Abstract
Objective
Organised cervical cancer screening was started in Estonia in 2006, but participation is still low. Human papillomavirus (HPV) self-sampling has proved to increase screening uptake. This study addressed the feasibility of HPV self-sampling and the acceptance of this method among long-term screening non-attenders.
Methods
A randomised intervention study was conducted in Estonia in 2020. Women born in 1958–1983 without a Pap smear in 2013–2019 were identified in the Estonian Health Insurance Fund database. From them, 12,000 women were randomly allocated to three equal-sized study groups. The opt-out group received a questionnaire and a Qvintip® sampling device by regular mail. Two opt-in groups received a questionnaire and an e-mail invitation to order a self-sampler online; one received Qvintip and the other Evalyn® Brush. Participantś background characteristics were obtained from the Population Register. The effect of covariates on participation rate was estimated with multivariate Poisson regression. Acceptance of self-sampling was analysed according to agreement with statements in the questionnaire.
Results
The overall participation rate was 16% with significant differences between opt-out (26%) and opt-in (11%) groups. Compared to the opt-out Qvintip group, adjusted relative risks for the Qvintip and Evalyn Brush opt-in groups were 0.41 (95% confidence interval (CI) 0.37–0.45) and 0.44 (95% CI 0.40–0.49), respectively. Participation was associated with living place, citizenship, and education. Self-sampling was well accepted: 98% agreed that it was easy to use, 88% preferred it as a screening method in future.
Conclusions
The results show the feasibility and good acceptance of HPV self-sampling among long-term screening non-attenders in Estonia.
Introduction
The Estonian Health Insurance Fund (EHIF) covers the costs of health services for the insured persons. 1 Nation-wide organised cervical cancer screening was introduced in Estonia in 2006. Until 2021, women with health insurance were invited to give a Pap smear every five years from the age of 30 up to the age of 55 years. 2
Nevertheless, the world standardised incidence of cervical cancer in Estonia, at 16.2 cases per 100,000, was the fourth highest in Europe in 2010 3 and the incidence was still rising in 2016. 4 One of the reasons for this is the low participation rate in the Pap-smear-based screening programme, which was 46.1% in 2019. 5 Starting from 2021, Pap smear was replaced by human papillomavirus (HPV) nucleic acid technology (NAT) as the primary screening test, opening up the opportunity to introduce HPV self-sampling, which may increase participation rate. 6 The accuracy of polymerase chain reaction-based self-samples is comparable to those taken by a physician. 7 In many European countries, the self-sampling method is already in use in cervical cancer screening programmes. 8 The acceptance of self-sampling compared to clinician-collected sampling has been studied previously; both have been found acceptable to women with no clear association with background factors. 9 Several studies have proved that self-sampling can increase the screening coverage in under-screened populations.10–12
The aim of this study was to assess the acceptability of HPV self-sampling among long-term non-attenders in the Estonian cervical cancer screening programme, women’s evaluations of the self-sampling procedure and different devices, and willingness to use self-sampling in the future.
Methods
Study sample
The study population comprised 65,218 women born between 1958 and 1983 who did not undergo a Pap smear test during 2013–2019. Data were obtained from the EHIF database, which is an administrative database of claims from Estonian healthcare providers, and covers information on each medical service, as well as on prescription medicines, medical devices and benefits for incapacity to work. 13
Data were individually linked with the Estonian Cancer Registry to exclude women with a diagnosis of uterine cancer (International Classification of Diseases 10th Revision (ICD-10) ICD-10 codes C53-55, D06), and with the Estonian Cancer Screening Registry to exclude women with a recent HPV test, colposcopy or Pap smear not reported to the EHIF (Figure 1). Additional information on the woman’s home and email address, education, ethnicity, citizenship, and native language was obtained from the Estonian Population Registry. Women currently residing abroad and those with missing postal or email address were also excluded (Figure 1). From the remaining 41,587 women, a random sample of 12,000 was drawn who were randomly allocated into three study groups (1:1:1) and invited to participate. The sample size was calculated to detect 4% of difference between the study groups at a statistical significance level of 0.05 with 90% power, assuming 30% maximum participation rate in one group. Two different self-sampling devices were used in this study: Qvintip® swab (Aprovix AB, Uppsala, Sweden) and Evalyn® Brush (Rovers Medical Devices B.V, Oss, the Netherlands). In the opt-out group, an HPV self-sampling kit (Qvintip) was sent to the participants’ home address by ordinary mail; in two opt-in groups the participants were sent an invitation by email and were offered an option to order a self-sampling kit from a website. In the latter two groups, women received either Qvintip or Evalyn Brush; the women did not know the type while ordering. Evalyn Brush could not be used in the opt-out group, its size being too big for Estonian mailboxes. Women in the opt-in groups who were unable to use the website for placing the order had the option to do it by phone or by e-mail.
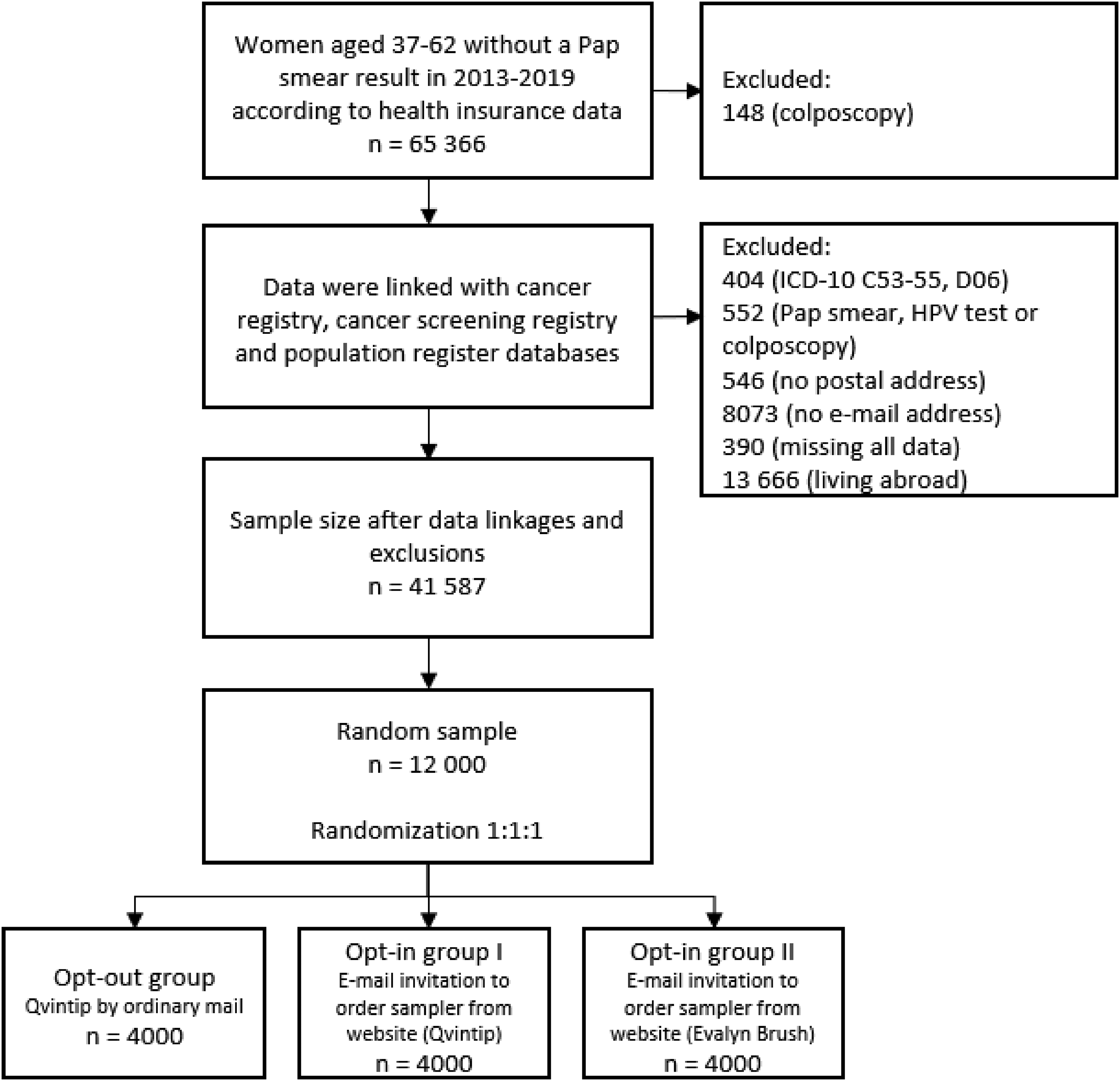
Flowchart of study design.
Study invitations were sent out by ordinary mail to the women in the opt-out group and by e-mail to those in the opt-in groups from August through October 2020. The invitation letter disclosed study information, explained the voluntary nature of participation, and asked for informed consent to participate. No signed informed consent was taken; the agreement to participate was indicated by return of a questionnaire and/or the self-sample. Women were offered the opportunity to perform an HPV self-sampling test free of charge regardless of their health insurance status. The self-sampling kit included a prepaid and preaddressed envelope for returning the used device to the laboratory for analysis.
All invitees received also a questionnaire (Appendix 1) containing 10 statements about their user experience and willingness to use HPV self-sampling in the future. Experience from previous studies was used to draft the questionnaire.14,15 The options for answering were the following: agree, disagree, cannot say. All invitees were asked which way they would prefer to be informed about a positive high-risk HPV (hrHPV) test result (phone, mail, or encrypted e-mail). The study invitations, information letters and questionnaires were in Estonian, Russian and English. The invitations were sent in four batches to the opt-out group and eight batches to the opt-in groups during the period of nine weeks. About three weeks later, all the invited women who had returned neither the self-sample nor the questionnaire (opt-out group) or had not ordered an HPV test kit (opt-in groups) were sent a reminder.
Analysis of HPV self-samples
One central laboratory (Tartu University Clinic Central Lab) analysed all the HPV self-samples. The dry self-sampling devices were transferred after arrival into 20 mL of ThinPrep medium prior to hrHPV testing. Samples were then analysed using the Alinity
Informing women about the results
All study participants could view their test results on the central Electronic Health Record (www.digilugu.ee) by logging in using digital identification. The test results became available within 2 weeks of sending the sample. All participants who tested positive for hrHPV were contacted in person using their preferred contact method as indicated in the questionnaire. If hrHPV infection was detected, additional examinations according to the national guidelines were offered (Pap smear in the clinic first, followed by colposcopy in the case of an abnormal Pap-smear result). The costs of all additional examinations and necessary treatment were covered by the EHIF, even if the person had currently no health insurance.
All participants had the option to ask for information by e-mail or phone. Women with a negative result were encouraged to continue to participate in the organised cervical cancer screening programme.
Primary and secondary outcomes
The primary outcome was the participation rate in different study arms, and the secondary outcome was the evaluation of self-sample device user experience. Participation was defined as having sent an HPV self-sample for analysis. The assessment of user experience was based on questionnaires, where at least one of the 10 provided statements was answered.
Statistical Analyses
Participation rates in different study groups were analysed using the intention-to-treat principle. Differences between groups were explored using the χ2 test. Poisson regression with a robust error variance was used to estimate the effect of study group and other covariates on participation rate. The robust Poisson method was used to avoid a possible overestimation of relationships between variables with logistic regression in case the outcome is not rare. Unadjusted and adjusted relative risks (RR) were calculated with 95% confidence intervals (CI). Covariates used were the study group, age group, place of residence, ethnicity, citizenship, native language, education, and interruption in health insurance. The age groups used were 37–43, 44–50, 51–56, and 57–62 years. Place of residence was categorised according to NUTS3 levels by Statistics Estonia.
16
The NUTS (Nomenclature des Unités territoriales statistiques) classification is a hierarchical system for dividing up the economic territory of the EU and the UK for regional statistics; NUTS3 is used for small regions. Ethnicity was categorized as Estonian, other or unknown. Citizenship was grouped as Estonian, other or undetermined. Native language was grouped as Estonian, other or unknown, and education as higher, secondary vocational, secondary, basic or unknown. Interruption in health insurance was categorised as a binary variable, with persons without health insurance as having a period without insurance longer than 6 months. Factors with a statistical significance level of
User experience with HPV self-sampling and willingness to use it in the future were analysed according to agreement with the statements in the questionnaire.
All analyses were conducted with Stata 14.2 (STATA Corp, College Station, TX). The random number generator was used in Stata for study sample selection and group allocation.
Ethics
The study protocol was approved by the Research Ethics Committee of the National Institute for Health Development (No 286, 20 April 2020).
Results
Characteristics of study population
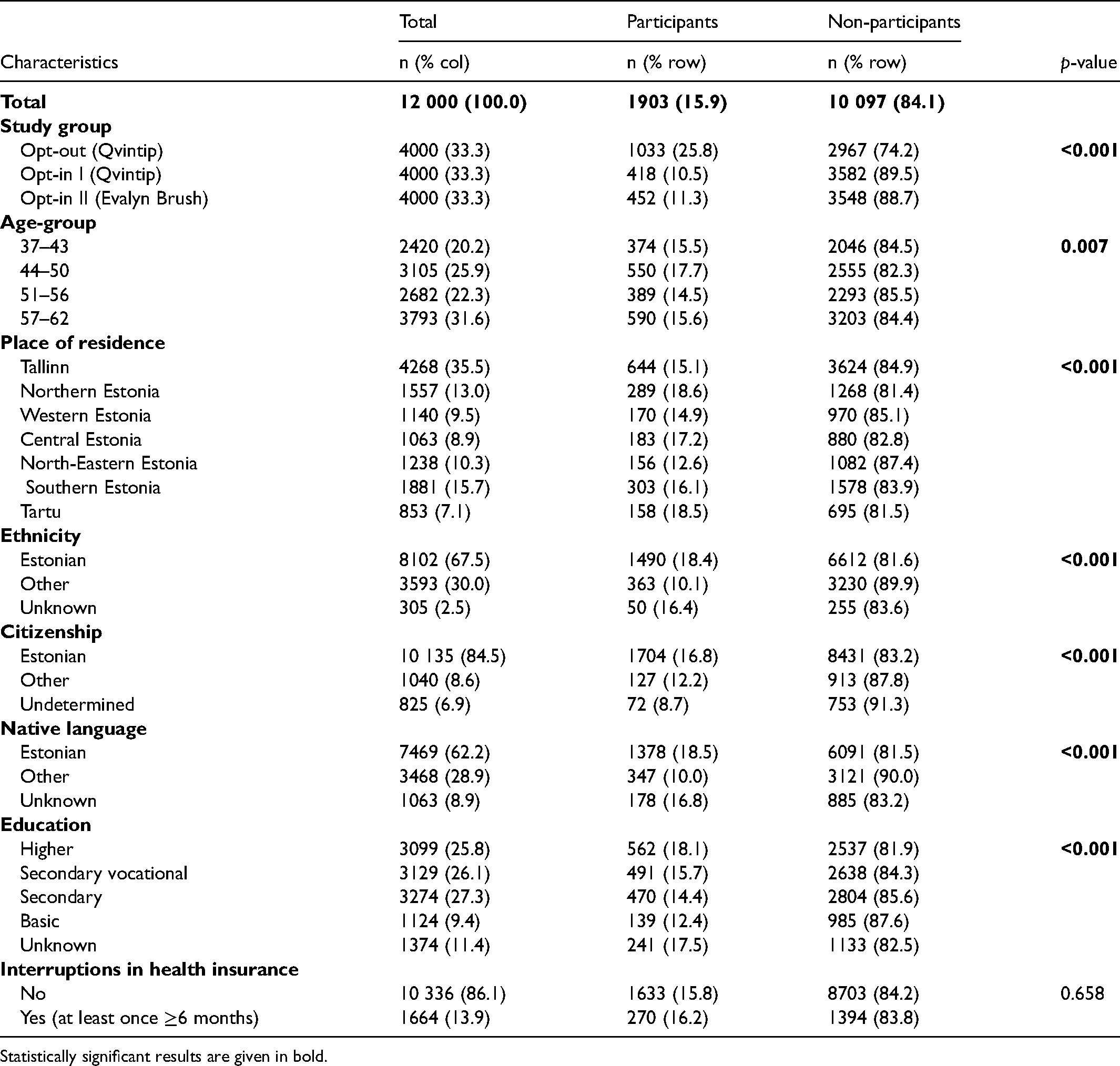
Table 1 shows the baseline characteristics of the study population. Mean age was 51.1 years; 31.6% were aged between 57 and 62. More than a third of the sample were residents of the Estonian capital city, Tallinn. By ethnicity 67.5% were Estonians, and approximately 85% were Estonian citizens. Secondary education was marked as the highest education level for 27.3%. For 13.9% there were interruption(s) in health insurance at least once for ≥ 6 months during 2013–2019.
Baseline characteristics of study population.
Statistically significant results are given in bold.
Altogether, 392 (3.3%) women were excluded from further correspondence after the initial invitation letter. Among them, 259 (2.2%) women refused to participate, 107 (2.7%) in opt-out and 152 (1.9%) in opt-in groups, respectively (Figure 2). Main reasons for non-participation were a previous hysterectomy, living abroad, recent screening, disability, special needs, and other health-related issues. Additional reasons were also pregnancy, childbirth, virginity, being overweight, religious considerations, logistical problems related to picking up the parcel, or other. In addition, in the opt-out group, 133 (3.3%) mail packages were returned unopened due to wrong address, missing or too small mailbox, etc.
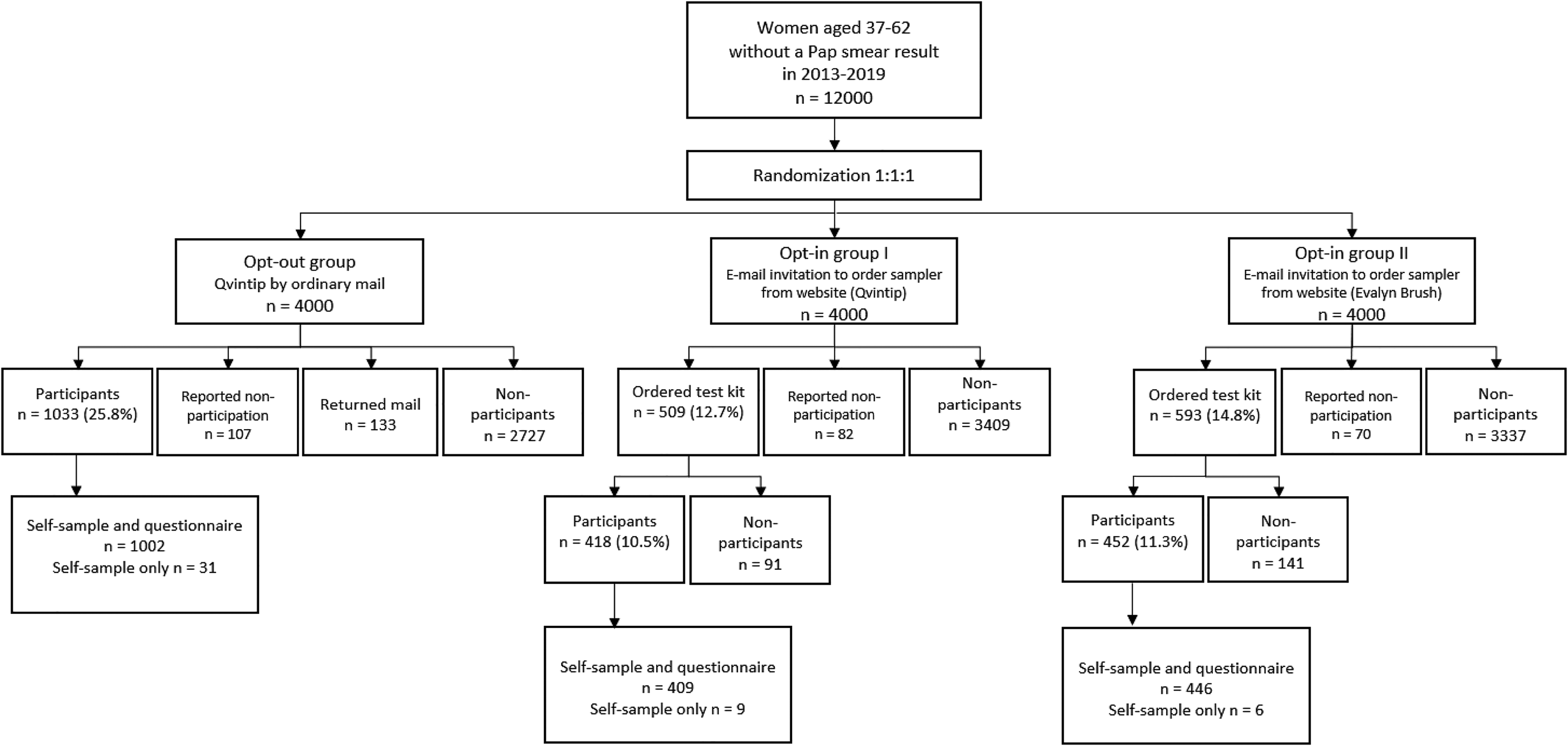
Flowchart of participation.
Participation rate
Out of the 12,000 enrolled women, 1903 (16%) participated in the study and sent the self-collected sample for analysis (Table 1, Figure 2). The mean age of the study participants was 51 years. There was a significant difference in participation rate between the opt-out group (n = 1033, 25.8%) and the two opt-in groups (n = 870, 10.9%). The participation rate was highest in age group 44–50 years, and women living in Northern Estonia, of Estonian ethnicity or citizenship, speaking Estonian as native language, and those with the highest educational level (Table 1).
In univariate analysis, the participation rate was significantly associated with age, place of residence, ethnicity, citizenship, native language, the level of education, and was not associated with interruptions in health insurance during the study period (Table 2). After adjustment in multivariate analysis, participation was significantly associated with study group, place of residence, citizenship, and the level of education. Compared to the Qvintip opt-out group, adjusted RRs for the opt-in groups were 0.41 (95% CI 0.37–0.45) for Qvintip and 0.44 (95% CI 0.40–0.49) for Evalyn Brush. Women living Northern Estonia were 21% more likely to participate in the study than women living in the capital city, Tallinn. Compared to Estonian citizens, the foreign citizens were 27% less likely, and women with undetermined citizenship 42% less likely, to participate. Women with primary or basic education were 32% less likely to participate than women with higher education (Table 2).
Probability of participation in different study groups.
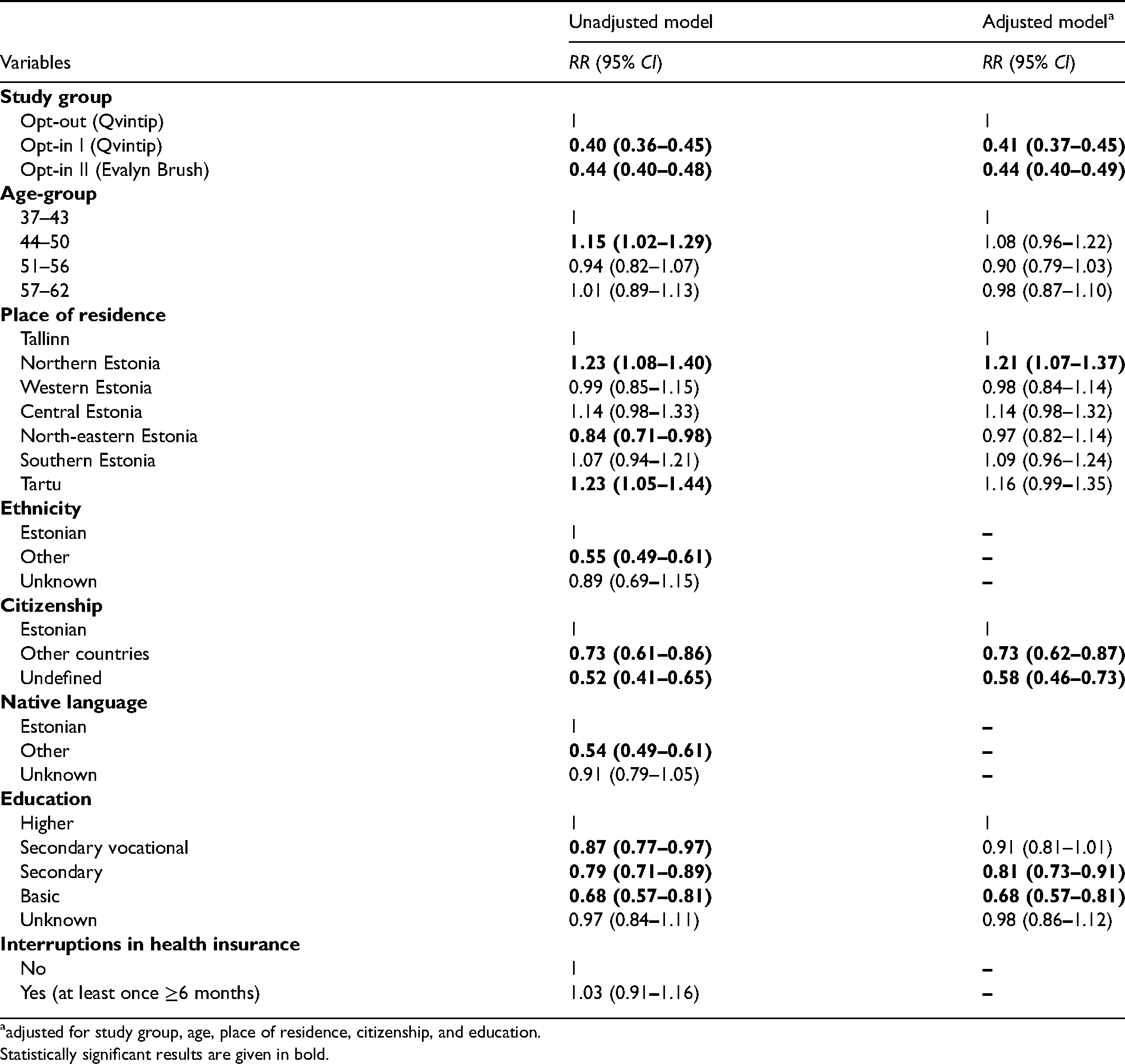
adjusted for study group, age, place of residence, citizenship, and education.
Statistically significant results are given in bold.
User experience evaluation and willingness to use self-sampling in the future
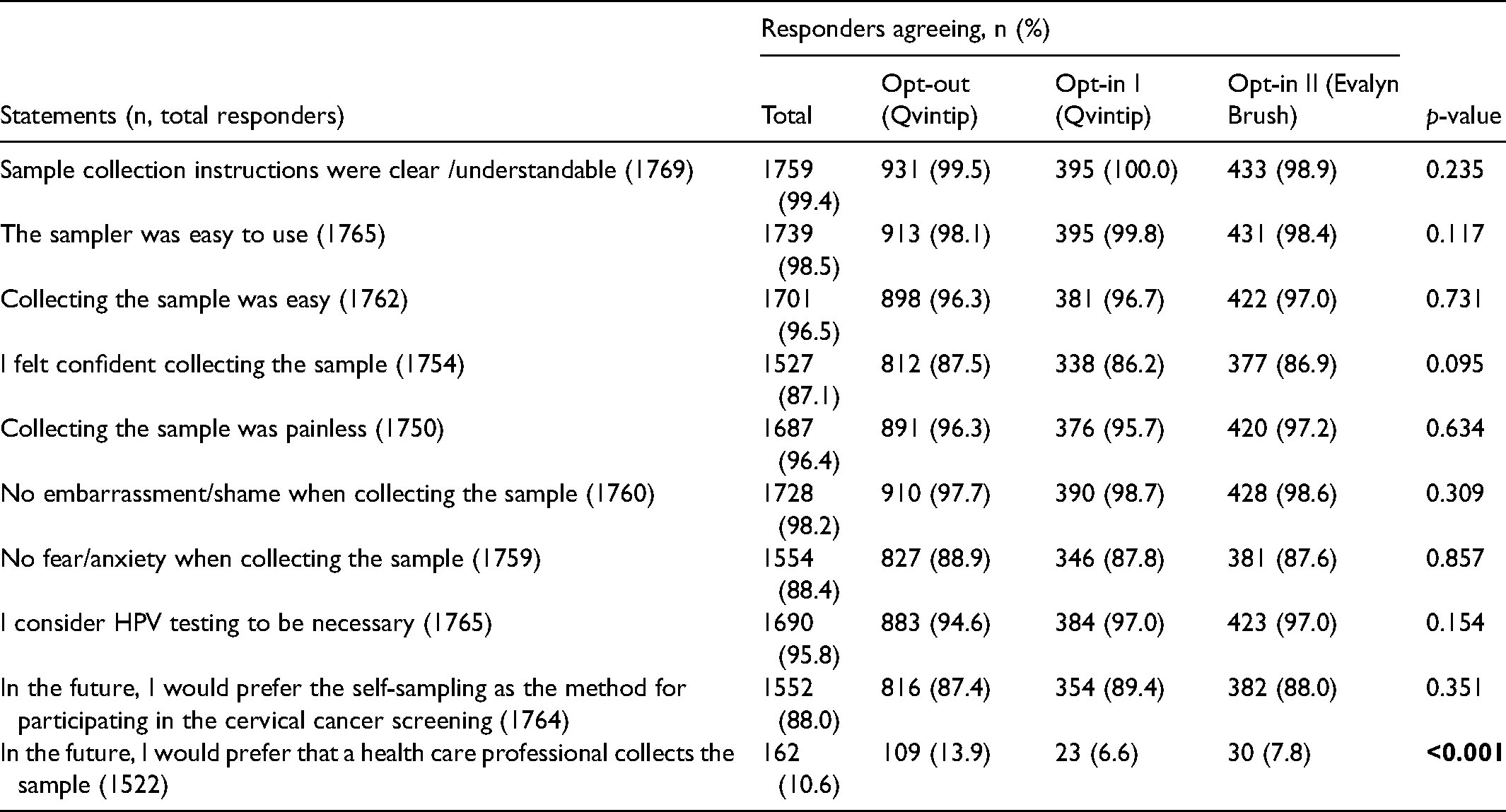
Out of 1903 women who sent the self-sample for analysis, 1857 (97.6%) also filled in the questionnaire. The analysis of user experience is based on their responses. It shows that self-sampling was well accepted: 99.4% agreed that the sampling instructions were clear and for 98.5% the self-sampling device was easy to use; 98.2% did not feel any embarrassment and 87.1% felt secure during the sampling procedure; 88% preferred self-sampling as a screening method in the future. The user experience did not differ among the study groups (Table 3). As positive aspects of self-sampling, many women considered it important that testing could be performed at a suitable time and place (73.2%), there was no need to go to the doctor (57%), and it offered privacy (46.2%). Additionally, it was noted that self-sampling was a quick, convenient and painless method that saved time; it could be done in a safe environment; it was suitable for people with reduced mobility; and a preferable testing method due to the severe acute respiratory syndrome coronavirus 2 pandemic.
Experience with human papillomavirus self-sampling.
HPV test results
The overall hrHPV positivity rate in the study was 10% (n = 191). Among the positive results, HPV16 was positive in 44 cases, HPV18 in nine cases, HPV45 in 13 cases, HPV31/33/52/58 in 50 cases, and HPV35/39/51/56/59/66/68 in 91 cases (some women tested positive for more than one hrHPV type).
Discussion
In this randomised intervention study in Estonia, HPV self-sampling was well accepted by long-term non-attenders in organised cervical cancer screening, with a large proportion of women who participated willing to use it to participate in cervical cancer screening in the future. These results are similar to findings from several other countries.8,14,17
This was the first attempt to use HPV self-sampling in Estonia among a random sample of long-term non-attenders in cervical cancer screening, and the participants were randomly allocated to study groups. Different outreach methods and self-sampling devices were tested.
The strengths of the study are the randomised design and the sample size, and the fact that women without health insurance who had not previously had the option of free-of-charge testing were included. The weaknesses of the study were the inability to check individually which invitations reached the target population in each study group, and that the study population might have differed from regular screening attenders; so, the results are not generalisable to the screening programme.
No comparison between self-collected and physician-collected samples was used, as we assumed no significant difference regarding the sensitivity for cervical intraepithelial neoplasia (CIN2 + or CIN3 + ) and specificity of hrHPV testing for self- versus clinician samples and Evalyn versus Qvintip. 18 Women’s level of confidence in test accuracy was not assessed in the questionnaire.
The participation rate was notably higher in the opt-out group. Participation in our study also depended on place of residence, citizenship, and education, but did not depend on age, and a vast majority of women in the opt-in groups were able to place the order via the website indicating good information technology (IT) literacy among the target population. Although women‘s preferences regarding the self-sampling device have varied in other trials,18–20 we found no difference in user preferences between Qvintip and Evalyn.
Participation rates in the opt-in groups of our study might have been underestimated, as it is difficult to check how many e-mails in these groups did not reach the study participants. Approximately 50% of electronic letters were not opened by the study participants. We do not know the reason for this. In the opt-out group, 3.3% of packages were returned due to wrong address, lack of mailbox, etc. Our results nevertheless confirmed that either opt-out or opt-in for HPV self-sampling is a possibility to include long-term non-attenders in screening, whose cervical cancer risk is higher than among women who regularly attend screening. 21
Higher participation rates among women with higher education have been noted in other studies as well. 22 Higher acceptance of self-sampling in Northern Estonia could be explained by a higher proportion of women with higher education in this region. In line with other studies, women with non-Estonian citizenship had a lower participation rate. 23
As a follow-up, we aim to collect data about women who tested positive for hrHPV in our study. Several studies have shown that the majority of women who have been underscreened but who tested HPV positive from a self-obtained sample will visit a clinic for follow-up diagnosis and management. 8
The study results might not reveal the real effect of HPV self-sampling on participation rates in cervical screening, because regular participants might differ from long-term non-attenders. As the next step, self-sampling will be piloted in the organised screening programme to identify optimal dissemination strategy and provide data for a cost-effectiveness analysis. The opt-out strategy has proved to be more effective in increasing participation rates, but the potential cost-effectiveness is a key consideration. 24 There is still a need to increase participation among women with lower education and non-Estonian ethnicity and citizenship, and relevant qualitative studies should be planned.
While implementing HPV self-sampling in organised cervical cancer screening, there will also be a need to collect the unused self-sampling device kits to reduce the ecological footprint of the programme.
Conclusions
The study demonstrates the feasibility of HPV self-sampling in Estonia. HPV self-sampling proved to be acceptable to long-term non-attenders in the cervical cancer screening programme. A vast majority of participants found it to be convenient and easy, and would like to use it in the future as part of organised screening. Participation rates in the opt-out group were higher than in the opt-in groups. There was no difference in the user experience between devices.
Supplemental Material
sj-docx-1-msc-10.1177_09691413211052499 - Supplemental material for Human papillomavirus self-sampling for long-term non-attenders in cervical cancer screening: A randomised feasibility study in Estonia
Supplemental material, sj-docx-1-msc-10.1177_09691413211052499 for Human papillomavirus self-sampling for long-term non-attenders in cervical cancer screening: A randomised feasibility study in Estonia by Piret Veerus, Reeli Hallik, Jaak Jänes, Kai Jõers, Keiu Paapsi, Kaia Laidra and Kaire Innos in Journal of Medical Screening
Footnotes
Acknowledgements
We thank every woman who participated in the study and the clinical staff at the clinics who helped us to conduct this study.
Funding
The study was funded by the Estonian Research Council grant No PRG722 and by the Ministry of Social Affairs. The Estonian Health Insurance Fund paid for all additional investigations, including for participants without health insurance.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by the Eesti Teadusagentuur (Grant no. PRG722).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
