Abstract
Background
Lymphomatosis cerebri (LC) is a rare manifestation of primary central nervous system lymphoma (PCNSL) with only a few cases reported in the literature, appearing as diffuse infiltrating process rather than a solitary mass. It is a non-Hodgkin’s type of lymphoma and is usually of the B-cell type origin.
Purpose
We intend to report this unique case of LC which came across as a diagnostic challenge.
Methods
A 53-year-old gentleman presented with complaints of two episodes of seizures 24 h apart followed by postictal confusion for 10–15 min. He underwent multiple MRI scans and underwent a biopsy of the lesion which reported infection, but he did not benefit from the treatment.
Result
The imaging was reviewed, suspicion of LC was raised and a review of histopathology was requested which later confirmed primary CNS lymphoma.
Conclusion
LC is a rare but established manifestation of PCNSL which mimics multiple other conditions. Understanding of the imaging pattern is important in making the diagnosis and differentiating it from other mimic conditions.
Introduction
Lymphomatosis cerebri (LC) is a rare manifestation of primary central nervous system lymphoma (PCNSL) with only a few cases reported in the literature. It differs from typical PCNSL by appearing as a diffusely infiltrating process rather than a solitary space-occupying lesion. 1 It is a non-Hodgkin’s lymphoma and is usually of B-cell origin. 2 LC mimics other diffuse white matter conditions such as demyelination, infection and gliomatosis cerebri, making the diagnosis challenging and prone to misdiagnosis. The histopathology sample is usually small, secondary to the ill-defined and scattered nature of lesions, which leads to a low yield and adds to difficulty in diagnosis. We present a case of a middle-aged immunocompetent gentleman who presented with seizures for 3 months and ultimately was pathologically proven to have LC. Approval was taken from the Ethical Review Committee of our institution (Aga Khan University) with reference number 2023-9036-25823. The need for consent was waived as all the information has been retrospectively taken from electronic medical records and the patient’s identity has not been revealed. No photographs have been used.
Case Summary
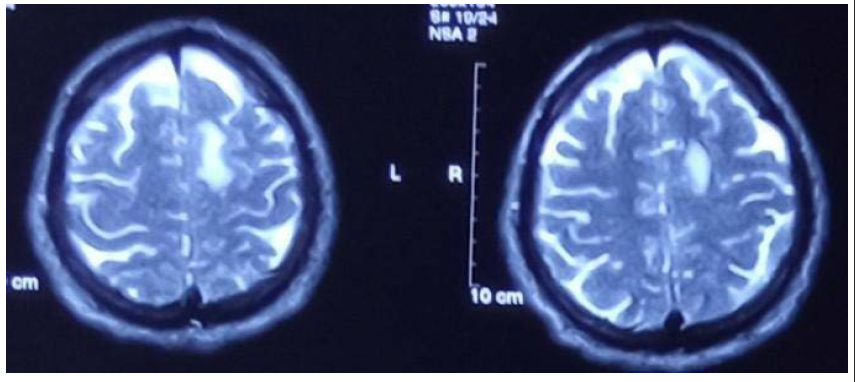
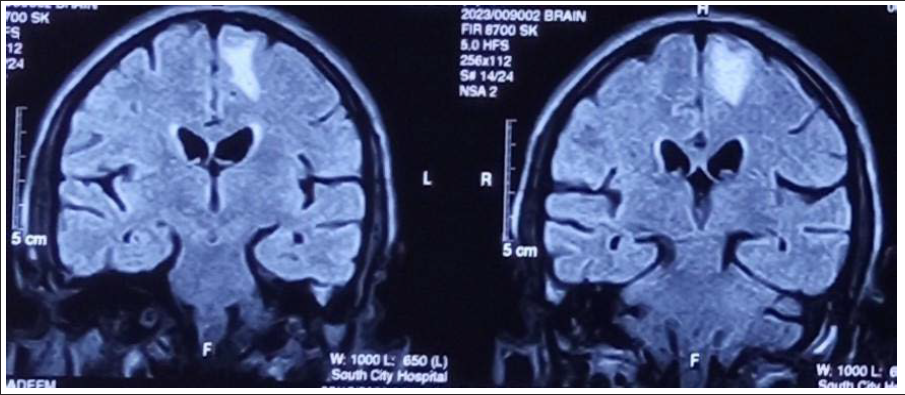
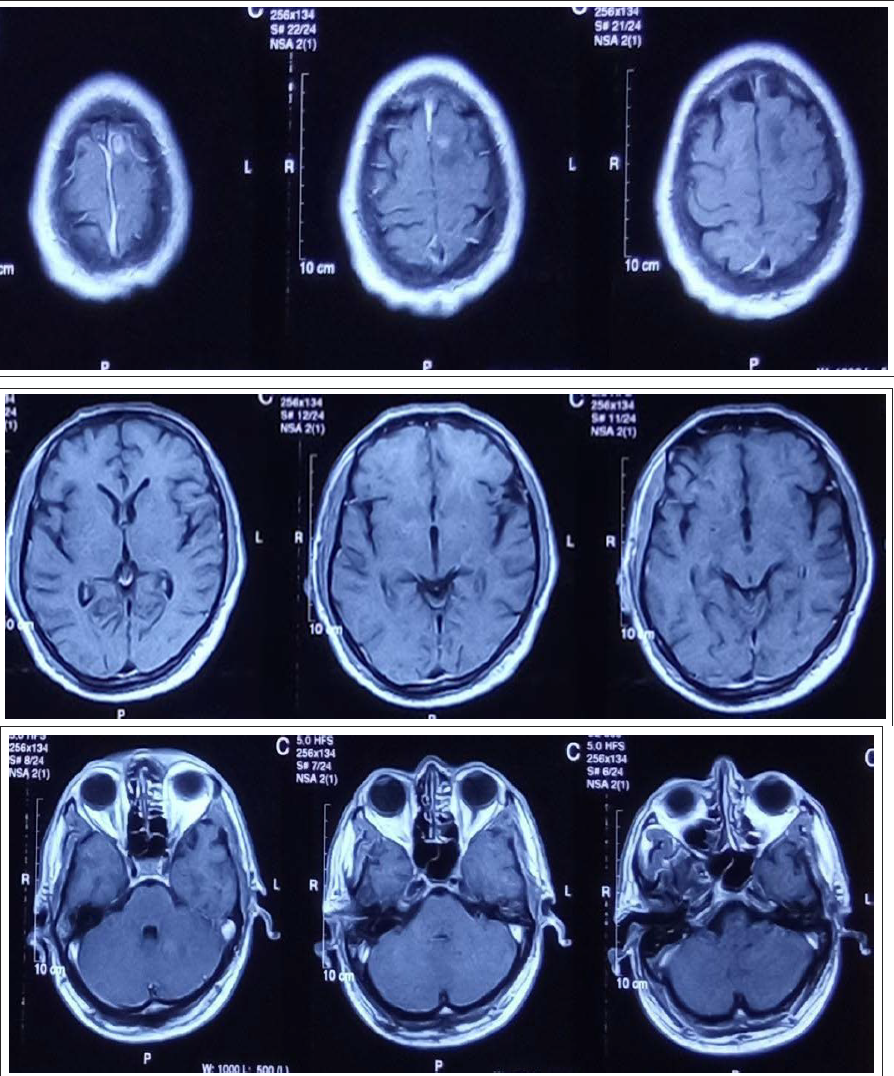
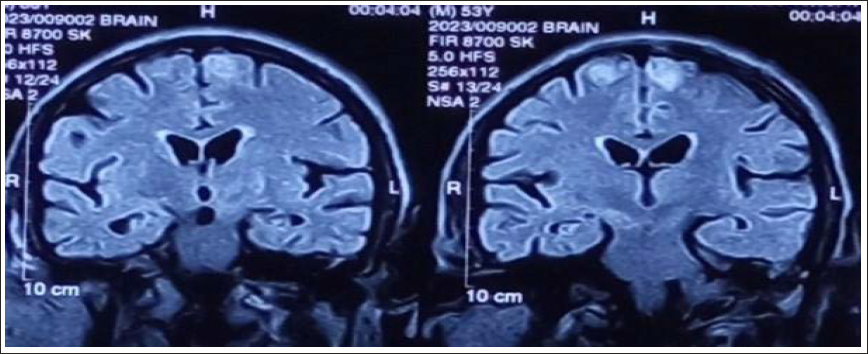
A 53-year-old gentleman presented with complaints of two episodes of seizures 24 h apart followed by postictal confusion for 10–15 min. He also complained of restlessness, decreased sleep and increased appetite for 2–3 weeks. He initially consulted a neurologist outside of our centre in March 2023 who ordered an MRI brain which showed an ill-defined T2 hyperintense lesion in the left frontal lobe at the vertex along with a few ill-defined lesions in the basal ganglia, left cerebellar hemisphere and left middle cerebellar peduncle (Figures 1–4). On post-contrast sequences, the frontal lobe lesion showed minimal enhancement, while the cerebellar lesions showed no post-contrast enhancement (Figure 5).
Axial T2-Weighted Images Showing an Ill-Defined Hyperintense Lesion in the Left Frontal Lobe Parasagittal Cortex.
Axial T2-Weighted Images Showing Ill-Defined Hyperintense Lesions in Bilateral Basal Ganglia.
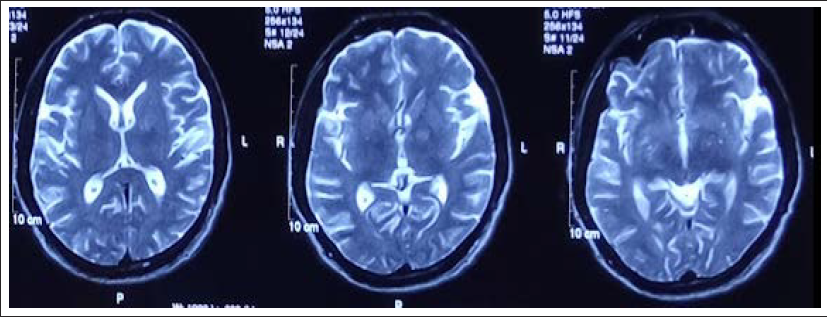
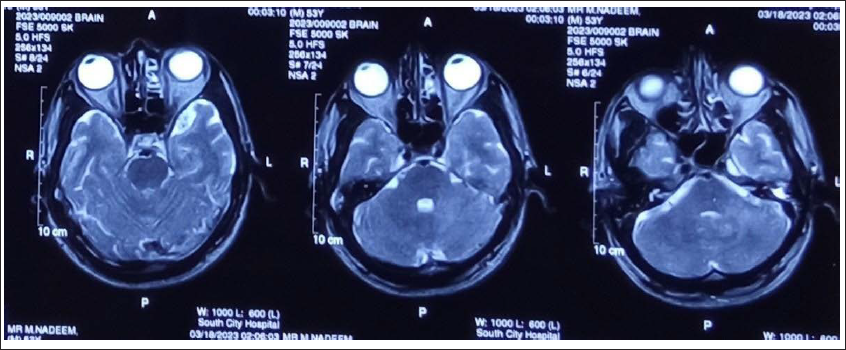
Axial T2-Weighted Images Showing Ill-Defined Hyperintense Lesions in the Left Cerebellar Hemisphere and Middle Cerebellar Peduncle.
Coronal FLAIR Images Showing Ill-Defined Hyperintense Lesion in the Left Frontal Lobe.
Axial T1 Post-Contrast Sections Showing Minimal Enhancement in the Left Frontal Lobe Lesion and Lack of Enhancement in the Rest of the Lesions.
He underwent a biopsy of the left frontal lesion. While awaiting the histopathology, the patient was conservatively managed and discharged home. However, his condition did not improve. He got a follow-up MRI brain in May 2023 which showed no change in the appearance of the previously seen lesions; however, a new lesion developed on this examination in the right medial frontal gyrus (Figures 6 and 7).
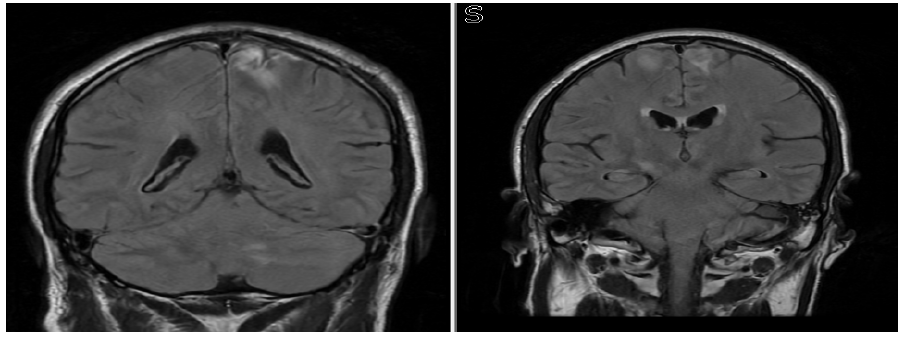
Coronal FLAIR Images of the Same Patient From Follow-Up MRI Done After 2 Months Showing Development of a New Lesion in the Right Frontal Medial Frontal Gyrus.
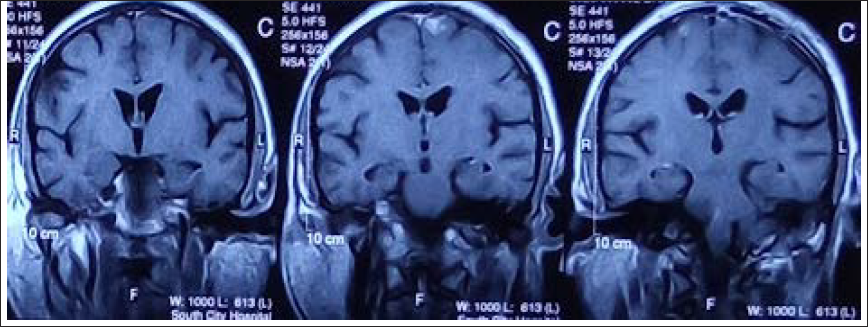
Coronal T1 Post-Contrast Images Showing Lack of Enhancement in the Right Frontal Lesion. Subtle Enhancement in the Left Frontal Lesion is Re-demonstrated.
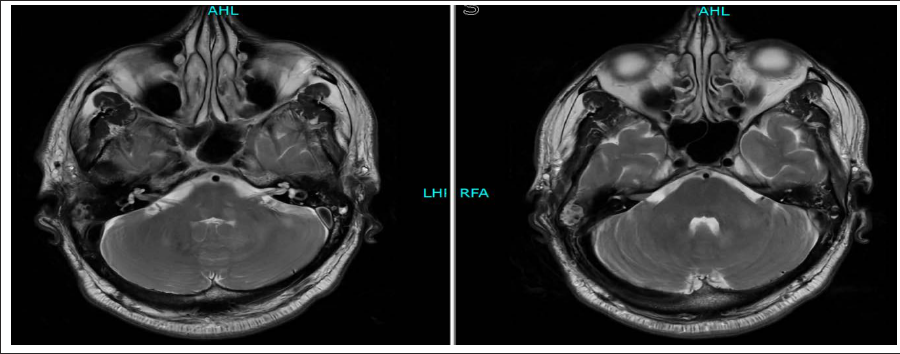
Then the patient presented in the ER in our centre and underwent another MRI brain scan which showed subtle improvement in the right frontal lobe lesion. The other lesions were stable. The complete examination carried out at our centre also included diffusion-weighted imaging which showed a lack of diffusion restriction in the lesions (Figures 8–10).
Axial T2-Weighted Images Showing Stable Left Cerebellar Lesions.
Diffusion Weighted Imaging Showing Lack of Diffusion Restriction Within the Lesions.
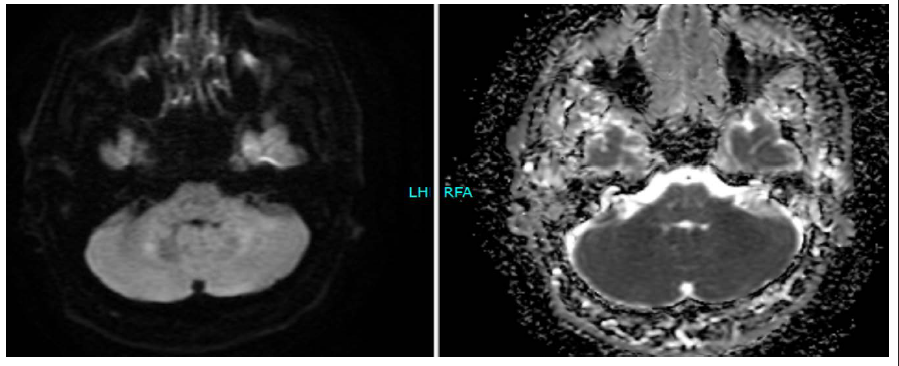
Coronal FLAIR Images Showing Improvement in Right Frontal Lobe Lesion and Stable Left Cerebellar Lesion.
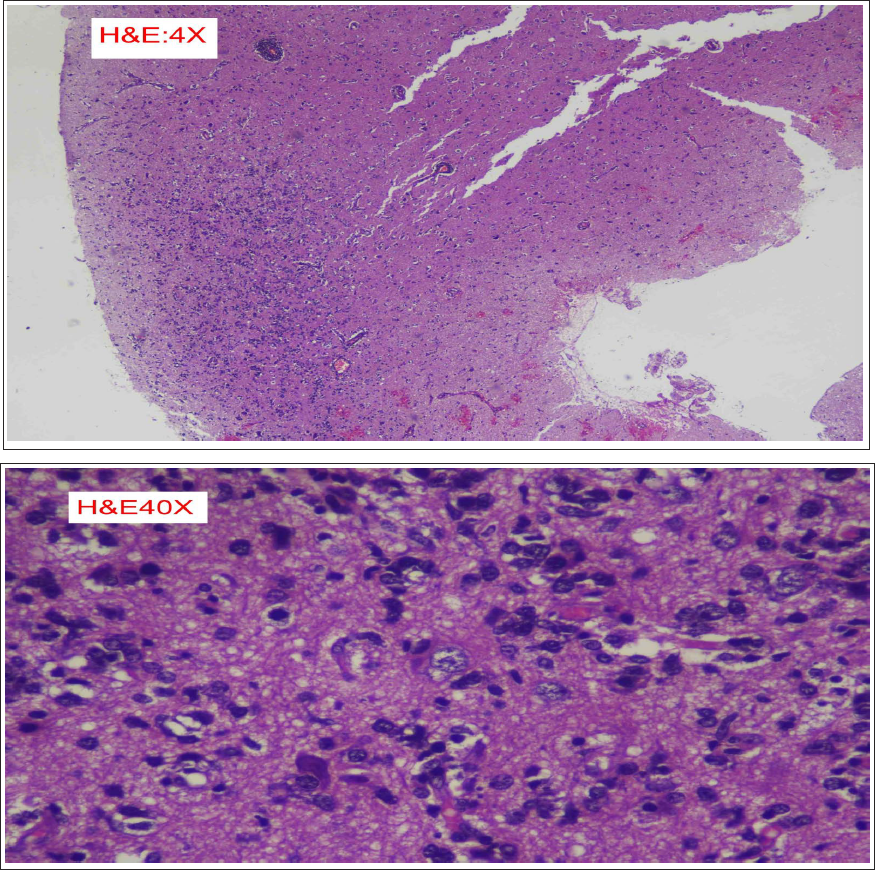
The imaging was then reviewed by a neuroradiologist at our centre, and suspicion of LC was proposed. Histopathology confirmed primary CNS lymphoma (Figures 11–13).
Brain Tissue Showing Neoplastic Lymphoid Infiltrate.
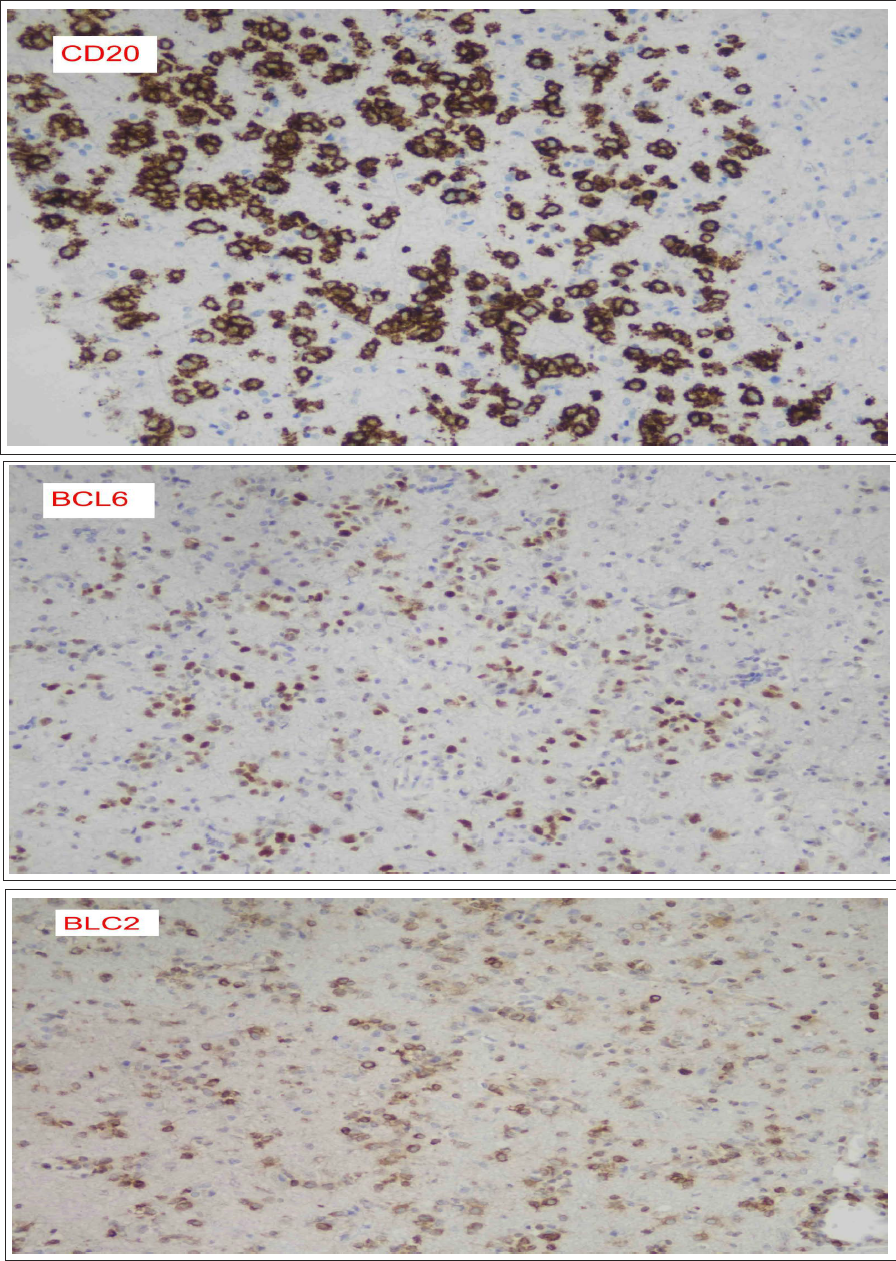
Neoplastic Lymphoid Cells Are Positive for CD20, BCL2 and BCL6 Immunostains.
Ki-67 Immunostain Shows a High Proliferative Index.
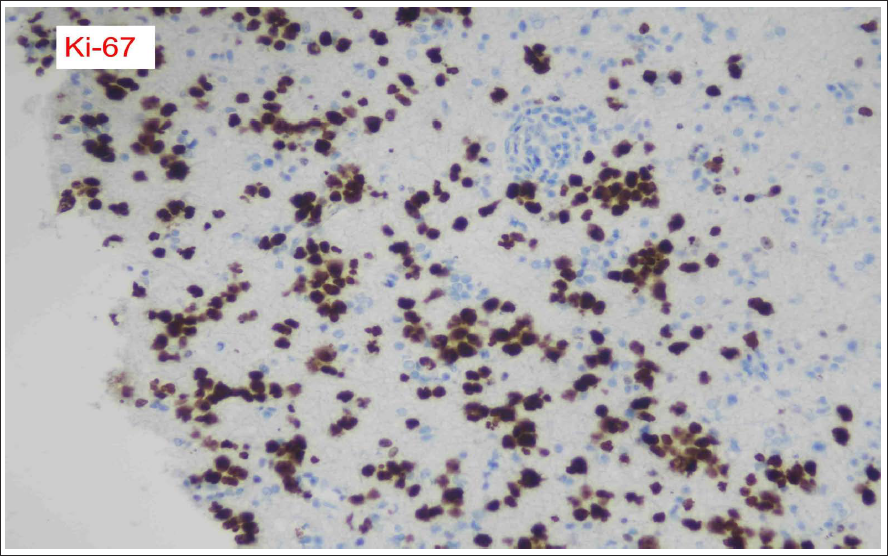
The patient was then diagnosed as a case of LC based on the involvement of more than three anatomical regions, lack of enhancement and minimal enhancement in the lesions and pathological diagnosis of primary CNS lymphoma. 3
Discussion
LC is an extremely rare subtype of PCNSL. Approximately 72% of LC cases are diffuse large B-cell lymphoma, while 28% are T-cell lymphoma.4, 5 Middle-aged and elderly males are most commonly affected, although females and younger adults may also be afflicted. 4
The conventional appearance of PCNSL is that of solitary or multiple discrete intra-axial lesions predominantly in a periventricular distribution which almost always show post-contrast enhancement and restricted diffusion. 6 LC, on the contrary, presents with a lack of focal mass formation, diffusely infiltrating both cerebral hemispheres in an asymmetric fashion, preferentially affecting the deep white matter, cerebral cortex/subcortical white matter, basal ganglia, thalamus and cerebellum. 4 There is typically faint or no contrast enhancement; DWI may show faint restricted diffusion or even facilitated diffusion.4, 7 Over the course of time, though, published series have shown LC to have progressive contrast enhancement and restricted diffusion as the blood–brain barrier breaks down and cellular material becomes more compact.4, 7
LC poses a diagnostic challenge as the imaging features are not entirely specific and overlap with a number of different pathologies. The differential diagnoses for this condition includes demyelinating disease, encephalitis and infiltrative tumor such as gliomatosis (12). These entities may involve similar brain regions to LC, however demyelinating disease such as multiple sclerosis tends to cluster around the callososeptal interface and may show rings of incomplete enhancement in the active phase. Autoimmune encephalitis may preferentially affect the limbic system such as the temporal lobes, while gliomatosis tends to be more confluent and mass like. Ultimately, confident diagnose may be lacking without biopsy. In a large systemic review of 45 cases with pathology proven lymphomatosis cerebri, all cases were misdiagnosed prior to biopsy or autopsy (11, Li onco letters).
Bakshi et al. reported two of the very first few cases of LC which presented as diffusely infiltrating non-mass-like lesions without contrast enhancement suggesting an intact blood–brain barrier. 8 They also proposed it as one of the differential diagnoses for rapidly progressive dementia. Patients with LC may have variable clinical symptoms such as gait disturbance, focal weakness, decline of cognitive function, memory disturbance, personality changes, dementia, anorexia, orthostatic hypotension, seizures, paraparesis and weight loss. Of these, the most common presenting symptoms are cognitive decline (59.5%), gait disturbances (54.8%) and behavioural changes (50%). 9 Pandit et al. 10 endorsed a similar clinical presentation of LC presenting with rapidly declining cognition and dementia in their case.
Previous studies of PCNSLs have shown that variations in the expression of LFA-1 (CD11a, α1β2) may be responsible for differences in individual tumour cell permeation of the brain. Loosely infiltrating cells in PCNSL showed less immunostaining for LFA-1 than did lymphoma cells within compact cell clusters. 11 Although it has not been studied in LC, it is assumed that a similar phenomenon is behind it. Rollins et al. demonstrated that although activation of microglial cells and astrocytes was prominent in areas of lymphomatous white matter infiltrates, no large areas of demyelination or extensive overt myelin breakdown were present. 12
As with most typical lymphomas, LC shows shrinkage or even disappearance after steroid administration due to cellular apoptosis. This may mislead clinicians into thinking this is an inflammatory rather than a tumoural condition; however, such a response is temporary and may even lead to negative biopsy results. As with all PCNSLs, high-dose Methotrexate-based chemotherapy followed by whole-brain irradiation is currently recommended for its management. The overall prognosis for LC is worse than typical PCNSL and 5-year survival after chemoradiotherapy remains at 20%–25%. 13 Most LC patients do not make it past 6 months after the initial onset of symptoms.
Conclusion
LC is a rare variant of PCNSL and should be considered when encountering a diffusely infiltrating process affecting both cerebral hemispheres in an asymmetric fashion, preferentially affecting the deep white matter, cerebral cortex/subcortical white matter and deep grey matter, with a lack of significant restricted diffusion on DWI and faint or absent contrast enhancement.
Abbreviations
LC, Lymphomatosis cerebri; PCNSL, primary central nervous system lymphoma.
Authors’ Contribution
The conceptualisation was done by SK and FM. The literature and drafting of the manuscript were conducted by SK. Radiological images have been collected by SK and FM. Clinical background was provided by DK. Histopathology images and details were provided by KM. The editing and supervision were performed by FM and RCC. All authors have read and agreed to the final version of the manuscript.
Statement of Ethics
Approval of the study was provided by the Ethical Review Committee of Aga Khan Hospital on 27-07-2023 (2023-9036-25823).
Footnotes
Declaration of Conflicting Interests
The authors declare no potential conflicts of interest concerning the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
