Abstract
Background
Parkinson’s disease (PD) is typified by inflammation of dopaminergic neurons leading to the release of various inflammatory mediators. These mediators activate the transcription factor NF-κB, which in turn activates inducible nitric oxide synthase (iNOS), leading to increased inflammation.
Purpose
This study was intended to study the effect of combination of mangiferin, a specific inhibitor of NF-κB with low-dose nitric oxide (NO) modulators.
Methods
A total of eight Wistar rats weighing 200–250 g were used in each group. Stereotactic surgery was performed to induce 6-hydroxydopamine (6-OHDA) lesions. The treatment period extended from day 14 to day 42, during which time behavioral tests were performed to evaluate the effects of mangiferin and its combination with NO modulators. On day 42, the brains of the rats were removed for biochemical and molecular analyzes.
Results
Mangiferin significantly improved locomotor activity and decreased inflammatory chemokines levels in rats with 6-OHDA lesions. Mangiferin therapy decreased myeloperoxidase (MPO) levels and reduced oxidative stress. In particular, caspase-3, caspase-9 and COX-2 activities were significantly reduced after the mangiferin treatment. A combination of 45-µg mangiferin and 10-mg/kg L-NAME showed the greatest improvement in locomotor, behavioral, biochemical, and molecular parameters impaired by 6-OHDA.
Conclusion
In this study, mangiferin was found to protect rats with 6-OHDA lesions by inhibiting inflammation causing chemokines such as TNF-α and IL-6. Besides, the grouping of iNOS inhibitor L-NAME at a dose of 10 mg/kg with 45-µg mangiferin enhanced the anti-inflammatory and anti-Parkinsonian activity of mangiferin. Consequently, the combination therapy of mangiferin and L-NAME is promising for the treatment of PD. However, clinical trials will be required to evaluate the efficacy of this combination therapy in humans.
Introduction
Parkinsonism is a complex and advancing condition characterized by the gradual deterioration of the dopamine-generating neurons in the striatum area of the brain. This predetermined demise of cells results in numerous indications, including tremors, hindered motor management, and rigidity. Microglial cells, specialized immune cells of the brain, become active in response to neuroinflammation, which can eventually cause the demise of dopaminergic neurons. This revelation has paved the way for novel approaches to treating Parkinson’s disease (PD), such as employing anti-inflammatory medications and immunomodulators.1–10
NF-κB regulates inflammation and impacts the production of various chemokines and other agents that cause inflammation. NF-κB triggers the activation of several genes, including chemokines such as TNF-α, IL-1β, and IL-6, which play a role in the innate and adaptive immune response and aid in the attraction and activation of immune cells such as neutrophils, macrophages, and T cells.11–16
The inducible nitric oxide synthase (iNOS) gene’s production of nitric oxide (NO) is controlled by NF-κB through binding to specific sites in its promoter region. The elimination of these binding sites has been found to hinder iNOS transcription in reaction to chemokines, leading to a reduction in NO production. NO is a crucial signaling molecule involved in several physiological and pathological processes, such as the regulation of blood pressure, neurotransmission, and immune system functions. Notably, NO has been discovered to regulate the activity of NF-κB, which is an important immune response regulator. Interestingly, chemokines can activate iNOS to produce reactive nitrogen species, which promotes the continuous activation of NF-κB. This reciprocal relationship between NO and NF-κB is crucial in regulating immune responses and resolving inflammation.17–30
It is essential to have a thorough comprehension of the intricate interplay between NO and various other signaling molecules to make progress in the creation of fresh treatments for PD and other neurodegenerative ailments.31–33
Furthermore, the research has indicated that mangiferin possesses the capability to restrain the production of chemokines such as IL-1, IL-6, IL-12, and TNF-α, along with NO in macrophages and phagocytes. Its anti-inflammatory characteristics are akin to those of aspirin, indomethacin, and ibuprofen and impact the cyclooxygenase (COX)-2 enzyme. All in all, these findings indicate that mangiferin holds great potential as a curative measure for inflammatory ailments.34, 35
The aim of this research was to examine whether a combination of mangiferin and NO modulators could provide a remedial effect in rats with 6-OHDA lesions. The study had specific objectives:
To assess the impact of 6-OHDA lesions on levels of chemokines (TNF-α, IL-4, IL-6, and IL-1β) and COX-2 activity in the striatum of rats. This assessment aimed to indicate the degree of inflammation and neurodegeneration in PD. To investigate the impact of mangiferin, a natural anti-inflammatory substance, on the levels of chemokines such as TNF-α and COX-2 activity in rats with 6-OHDA-induced Parkinsonism. The objective was to evaluate the potential therapeutic efficacy of mangiferin in reducing inflammation and preventing further neuronal damage. To examine the effect of levodopa, a commonly used pharmacological treatment for PD, on the levels of pro-inflammatory chemokines and COX-2 activities in rats with 6-OHDA-induced lesions. To explore whether combining mangiferin with a NO modulator could have synergistic effects in reducing inflammation and improving motor function in rats with 6-OHDA-induced lesions.
In essence, our objective with this research was to enhance our comprehension of the underlying pathways that drive the progression of PD, while exploring the possible remedial advantages of different remedies. These remedies comprise of both organic compounds and pharmaceutical therapies, with a particular emphasis on their capacity to alleviate inflammation and enhance motor abilities. Our aim with this study is to augment the existing knowledge base and establish a foundation for more effective treatment protocols for those impacted by PD.
Methods
Animals
This study involved the selection of male Wistar rats with a weight range of 200–250 g. They were kept in groups of 3–5 rats in cages that were ventilated individually and followed a 12-h light–dark cycle. The rats had free access to standard food and water. The temperature in the cages was maintained at 21°C –22°C and humidity at 50%–55%. The Institutional Animal Ethics Committee (IAEC) of King George Medical University (KGMU), Lucknow granted ethical approval for the study. The study was conducted in compliance with IAEC guidelines and approval was obtained before the study began, with reference number 84/IAEC/Pharma/2017.
Pre-Operative Care
Upon arrival at the facility, rats underwent a period of adaptation lasting at least 1 week before the commencement of the experiment. This critical adaptation phase aimed to help the rats adjust to their new surroundings, promote their welfare, and enhance the reliability of the experimental outcomes. Prior to the surgical intervention, each rat was meticulously distinguished based on criteria such as age, health, and weight, and handled with utmost care to minimize any potential stress during the procedure.
6-OHDA Surgery in Rats
Surgical Environment and Aseptic Technique
Stringent precautions were implemented to establish a germ-free surgical milieu, which involved segregating the operational zone from routine laboratory operations. Prior to the commencement of any surgical interventions, all surfaces underwent a meticulous decontamination process with 70% ethanol. The surgical equipment was subjected to appropriate sterilization protocols to guarantee their sterility and efficiency. In order to avoid anesthesia-induced hypothermia, a warming pad was positioned beneath one side of the cage to ensure the rat’s comfort and welfare.
Preparation of Drugs for Surgery
The 6-OHDA solution was newly made immediately prior to the surgical procedures to ensure its effectiveness. In order to maintain the stability of 6-OHDA bromide (6-OHDA.Br) and prevent it from oxidizing into an inactive form, it was dissolved in a sterile saline solution with 0.2% ascorbic acid. To create the 0.2% ascorbic acid solution, precisely 0.2 g of ascorbic acid was weighed and transferred into a graduated cylinder. Sterile saline solution (0.9%) was then added to the measuring cylinder to make the volume reach 1 L.
To prepare a working concentration of 6-OHDA.HBr, 5 mg of the drug was mixed with 0.8 ml of sterile saline and 30 mM 6-OHDA containing 0.03% ascorbic acid was achieved. The pH of the solution was maintained at five throughout the procedure. The prepared solution was divided into aliquots and stored at –20°C until needed for use.
6-OHDA Administration 36
The surgical site and equipment were meticulously disinfected with 70% ethanol to establish a sterile setting.
The Hamilton needle was attached to the syringe and filled with sterile saline solution containing ascorbic acid.
The animal was anesthetized with a mixture of ketamine and xylazine to achieve unconsciousness.
The animal’s head was cautiously shaved and sanitized with 70% ethanol to ensure sanitation.
The animal was positioned under the stereotactic frame with the skull firmly secured to prevent any movement during the procedure.
The stereotactic coordinates for the substantia nigra region were determined using the reference book “Paxinos and Watson” (Figure 1), The Rat Brain in Coordinates.” These coordinates functioned as a guide for precise targeting during surgery. 37
The skin was incised in the center and the fascia covering the bone was cautiously removed to locate the bregma, a specific anatomical marker.
The needle was accurately positioned over the bregma using the provided stereotactic coordinates.
A borehole was drilled into the rat’s brain and a small puncture was made in the meninges, the protective membrane surrounding the brain.
The 6-OHDA-HBr solution was prepared and a micro-syringe was loaded with the solution.
The syringe was firmly attached to the pump and the needle was connected to the Hamilton needle to ensure an appropriate delivery system.
The infusion pump was started at a regulated flow rate of 0.5 µL/min to administer the 6-OHDA-HBr solution.
The needle was left in the syringe for 5 min before it was slowly removed.
Rats were cannulated with a 20-gauge needle for future treatments or procedures.
After a 14-day period following the injection of 6-OHDA, the rats were selected for further study based on the occurrence of opposite rotations when subjected to an apomorphine test, a commonly used test to evaluate lesion efficacy.
Rat Brain Co-ordinates for Substantia Nigra Pars Compacta Dorsal Part.
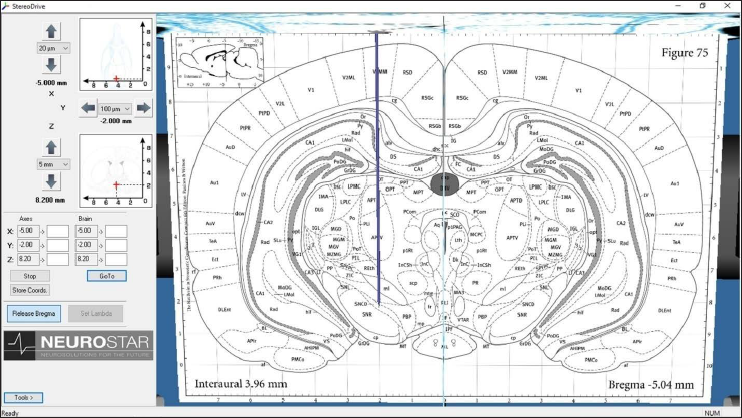
The proposed plan for therapy involved the use of mangiferin at a quantity of 45 µg through a cannula that was implanted in the brain. This therapy was given as a standalone measure or combined with low-dose NO modulators, namely L-Arginine (100 mg/kg) and L-NAME (10 mg/kg). The treatment schedule lasted from day 14 to day 42, amounting to duration of 28 days. The remaining treatments were given intraperitoneally (i.p.).
Following mangiferin therapy, the rats were subjected to behavioral testing to assess any alterations in locomotor function in the presence of 6-OHDA lesions. At the end of the research, the rats were humanely put to sleep using high-dose anesthesia (pentobarbital 100 mg/kg) and their brains were carefully extracted for further biochemical and molecular analyses.
Drugs and Chemicals
6-OHDA, mangiferin, levodopa, 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES), magnesium chloride (MgCl2), ethylenediaminetetraacetic acid (EDTA), hexadecyl-trimethyl-ammonium bromide, o-dianisidine dihydrochloride, phenylmethylsulfonyl fluoride (PMSF), tri-pyridyl triazine, apomorphine, pyrogallol, and Griess reagent were procured from Sigma (Millipore Sigma, Burlington, USA). Enzyme-linked immunosorbent assay (ELISA) kits for the quantification of NF-κB, TNF-α, IL-1, IL-4, and IL-6 were obtained from Cloud Clone Corp (Cloud Clone Corp, Katy, Texas, USA). Caspase-3 and caspase-9 were detected using a commercially available assay kit for caspases (BioVisionInc, Milpitas, California, USA). Cyclooxygenase levels in rat brain tissue were measured using an ELISA kit (CUSABIO Houston, Texas, USA).
Behavioral and Locomotor Analysis
Animal Activity Meter: Opto-Varimex-5 Auto-Track
A Columbus Instruments (USA) animal behavior measuring device was utilized to gauge various parameters, such as the length of total movement, rest duration, and stereotypical behavior duration. The device integrates auto-tracking technology, which employs sophisticated techniques for measuring motor activity and monitoring the distance covered by animals for behavioral analysis. The device comprises a sequence of infrared photocells placed strategically throughout the testing area to sense motion, while a separate sequence of photocells positioned above the animal detects vertical motion. By simultaneously tracking the deflection of the beam along the horizontal axes (X and Y), the device generates precise coordinates that indicate the animal’s position. By scrutinizing the recorded patterns, subtle modifications in the animals conduct during the experiment can be identified and assessed.
Any-maze Video Tracking System
The conduct and movement of the rodents were evaluated using the Any-Maze video tracking system from Stoelting (USA). The rats were positioned in a particular area of an unenclosed space for a brief duration. Their activities were recorded by a camera mounted above, while the Any-Maze program was utilized to scrutinize the gathered data. The software was used to measure different factors including the distance covered, average pace, length of immobility, and number of immobility episodes. This mechanism facilitated an extensive appraisal of the actions and mobility patterns of the rats throughout the test phase.
Cylinder Test
To evaluate the imbalance of the musculoskeletal system in rats, we executed the cylinder examination. This examination is a well-established technique for evaluating locomotor imbalance in animal models of central nervous system ailments. Throughout the examination, the rats were positioned in a transparent plastic cylinder with the upper part open. As they stood up against the wall of the arena, the movement of the rats’ forelimbs was monitored and documented.
In this examination, forelimb participation was determined by analyzing whether the complete palm of the forearm was in contact with the wall of the arena, displaying body support. For each creature, a total of 20 contacts were documented during the 5 minutes session of the right paw contact with wall of cylinder. The percentage of contacts between the impacted and unaffected forelimbs was then determined as a fraction of the total contacts.
This analysis allowed us to evaluate imbalance in locomotion by determining the variations in forelimb usage between the impacted and the unaffected sides. The cylinder examination provides valuable perception into motor function and imbalance in rats throughout the experimental period.
Grip Strength Meter
We utilized a grip strength meter manufactured by Columbus Instruments (USA) to gauge the forelimb grip strength of rats. This standardized evaluation is frequently employed to evaluate neuromuscular performance and identify motor neurotoxicity.
In the course of the evaluation, the rat was placed on a metallic grid or pull bar fastened to the device for gripping. Subsequently, the rat was delicately pulled backwards in a horizontal direction, prompting the animal to exert maximum force to maintain the grip. The Newton measurement recorded the rat’s applied force.
By quantifying the grip force of the forelimb, we were able to examine the power and muscle function of the rats’ forelimbs. This appraisal provides us with valuable information regarding their motor abilities and enables us to track any changes in neuromuscular function that may occur during the experiment.
Cook’s Pole Climbing Test
The Cook’s Test of Pole Climbing was employed to evaluate the delay in climbing a wooden pole situated at the center of the equipment in rats that were paralyzed due to 6-OHDA. The pole acted as a safe zone for the rodents, preventing them from receiving any shocks. The duration taken by each rat to climb the pole was meticulously noted and documented during the experiment. This enabled us to determine the rats’ physical coordination and dexterity and to detect any alterations in their climbing capacity due to the 6-OHDA lesion. 37
Stepping Test
The tap test was utilized to evaluate the absence of initiation in the forepaw on the opposite side. This particular test quantifies the number of steps taken by rats using their forelegs on an exposed table that is 1.2 m long. The test was conducted during daylight hours, and every attempt was documented with a digital recording device.
To initiate the test, the rat was permitted to get comfortable on the edge of the table for approximately 2 sec, generally with all four limbs resting on the surface. At that point, the experimenter would raise the rat’s tail and lift its hind legs, leaving only the front legs touching the table. The rat was then pulled back at a steady pace of around 1 m in 3–4 sec, covering a total distance of 1 m. The rat was then moved back to its original position.
Throughout this backward movement, the number of adapted steps taken by both limbs was carefully monitored and recorded. These findings demonstrated that there were no changes in motor coordination or movement initiation in rats that had been lesioned with 6-OHDA, providing insight into the absence of contralateral forepaw initiation.
Estimation of Oxidative Stress Markers
Tissue Homogenate
To standardize rat cerebral tissue, 100 mg of tissue from every group was dissolved in 1 mL of phosphate-buffered saline (PBS) with a pH of 7.4. The amalgam was then rotated at 3,500 rpm for 10 min. Subsequently, the resulting supernatant, which contained the dissolved constituents, was meticulously gathered and conserved at –20°C for subsequent scrutiny.
Malondialdehyde Levels in Brain Tissue Homogenate
To assess the amount of malondialdehyde (MDA) present in the rat brain homogenate, a TBARS (thiobarbituric acid [TBA] reactive substance) test was conducted. Initially, 100 mg of rat brain tissue from each group was pulverized in 1 mL of 0.01 M PBS with a pH of 7.4. The resultant homogenate was subjected to centrifugation at 3500 rpm for 10 min, and the supernatant was collected and preserved at –20°C for future analysis.
To determine the MDA concentration, a 200-µL sample of the supernatant was mixed with 200 µL of 10% trichloroacetic acid (TCA) and 200 µL of 0.67% TBA. The mixture was then incubated at 95°C for 45 min. Once cooled, 400 µL of butanol was added to the mixture, and it was centrifuged at 3,000 rpm for 10 min. The optical density of the butanol layer was measured at 532 nm, and the MDA concentration was calculated using a standard curve of tetramethoxypropane. This technique enables the quantification of MDA, which is indicative of lipid per oxidation, in the homogenate of rat brain tissue.
Myeloperoxidase Assay in Brain Tissue Homogenate
To assess microglia activity, the myeloperoxidase (MPO) assay was performed following the method described by Barone et al. The rats were euthanized with a high dose of pentobarbitone sodium (100 mg/kg i.p.) and their brains were extracted and homogenized. 39 The resulting supernatant was examined for MPO at a wavelength of 460 nm and the results were expressed in mU/gm wet tissue weight.
Superoxide Dismutase Activity in Brain Tissue Homogenate
One commonly employed technique to assess the function of superoxide dismutase (SOD) in rat homogenate is the procedure outlined by Marklund and Marklund.
38
The Marklund and Marklund technique for determining SOD activity is based on the suppression of pyrogallol autooxidation by SOD, which can be quantified spectrophotometrically at 420 nm. A brief outline of the procedure is as follows:
Prepare a cold 50 mM Tris-HCl buffer (pH 8.2). Homogenize rat brain tissue in the cold buffer and centrifuge the homogenate at 10,000 g for 20 min at 4°C. Collect the supernatant and transfer it to a new tube on ice. Add 0.1 mM EDTA to the supernatant to eliminate interfering metal ions. In a cuvette, prepare a reaction mixture containing 50 mM Tris-HCl buffer (pH 8.2), 0.1 mM EDTA, 1 mM pyrogallol, and the sample. Thoroughly mix the reaction mixture and incubate it at 37°C for 5 min. Stop the reaction by adding 1 M HCl and measure the absorbance at 420 nm against a blank that contains all reagents except for the sample. Using the formula SOD activity = [(A1 ̶ A2)/(A0 ̶ A2)] × V/t × DF, determine the SOD activity, where A0 represents the blank absorbance, A1 represents the sample absorbance, A2 represents the sample absorbance without pyrogallol, V represents the sample volume added, t represents the reaction time, and DF represents the dilution factor.
Catalase Assay in Brain Tissue Homogenate
The catalase activity in rat brain tissue homogenate was determined using the method described by Sinha et al. 39 and Aebi et al. 40
Below is a summary of the procedure:
Prepare a chilled 50-mM potassium phosphate buffer (pH 7.0). Homogenize rat brain tissue in cold buffer and centrifuge the mixture at an appropriate speed and temperature to obtain a clear supernatant. Create a reaction mixture by combining the supernatant, 50 mM potassium phosphate buffer (pH 7.0), and a suitable concentration of hydrogen peroxide (H2O2). Incubate the reaction mixture for a designated time at a suitable temperature. Stop the reaction by adding an appropriate stop solution, such as 10% sodium azide or 5% dichromate. Use a spectrophotometer to measure the absorbance of the reaction mixture at a specific wavelength, usually 240 nm. Express the Catalase activity as units per milligram of protein, where the protein content in the homogenate is determined using an appropriate protein assay method like the Lowry method.
Total Antioxidant Capacity Assay
The FRAP method was employed to determine the aggregate antioxidant capacity of the striatum. The protocol involved the ensuing steps:
Prepare the FRAP reagent by blending 12.5 mL of 0.3 mol/L acetate buffer (CH3COOH:CH3COONa), pH 3.6, with an equivalent volume of methanol. Introduce 2.5 mL of 10 mmol/L (2, 4, 6-tris (2-pyridyl)-s-triazine (TPTZ) and 2.5 mL of 0.02 mol/L FeCl3-6H2O in 0.04 mol/L HCl to the solution. Combine 225 mL of 50% methanol in water and 75 µL of the supernatant from the tissue homogenate to the mixture. Measure the absorbance of the mixture at 593 nm immediately after the initiation of the reaction and after 1 min. Maintain the reaction temperature at 37°C during the measurement.
Pro-inflammatory (TNF-α, IL-1β, IL-4 and IL-6) Chemokines Estimation
At the end of the research, high doses of anesthesia were administered to all the creatures to euthanize them. Subsequently, their brains were extracted and pulverized. ELISA kits, which were obtainable from Cloud Clone Corp. Provider, were utilized to determine the levels of pro-inflammatory chemokines TNF-α, IL-1β, IL-4, and IL-6 in the brain tissue. These ELISA kits facilitated the quantitative measurement of chemokine levels in the tissues. The outcomes were expressed as picograms (pg) of chemokine per microgram (µg) of tissue.
Estimation of NF-κB
The rats were put down with a potent anesthetic before their brains were separated and blended in a 10% solution of PBS. An ELISA kit was procured from Cloud Clone Corp., situated in Katy, Texas, USA, to gauge the levels of NF-κB in the homogenized brain tissue. This ELISA kit is readily available in the market and provides an accurate measure of NF-κB levels in brain tissue. The data was expressed in picograms (pg) of NF-κB per gram (g) of tissue.
Caspase-3 Activity
To evaluate the caspase-3 function in homogenized brain tissue, a fluorometric analysis kit from BioVisionInc Inc., USA, was employed. The evaluation adhered to the manufacturer’s instructions. The fluorometer measured caspase-3 activity at an excitation wavelength of 360 nm and an emission wavelength of 460 nm. The levels of caspase-3 activity were expressed in picograms (pg) per gram (g) of tissue.
Caspase-9 Activity
In order to assess caspase-9 activity in brain tissue homogenates, a fluorometric assay kit from BioVision Inc., USA, was employed. The brain tissue homogenates were initially resuspended in lysis buffer and then homogenized again. Caspase-9 activity was subsequently evaluated using the fluorometric assay kit in accordance with the guidelines of the manufacturer.
The caspase-9 activity was ultimately measured using a fluorometer that had an excitation wavelength of 400 nm and an emission wavelength of 505 nm. The levels of caspase-9 activity were expressed in pg/g of tissue.
COX Activity
The COX-2 activity in rat brain tissue homogenates was gauged by making use of a COX-2 ELISA kit procured from CUSABIO, a Houston, Texas, USA-based enterprise. The assay was executed in line with the manufacturer’s guidelines.
The specified protocol was followed to prepare brain tissue homogenates, and an ELISA kit was employed to determine COX-2 activity. The assay outcomes were expressed in pg/g of tissue, denoting the degree of COX-2 activity present in rat brain tissue.
NO Estimation
In this research, cerebral tissue samples were homogenized and processed with Griess agent as per recognized procedures. The interaction between NO and Griess agent led to the creation of a hued compound, which was evaluated through spectrophotometry. The outcome of the measurement was presented as µg NO/mg rat cerebral tissue, thereby revealing the NO proportion in cerebral tissue of 6-OHDA-lesioned rats. This indicated the degree of NO generation or function under experimental circumstances.
Statistics
The collected data underwent statistical examination through both two-way and one-way analysis of variance (ANOVA), as applicable. Subsequently, multiple group comparisons were conducted using the Newman–Keuls post-hoc test. GraphPad Prism 6.0 was utilized for the statistical analysis.
A
This statistical methodology enables the assessment of significant variances between experimental groups and supports the formulation of conclusions based on the observed outcomes.
Results
Effect of Mangiferin and its Interaction with NO Modulators on 6-OHDA-induced Changes in Behavioral and Locomotor Changes
Effect of mangiferin and NO modulators on distance travelled, average speed, time mobile, and number of mobile episodes in 6-OHDA-lesioned rats
The impact of mangiferin (45 µg) on locomotor parameters was assessed in rats with Parkinsonism induced by 6-OHDA after 42 days of lesion. The locomotor parameters that were evaluated included the number of mobile and freezing episodes, distance covered, time spent mobile, time spent stationary, time spent stationary, and the average speed.
Following the 6-OHDA lesion, there was a significant decrease in locomotor parameters, including a reduction in the number of mobile and freezing episodes, distance covered, time spent mobile, time spent stationary, time spent stationary, and mean speed (
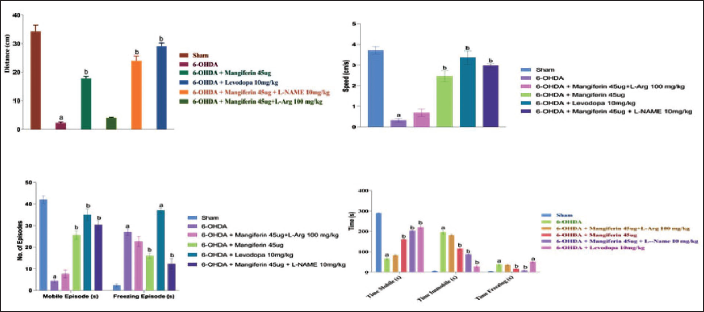
In rats treated with mangiferin (45 µg), there was a notable increase in mobile time and a decrease in stationary time and immobilization time (
Rats treated with a combination of mangiferin 45 µg and L-Arg (100 mg/kg) did not exhibit significant changes in locomotor parameters. However, the maximal improvement in all parameters was observed in rats treated with a combination of a low-dose iNOS inhibitor L-NAME (10 mg/kg) and mangiferin (45 µg). The results obtained after treatment with L-NAME 10 mg/kg and mangiferin 45 µg were similar to those observed with levodopa. These findings, as shown in Figure 2, demonstrate the advantageous effects of mangiferin, either alone or in combination with other agents, in enhancing locomotor parameters in the rat model of 6-OHDA-induced Parkinsonism.
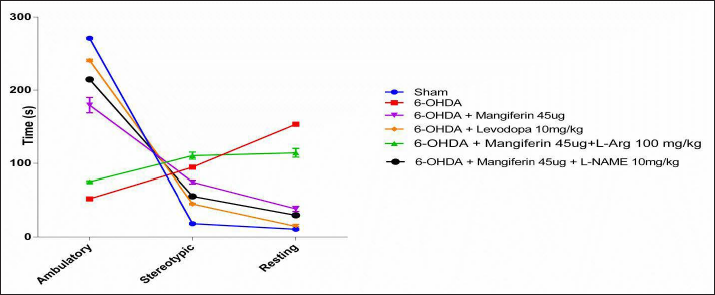
Effect of Mangiferin and its Interaction with NO Modulators on Ambulatory, Stereotypic and Resting Time in 6-OHDA-Lesioned Rats
The rat’s ambulatory, repetitive, and inactive periods were measured using an Opto-varimax 5 Columbus Instruments activity meter after 42 days of 6-OHDA-induced PD. Parkinsonian rats with one-sided 6-OHDA-induced symptoms displayed a significant drop-in ambulatory time and a rise in repetitive and inactive periods.
The effects caused by 6-OHDA in rats with one-sided Parkinsonism were effectively reversed with the administration of 45 µg of mangiferin (
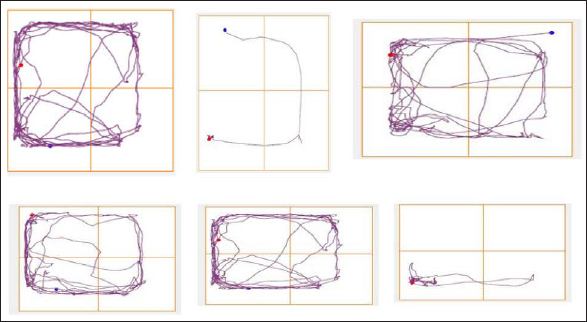
Effect of Mangiferin and its Interaction with NO Modulators on 6-OHDA-induced Changes in Track Plot of Rats
The tracking records of rats with unilateral 6-OHDA-induced Parkinsonism indicated a noteworthy decline in locomotor activity. However, the use of mangiferin (45 µg) for 28 days showed a significant improvement in locomotor activity, as confirmed by the heat map analysis. Likewise, the administration of levodopa (10 mg/kg) also resulted in a considerable enhancement of locomotor activity, as depicted in the heat maps.
Conversely, the combined use of mangiferin (45 µg) and L-Arg (100 mg/kg) did not exhibit any significant effects on the locomotor changes caused by 6-OHDA. Nonetheless, when mangiferin (45 µg) was paired with a low-dose iNOS inhibitor L-NAME (10 mg/kg), there was a significant improvement in locomotor activity, reaching a level comparable to the locomotor changes induced by levodopa (10 mg/kg), as illustrated in Figure 4. These findings suggest that the combination therapy of mangiferin and L-NAME has the potential to enhance locomotor activity in a rat model of unilateral 6-OHDA-induced Parkinsonism (Figure 4).
Effect of Mangiferin and its Interaction with NO Modulators on Sensorimotor Forelimb Function in 6-OHDA-lesioned Rats
To evaluate the asymmetry of forelimb function in rats with unilateral 6-OHDA-induced Parkinsonism, their spontaneous use of the opposite forepaw was examined. The results revealed a substantial decrease in the spontaneous use of the contralateral forelimb in these rats. However, treatment with mangiferin (45 µg) for 28 days significantly enhanced the use of the contralateral forelimb, as demonstrated by an increase in its spontaneous use in treated rats. Similarly, levodopa (10 mg/kg) treatment also boosted the use of the contralateral forelimb in rats with 6-OHDA-induced Parkinsonism.
Moreover, the combined administration of L-NAME (10 mg/kg) and mangiferin (45 µg) also significantly improved the contralateral forelimb’s use (
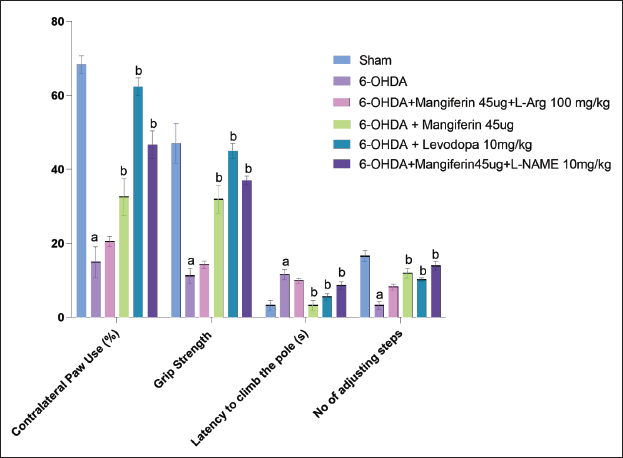
Effect of Mangiferin and its Interaction with NO Modulators on Grip Strength of 6-OHDA-lesioned Rats
The grip strength of rats was evaluated 42 days after 6-OHDA lesion using a grip strength meter (Columbus Instruments, USA). A significant reduction in grip strength was observed in rats with 6-OHDA lesion when compared to the sham group, indicating a decline in motor function. However, daily administration of mangiferin (45 µg) significantly increased the grip strength in parkinsonian rats, indicating an improvement in motor function.
Similarly, treatment with levodopa (10 mg/kg) also significantly increased grip strength in injured rats, further supporting the improvement in motor function. Combinatorial therapy with L-NAME (10 mg/kg) and mangiferin (45 µg) resulted in a significant improvement in grip strength in parkinsonian rats, suggesting a synergistic effect between the two treatments.
Conversely, rats treated with mangiferin (45 µg) and L-Arg (100 mg/kg) showed no significant change in grip strength when compared to the parkinsonian group. These findings demonstrate that the combination of mangiferin and L-Arg did not effectively improve grip strength in this model of PD.
Overall, the results indicate that mangiferin, alone and in conjunction with L-NAME, can increase grip strength and improve motor function in rats with 6-OHDA-induced Parkinsonism, whereas the combination with L-Arg did not produce significant benefits.
The study investigated the effect of mangiferin and its interaction with NO modulators on the Cook’s pole climbing test in 6-OHDA-lesioned rats. Lesion with 6-OHDA significantly increased the time required for rats to climb the pole when compared to sham-operated rats, indicating motor impairment. However, daily treatment with mangiferin (45 µg) for 28 days led to a significant reduction in pole climbing time in rats with Parkinsonism, indicating an improvement in motor function.
Similarly, rats treated with levodopa (10 mg/kg) also showed a significant reduction in the time required to climb the pole, further supporting the improvement in motor function. Combinatorial therapy with L-NAME (10 mg/kg) and mangiferin (45 µg) also significantly reduced the time required to climb the pole in rats with Parkinsonism, suggesting a synergistic effect between the two treatments.
On the other hand, the combination of L-Arg (100 mg/kg) and mangiferin (45 µg) produced no significant effect (Figure 5).
Effect of Mangiferin and its Interaction with NO Modulators on Forelimb Akinesia in 6-OHDA-lesioned Rats
The stepping test was used to evaluate forelimb akinesia in rats with 6-OHDA lesions. This test measures the number of adjusting steps taken by the contralateral forelimb. The results demonstrated a significant decrease in the number of adjusting steps, indicating forelimb akinesia in rats with PD.
Nonetheless, administering mangiferin (45 µg) daily considerably enhanced forelimb akinesia by elevating the number of adjusting steps taken by the contralateral forelimb in lesioned rats. This indicates that mangiferin efficiently mitigates the motor impairment associated with forelimb akinesia in this PD model.
Similarly, administering levodopa (10 mg/kg) to rats also resulted in a notable decrease in forelimb akinesia, further supporting the improvement in motor function with this medication.
Moreover, the combination of L-NAME (10 mg/kg) and mangiferin (45 µg) therapy showed a significant decrease in forelimb akinesia in rats. This indicates a synergistic effect between the two treatments. This combination therapy is effective in enhancing motor performance in the stepping test.
The results highlight the favorable effects of mangiferin, both alone and in combination with L-NAME, in reducing forelimb akinesia and improving motor function in rats with 6-OHDA-induced Parkinsonism. These findings suggest that mangiferin and combination therapy have the potential to be promising therapeutic strategies for managing the motor symptoms linked with PD.
Effect of Mangiferin and its Interaction with NO Modulators on 6-OHDA-induced Changes in Biochemical Parameters
Effect of mangiferin and its interaction with NO modulators on MDA concentration in 6-OHDA-lesioned rats
The levels of MDA, an indicator of oxidative stress, in rat brain tissue homogenates were evaluated. The outcomes demonstrate that mangiferin treatment alone or in combination with L-Arg and L-NAME significantly decreased MDA concentration in brain tissue (
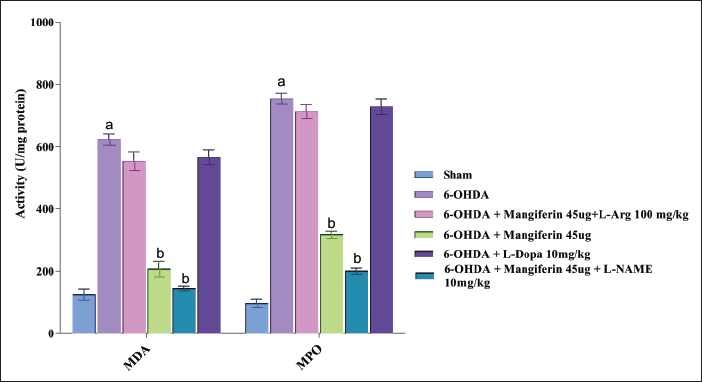
These results indicate that mangiferin, particularly in combination with NO modulators, possesses antioxidant properties by decreasing MDA levels in the brain tissue of rats with 6-OHDA-induced Parkinsonism. The combination therapy of levodopa and mangiferin holds promise as a potential approach to enhance antioxidant capacity and alleviate oxidative stress associated with PD (Figure 6).
Effect of Mangiferin and its Interaction with NO Modulators on Myeloperoxidase Activity in 6-OHDA-lesioned Rats
Administering mangiferin (45 µg) for a duration of 28 days led to a considerable reduction in MPO activity in rats that had been induced with Parkinsonism using 6-OHDA. Additionally, when mangiferin was coupled with L-NAME (10 mg/kg), MPO levels were further diminished (
On the other hand, the administration of levodopa (10 mg/kg) did not substantially impact MPO activity, suggesting that it does not affect MPO levels directly. Similarly, the combination of mangiferin (45 µg) and L-Arg (100 mg/kg) did not produce a significant effect on MPO activity in rats with 6-OHDA lesions.
These findings suggest that mangiferin treatment alone can effectively decrease MPO activity, indicating a potential anti-inflammatory effect. Furthermore, the combination of mangiferin and L-NAME leads to a reduction in MPO concentration, indicating a cooperative effect on the inflammatory response. However, it is crucial to note that in this model of Parkinsonism, neither levodopa nor the combination of mangiferin and L-Arg had a significant impact on MPO activity (Figure 6).
Effect of Mangiferin and its Interaction with NO Modulators on Oxidative Stress (SOD and Catalase activity) in 6-OHDA-lesioned Rats
The function of SOD and catalase, enzymes that fight oxidative stress, was gauged in rats with Parkinsonism induced by 6-OHDA. It was observed that the activities of both SOD and catalase were markedly decreased in these rats, indicating heightened oxidative stress.
Nevertheless, the administration of mangiferin (45 µg) for 28 days substantially increased the function of both SOD and catalase in Parkinsonian rats. This indicates that mangiferin possesses antioxidant characteristics and can effectively combat oxidative stress in this model.
In contrast, the treatment of levodopa (10 mg/kg) did not significantly impact the function of SOD or catalase, which implies that it does not have a direct effect on the antioxidant defense mechanism.
Interestingly, when levodopa was combined with mangiferin, there was a noteworthy increase in SOD function in rats with parkinsonian symptoms (
Furthermore, the combination of mangiferin with L-NAME (an inhibitor of NO synthase) considerably reduced oxidative stress in rats, as evidenced by increased function of both SOD and catalase. This suggests that L-NAME augments the beneficial effects of mangiferin in reducing oxidative stress.
On the other hand, the combination of mangiferin with L-Arg (an amino acid that acts as a precursor of NO synthesis) did not significantly impact oxidative stress, as indicated by no alteration in the function of SOD and catalase. This suggests that L-Arg may interfere with the antioxidant effects of mangiferin (Figure 7).
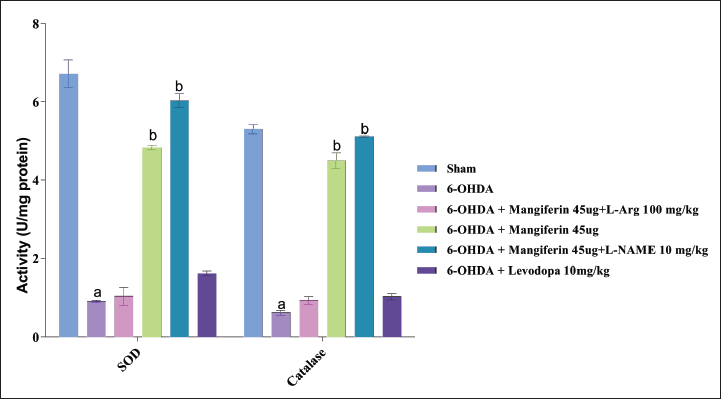
Overall, the findings highlight the intricate interplay between mangiferin and NO mediators in the context of oxidative stress. The combination of mangiferin with L-NAME enhanced its antioxidant effects, while combination with L-Arg did not show a significant effect. These results suggest that the specific mediator involved plays a role in modulating the antioxidant properties of mangiferin (Figure 7).
Effect of Mangiferin and its Interaction with NO Modulators on NO Concentration in 6-OHDA-lesioned Rats
In the PD model induced by 6-OHDA, rats exhibit elevated levels of NO. Nonetheless, the administration of mangiferin (45 µg) for 28 days significantly decreased NO levels in the brain tissue homogenates of these rats, indicating that mangiferin has the potential to reverse the 6-OHDA-induced increase in NO.
In contrast, treatment with levodopa (10 mg/kg) did not have a significant impact on NO levels, implying that levodopa does not modulate the NO pathway in the same way as mangiferin.
Remarkably, the simultaneous administration of mangiferin (45 µg) and the iNOS inhibitor L-NAME (10 mg/kg) resulted in the maximum reduction of NO levels. This suggests that L-NAME enhances the effect of mangiferin in reducing NO concentrations. Conversely, the combined use of mangiferin (45 µg) and L-Arg (100 mg/kg) did not produce a significant effect on NO concentration (Figure 8).
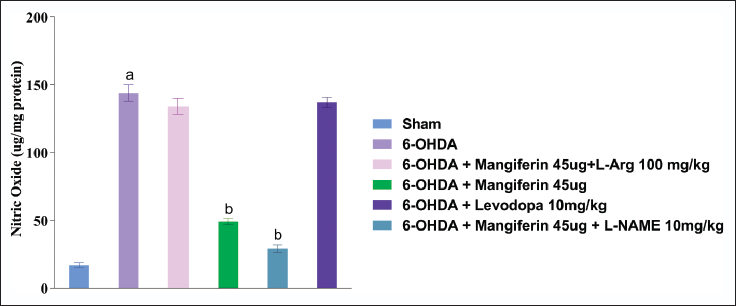
These results indicate that mangiferin directly impacts the NO pathway and effectively reduces NO levels in the context of 6-OHDA-induced PD. The combination with L-NAME amplifies this effect, while the combination with L-Arg does not significantly affect NO levels.
Effect of Mangiferin and its Interaction with NO Modulators on 6-OHDA-induced Changes in Molecular Parameters
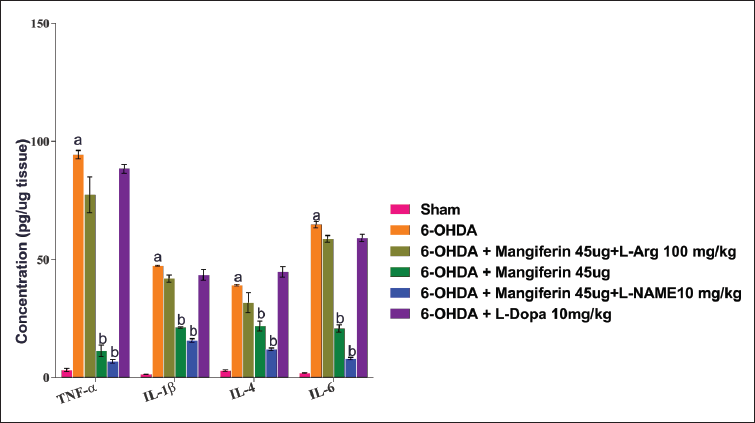
Effect of mangiferin and its interaction with NO modulators on pro-inflammatory chemokine concentration in 6-OHDA-lesioned rats
This study aimed to evaluate the levels of pro-inflammatory chemokines in rats with 6-OHDA lesions and their modulation by mangiferin and NO modulators. The results indicated that the concentration of pro-inflammatory chemokines was significantly increased in the brain tissue homogenates of rats with 6-OHDA lesioning.
After a 28-day treatment with mangiferin (45 µg), the concentration of pro-inflammatory chemokines in the lesioned rats was significantly reduced, demonstrating its anti-inflammatory properties. These findings suggest that mangiferin has the potential to alleviate the neuroinflammatory response associated with 6-OHDA lesioning.
In contrast, treatment with levodopa (10 mg/kg) did not significantly affect the concentration of pro-inflammatory chemokines in the brain tissue homogenates of rats with 6-OHDA lesioning. This suggests that levodopa does not directly modulate the neuroinflammatory response in the same way as mangiferin. However, levodopa’s primary therapeutic action is to replenish dopamine levels in the brain, thereby relieving motor symptoms associated with PD.
Furthermore, the study investigated the interaction between mangiferin and NO modulators. The combination of mangiferin (45 µg) with the iNOS inhibitor L-NAME (10 mg/kg) resulted in a further reduction in pro-inflammatory chemokine concentration when compared to mangiferin treatment alone. This indicates that L-NAME enhances the anti-inflammatory effect of mangiferin in the context of 6-OHDA lesioning.
However, the combination of mangiferin (45 µg) with L-Arg (100 mg/kg) did not significantly affect pro-inflammatory chemokine concentration. This suggests that L-Arg does not modulate the anti-inflammatory properties of mangiferin in this model.
Overall, these findings suggest that mangiferin has anti-inflammatory effects and can reduce the concentration of pro-inflammatory chemokines in rats with 6-OHDA lesioning. The combination with the iNOS inhibitor L-NAME enhances this effect, while the combination with L-Arg does not significantly impact pro-inflammatory chemokine concentration (Figure 9).
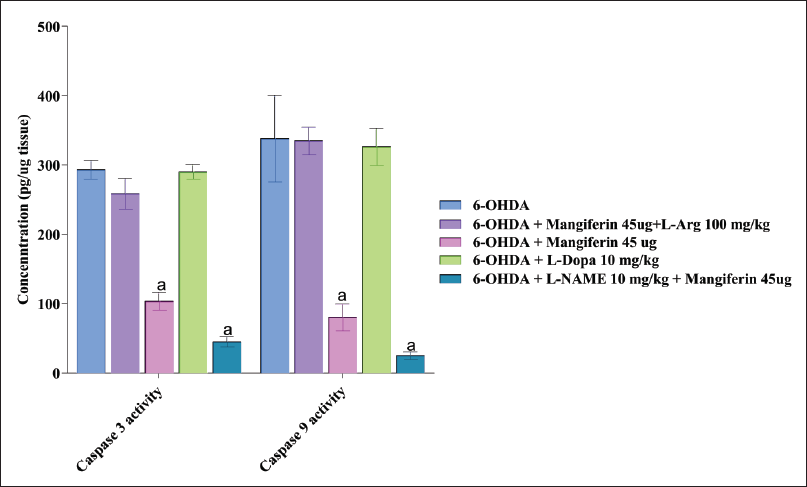
Effect of Mangiferin and its Interaction with NO Modulators on Caspases Activity in 6-OHDA-lesioned Rats
In rats that underwent 6-OHDA lesioning, there was a significant increase in the activity of caspase-3 and caspase-9, which are indicative of apoptotic cell death. However, the administration of mangiferin (45 µg) for 28 days effectively reversed the heightened caspase activity caused by 6-OHDA. This indicates that mangiferin has the potential to prevent apoptotic cell death in the context of PD.
On the other hand, treatment with levodopa (10 mg/kg) or a combination of mangiferin (45 µg) and L-Arg (100 mg/kg) did not show any significant effect on caspase activity. This suggests that these treatments do not directly affect apoptotic pathways in the same way that mangiferin does.
Of note, the combination of mangiferin (45 µg) with the iNOS inhibitor L-NAME resulted in a marked enhancement of mangiferin’s caspase-inhibiting property. This implies that L-NAME amplifies the anti-apoptotic effect of mangiferin in this model (
These findings underscore the potential of mangiferin as a neuroprotective agent in PD by preventing apoptotic cell death. Furthermore, the combination with L-NAME may offer a promising therapeutic strategy to further augment the anti-apoptotic effects of mangiferin. Continued research is necessary to fully comprehend the underlying mechanisms and explore the clinical implications of these findings (Figure 10).
Effect of Mangiferin and its Interaction with NO Modulators on COX-2 Activity in 6-OHDA-lesioned Rats
In rats, 6-OHDA damage leads to an increase in inflammation in the striatum, which can be seen through the heightened activity of the pro-inflammatory enzyme COX-2. However, the administration of mangiferin (45 µg) results in a significant reduction in the 6-OHDA-induced increase in COX-2 activity, indicating its anti-inflammatory properties.
On the other hand, neither levodopa (10 mg/kg) nor the combination of mangiferin (45 µg) and L-Arg (100 mg/kg) appeared to have any significant effect on COX-2 activity. This suggests that levodopa and L-Arg may not play a direct role in modulating the inflammatory pathways that regulate COX-2.
Interestingly, the rats that received a combination of mangiferin (45 µg) and L-NAME (10 mg/kg) showed the most significant improvement in COX-2 activity when compared to those who only received L-NAME (
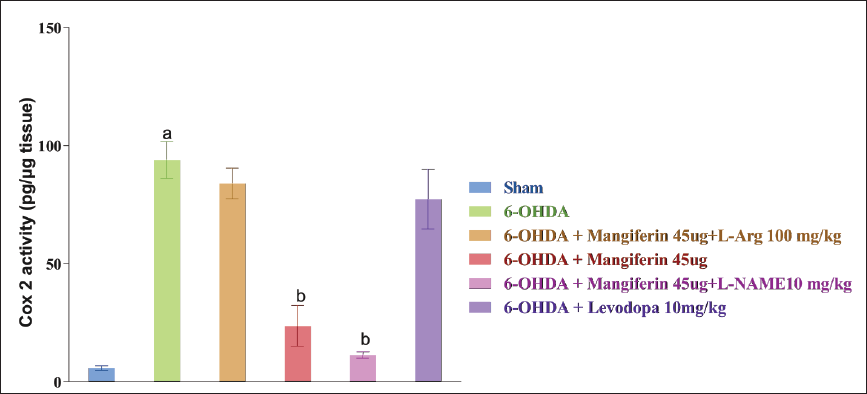
The results of this study highlight the potential of mangiferin as an anti-inflammatory agent in the context of PD, especially in reducing COX-2 activity. Further research is necessary to fully understand the mechanisms involved in these findings and to explore the potential interactions between levodopa, mangiferin, and L-Arg in modulating inflammation.
Discussion
PD is characterized by the development of one-sided tremors that gradually increase in severity. Other symptoms include muscle stiffness, bradykinesia, and disturbances in sleep. Although the exact cause of PD is not yet fully understood, scientific research indicates that inflammatory processes and oxidative stress may play a part in its development.
Our study in 2021 investigated the neuroprotective properties of mangiferin in a rodent model with 6-OHDA lesions. These compounds seem to function by increasing oxidative stress and inflammatory cascades. Mangiferin has antioxidant properties and is effective in combating free radicals, which helps to protect the body from further harm. Our study aimed to evaluate the therapeutic benefits of combining a low dose of mangiferin (45 µg) with a NO modulator for PD.
Our findings revealed that mangiferin (45 µg) significantly improved locomotor impairment in rats with 6-OHDA lesions by rescuing dopaminergic neurons. The combination of mangiferin (45 µg) and L-NAME (10 mg/kg) resulted in further improvement in locomotor parameters, indicating the inhibitory impact of L-NAME on iNOS. Mangiferin enhances seizure strength and behavioral changes linked to 6-OHDA treatment, suggesting cognitive and motor benefits for PD patients.
The improvement noted in behavioral parameters following treatment with mangiferin (45 µg) may be due to the presence of dopamine, a vital neurotransmitter that regulates mood and behavior. By increasing dopamine availability, mangiferin (45 µg) has the potential to enhance mood and behavioral outcomes in individuals with PD. In summary, our findings support the use of mangiferin (45 µg) and L-NAME (10 mg/kg) combination therapy as a promising treatment for PD, indicating improvements in locomotor parameters and potential additional benefits in cognitive resolution and mood.
PD is characterized by the appearance of tremors on one side of the body that worsen over time. Other symptoms include muscle rigidity, bradykinesia, and sleep disturbances. While the exact cause of Parkinsons is not yet fully understood, scientific evidence suggests that oxidative stress and inflammatory processes may play a role in its development.
Our 2021 research explored the neuroprotective properties of mangiferin in a rodent model with 6-OHDA lesions. These compounds appear to work by increasing oxidative stress and inflammatory cascades. Mangiferin is an antioxidant that effectively fights free radicals and protects the body against further damage. Our study aimed to evaluate the therapeutic effectiveness of combining a low dose of mangiferin (45 µg) with a NO modulator for PD.
Our findings showed that mangiferin (45 µg) significantly improved locomotor impairment in rats with 6-OHDA lesions by rescuing dopaminergic neurons. The combination of mangiferin (45 µg) and L-NAME (10 mg/kg) resulted in further improvement in locomotor parameters, indicating the inhibitory effect of L-NAME on iNOS. Mangiferin also improved seizure strength and behavioral changes associated with 6-OHDA treatment, suggesting cognitive and motor benefits for PD patients.
The improvement observed in behavioral parameters after treatment with mangiferin (45 µg) may be due to the presence of dopamine, a key neurotransmitter that regulates mood and behavior. By increasing dopamine availability, mangiferin (45 µg) has the potential to improve mood and behavioral outcomes in people with PD. Overall, our results support combining mangiferin (45 µg) and L-NAME (10 mg/kg) as a promising treatment for PD, suggesting improvements in locomotor parameters and potential additional benefits in cognitive and mood resolution.
Furthermore, the transcription factor NF-κB assumes a pivotal function in regulating gene expression of diverse anti-inflammatory reactions by serving as a chief controller that stimulates the generation of inflammatory agents. Our investigation demonstrates that administering mangiferin (45 µg) and L-NAME (10 mg/kg) together efficiently curbs the manifestation of NF-κB, a hopeful remedy for PD.20, 18 Hence, NF-κB performs a crucial function in regulating the expression of genes responsible for producing pro-inflammatory chemokines, highlighting its pivotal role in controlling the inflammatory reaction.41–43
Mangiferin exhibited potent inhibitory effects on NF-κB, leading to a significant reduction in TNF-α and IL-6 serum levels, as demonstrated in our investigation. The anti-inflammatory and anti-Parkinsonian properties of mangiferin may be attributed, in part, to its capacity to hinder NO synthesis. Our results propose that the cooperative use of mangiferin and the low-dose NO modulator L-NAME potentiates the anti-inflammatory and anti-Parkinsonian effects of mangiferin. L-NAME serves as an anti-inflammatory agent by obstructing the NO-NOS pathway, which, in turn, suppresses inflammation by inhibiting the activation of different inflammatory mediators such as cyclooxygenase. 44
More research is necessary to completely elucidate the curative impacts of mangiferin in PD. Additional research could assess the capacity of mangiferin to hinder disease progression by inspecting the long-term outcomes of its treatment.
Moreover, the investigation of the intricate mechanism underlying the neuroprotective characteristics of mangiferin necessitates further research, incorporating the possibility of other signaling pathways aside from the established NF-κB pathway.
Despite the encouraging outcomes of the present study, further research is imperative to investigate the therapeutic potential of mangiferin in PD. Such research holds the promise of illuminating the complex mechanisms involved in the pathogenesis of the disease and may pave the way for novel therapeutic interventions aimed at enhancing the quality of life of individuals afflicted with PD.
Conclusion
In conclusion, we demonstrate that mangiferin has neuroprotective effects in 6-OHDA-lesioned mice, possibly through its ability to reduce oxidative stress and inflammation. The ability of mangiferin to inhibit NF-κB, reduce pro-inflammatory chemokines such as TNF-α and IL-6, and reduce NO production by macrophages suggests that it may be a potential therapeutic agent for PD. In addition, the combination of mangiferin and the iNOS inhibitor L-NAME appears to have a synergistic effect in protecting against inflammation. Future studies should explore the underlying mechanisms of this synergism and evaluate the optimal dose and regimen for this combination therapy. Future research into the use of mangiferin and L-NAME as a synergistic treatment for PD will provide valuable insight to expand treatment options for patients. To conclude, we have shown that mangiferin possesses neuroprotective properties in mice with 6-OHDA-induced lesions, potentially by reducing oxidative stress and inflammation. The capacity of mangiferin to hinder NF-κB, decrease the production of pro-inflammatory chemokines such as TNF-α and IL-6, and lower NO levels in macrophages suggest that it could be a promising therapeutic agent for PD. Moreover, the combination of mangiferin and the iNOS inhibitor L-NAME seems to have a synergistic effect in protecting against inflammation. Further investigations should delve into the underlying mechanisms of this synergism and determine the optimal dosage and regimen for this combination therapy. Prospective research on the application of mangiferin and L-NAME as a synergistic treatment for PD will provide valuable insights to broaden the array of treatment options available for patients.
Footnotes
Availability of Data and Materials
The authors confirm that the data supporting the findings of this study are available within the manuscript.
Authors’ Contribution
Tiwari PC, Manju J. Chaudhary and Rishi Pal conceived the experiments. Tiwari PC carried out experiments and was guided by Rishi Pal and Manju J. Chaudhary. Tiwari PC wrote the manuscript with the help of Manju J. Chaudhary and Rishi Pal supervised the manuscript. All authors provided critical feedback and helped shape the research, analysis, and manuscript.
Statement of Ethics
Before starting the study, necessary approval from the IAEC of King George’s Medical University (KGMU), Lucknow was obtained with approval letter No. 84/IAEC/Pharma/2017.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Authors acknowledge Senior Research Fellowship provided to Mr. Prafulla Chandra Tiwari by Indian Council of Medical Research with file no. 45/34-2014/PHA-BMS.
Human Rights
This manuscript does not contain any studies on human volunteers or human tissue samples.
Animal Rights
All experiments were done according to the guidelines of the IAEC of KGMU and the Prevention of Cruelty to Animals Act 1960 of the Government of India.
