Abstract
Background:
Coma after acute brain structural injury (ABI) are associated with high mortality and disability. Somatosensory evoked potentials (SSEP) N20 and N70 are used to predict prognosis.
Purpose:
We assessed the utility of SSEP (N20-N70) as an early indicator of long-term functional prognosis in these patients.
Methods:
We conducted a retrospective cohort study of patients admitted to the intensive care unit (ICU) with a diagnosis of coma after ABI (n=60). An SSEP study including N20 and N70 was performed 24–72 hours after coma onset. Functional recovery was evaluated 6 to 12 months later using the Modified Glasgow Scale (mGS). The study was approved by our local research ethics committee.
Results:
The absence of N20 (41% specificity=100%) or N70 (78%) was a strong indicator of a poor outcome. In contrast, the presence of N70 was an indicator of a good outcome (specificity=64.2% sensitivity=91.3%).
Conclusion:
SSEP N20 and N70 are useful early prognostic markers with high specificity (N20) and sensitivity (N70). N70 has potential additional value for improving the prediction of good functional outcomes in the long term.
Introduction
The long-term neurological prognosis of patients admitted to the intensive care unit (ICU) with coma after cardiac arrest (CA) and other causes of coma after severe acute brain injury (ABI) are uncertain in most cases. 1
Hypoxic-ischemic encephalopathy can occur after CA and remains one of the leading causes of death and disability in these patients. 2 Post-CA syndrome is characterized by diffuse brain damage, myocardial dysfunction, and systemic ischemia-reperfusion response. 1
ABI is considered an emergency3, 4 because its most frequent causes are traumatic brain injury and cerebrovascular disease, and both conditions benefit from specific treatments that could potentially reverse the damage.
To evaluate the likelihood of patient recovery, the European Resuscitation Council 5 and French recommendations for clinical neurophysiology 6 describe techniques that are useful for predicting neurological prognosis. Neurophysiological tests such as somatosensory evoked potentials (SSEP N20) and electroencephalogram (EEG) are the most frequently used techniques. When combined, they provide diagnostic and prognostic information for patients with coma. 7 None of these tests is perfect, and it is recommended that a combination of tests is applied before making the drastic decision to stop life support.8, 9
SSEP reflect cortical function, as they indicate the integrity of the somatosensory pathway. 10 It is widely known that the absence of N20 potentials has a high predictive value for poor neurological prognosis;10–13 however, its utility in predicting good prognoses is limited.8, 9, 14
Previous studies15, 16 have suggested that the N70 SSEP could improve prognostic evaluation as an indicator of cortical function, especially in patients with coma after CA, to a lesser extent in patients with coma after ABI, because it is generated by cortico-cortical interactions with the ascending reticular activating system. 9 N70 is particularly vulnerable to hypoxemia.16, 17 The SSEP study should be conducted at least 24 h after injury onset, as earlier determinations can be interfered with by the cooling-off period, 18 increasing the false positive rate (FPr).
The usefulness of the N70 potentials in patients with coma secondary to structural lesions has not been clearly established. It is essential to consider that the mechanism of injury because of etiology is different from coma after CA. Several authors describe a high specificity (Sp) of N20 for predicting poor prognosis.9, 19, 20
Our primary objective is to analyze the accuracy of N70 SSEP in combination with N20 SSEP in predicting long-term neurological outcomes in patients who have suffered ABI.
Methods
We conducted a retrospective analysis with prospective recruitment at a single academic hospital center. The General University Hospital of Albacete maintains a prospective registry of all tests performed on critically ill patients. We used this registry to identify patients treated for three consecutive years.
Patients
Patients who were diagnosed with coma after ABI and over 18 years of age were consecutively selected when admitted to the ICU. Data analysis was conducted for three consecutive years. We excluded underage patients, those who did not live in the hospital area (to avoid problems in follow-up), and those who previously had a neurodegenerative disease (Figure 1).
Methodology Inclusion, Exclusion of Patients, and Procedures.
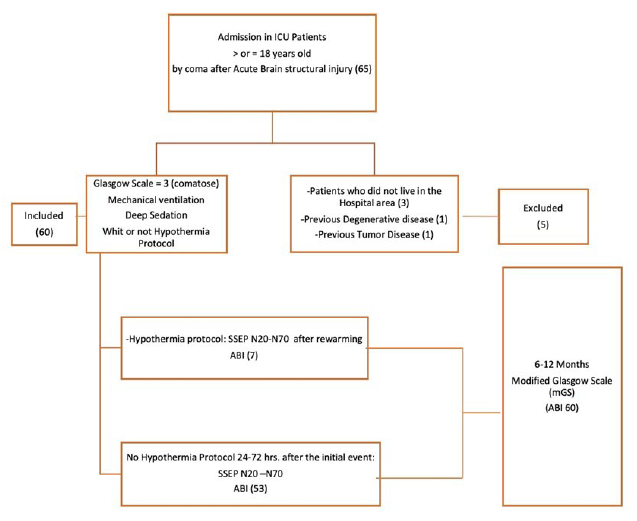
Procedures and Variables
All patients underwent a complete neurophysiological study as a routine practice. The study included N20 and N70 SSEP performed 24 h to 72 h (24–48 h or 48–72 h) and always following rewarming if hypothermia was administered as recommended. 21 Therapeutic hypothermia (TH) was applied according to our local protocol (target temperature of 33°C for 24–48 h). The Glasgow scale was assessed prior to the performance of the SSEP: ocular aperture, verbal response, and motor response. All patients were assessed for brainstem reflexes (photomotor, corneal, and oculocephalic).
The study of SSEP was conducted using XLTEK-Protector equipment according to a standard protocol with monopolar subdermal needle electrodes applied at the bilateral Erb point, the spinous process of C7 with anterior cervical reference, the bilateral shoulder and according to the international system 10 to 20 in Fz, C3, and C4 15 with the reference in the ipsilateral auricular lobe. In all cases, stimulation was performed in the bilateral median nerve in the wrist with filters 30 Hz to 3000 Hz (Erb-C5) 1 Hz to 250 Hz (cortical recordings) analysis time of 100 ms. 22 Two block stimulations were performed. If the averages presented a significant artifact, a muscle relaxant was administered. Peripheral (N9, N13, and P14) and cortical responses (N20 and N70) were obtained. For this study, N20 and N70 SSEP results were dichotomized in the presence (when bilaterally present) or absence (absent on at least one side) of N20 and N70, respectively. Unilateral absence is an abnormal response (level 3, clinical interpretation of median nerve SSEP modified from Cruccu) 23 ; therefore, the responses are dichotomized as bilateral present and unilateral or bilateral absent, and amplitude was not considered. The maximum latency N70 was 110 msec.
For the EEG recording, subdermal needle electrodes were used according to the reduced international 10 to 20 system and electrocardiogram, and 20 min of recording during sound and pain stimulation were obtained in all patients. In this study, EEG patterns were divided into favorable (mild, moderate, or severe encephalopathy) and unfavorable (critical or intercritical epileptic patterns, status epilepticus, burst suppression, and electrical silence)1, 11 and were systematically collected from each patient. Malignant rhythm and favorable patterns were identified in other studies. 10
We assessed the effects of the following variables on the neurological outcome: sex; age; treatment of sedation: fentanyl, midazolam, or fentanyl and midazolam; and treatment hypothermia: protocol for 24 h to 48 h cooling with noninvasive methods to a goal temperature of 32°C to 33°C, followed by progressive normothermia to 35°C to 36.5°C. Performance of SSEP 24-72 hours after coma, duration after CA (considered prolonged if longer than 20 min), place of CA (hospital or out-hospital), and type of brain injury (traumatic, subarachnoid hemorrhage, others: stroke, brain vascular malformations), temperature, and blood test parameters (glucose >200 mg, sodium, potassium, urea, and creatinine).
Finally, the patients’ long-term functional and neurological outcomes were evaluated between 6 months and 12 months after the initial assessment. For this purpose, we used the modified Glasgow Scale (mGS) 24 similar to other studies25, 26: 1 = death, 2 = persistent vegetative state, 3 = severe disability (conscious but dependent), 4 = moderate disability (disabled but independent), and 5 = no disability. An mGS score between 1 and 3 was considered to indicate an unfavorable outcome, and an mGS score of 4 to 5 was considered a good outcome.
Data Collection
In our center, the patients’ medical information was included in a computerized hospital registry that was under the supervision of the documentation service. We reviewed these electronic medical records to obtain the information required for the study, and follow-up was performed by telephone interview. Data were analyzed in protected clinical data systems and de-identified for final storage.
Ethical Aspects
The relatives of the studied patients gave written informed consent for the neurophysiological tests that were performed during the patients’ stay in the ICU. The study was approved by our local research ethics committee. Confidentiality of the information was maintained throughout the study.
Statistical Analysis
In the descriptive analysis, the mean and standard deviation are provided for normal quantitative variables, and the number of patients and percentages are shown for qualitative variables.
For the statistical analysis, many variables were used in a dichotomous fashion, taking into account previous studies and guidelines (Sandroni, 2014). 27 These include N20/N70 (presence vs. absence), hyperglycemia (glucose level > 200 mg/dL), sedative medication (yes: fentanyl, midazolam, or fentanyl and midazolam or not), functional outcome (unfavorable vs. good), duration of CA (prolonged > 20 min or not), place of CA (out-of-hospital vs. intrahospital), and TH protocol (yes vs. no). The chi-squared test and Fisher’s exact test were used as appropriate to evaluate the associations of these dichotomic variables with functional outcomes. To assess quantitative variables, the Student’s t-test was used. For all comparisons, the level of significance was P < .05, and statistical precision was determined by the 95% confidence interval (CI). We used binary logistic regression in relation to neurological prognosis.
The diagnostic accuracy for predicting long-term functional outcomes using each test (N20, N70) was estimated using sensitivity (Se), Sp, positive predictive value, negative predictive value, and FPr; the values for each of these parameters are shown with their 95% CIs.
The data were analyzed using SPSS Software v.22 (SPSS Inc., Chicago, IL, USA).
Results
Characteristics of the Study Population in Acute Brain Structural Injury (n = 60).
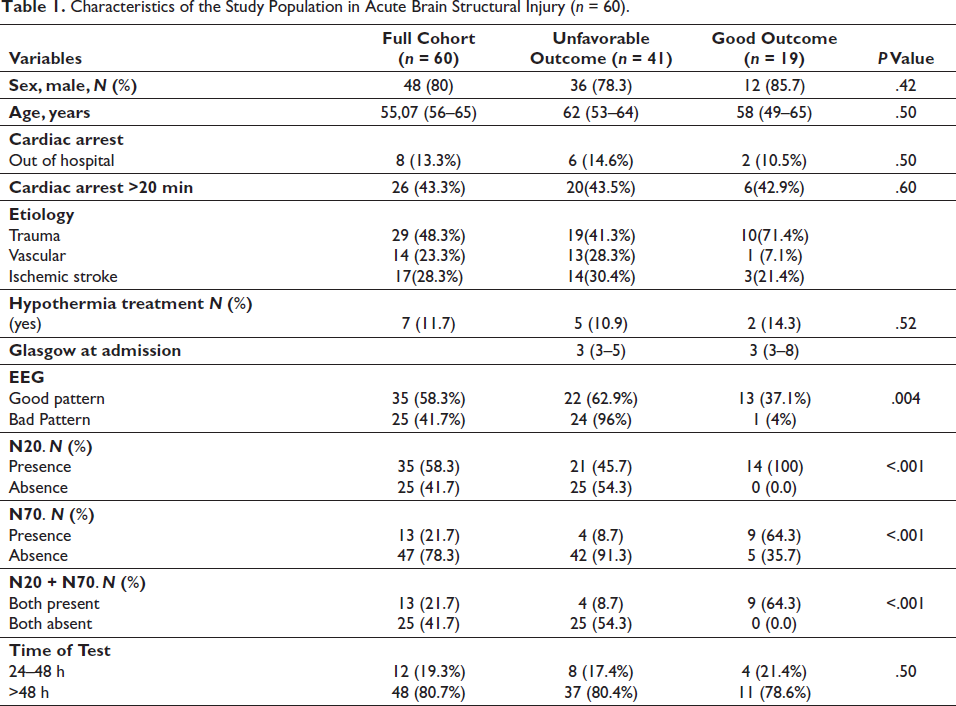
Diagnostic Accuracy of Somatosensory Evoked Potentials (SSEP) (N20 and N70) and EEG for Long-Term Outcome Prediction in Patients with Coma After Acute Brain Structural Injury (ABI).
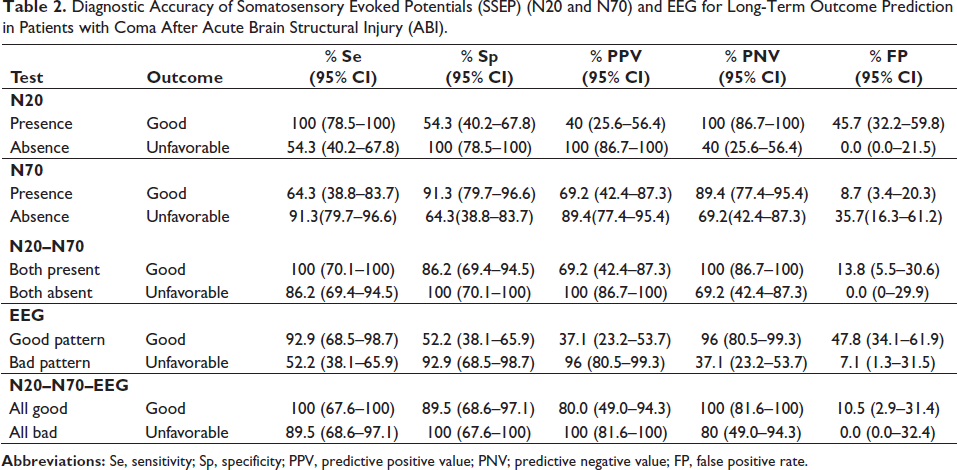
EEG alone was a worse predictor of a negative outcome in ABI than in CA (Sp 92.9, FP 7.1%); one patient with an ominous EEG pattern achieved full recovery. He was 89 years old with traumatic brain injury, and both N20 and N70 were present in his SSEP.
Logistic Regression Modified Glasgow Coma Scale and Predictor to Good Outcome in Patients with Coma After Acute Brain Structural Injury (ABI).
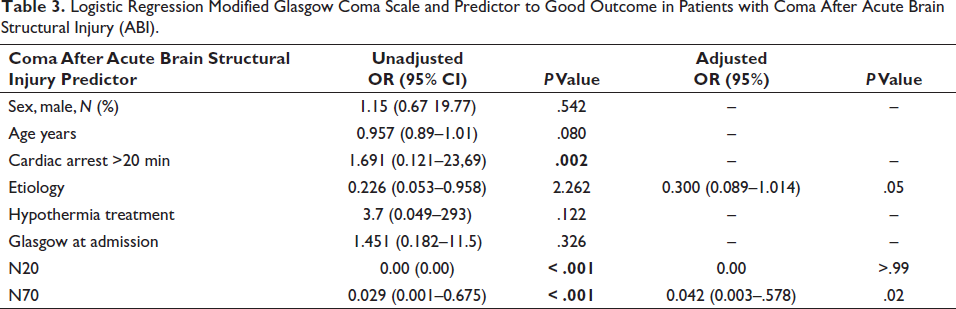
The predictors of mortality in ABI were older age >60 years (mean age of survivors vs. deceased patients: 49.23% vs. 60.2%, P = .14), N20 absence, and an unfavorable EEG pattern (OR 3.3 (1.8–5.9) P-value < .001).
Discussion
In our study, the absence of N20 with a specificity of 100% (0% FP) was validated as an optimal marker of poor functional prognosis from 6 months to 12 months after admission in patients with postanoxic coma after ABI, in concordance with previous studies.16, 28 However, the presence of the N20 response does not allow for the prediction of a good prognosis in these patients, 5 and this is where N70 proves to be an added value because its presence increases the possibility of a favorable neurological evolution after ABI (64.3% Se). Because in itself it represents the preservation of cortico-cortical synapses which are essential for arousal and recovery of coma as described by Lachance et al. 10 It should be noted that in our study the absence of the short latency potential N20 already indicates the absence of the long latency potential N70. Their combined presence (when the long-latency potential accompanies the short-latency potential) increases the Se 100% for good prognosis and Sp 100% with an FPr of 0 for poor prognosis; therefore, the N70 potential gives an added value to the evaluation of the critical patient after ABI.
The N70 response was previously analyzed by Madl et al. 15 in 162 patients with postanoxic coma but only for coma after CA. They found that the absence of this potential had an Sp of 97% and an Se of 94% in predicting a poor functional prognosis. In our series, we found that N70 had a similar Se (91.3%) but a lower Sp (64.3%); we hypothesized that this could be because of differences in the times of evaluation and cooling-off periods and etiology used in our study and the study of Madl et al. 15
In the study of Logi, 19 the absence of N20 had an Sp 60% to 77% and an Se 31% to 33% in correlation with poor functional prognosis; in our study, we had an Sp 100% and an Se 54.3%. The main difference in our study is that the time of PESS was between days 1 and 46.
Sherman et al. 29 proposed that the absence of N70 has an Se of 62% and an Sp of 100%. Their study differed significantly from ours in two respects: in their study, N70 presence was accepted up to 176 msec (we considered this to be too long, and in our study, this was recorded as absence), and SSEP evaluation was performed earlier than in our study (12 h after CA), possibly resulting in more false positives and false negatives and the etiology is only CA.
We found that the Se of N20 was 54.3%, while that of N70 was 91.3% for an excellent functional outcome, indicating that N70 presence is more reliable than N20 presence. 3 The data presented in our study are in concordance with those of previous reports, 17 corroborating the existence of confounding factors SSEP as for EEG related to structural damage that could interfere with the somatosensory pathway and alter SSEP acquisition (e.g., transient absence of N20) in up to 15% of cases. 30 These disruptions are because of confounding factors such as edema, space-occupying lesions, increased intracranial pressure, and even the placement of electrodes, which are often conditioned on craniectomies or bone injuries. However, this does not change the prognostic value of these tests because most of these patients have severe neurological sequelae. 31 EEG controls are needed according to the patient’s evolution. Favorable EEG patterns are related to a high Se 92.9% for good functional recovery; in our study, additionally, the presence of PESS correlates with a good long-term functional prognosis.
Finally, among the limitations of the study is its retrospective design. We were not able to evaluate other important prognostic factors, such as amplitudes because when the study was designed it was not planned and the measurements were not taken; decreased SSEP (N20) is associated with poor outcomes after CA, 32 neuroimaging 33 and neuron-specific enolase, or other tests, such as P300, or MMN. Another significant limitation of this type of study is the “self-fulfilling prophecy” bias. Although it is impossible to entirely exclude the possibility that the results obtained for SSEP could have led to the interruption of ICU care and therefore affected the odds of an unfavorable outcome, as far as we know, at the time this study was conducted in our hospital, the SSEP result carried little weight in decisions regarding withdrawal or maintenance of therapy. Another critical factor is TH, which can affect SSEP. We were careful to always obtain SSEP after the patient’s temperature was normalized. Lastly, the sample size, as well as the data analysis, could not be extended.
Conclusion
In patients with coma from structural damage with the bilateral presence of N20, the determination of SSEP N70 is an early prognostic marker of good functional outcome in the long term. This represents a simple technique that has proven to be an excellent complement to previously established prognostic markers such as N20 and EEG. The information presented here should be of great value and can complement the multimodal decision when determining the long-term prognosis to limit therapeutic effort.
Footnotes
Acknowledgments
We thank all the doctors of the Clinical Neurophysiology Department of University General Hospital of Albacete, especially Dr Pardal for encouraging this work at a specific moment of my training and Dr Lorena Rossi for her support. We also thank the nursing staff for their collaboration.
Authors’ Contribution
AA initiated and concluded the study, DM performed the SSEP tests, and TS supervised the design of the research and the neurological approach. EF contributed to the statistical design, reviewed the manuscript and supervised its English translation. ID conducted part of the analytical study and the review of the paper. EG oversaw the statistical study and its development. All authors read and approved the final manuscript.
Statement of Ethics
Ethical approval was obtained from the institutional ethics committee. The relatives of the patients studied gave written informed consent for the administration of the neurophysiological tests that were performed during the patients’ stay in the ICU on admission to the critical care unit. Confidentiality of information was maintained throughout the study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
