Abstract
Background
Measuring pain objectively or quantitatively is difficult. Since urine oxytocin (OXT) concentration has been reported to be potentially associated with chronic pain.
Purpose
We conducted a pilot study to evaluate the relationship between changes in numerical rating scale (NRS) score following nerve block and urine OXT concentration in patients with chronic pain.
Methods
We investigated 19 chronic pain patients who were admitted to our hospital for nerve intervention. NRS score and urine OXT concentration were assessed before and after the nerve block.
Results
NRS scores were 7 [6, 9] and 2 [1, 4] (median [interquartile range]) before and after the treatment intervention, respectively. Urinary OXT levels were 523 [293, 743] and 531 [308, 887] pg/mL (median [interquartile range]) before and after the treatment, respectively. We compared the group with greater improvement in NRS after the treatment intervention and those with less improvement. The group with greater improvement in NRS after the treatment intervention showed a greater increase in OXT level. However, baseline urine OXT concentrations were highly variable in patients with chronic pain and the changes in urinary OXT concentrations were also variable.
Conclusion
Based on this pilot study, the utility of urine OXT concentration as an objective biomarker of chronic pain needs further investigation.
Introduction
Numerical rating scales (NRS) or verbal rating scales are often used for the subjective assessment of pain intensity in patients with chronic pain. Although the visual analogue scale and face rating scale have also been used as subjective scales for pain assessment, a definite index for pain intensity has not been established because each patient’s complaint of pain is variable. Quantitative evaluation of pain using devices such as PAINVISION (NIPRO co., Tokyo, Japan) is often used for objective assessment of pain, although there is, so far, no established standard technique for pain assessment because of the subjective variability. 1
Oxytocin (OXT) is synthesised in the hypothalamus and carried to the posterior pituitary gland for release into the bloodstream. OXT in the blood can be finally excreted in urine. 2 Previously, the function of OXT had been only evaluated in relation to reproductive biology in females and males, however, OXT has been reported to be a neurotransmitter in association with emotion, sexual behaviour, neuropsychiatric disorders and social behaviour. 3 The role of OXT in humans is associated with exerting anti-depressive, anti-anxiety and anti-stress effects, as well as adjustment of the autonomic nervous system. OXT also responds to the signal from periaqueductal grey matter and exerts analgesic effects via adjustment for the secretion of endogenous opioids, although the detailed mechanism remains unclear. 4 Previous studies on the association between chronic pain and OXT have shown that OXT is indirectly associated with the pain process and modulation in the brain and that it decreases the sensitivity of peripheral tissues to nociceptive stimuli via direct analgesic effect.5, 6 OXT levels can be measured using several methods in various body fluids, such as saliva, serum, cerebrospinal fluid and urine.
In this study, we focused on the relationship between OXT and chronic pain, assuming that endogenous OXT levels would change after an intervention to relieve pain in patients with chronic pain, to evaluate urinary OXT concentration as a noninvasive biomarker of pain intensity or a predictor of responsiveness in a chronic pain patient to interventional treatments such as nerve blocks.
Methods
Data Source, Study Design and Setting and Participants
This prospective observational study was conducted at Okayama University Hospital. We obtained our data from the electronic medical records at Okayama University Hospital, an institutional data repository that includes robust perioperative data and clinical details about all patients. All patient data were de-identified at the hospital, and the anonymous data were collected to create a database. The ethics committee of our institution (Okayama University Graduate School of Medicine, Dentistry and Pharmaceutical Sciences and Okayama University Hospital) approved the study design (approval No. 1607-014). All procedures were performed in accordance with relevant guidelines and regulations. We obtained informed consent from all patients for study participation. We included chronic pain patients who underwent nerve block and intravenous lidocaine drip (2 mg/kg) at our hospital between 1 January 2017 and 31 December 2019. We excluded patients younger than 20 years and older than 90 years of age. We also excluded pregnant patients and those with decreased kidney function (serum creatinine >1.5 mg/dL). We obtained patient demographic data, including age, sex, body mass index and results of psychology consultations from the database. We also collected detailed data on the patient’s disease and nerve block. Urine samples were collected for urine OXT measurement. NRS pain scores were assessed from 0 (no pain) to 10 (worst possible pain) before and after the nerve block intervention.
Interventions
Nerve blocks included peripheral blocks, epidural blocks, facet blocks and nerve root blocks. Peripheral blocks included ultrasound-guided infraorbital nerve block, cheek nerve block and transversus abdominis plane (TAP) block. Epidural block, facet block and nerve root blocks were performed under contrast X-ray guidance. We used thermocoagulation (90°C, 180 seconds) in order to extend the analgesic effects of the blocks. Spinal cord stimulation was conducted under X-ray guidance to adjust the position of the catheter tip to enable accurate stimulation of the patient’s pain. Intravenous lidocaine (2 mg/kg) was administered to only one patient, who required pain relief for postherpetic pain.
Outcomes
The primary outcome evaluated in this study was a change in NRS scores before and after the nerve block intervention. The secondary outcome was the change in urine OXT level before and after the intervention. The intervention was defined as being satisfactorily effective if there was a decrease in the NRS (∆NRS) score to greater than or equal to 5.
Measurement of Urine OXT Concentration
The urine sample was stored for 12 hours before the pain treatment intervention and 12 hours after the intervention, and part of the sample was frozen and stored. The concentration of OXT in urine was measured using an OXT ELISA kit (Enzo Life Sciences, Inc., Farmingdale, NY, USA) according to the manufacturer’s instructions. The samples were measured without any dilution and column extraction.
Statistical Analysis
Values are expressed and mean ± standard deviation or median [interquartile range]. We used Fisher’s exact test to analyse categorical data and a t-test, Wilcoxon’s signed rank test or Mann–Whitney U-test to compare numerical data. The threshold for significance was set at p < .05. R (version 3.6.3, The R Foundation for Statistical Computing, Vienna, Austria) was used for all analyses.
Results
Nineteen patients (nine males and 10 females, mean age 65.2 ± 18.7 years old) were included in this study. The causes of pain were lumbar spinal canal stenosis in five patients, trigeminal neuralgia in four patients, cervical myelopathy in two and other causes in six patients (Table 1).
Patient Characteristics.
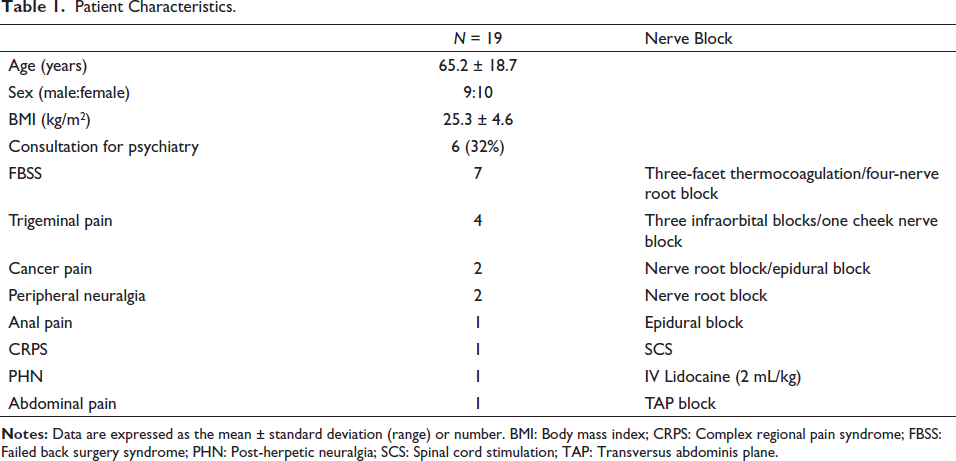
Among seven patients with failed back surgery syndrome (FBSS), three received thermocoagulation for facet pain and four received root blocks for nerve root syndromes. Among the four patients with trigeminal pain, three underwent thermocoagulation of the infraorbital nerve and one underwent thermocoagulation of the cheek nerve. Among the two patients with cancer pain, one received continuous epidural block and one received nerve root block. We conducted two diagnostic root blocks in patients with peripheral neuropathy. One each of four patients (anal pain, complex regional pain syndrome [CRPS], acute herpes zoster pain and abdominal pain) received epidural block, spinal cord stimulation, intravenous lidocaine and TAP block (Table 1).
NRS score before the treatment intervention was 7 [6, 9]. NRS was significantly decreased after the treatment to 2 [1, 4] (p < .01) (Figure 1A).
Changes in NRS After Treatment Intervention. (A) Changes in NRS in All Patients. (B) NRS in the Patients with Greater Improvement After the Treatment. (C) NRS in the Patients with Less Improvement After the Treatment. NRS; Numerical Rating Scale (0–10). Data are Expressed as a Tukey Plot and Each Score. **; p < .01 and *; p < .05 by Wilcoxon’s Signed Rank Test.
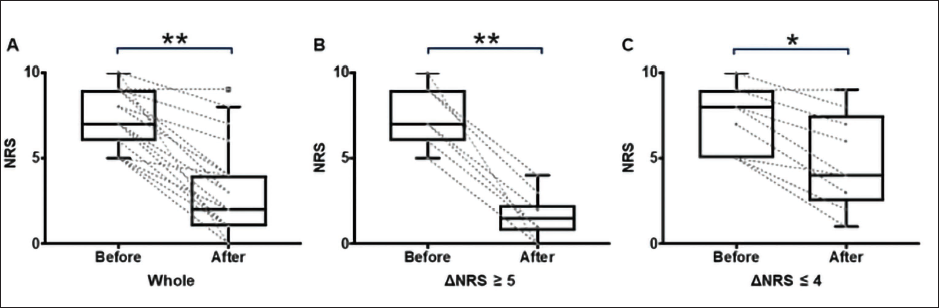
The intervention was satisfactorily effective in 10 patients (53%) (defined as a decrease in NRS score greater than or equal to 5) (Figure 1B and C), while the effects of treatment were less in the other 9 patients.
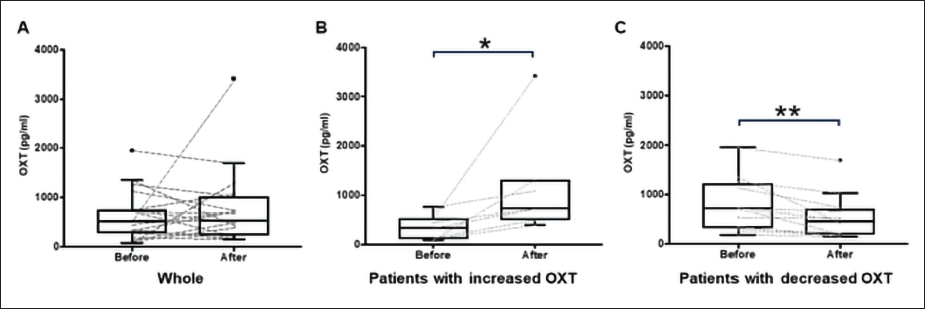
Urinary OXT levels were 522.9 [292.5, 742.9] pg/mL before the treatment and 531.0 [308.2, 887.0] pg/mL after the treatment (Figure 2A), with much variability in the trend of OXT levels. Urine OXT was increased from 328.6 [118.4, 483.3] to 723.4 [604.4, 1199.3] in 7 patients and decreased from 715.2 [343.5, 1170.0] to 454.7 [203.1, 687.7] in 12 patients (Figure 2B and C).
Changes in Urine OXT After Treatment Intervention. (A) Changes in Urine OXT in All Patients. (B) Urine OXT Levels were Increased in 7 Patients After the Treatments. (C) Urine OXT were Decreased in 12 Patients After the Treatments. Data are Expressed as Tukey Plots and Each Concentration. **; p < .01 and *; p < .05 by Wilcoxon’s Signed Rank Test.
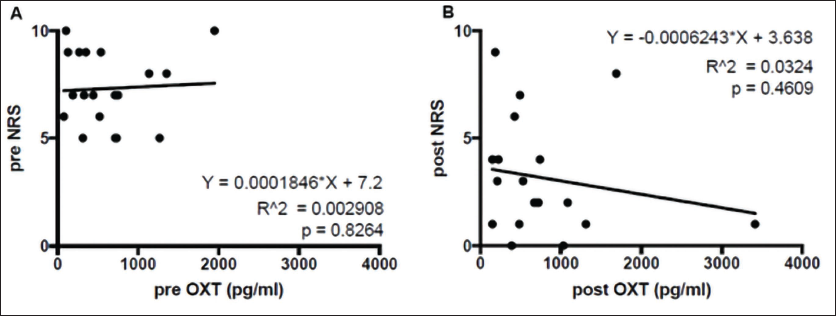
Additionally, there was no significant correlation between the NRS score and OXT concentration before and after the intervention (Figure 3).
Correlation Between Urine OXT and NRS Before and After the Treatment. (A) Scatter Plot of Urine OXT and NRS Before the Treatments. (B) Scatter Plot of Urine OXT and NRS After the Treatments.
However, a comparison of the group with greater NRS improvement after the treatment intervention (∆NRS ≥ 5 group) versus the group with less improvement after the treatment (∆NRS ≤ 4 group) showed that the changes in OXT (∆OXT) were significantly larger in ∆NRS ≥ 5 group (Table 2).
Comparison Between Groups with Greater Versus Less NRS Improvement After the Intervention.
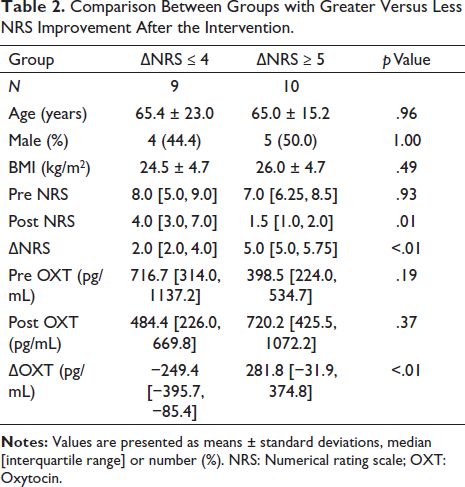
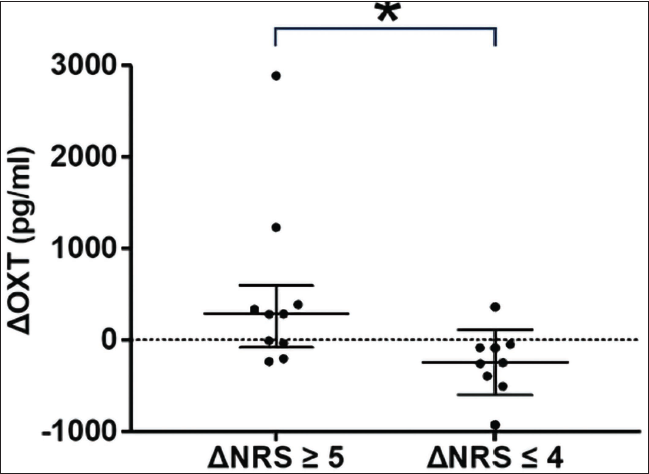
The median of ∆OXT in ∆NRS ≥ 5 group was a positive number, while that in ∆NRS ≤ 4 group was a negative number (Figure 4).
Comparison of Changes in OXT Level After the Treatments (∆OXT) Between the Groups with Greater NRS Improvement (∆NRS ≥ 5 Group) and Less Improvement (∆NRS ≤ 4 Group). Bars are Median and Interquartile Range. *; p = .007 by Mann–Whitney U-test.
Discussion
In the present study, we investigated urine OXT concentrations in 19 patients with chronic pain before and after interventions to decrease the pain. To our knowledge, this study was the first to investigate the change in urine OXT concentration after pain intervention for patients with chronic pain. Most patients tended to have a decrease in NRS scores after the nerve block. We found no significant correlation between NRS scores pre- and post-treatment and urinary OXT concentrations. However, the group with increased urinary OXT after the treatment intervention showed a greater decrease in NRS score. This suggests that OXT concentration before a pain-relieving intervention might be a predictive tool regarding the efficacy of the intervention.
The role of OXT in chronic pain has not been clarified, although OXT might be associated with mental and physical pain in the process of chronic pain extension in cases with peripheral and central sensitisation. 7 We found that the group with greater improvement in NRS after the treatment intervention showed a greater increase in OXT level. We thought that higher OXT concentration would be associated with more emotional feeling, as this result there was a positive relationship between greater improvement in NRS and greater increase in OXT level. 8
OXT concentration is typically measured in serum and saliva, and only a few reports exist regarding its measurement in urine. Measuring OXT in urine might be advantageous as compared to its measurement in plasma in terms of non-invasiveness. However, there is no conclusive evidence regarding the correlation between plasma and urine OXT levels. Despite this, we used urine samples for evaluating OXT concentration due to its non-invasiveness. We found that since the normal value ranges widely, it is difficult to determine the baseline value of urinary OXT. Additionally, hormonal balance and age might influence OXT concentration in individual patients. However, in this study, we focused more on the change or trend of OXT levels from baseline values rather than on its absolute value. Two previous pilot studies have evaluated urine OXT concentrations in humans. Feldman et al. investigated urinary, plasma and salivary OXT levels in 112 parents and found a correlation between urine OXT levels and anxiety and parenting stress among mothers. 9 An exploratory analysis demonstrated a positive correlation between plasma and urinary OXT levels corrected with serum creatinine values, although two samples with extreme urinary OXT concentrations were excluded out of 18 participants. 10 In our data, there were two abnormal values of urine OXT, but even after excluding these two data points, we could not find any significant correlation between the NRS score and OXT.
Tracy et al. reported that in older chronic pain patients, intranasal OXT might attenuate pain intensity in the process of chronic pain. 11 However, our patients had higher OXT levels than those in Lussier’s report, since older patients might have lower OXT concentrations. In our study, there were six patients over 80 years of age and their individual OXT concentrations were 717, 734, 539, 314, 271 and 106 pg/mL (in a 91-year-old man with FBSS, 82-year-old female with FBSS, 88-year-old female with trigeminal pain, 86-year-old male with postherpetic pain, 81-year-old female with FBSS and an 87-year-old male with trigeminal pain, respectively). Hence, since the OXT concentrations of older patients were so variable, and it was difficult to explain the reason for the variability, evaluation of urine OXT in a larger cohort is required to reach a definitive conclusion.
This study had several limitations. First, this was a pilot study at a single centre and included a relatively small sample size. Second, the disease for which the nerve block was performed was also variable. This is a common limitation among chronic pain studies because chronic pain is a condition with many variables such as causative diseases, affected periods and personal/social factors. Third, a method to measure urine OXT is not standardised. Currently, there is a controversy regarding the ideal method, such as liquid chromatography, radio-immunoassay and enzyme-linked immunosorbent assay. 12 The first and the second require high-end equipment or harmful reagents. We employed the last because this is a pilot study and feasibility for larger studies has precedence. Fourth, we could not find the specificity for monitoring chronic pain.
Conclusion
This study was a pilot study regarding urine OXT concentration. We confirmed that although OXT can be measured non-invasively in urine, its concentration is variable in patients with chronic pain. In addition, although NRS scores decreased significantly following nerve block in all the patients in this study, the change in urine OXT concentration was variable. Hence, the potential of urine OXT concentration as an objective biomarker of chronic pain needs further investigation.
Footnotes
Abbreviations
BMI: body mass index
CRPS: complex regional pain syndrome
FBSS: failed back surgery syndrome
NRS: numerical rating scale
TAP: transversus abdominis plane
Acknowledgement
We sincerely appreciated our pain team staff.
Authors’ Contribution
DO collected our data and wrote our manuscripts. TM collected our data and wrote our manuscripts. YM wrote and reviewed our manuscripts. RK and HM reviewed our manuscripts.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Patient Consent
We obtained informed consent from all patients for study participation.
Statement of Ethics
The ethics committee of our institution (Okayama University Graduate School of Medicine, Dentistry and Pharmaceutical Sciences and Okayama University Hospital) approved the study design (approval No. 1607-014). All procedures were performed in accordance with relevant guidelines and regulations.
