Abstract
Background
Physically Disabled women face multiple barriers to cervical screening, contributing to lower uptake and increased health inequalities. Human papillomavirus self-sampling has been shown to increase screening participation in under-screened populations, but little is known about its acceptability for Disabled women.
Methods
A cross-sectional online survey was conducted with 1493 UK-based participants who identified as having a physical disability, impairment, condition, or difference that makes cervical screening difficult or impossible. Participants completed questions on the acceptability of human papillomavirus self-sampling, attitudes and beliefs relating to self-sampling, and future screening preferences. Descriptive statistics and multinomial logistic regression were used to analyse responses.
Results
Most participants reported that they would be able to carry out self-sampling themselves (63.3%) and would be willing for a healthcare professional to use a self-sampling kit on their behalf (59.1%). Many (70.5%) had concerns about not performing the test correctly. Around half (53.0%) would prefer self-sampling at home if offered a screening choice. Women who had never attended screening, or who had delayed/missed appointments, were significantly more likely to prefer self-sampling (odds ratios 13.11 and 5.25, respectively) than women who had always attended. Approximately a fifth of participants (18.7%) would prefer a non-speculum clinician-taken test.
Conclusion
Human papillomavirus self-sampling was acceptable to many physically Disabled women and preferred over conventional screening, particularly among those who had delayed/missed screening or never attended. Implementation should include tailored accessible instructions to support human papillomavirus self-sampling, disability-informed clinical support, and consideration of non-speculum clinician-taken samples to ensure equitable access and reduce inequalities in cervical screening.
Keywords
Introduction
Persistent infection with high-risk human papillomavirus (HPV) is responsible for more than 90% of cases of cervical cancer. 1 In addition to primary HPV prevention through HPV vaccination, which has been available in the UK since 2008, cervical screening provides secondary prevention by allowing for the detection of HPV and precancerous cell changes, the latter of which can then be treated. In the UK, women and people with a cervix (henceforth ‘women’) aged 25–64 are eligible for cervical screening every 5 years as part of the NHS cervical screening programme. 2 Despite the programme saving an estimated 5000 lives annually, 3 there are documented inequalities in uptake of cervical screening; for example, by socioeconomic status and ethnicity. 4 HPV self-sampling (which allows women to take a vaginal swab themselves and does not require a speculum) has the potential to address some of the barriers to screening.
In 2021, UK researchers conducted a large-scale feasibility trial, YouScreen, inviting more than 27,000 women who were overdue for cervical screening to use an HPV self-sampling kit. Of the 2712 women who used the self-sampling kit and completed a questionnaire about the kit, 71.3% indicated they would opt for self-sampling in the future. 5 The UK HPValidate trial (which ran from 2021 to 2023 and aimed to find out if vaginal self-tests were as accurate as clinician-assisted sampling in identifying HPV 6 ) included an embedded sub-study that tested user experience and acceptability of HPV self-sampling. The authors found that 85% of women would like to be given a choice between self-sampling or clinician-based sampling and 71.7% of participants stated this would improve cervical screening.6,7 The results of these trials informed a recent National Screening Committee consultation and the decision by NHS England to roll out self-sampling for those overdue their appointment in early 2026. 8 In the future, self-sampling may be offered to all women at the point of invitation.
While the findings from YouScreen and HPValidate, as well as from the introduction of HPV self-sampling in other countries such as Australia and France,9,10 suggest that self-sampling is acceptable to many under-screened women, these data do not include information about physical disability. However, research suggests that healthcare systems and environments often create barriers to cervical screening for Disabled women. Such barriers include difficulty getting transport to appointments, lack of accessible facilities in clinics, difficulty getting into position for speculum insertion on the examination table, and healthcare practitioners’ lack of knowledge of physical disability.11,12 Barriers such as these lead to lower screening uptake in this population.13,14 While HPV self-sampling may increase screening uptake for physically Disabled women, there has been very little research to understand the extent to which this is the case and any challenges that this population may face, with only two qualitative studies to date that we are aware of. The first is a 2020 Danish report in which seven wheelchair users were interviewed about HPV self-sampling. 15 The women reported that self-sampling would make it easier for some women to access cervical screening, but it would not be suitable for all Disabled women. The second study consisted of interviews with 56 physically Disabled women in the US who tried HPV self-sampling and suggests that self-sampling may be acceptable to some Disabled women as an alternative to clinician-led cervical screening. 11
The present study is the first survey to explore the acceptability of, and attitudes towards, HPV self-sampling among physically Disabled women. There are more than 8 million Disabled women in the UK 16 and so this is an important and overlooked dimension of inequality to consider when implementing this new technology within the national screening programme. Our study was informed by substantial patient and public involvement (PPI) work, which resulted in adoption of the social model of disability 17 to inform the recruitment strategy, whereby women who identified as having a ‘physical disability, impairment, condition or difference’ were invited to take part.
The aim of this study was to investigate the acceptability of, and attitudes towards, HPV self-sampling for women with a physical disability, impairment, condition or difference. This was explored through two research questions:
To what extent is HPV self-sampling acceptable for women with a physical disability, impairment, condition, or difference? What factors affect their preferences regarding future screening?
Methods
Patient and public involvement (PPI)
One of the co-authors, SR, was the PPI lead for this project. We additionally recruited 14 physically Disabled women (aged 26–63) as our stakeholder advisory group. SS and EK met regularly with SR, and we met with the stakeholder advisory group three times to develop the materials. We presented the advisory group with a first draft of the survey, and they helped us to develop the recruitment method, refine the questions and publicize the study. They also were invited to comment on a draft of this article and their comments have been incorporated and acknowledged.
Design
The data reported here were collected as part of a larger cross-sectional survey that explored current screening attendance and preferences, as well as attitudes and preferences relating to self-sampling. Only the self-sampling aspect of this survey is reported here. We conducted the survey using two recruitment approaches: (i) an online panel (Prolific) and (ii) advertising via charities and existing links. The first wave of data was collected in July 2024, and the second was collected between September 2024 and May 2025. The survey was delivered on the online survey platform Qualtrics.
Participants
Eligibility criteria: To be eligible for the survey, participants were asked to identify as having a physical disability, impairment, condition or difference that makes cervical screening challenging or impossible, be aged 25–64 (screening age), be living in the UK, and to have previously been invited to cervical screening (even if they did not attend). Participants were not eligible to take part if they had received a total hysterectomy (to remove all of the womb and cervix). Those who took part via Prolific received the eligibility criteria in a short survey format to assess suitability to access the main survey. The main survey included the eligibility criteria as questions that all participants were required to answer before completing the full survey; these questions also served as screening questions for those individuals recruited through charities.
We aimed to recruit a sample of 1500 participants so as to provide precise estimates of proportions with a margin of error no worse than ± 0.025, at a 95% confidence level using an asymptotic method, and greater precision for estimates of continuous variables.
Online panel recruitment: A total of 1439 participants were recruited through the online panel company Prolific. The first wave of Prolific participants (n = 1000) was recruited in July 2024. We returned to Prolific for a second wave of recruitment (n = 439) in March 2025 due to low uptake of recruitment using alternative methods such as charities and PPI links. Participants were paid £0.10 for completing the eligibility survey and £3.00 for completing the main survey. For the main study, participants who failed more than one attention check question (out of four) were not reimbursed, and their data were excluded from analysis.
Charity-based/PPI links recruitment: To ensure representation of a range of disabilities and conditions, we aimed to supplement data collected via Prolific by recruiting extra people through relevant charities (e.g. SCOPE, Spinal Injuries Association, and Brittle Bone Society) and our PPI stakeholder members’ personal networks; n = 54 participants were recruited using this method. Participants who were recruited through this method completed the main survey on Qualtrics and were reimbursed with a £5.00 voucher.
Measures
The full survey is available at: https://osf.io/fbmd2. All participants were presented with a participant information sheet outlining the nature of the study and were required to provide online consent.
To explore HPV self-sampling, participants were first presented with a short extract explaining what self-sampling is, including an image of what a self-sampling kit might look like. Participants were then asked the question, ‘Do you think you would be able to do self-sampling yourself?’. Those who did not answer ‘no’ were presented with the seven items in the ‘Concerns’ and ‘Autonomy’ subscales from the HPV Self-sampling Attitudes and Beliefs scale; 18 the items were answered on a 5-point Likert scale: ‘strongly disagree’– ‘strongly agree’.
Participants were then asked two further questions related to preferences about self-sampling: ‘Would you be willing to have someone e.g. a family member/personal assistant/carer assist you with the self-sampling test?’ and ‘Would you be willing to have a doctor or nurse use the self-sampling kit to take your sample?’ Participants were finally asked about their future screening preferences with an item adapted from a study by Drysdale et al.: 19 ‘If you were offered a choice of screening, which would you choose?’ with the following options: ‘Screening done in the same way as now by a nurse or doctor (with a speculum)’; ‘A nurse or doctor using the self-sampling kit to take my sample (without a speculum)’; ‘Self-sampling kit to do myself at home’; ‘I don't know which screening option I would choose’; ‘Not applicable – I wouldn’t have any cervical screening at all’; ‘Prefer not to say’.
Participants were asked: ‘Does your physical disability, condition, impairment or difference affect you in any of the following areas?’ This question was based on the 10-item Impairment Harmonised Standard 20 with additional items adapted from the International Classification of Functioning, Disability, and Health 21 to include pain, sexual function and urinary function. Participants were then asked if they used assistive devices. If they answered ‘yes’, they were provided with a list of options from the Melbourne School of Population and Global Health. 22
Six items modified from Jo's Cervical Cancer Trust 23 survey relating to cervical screening status were provided to measure participants’ previous experience and future intentions relating to cervical screening attendance. Participants were asked ‘Which of the following best describes your previous participation in cervical screening?’ and were provided with response options ‘I have never attended’, ‘I have attended but sometimes delayed or missed my screening appointment (more than 6 months)’, ‘I have always attended when invited (within 6 months)’. If participants selected ‘I have never attended’, this would filter to a question asking if this was due to their physical disability, impairment, condition or difference. This question was also asked of participants who selected ‘I have sometimes delayed or missed screening’.
Socio-demographic questions were asked to record participants’ gender and ethnic background (wording taken from UK Census, 2021 24 ), location, religion, education (wording taken from YouGov, 2020 25 ), household income, relationship status, and language.
Finally, a debrief was provided, directing participants to The Eve Appeal website and helpline (eveappeal.org.uk) for further support and advising participants to contact their GP if they have any questions.
Data analysis
We calculated descriptive statistics for participant characteristics, previous screening participation, practicality and preferences relating to self-sampling, and attitudes and beliefs relating to self-sampling.
We used multinomial logistic regression to explore preferences for future screening. Variables representing demographic characteristics, previous screening attendance and aspects of disability were specified a priori as predictor variables and entered as blocks in the model. These predictors were checked for collinearity. The outcome variable comprised three preferences: (i) ‘A nurse or doctor using the self-sampling kit to take my sample (without a speculum)’; (ii) ‘Self-sampling kit to do myself at home’; (iii) ‘Screening done in the same way as now by a nurse or doctor (with a speculum)’. The last of these was the reference category, such that the odds ratios (ORs) from the analysis indicate how much larger (ORs > 1) or smaller (ORs < 1) the odds were of preferring self-sampling with a nurse or doctor, or of self-sampling done at home, than the odds of preferring screening done in the usual way.
The goodness-of-fit of the full model was evaluated via a deviance chi-square test (where a non-significant test indicates satisfactory goodness-of-fit), and the percentage of correctly classified cases was calculated. The explanatory power of the model was measured using the Nagelkerke pseudo-R2 statistic (range 0–1), indicating the degree to which values of the outcome variable can be explained by the model. 26 For each predictor variable, a p value was calculated via a likelihood ratio test. Statistical significance was set at p ≤ 0.05 (two-tailed), and 95% confidence intervals were derived for all ORs. Analyses were conducted in IBM SPSS Statistics v 30.
Ethical approval
This study was granted ethical approval from Keele University Research Ethics Committee (REF: REC Project Reference 0787) and the University of Sheffield School of Psychology research ethics committee (REF: 064385).
Results
Of the 1602 respondents to the survey, 109 were excluded due to either incomplete survey responses (n = 88), more than one failed attention check (n = 8), or on the basis of ineligibility (n = 13). After exclusions, there were 1493 participants included in the analysis. Participant characteristics are shown in Table 1. The mean (SD) age was 41.71 (10.96) years. All respondents were female by sex, with 98.1% identifying as women. A large majority (89.7%) were of white ethnicity, with 89.3% identifying as English, Welsh, Scottish, Northern Irish, or British. Most of the respondents (60.8%) held a degree-level qualification or higher, and 84.3% lived in England. The majority (61.1%) were married or living with a partner, and 22.6% were single. The dataset is available at: https://osf.io/ufx8r/files/dm7cs.
Participant characteristics.

Percentages may not sum to exactly 100 owing to rounding. SD: standard deviation.
Undergraduate (e.g. BA and BSc) or postgraduate (e.g. MA, MSc and PhD) degree or other technical, professional or higher qualification.
In terms of previous screening participation, 660 participants (44.2%) had always attended, 698 (46.8%) had sometimes delayed or missed an appointment, and 131 (8.8%) had never attended.
Practicality and preferences relating to self-sampling
Regarding self-sampling, 63.3% believed they could perform it themselves, 23.8% were unsure, and 9.4% said they could not (Table 2). Among those who did not answer ‘yes' to being able to perform self-sampling (n = 548), 24.5% might be willing and 16.6% would be willing to have assistance from someone they know. Over half of all respondents (59.1%) would be willing to have a healthcare professional use the self-sampling kit.
Practicality and preferences relating to self-sampling, n (%).

This question only posed to those answering ‘no’, ‘maybe’ or ‘don’t know’ to the question on being ‘able to do self-sampling yourself’.
Attitudes and beliefs relating to self-sampling
Among the 1353 participants who responded to the Tatar et al. 18 attitude items (Table 3), 70.5% expressed some level of concern about not performing the self-sampling correctly, while fewer reported concerns about harming oneself (14.1%) or infection (14.5%). Most (90.5%) disagreed/strongly disagreed that they would feel embarrassed doing self-sampling. Many (77.2%) agreed/strongly agreed it would save them travelling to a healthcare professional, and 86.5% agreed/strongly agreed it would give them greater control over their body. Additionally, 71.9% agreed/strongly agreed they would be more comfortable performing the swab themselves compared to having it done by a healthcare professional.
Attitudes and beliefs relating to self-sampling, n (%).

Median categories are italicized. Denominator n = 1353 (respondents who said they would not be able to do self-sampling excluded). HPV: human papillomavirus.
Preferences for screening
The most favoured screening method was a self-sampling kit to use at home (53.0%), followed by a healthcare professional using the kit without a speculum (18.7%), and conventional screening with a speculum (17.1%); see Table 4.
Preferred method of screening (n = 1493).

The multinomial regression model (Table 5) showed adequate fit to the data (p = 0.944), correctly classified 805/1319 (61.0%) of cases, and explained 17.1% of the values of the outcome variable (Nagelkerke R2 = 0.171). Ethnicity (Block 1) was a non-significant predictor and explained <0.5% of the variability of the outcome values. Previous screening practice (Block 2) was a significant predictor (p < 0.001), explaining 12.2% of the variability of the outcome values. Specifically, those who had never attended screening and those who had sometimes delayed or missed screening were 2.97 times and 2.65 times more likely, respectively, to favour a nurse or doctor using a self-sampling kit over screening done in the usual way than were those who had always attended. Never-attenders and those delaying or missing screening were 13.11 times and 5.25 times more likely, respectively, to favour using a self-sampling kit at home over screening done in the usual way than were always-attenders.
Multinomial logistic regression for screening preferences; n = 1319.
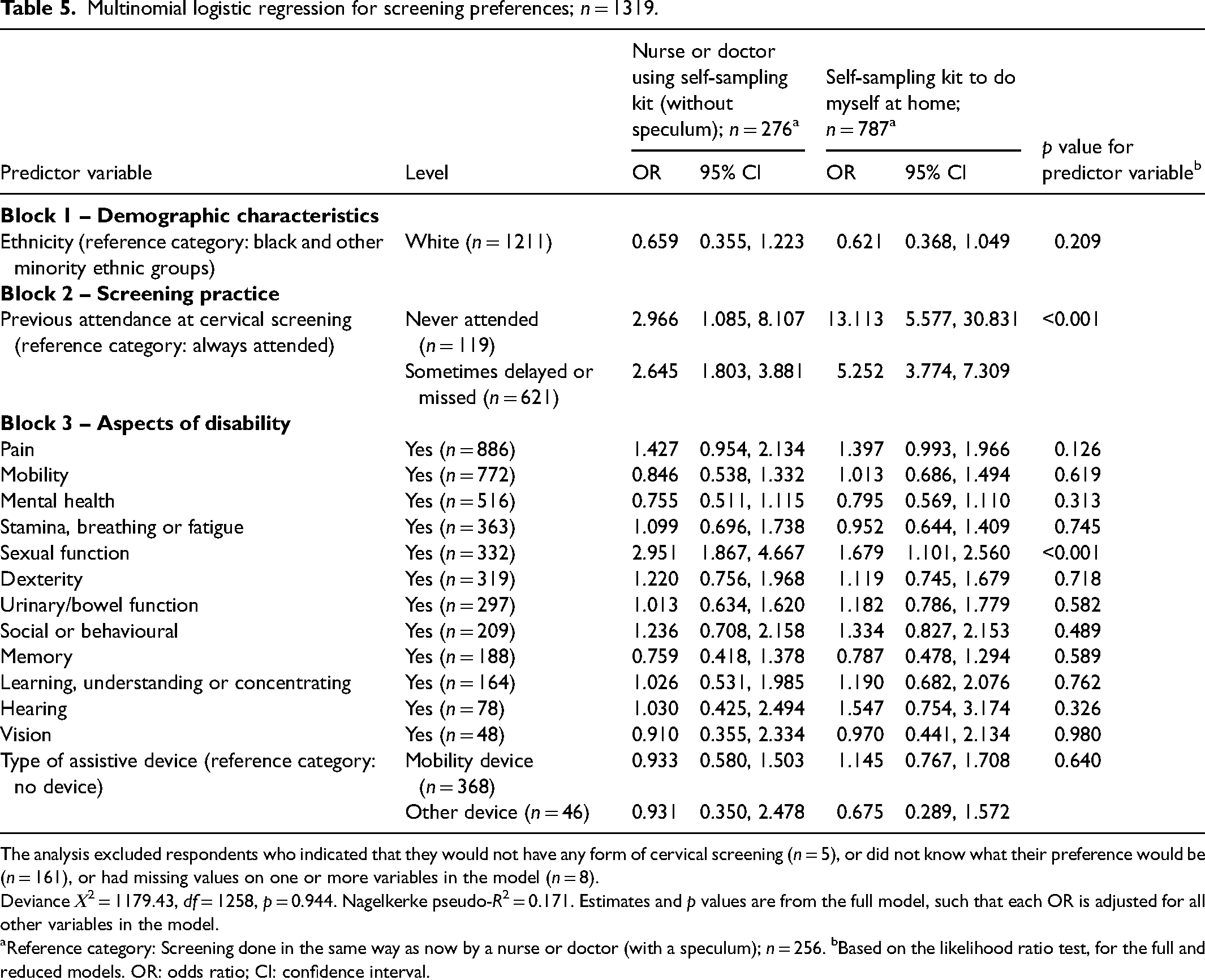
The analysis excluded respondents who indicated that they would not have any form of cervical screening (n = 5), or did not know what their preference would be (n = 161), or had missing values on one or more variables in the model (n = 8).
Deviance Χ2 = 1179.43, df = 1258, p = 0.944. Nagelkerke pseudo-R2 = 0.171. Estimates and p values are from the full model, such that each OR is adjusted for all other variables in the model.
Reference category: Screening done in the same way as now by a nurse or doctor (with a speculum); n = 256. bBased on the likelihood ratio test, for the full and reduced models. OR: odds ratio; CI: confidence interval.
Among the variables representing aspects of disability in Block 3, only sexual function was a significant predictor (p < 0.001). Those with a disability relating to sexual function were 2.95 times more likely than those without to favour a nurse or doctor using a self-sampling kit over screening done in the usual way, and 1.68 times more likely to favour using a self-sampling kit at home over screening done in the usual way. Collectively, the variables relating to aspects of disability in Block 3 explained only 3.9% of the values of the variability in the outcome variable.
Discussion
To our knowledge, this is the first study to quantitatively explore the acceptability to physically Disabled women of HPV self-sampling. Over half of the participants in this study reported that they would be able to carry out self-sampling, and around half expressed a preference for this method over other methods, including conventional speculum-based screening. These findings are consistent with previous qualitative studies with physically Disabled women, which suggest that self-sampling could alleviate barriers associated with clinician-led screening.11,15 Importantly, women who had never attended screening, or who had delayed or missed appointments, were markedly and significantly more likely to prefer self-sampling than those who had always attended. This finding mirrors results from studies in under-screened populations7,19 and further supports NHS England's planned roll out of self-sampling to under-screened women.
With the exception of sexual function, specific types of disability did not predict screening preference, underlining the fact that different options need to be available for women to be able to select the screening method that best suits them. Those participants whose disability was related at least in part to sexual function expressed preferences (for non-speculum clinician sampling and for self-sampling) that may reflect concerns linked to the use of a speculum, possibly related to heightened discomfort on its insertion or its association with penetration. 27
While use of a self-sampling kit at home was the most favoured option, almost a fifth of our participants expressed a preference for non-speculum clinician sampling, and a similar number would prefer conventional speculum sampling. These findings reinforce the important message that there is not one solution to making cervical screening more accessible for physically Disabled women, and further work is needed to increase the inclusivity of screening. We are not aware of planned work to introduce non-speculum clinician sampling, although we note that Landy et al. 28 found that offering non-speculum clinician-sampling and self-sampling to older women with lapsed screening attendance increased uptake, with non-speculum sampling being favoured by those who dislike the speculum but prefer a clinician to take a sample.
Concerns about performing the test correctly were widespread among our participants, highlighting the need for accessible, tailored instructions and reassurance from healthcare professionals. While this is frequently a concern for non-disabled women too, currently, the instructions for self-sampling kits typically use images of non-disabled bodies, which are often shown in weight-bearing positions to demonstrate how to insert the swab. The planned roll out of HPV self-sampling in the UK to women who never or rarely attend their routine cervical screening appointments is an ideal opportunity to consult with Disabled women to ensure the kits and their instructions are as accessible as possible.
Reassuringly, most participants in our study did not report concerns about harming themselves, getting an infection, or feeling embarrassed using the kits. Furthermore, many indicated that the kits would offer empowerment, with many participants agreeing that if they did self-sampling, they would be more in control of their body. These findings suggest that self-sampling offers a very acceptable alternative to conventional sampling for many physically Disabled women.
Strengths and limitations
This is the first study to systematically explore the acceptability of HPV self-sampling among physically Disabled women. We were able to recruit a substantial number of participants in this overlooked demographic. Previous trials in the UK and internationally have shown that HPV self-sampling is acceptable and can improve uptake among under-screened groups5–7,9,10 but did not include Disabled populations. Given that Disabled women face many barriers to cervical screening – for example, see Vinson et al. 11 – our findings address a major evidence gap12,13 and provide important new evidence that HPV self-sampling could help to reduce these barriers to make cervical screening more accessible for some physically Disabled women.
Both the design and recruitment process of the study were informed by extensive patient and public involvement, and as a result, the research is grounded in the social model of disability. 17 Using this as the lens through which we designed the study meant that we asked women to self-identify as having a physical disability, impairment, condition or difference. This enabled us to bypass challenges associated with the absence of systematic reporting about disability in primary care health records and access women with a wide range of physical disabilities and conditions, including those that are less visible.
In terms of limitations, most participants were recruited via an online panel, which may have resulted in over-representation of women with higher digital literacy and education. The cross-sectional design explored attitudes and intentions but did not assess actual use of self-sampling kits, or how collection and return would work for Disabled women, which is something that would benefit from future research. Lastly, non-speculum clinician-taken sampling is not currently available in the UK and, in its absence, we cannot be sure what the 18.7% of women who preferred this option over self-sampling or speculum sampling would choose.
Implications for practice
To ensure the successful implementation of HPV self-sampling and maximize uptake among under- and never-screened physically Disabled women, it will be important to ensure that self-sampling kits and instructions are accessible for those who are able to, and wish to, use them, as concerns about correct use were common in our participants. Importantly, self-sampling will not be a suitable option for all physically Disabled women and other options, such as non-speculum clinician sampling, should also be considered. Training for healthcare professionals should emphasize disability confidence and inclusive communication, enabling them to actively remove barriers and support women who require assistance. Crucially, service models should offer a choice based on the patient's individual needs and preferences between self-sampling, non-speculum clinician-assisted self-sampling, and conventional screening. Finally, Disabled people should be meaningfully involved in the roll out of self-sampling, ensuring that it contributes to reducing the barriers currently faced rather than creating new ones.
Conclusion
This study provides the first quantitative evidence on the acceptability of HPV self-sampling among physically Disabled women. The findings show that self-sampling would be acceptable to many Disabled women and could prove to be a preferred option to conventional speculum-based screening, particularly among those who have previously missed or delayed attendance. Our findings highlight the importance of accessible instructions and reassurance from healthcare professionals to help meet patient needs and facilitate cervical screening. In order to reduce inequalities in preventive healthcare for Disabled women, ensuring the voices of Disabled women are considered in implementing the roll out of self sampling will be essential to this new aspect of the NHS cervical screening programme.
Footnotes
Acknowledgements
We would like to thank the following members of our PPI stakeholder group for their work in helping us develop the survey materials: Susan Bennett, Lynda Hesketh BEM., Emma Major, Ali Murray, Melody Powell, Roxanne Steel, plus a further eight members who prefer to remain anonymous. We would also like to thank the people who took part in the survey, as well as SCOPE, Spinal Injuries Association, Brittle Bone Society, Muscular Dystrophy UK, for circulating the survey, and The Eve Appeal for providing support to this project.
Ethical considerations
This study was granted ethical approval from Keele University Research Ethics Committee (REF: REC Project Reference 0787) and the University of Sheffield School of Psychology research ethics committee (REF: 064385).
Consent to participate
Participants had to consent to continue by selecting the relevant tick box on the online survey before being able to continue.
Author contributions
EK and SS: programmed the software; EK: collected the data; JS: conducted the statistical analysis and prepared the tables; EK, SS and JS: wrote the main manuscript text; SS, CAC, AF, CH, SR, JS and KWB contributed to the funding application. All authors contributed to conceptualization of the study and methodology and reviewed the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is funded by the UK National Institute for Health and Care Research (NIHR) under its Research for Patient Benefit (RfPB) Programme (Grant Reference Number NIHR204322). The views expressed are those of the authors and not necessarily those of the NIHR or the Department of Health and Social Care. CCG is part funded by the National Institute for Health and Care Research (NIHR) Applied Research Collaboration West Midlands (NIHR200165).
