footnotes
Conflict of Interest
The authors declared no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
