Abstract
Background
Non-attenders for cervical screening are at increased risk of cervical cancer. Studies offering self-sampling for high-risk Human Papillomavirus (HrHPV) testing have shown greater uptake than sending another invitation for cytology.
Objectives
To explore whether uptake would increase in a less diverse, more stable population than the previous English study, which demonstrated a lower response rate than other studies. The primary objective was whether non-attenders were more likely to respond to a postal invitation, including kit, to collect a self-sample compared with a further invitation for cytology screening. The secondary objective was whether women with an abnormal result would attend for follow-up.
Methods
6000 non-attenders for screening in this pragmatic, randomized (1:1) controlled trial in Newcastle-upon-Tyne were sent an HPV self-sample kit (intervention) or a further invitation for cytology screening (comparator).
Results
411(13%) responded to the intervention, returning a self-sample (247(8%)) or attending for cytology (164(5%)), compared with 183(6%) attending for cytology, relative risk 2.25 (95% CI 1.90–2.65) (comparator arm). Of those testing hrHPV positive (32(13%)), 19(59%) subsequently attended cytology screening. Of those in the intervention group who attended for cytology screening without returning an hrHPV self-sample (n = 164), 5% (n = 8) were referred for colposcopy - all attended. In the comparator group eight of the nine referred for colposcopy attended.
Conclusion
Persistent non-responders to invitations for cervical screening are significantly more likely to respond to a postal invitation to return a self-collected sample for HPV testing than a further invitation for cytology screening. However, just over half followed up on this positive HPV result.
Introduction
Women who do not attend for regular cervical screening are at increased risk of developing cervical cancer compared with regular attendees. 1 It has been estimated that the cervical screening programme in England and Wales has prevented up to 5000 deaths per year with three to five yearly routine cytology screening. 2 Attendance rates in the United Kingdom (UK) have consistently declined in recent years. 3 In 2012–2013 78.3% were screened at least once in the last five years, below the National Health Service Cervical Screening Programme’s (NHSCSP) target of 80%.3–6
Inequalities in the uptake of cancer screening have been well documented, with poor uptake associated with ethnic minority populations and social deprivation.7,8 Barriers to attendance cited by women include fear or embarrassment, pain, the gender of the sample taker, inconvenient appointment times, and difficulty arranging child-care or time off work.6,9,10,11 Self-collected sampling may overcome many of these barriers.
Clinician-collected samples for high-risk Human Papillomavirus (hrHPV) testing have been shown to offer better protection against cervical cancer than cytology alone.12–14 This procedure is being explored as the primary test for screening programmes, with the Netherlands the first national programme due to adopt it. 15 It also has potential as a test carried out on cervico-vaginal samples collected by women themselves, which has been shown to have similar sensitivity and specificity to clinician samples, although not with all hrHPV testing platforms.16–19 In addition, self-collected sampling, using devices such as swabs, brushes, tampons, or lavage, is broadly acceptable to women.19–23
In the era of HPV prophylactic vaccination screening programmes are likely to change. Participants in the school-based vaccination programme in the UK are due to be invited for screening as early as 2015. UK trials of HPV testing as primary screening are currently underway. The plan in the Netherlands is for HPV primary screening with clinician samples, and to offer self-sampling to non-responders. 15 This study should add useful information for changing screening strategies.
In a number of studies of self-collected sampling to increase screening in non-responders, the strategy appears more effective than sending another invitation for cytology.17,24–31 Five studies have reported response rates to self-sampling, ranging from 20 to 39%. However a sixth study, carried out in a district of London with a highly mobile population, of whom 65% were not from a white British ethnic background, showed a lower response rate of only 10.2%.29,32
The rationale for undertaking the present study in Newcastle upon Tyne was to explore whether targeting non-responders from a less diverse and more stable population would improve uptake. Newcastle upon Tyne has a population where 18% are not white British in contrast to 65% in the London study. 32
Methods
Design
This trial was a pragmatic, randomized controlled trial of 6000 women randomized 1:1 to an intervention group or a comparator group. The report of this clinical trial conforms to the CONSORT statement. 33
Participants
Participants were selected from women in the Newcastle upon Tyne region (a mixed urban/rural location typical of the UK) who had failed to attend for cervical cytology. These women were at point 4 on the North East Primary Care Services Agency (NEPCSA) cervical screening invitation letter schedule (Figure 1), and had received a minimum of two invitations to attend for cervical screening. They were not all on three to five yearly recall, as some were due for early cytology repeat. They were identified on 3 September 2012 via the Exeter National Health Applications and Infrastructures Services System (NHAISs) at NHS England, Durham, Darlington and Tees and working on behalf of Cumbria and Northumberland, Tyne and Wear Area Team (formerly NEPCSA). NHAIS holds the screening information of all women registered nationally with a General Practitioner (GP). Routinely, women due for screening are invited by the regional screening team. The GP will be informed of women due for screening, and will also be made aware of non-responders. They may, as a point of good practice, contact the women, but the process is primarily managed on a regional level.
North of England Commissioning Cervical screening invitation letter schedule.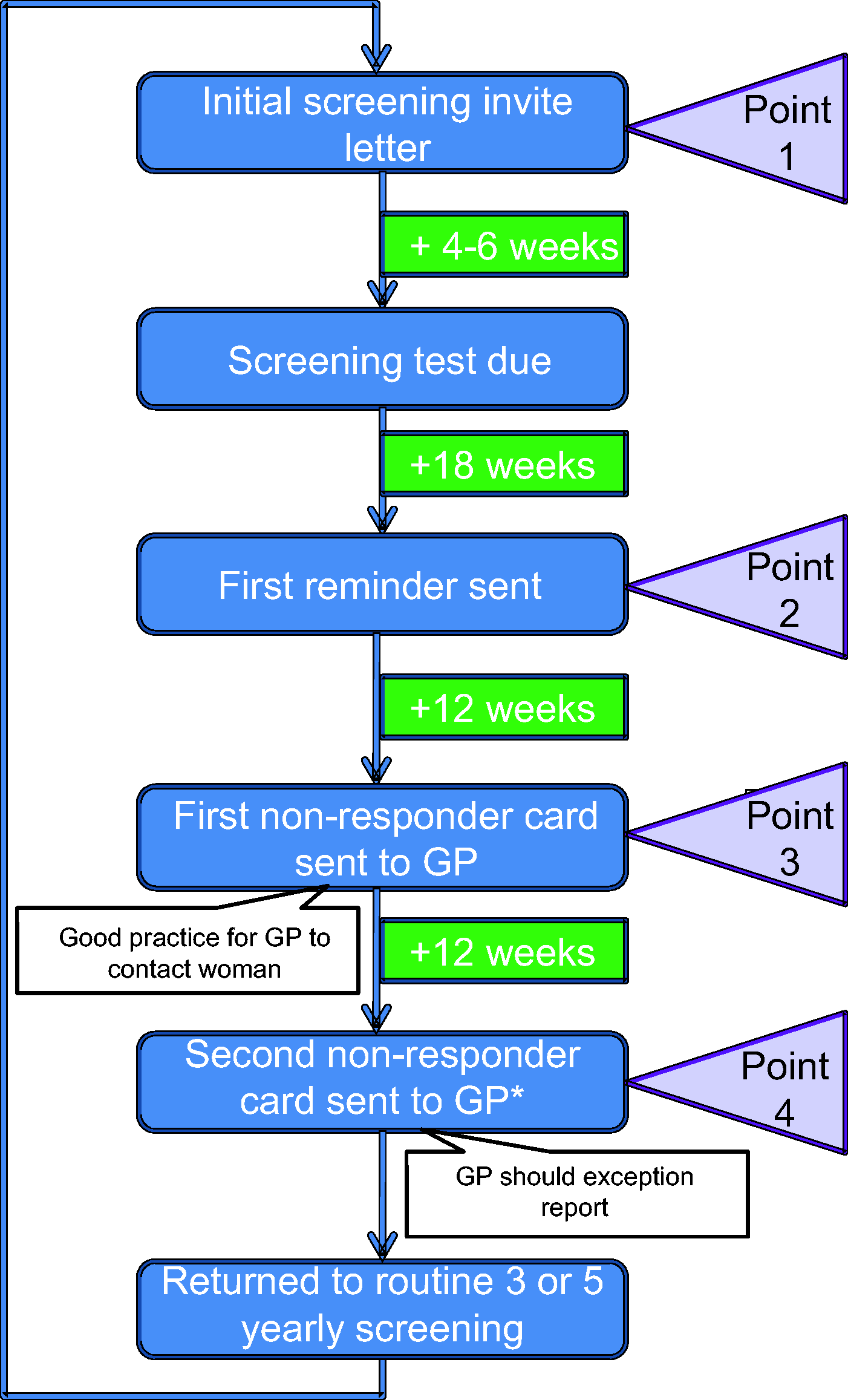
Intervention and Comparator Groups
The intervention group received, by post, a Dacron swab self-collection kit, specimen tube with specimen transport medium (1 ml), packaging material, trial information including a self-sample collection instruction sheet, and a consent form. They were asked to return their swab, consent form, and contact details within three months, which was deemed a reasonable response time. HrHPV samples returned to the Centre for Cancer Prevention (CCP), with a complete consent form and contact details, were tested using Qiagen Hybrid Capture®II technology (Relative Light Unit cutoff >1 indicating hrHPV positive). The hrHPV results were sent to the women by post and although all women were advised that they should still attend for screening, this was emphasized in the hrHPV positive group. All women who did not respond, either to the invitation or advice to follow up an hrHPV positive result with cytology, were returned to the appropriate point on the screening invitation letter schedule (Figure 1).
The comparator group received a further standard cervical cytology invitation letter by post from the NHS England screening team. They received their results in the usual way, by letter from the screening office. All women were advised that they should still attend for routine cytology screening, and were returned to the appropriate point on the screening invitation letter schedule (Figure 1) if they failed to do so. All who had abnormal cytology were managed routinely in accordance with the NHSCSP.
Objectives
The primary objective was to ascertain whether non-attenders were more likely to respond to a postal invitation to collect a self-sample for hrHPV testing by returning the test or attending for cytology screening, compared with a further invitation for routine cytology screening. The secondary objective was to ascertain whether women with abnormal results would attend for follow-up cytology or colposcopy, if necessary.
Outcomes
The primary outcome was the proportion of women responding to the intervention by returning a self-sample or attending for cytology screening, compared with the number of women sent a further standard invitation for cytology who attended within three months. Secondary outcomes were compliance to follow up, and included response to a positive hrHPV result by attending for cytology within three months, and attending for colposcopy in response to abnormal cytology within four months.
Sample size
The sample size of 3000 women in each of the two arms (intervention and comparator) is powered to detect a moderate improvement in initial response, and a substantial improvement in the proportions who attend colposcopy (after a positive screening test). Samples sizes were based on 5% type I error and assumptions made based on the results of the previous UK study. 19 The primary outcome was the proportion providing a screening sample, assumed to be 7.5% in the comparator arm and 10.0% in the intervention arm (based on response rates in the previous English study). 19 Allowing for 10% loss through not reaching the person with the invitation (for example no longer living at that address) and using a two-sided chi-squared test statistic, 2984 women in each arm were required to obtain 90% power. With 3000 patients in each arm and assuming 0.4% of those sent a reminder letter compared with 1.0% of those sent a self-sampling kit would attend for colposcopy (as a result of the test result), this would give 79.6% power to detect a difference (two-sided). 19
Randomization and blinding
As the trial aimed to evaluate an intervention in a population of non-responders, Zelen’s design was adopted. 34 By randomizing participants prior to consent, the potential response bias in the comparator group would be avoided. The local screening team identified the number of non-responders in a snapshot of all women at Point 4 on the NEPCSA cervical screening invitation letter schedule on 3 September 2012 (Figure 1). A randomized list of participant numbers (PNO) was produced at CCP using an in-house computer application and balanced in blocks of four without stratification. PNOs were allocated to the intervention group of 3000 (N) and comparator group of 3000 (C). The list of PNOs was emailed to the Newcastle upon Tyne team in a Microsoft Excel format and inserted into the list of women at Point 4, randomly allocating a PNO to the trial cohort of 6000 women. The names, addresses, and PNOs were securely emailed to the mailing company contracted to NHS North of Tyne, the Mailing House Group Limited, where the intervention packs and comparator group letters were prepared and dispatched. This ensured that the study team was blinded to the allocation, and that those who did not consent to participate in the study remained anonymous. Researchers had access to all data of consented participants.
Statistical methods
The proportions of women responding to the intervention (primary outcome) were calculated in the form of percentage of responders in each arm. The relative risks (RR) were also calculated, with 95% confidence intervals (CI). Secondary outcomes were similarly measured as a proportion of those responding in that category. Additional analyses of the influence of covariates were carried out using multivariate logistic regression.
Ethical Approval
This trial received ethical approval on 1 August 2012 from NHS Health Research Authority, NRES Committee North East – Sunderland (Reference 12/NE/0242).
Results
Recruitment and participant flow
All 6000 women were sent a self-sample collection pack or invitation to attend for screening on 10 September 2012. The final date for either attendance for cytology screening or self-collected sample return was 10 December 2012. The cohort responses are shown in Figure 2. The last colposcopy was performed on 21 January 2013, when the last person attending cytology underwent colposcopy within the prescribed timeframe.
Participant Flow Chart.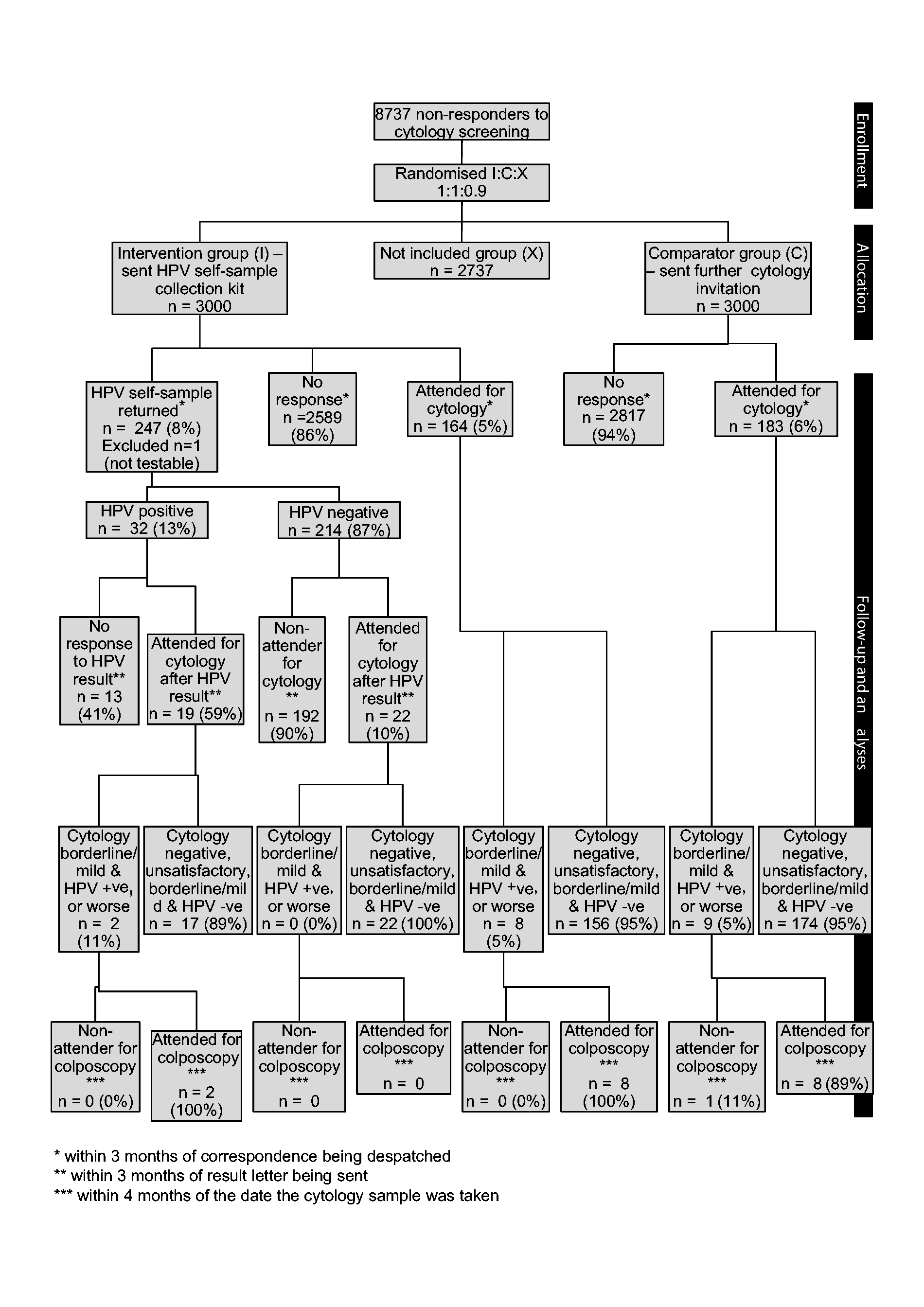
Baseline data
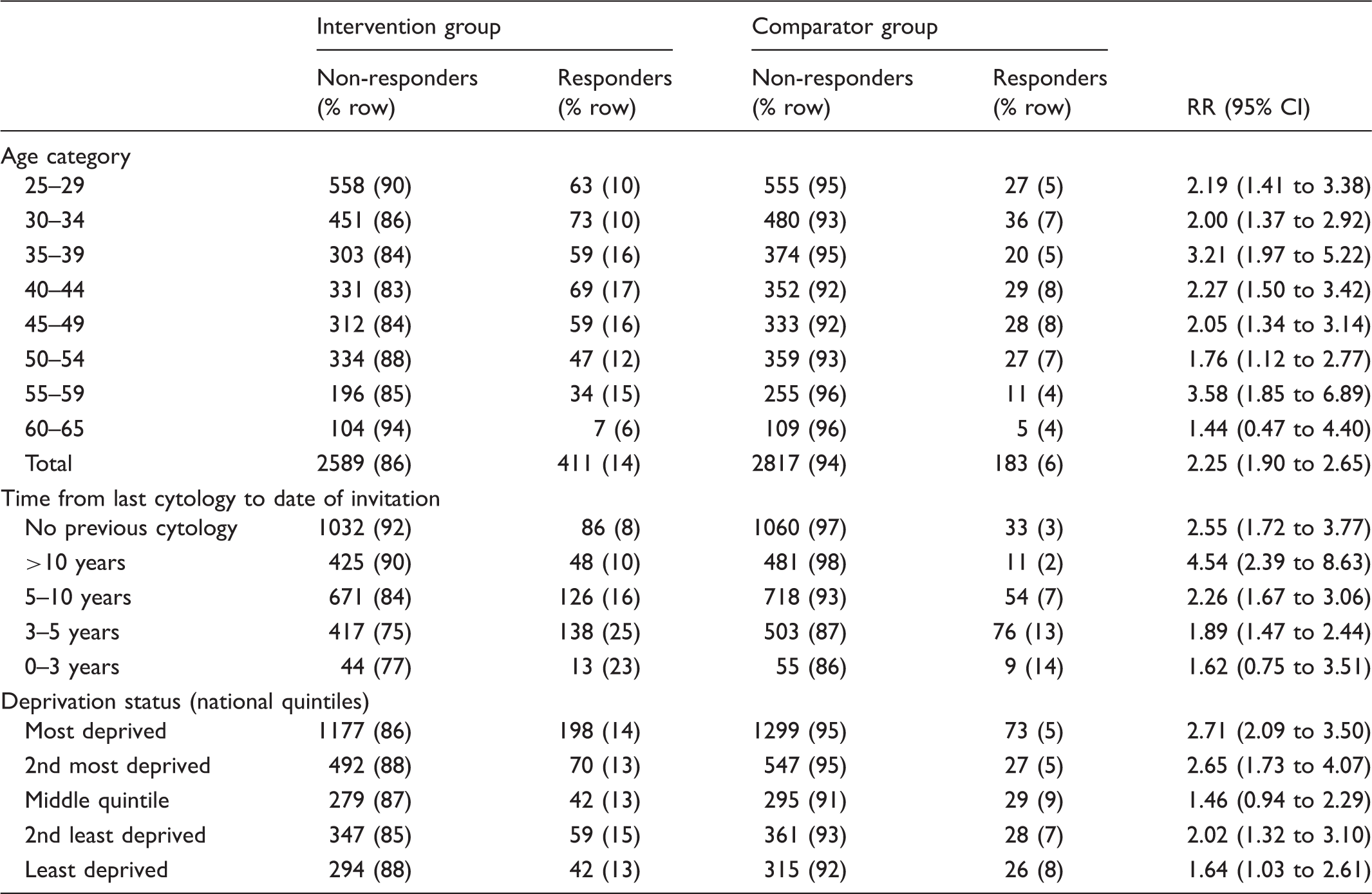
Demographics by allocation group and response.
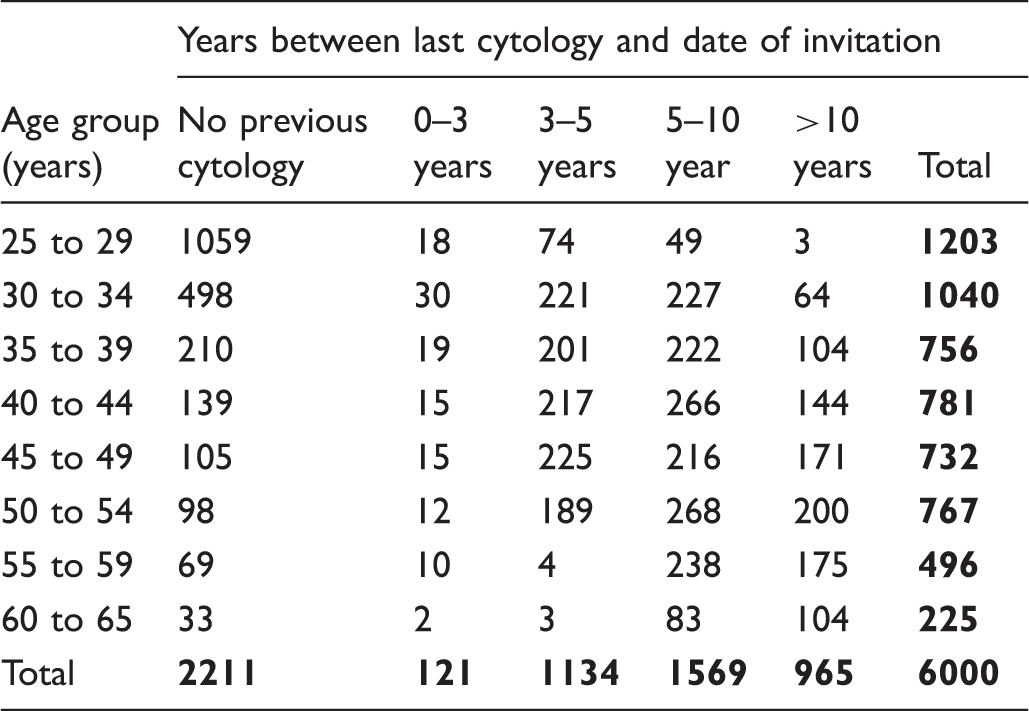
Time between last cytology and date of invitation by age category.
This was a pragmatic trial with the total cohort of 6000 women included in the primary outcome analysis. There were 438 (7%) letters and packs returned to sender, 226 from the comparator group and 212 from the intervention group. The reasons for return included movement out of the area (153 women), death, other reasons, or reasons not given. There were no reported adverse events in the intervention group.
Outcomes and estimations
The primary outcome showed 411 (13%) women in the intervention arm responded to the intervention by returning an hrHPV self-sample (247 (8%)) or attending for cytology (164 (5%)) (Figure 2). This compares with 183 (6%) in the comparator arm responding to a further invitation for cervical screening (RR 2.25 (95%CI 1.90 to 2.65). Women in the intervention arm were two times more likely to respond, and this remained when adjusting for age, deprivation status, or time since their last cytology (RR 2.24 (CI 1.90–2.64). When response rates were restricted to those whose invitations were not returned to the sender, the rates increase in both groups, with 9% (212 of 2788) returning a self-sample and 6% attending for cytology in the intervention group, and 7% (173 of 2744) attending for cytology in the comparator group. If the 28 women who attended for cytology within two months prior to the study invitation (whose attendance would not yet have been recorded on the Exeter system) were removed from the analysis, the figures remain similar: 14% response in the intervention group and 6% in the comparator (RR 2.00(1.69–2.34))
Cytology results.
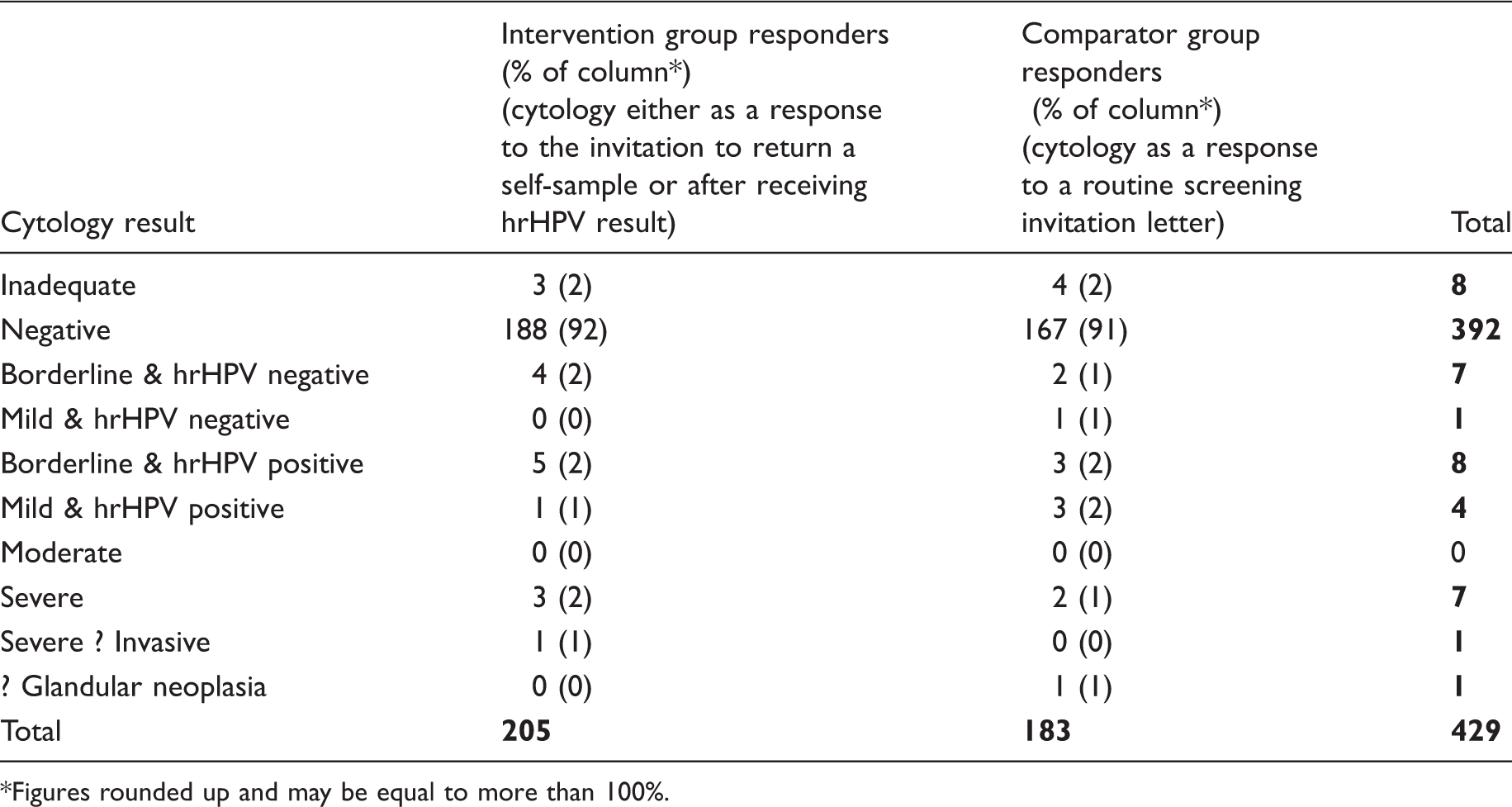
Figures rounded up and may be equal to more than 100%.
Histology results in women referred to colposcopy who attended within six months.
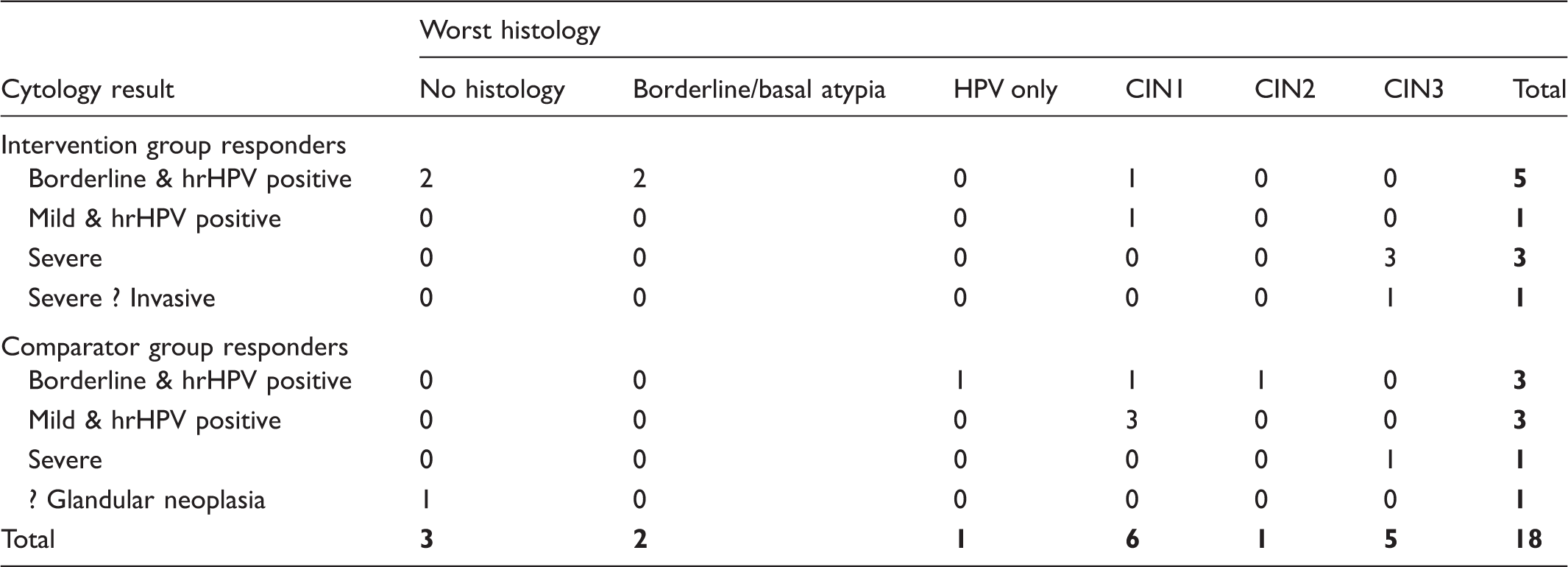
Of the women in the intervention group who responded by attending for cytology screening, rather than returning an hrHPV self-sample (n = 164), 5% (n = 8) were referred to colposcopy in accordance with NHSCSP guidelines, and all eight women (100%) attended. There were two referrals with high grade cytology – severe dyskaryosis and severe dyskaryosis with possible invasion. Both were found to have CINIII after LLETZ. In the comparator group, nine women required referral for colposcopy following abnormal cytology results. Three had cytology results of borderline dyskaryosis with hrHPV triage test positive. A further three had mild dyskaryosis and hrHPV positive, and two had severe dyskaryosis. The final woman had possible glandular neoplasia. Of these nine women, 89% (n = 8) attended for colposcopy. Two required LLETZ for CINII and CINIII. The one woman who did not attend colposcopy within three months did attend within six months, following a severely dyskaryotic smear, with CINIII confirmed on LLETZ.
Influence of covariates
The response rate in the intervention arm was similar across all deprivation quintiles (13–15%). This was not the same in the comparator group, where the two most deprived quintiles showed a 5% response rate, whereas the three least deprived showed 7–9%. Women in the intervention group were more likely to be responders than those in the comparator group in all age groups except those aged 60 to 65 (RR 1.44 (95% CI 0.47–4.40)). This could be a reflection of the small numbers in this age group (Table 1). The time since last cytology and response to intervention was statistically significant for both intervention and comparator groups, with those who had no previous cytology being less likely to respond (p < 0.01). Women in the intervention group were more likely to respond, except in those who had attended for cytology within the last five years (Table 1). Of those never screened, or not screened within the last ten years, 6% were responders to the interventions (Table 1).
Multivariate analyses showed increasing response with increasing age and the probability of responding increased with decreasing deprivation (p = 0.004), however, the relative risk for the interaction was 1.01 (1.00–1.02). There was no significant interaction (not shown) between age and time since last cytology (p = 0.088), nor deprivation and time since last cytology (p = 0.109).
Discussion
This pragmatic randomized controlled trial in non-responders to invitations to attend for cervical screening has shown a response rate to the intervention of posting a self-sample collection kit for HPV testing of 13%, compared with 6% of the comparator group, who responded to a further invitation to attend for cytology. Of this 13%, 8% returned a self-collected sample, and 5% chose to attend for cytology screening. Randomization to intervention or comparator group ensured balanced distribution of potentially influential factors. It is difficult to mitigate against the intervention group responding to a different type of correspondence, as it is to quantify whether being encouraged to still attend for cervical screening as part of the NHSCSP discouraged some women from returning a self-sample. This response rate, although higher than the previous UK study, is still lower than in other countries. 29 This may be a reflection of the different definitions of non-responders in the cohorts, or the interventions in some studies immediately preceding the self-collection kit, such as pre-invitation letters.26,31,35,36 The findings of this pragmatically designed trial can be generalized to the English NHSCSP, however, the outcomes can also be generalized to a wider population, when considered as part of the pool of evidence of self-collected cervico-vaginal sampling in non-responders.
Of the women testing hrHPV positive, only 59% attended for cytology, which raises concerns about the CIN which remains undetected and untreated. This response rate was lower than for the previous English study, possibly because the London study offered women cytology in a research clinic, whereas this study was more pragmatic and women were advised to arrange and attend for screening under their own auspices. The hrHPV positivity rate in this group was higher than that found in previous studies at 13%, compared with 8.3% in studies of a comparable age group in the UK using the same hrHPV test.29,37 This could be explained by this study cohort’s lower median age: 41(IQR 32–48) versus 47.5 (40.5–54.5) in the previous English study. However, less that 1% of women returning a self-sampling test required colposcopic examination and, although numbers were too small to be of great significance, high grade CIN was detected and treated in 100% of cases. In those undergoing cytology screening alone, 4.6% required colposcopy, and 25% of these women had high grade disease diagnosed and treated at colposcopy. The attendance rates for colposcopy were the same in both groups. This differed from the previous study in London, but again could be explained by offering women in the intervention group colposcopy at the same time as cytology in the research clinic. It could be hypothesized that the colposcopy attendance in this pragmatic study better replicates the non-study situation.
Deprivation is associated with non-attendance for cervical screening, and in Newcastle as a whole, it would be expected that 40% of the population would be in the two most deprived national quintiles. 38 In this study cohort of non-responders, however, this figure reaches 65%, which supports the association between deprivation and non-attendance for cervical screening. Deprivation does not appear to be a barrier to returning a self-sample, as 68% of responders in the self-sample group lived in the two most deprived quintiles, compared with 57% in both arms who attended for cytology screening first. The accessibility of self-sampling, without the need to take time off work, travel to the clinic, or arrange childcare, may be of benefit to those from lower resource settings.
This trial supports the increasing evidence that self-sampling for hrHPV testing is effective for women not currently being reached by organized screening programmes.16,17 However, there are cost implications in sending out self-collection kits to all women considered non-responders. Twelve test kits were sent for every one returned, and approximately 40% of the expenditure in the intervention arm was the cost of producing and sending kits. 39 This compared with 15% of the overall cost in the comparator arm of sending out repeat invitations. The estimated cost per high grade CIN detected was, however, similar in both the comparator and intervention arms (ratio 1:1.09). This was not the case when those in the intervention group who attended for cytology screening, rather than return a self-sample, and those testing HPV negative were excluded (ratio 1:1.36). A full economic analysis is required before such a strategy can be adopted.
HrHPV testing is being considered as the primary cervical screening method for the UK, where trials are currently underway, and in countries where cervical screening programmes are not yet established. 12 There is also interest in self-sampling for HPV testing as a method of primary screening in countries where resources and access to healthcare and healthcare professionals are limited.40,41 Although this is a reasonable option for non-responders to invitations for screening, it would be of value to explore more cost-effective approaches to target non-responders. When considered as primary screening, self-collection for HPV testing tends to have a lower sensitivity and specificity compared with clinician samples. A recent meta-analysis suggested that the pooled sensitivity for self-collected samples in primary screening for the detection of CIN2 or worse was 76% (95% CI 69 to 92) and specificity 86% (95% CI 83 to 89) versus 91% (95% CI 87 to 94) sensitivity and 88% (95% CI 85 to 91) specificity for clinician samples. 16 Although HPV testing from self-collected samples in its current form is not the preferred test to a clinician sample, there is evidence to suggest that selecting the correct test (Polymerase Chain Reaction based) or adapting the testing methodology for self-sampling (such as raising the cut-off point for positivity) could improve the sensitivity and specificity.16,42
Footnotes
Acknowledgements
Professors Jack Cuzick and Peter Sasieni (CCP), Melanie Ellis, Jan Scutt, Cheryl Murray, Christopher Purvis and Neil McCarthy on behalf of NHS England, Durham, Darlington and Tees and working on behalf of Cumbria, Northumberland, Tyne and Wear Area Team; Shona Haining on behalf of North of England Commissioning Support; Jilly Goodfellow on behalf of Royal Victoria Infirmary; Rebecca Landy, George Terry, and Linda Ho (CCP).
Conflicts of interest
Funding
Cancer Research UK Programme Grants (C569/A10404 and C8162/A10406)
Trial registration
UK Clinical Research Network (UKCRN): Portfolio Database ID: 13096
International Standard Randomised Controlled Trial Number (ISRCTN): 39154605
