Abstract
Objective
The performance of Cobas4800 cycle threshold value (Ct-value, reflecting viral load) combined with human papillomavirus (HPV) 16/18 genotyping was explored as a method of risk stratification to triage patients after primary HPV screening of self-collected samples.
Methods
The Chinese Multi-site Screening Trial database was reviewed, with focus on self-collected samples, using the results of Cobas4800 HPV assay. Quartiles of Ct-values of each genotype were used for grouping and developing screening algorithms. Diagnostic accuracy for paired comparisons between algorithms was obtained using McNemar’s test.
Results
A total of 10,498 women were included. The Ct-values of HPV16 and other high-risk HPV were inversely correlated with the severity of cervical lesions (p < 0.001). Risks for cervical intraepithelial neoplasia (CIN2+/CIN3+) were significantly stratified by Ct-values from channels detecting HPV16 and other high-risk HPV types. “HPV with HPV16/18 and reflex Ct <33.7” (algorithm G) achieved a favorable sensitivity to “HPV with atypical squamous cells of undetermined significance or worse (≥ASCUS)” (81.9% vs. 70.1% for CIN2+, p < 0.001), a comparable sensitivity to “HPV with HPV16/18 reflex cytology ≥ASCUS” (81.9% vs. 81.3% for CIN2+, p > 0.05), and resulted in a slightly lower specificity than the latter two algorithms (92.6% vs. 97.4% and 95.4% respectively for CIN2+, p < 0.05). However, algorithm G achieved a comparable sensitivity to HPV testing alone for CIN3+, and reduced the colposcopy referral rate from 13.7% for HPV testing alone to 8.4%.
Conclusions
HPV viral loads reflected by Ct-values are associated with the severity of cervical lesions. Ct-values with an appropriate cut-off of 33.7, combined with HPV16/18 genotyping, represent a promising triage of HPV-positive women particularly for self-collected samples.
Introduction
Cervical cancer screening programs have been proved effective in reducing incidence and mortality of cervical cancer and precancer worldwide. 1 Human papillomavirus (HPV) testing provides a more sensitive method for the detection of cervical lesions than cytology, and detects disease earlier.2–4 Moreover, HPV testing on self-samples has proved efficient and convenient for cervical screening, and significantly increases the coverage of cervical screening, particularly in low-resource areas. 5 However, the management of HPV-positive patients remains challenging due to the relatively low specificity of HPV testing in general. 6 The most common triage methods are cytology and genotyping. 6 However, high-quality cytology is often unavailable in low-resource areas. Given the differing risks associated with different genotypes,7–9 partial genotypes such as HPV16 and HPV18 are widely used to guide clinical management of HPV-positive women.4,6
HPV results and related viral load could be obtained from standard output and acquired at the same time by HPV assay on self-samples. However, the role of viral load with regard to cervical lesions remains controversial.7,10 A prior study from our group found that HPV viral load detected by HC-2 assay was associated with cervical lesion grades, potentially having value for the triage of HPV-positive women. 11 However, the lack of type-specific data in the study limited proof of value for secondary screening. A recent study shows that making more stringent cycle threshold (Ct, reflecting viral load) cutoffs on the three channels in Xpert HPV assay improves specificity with only modest losses in sensitivity. 12 Another recent study has confirmed that HBRT-H14, a RT-PCR assay, was as sensitive but less specific than HC2 for detecting cervical precancer at the predefined cutoff (Ct-value ≤40). 13 However, HBRT-H14 fulfilled international accuracy criteria for cervical cancer screening when lowering the cutoff of Ct-value for HPV16, HPV18 and other high-risk (hr) HPVs from the original values of 40 to 37, 35, and 35, respectively. 13 Cobas4800 HPV assay is a PCR assay and has been approved by the FDA; we can use the Ct-value as a rough approximation of the quantity of HPV DNA present or viral load.12,14 In the present study, using Cobas4800 Ct-values as a reflection of the viral load, we reviewed the Chinese Multi-site Screening Trial (CHIMUST) database, and focused on self-collected samples. We then explored the performance of viral load (reflected by Ct-value) as a method of risk stratification to triage patients after primary screening.
Methods
We reviewed the data from CHIMUST, a multisite, population-based, cross-sectional study that was conducted in six sites in China from August 2016 to January 2018 (Trial Registration Number: ChiCTR-EOC-16008456). A total of 10,885 nonpregnant women, aged 29 to 60, with an intact uterus, no history of pelvic radiation, and no cervical cancer screening within three years were enrolled. All the self-collected and clinician-collected samples were tested with multiple PCR-based hrHPV assays including Cobas4800 and SeqHPV (BGI Shenzhen, Shenzhen, China). The study protocol was approved by the Ethics Committee from Peking University Shenzhen Hospital (PUSH), Shenzhen, China (No. 2016001). All participants signed the informed consent.
Self-collected samples were prepared for the Cobas4800 assay (Roche, USA) per the manufacturer’s instructions. Cobas4800 assay detects 14 types of hrHPV in three channels: HPV16, HPV18, and 12 other types of hrHPV (Other hrHPV). Output from the assay includes a Ct-value from each of these three channels. Ct-value is the number of cycles needed until DNA amplification occurs exponentially in a PCR assay, and is inversely correlated with the log amount of target in the sample.14,15 Therefore, a high Ct-value means a low viral load (more cycles needed to become positive) and a low Ct-value indicates a high viral load. 12 Clinician-collected samples were prepared for cytological testing (ThinPrep, Hologic). The cytology slides were reported according to the Bethesda classification system by senior cytopathologists from PUSH. 16
Patients with HPV-positive results from any HPV assay in CHIMUST (Cobas4800, SeqHPV on either self-collected or clinician-collected samples) were referred for colposcopy-directed biopsy. Cytology was used for research purposes and not for colposcopy-referred requirements. 17 Biopsies under colposcopy were performed according to the Preventive Oncology International (POI) protocol. 18 All histology slides were interpreted blindly by PUSH pathologists as Normal, cervical intraepithelial neoplasia (CIN) 1, CIN 2, CIN 3, adenocarcinoma in situ (AIS), and cancers. Women with cytology ‘atypical squamous cells of undetermined significance or worse’ (≤ASCUS) and negative hrHPV results were considered to have a minimal risk of CIN and were classified as within normal limits. 18
Hierarchical typing results were grouped as HPV16, HPV18, Other hrHPV, and negative. CIN3+ and CIN2+ were used as the study endpoints. Spearman’s rank correlation coefficient (r) was used to test for linear trends between Ct-values and the severity of the cervical abnormality. Quartiles of Ct-values of each genotype were used for grouping and developing screening algorithms. A multivariate logistic regression model was used to estimate the relative risks (RRs) and 95% confidence intervals (CI) for CIN2+/CIN3+ according to genotypes, multiplicity of genotypes, and Ct-values, and adjusted for age at enrollment. Ct-values of HPV16 or HPV18 were also adjusted for related coinfection. p-Values of diagnostic accuracy for paired comparisons between algorithms were obtained using McNemar’s test. Analyses were carried out using SPSS software 24.0 (IBM, Chicago, IL, USA). All statistical tests were 2-sided, and p < 0.05 were considered statistically significant.
Results
Out of 10,885 women offered self-sampling for hrHPV testing in the study, 265 were excluded because of HPV-positive results without histology, 62 because HPV negative on all assays and cytology ≥LSIL therefore without histology, 60 women because invalid cytology or invalid HPV, leaving 10,498 cases available for analysis (Figure 1). The mean age of the study group was 44 years (range: 29–60 years). The positive rates of hrHPV, HPV16, HPV18, and Other hrHPV were 13.7%, 2.4%, 0.9%, and 10.5%, respectively. Of the total 10,498 women, 5 women had a cervical carcinoma, 49 were diagnosed with CIN3, 90 with CIN2, 159 with CIN1, and 10,212 as Normal.
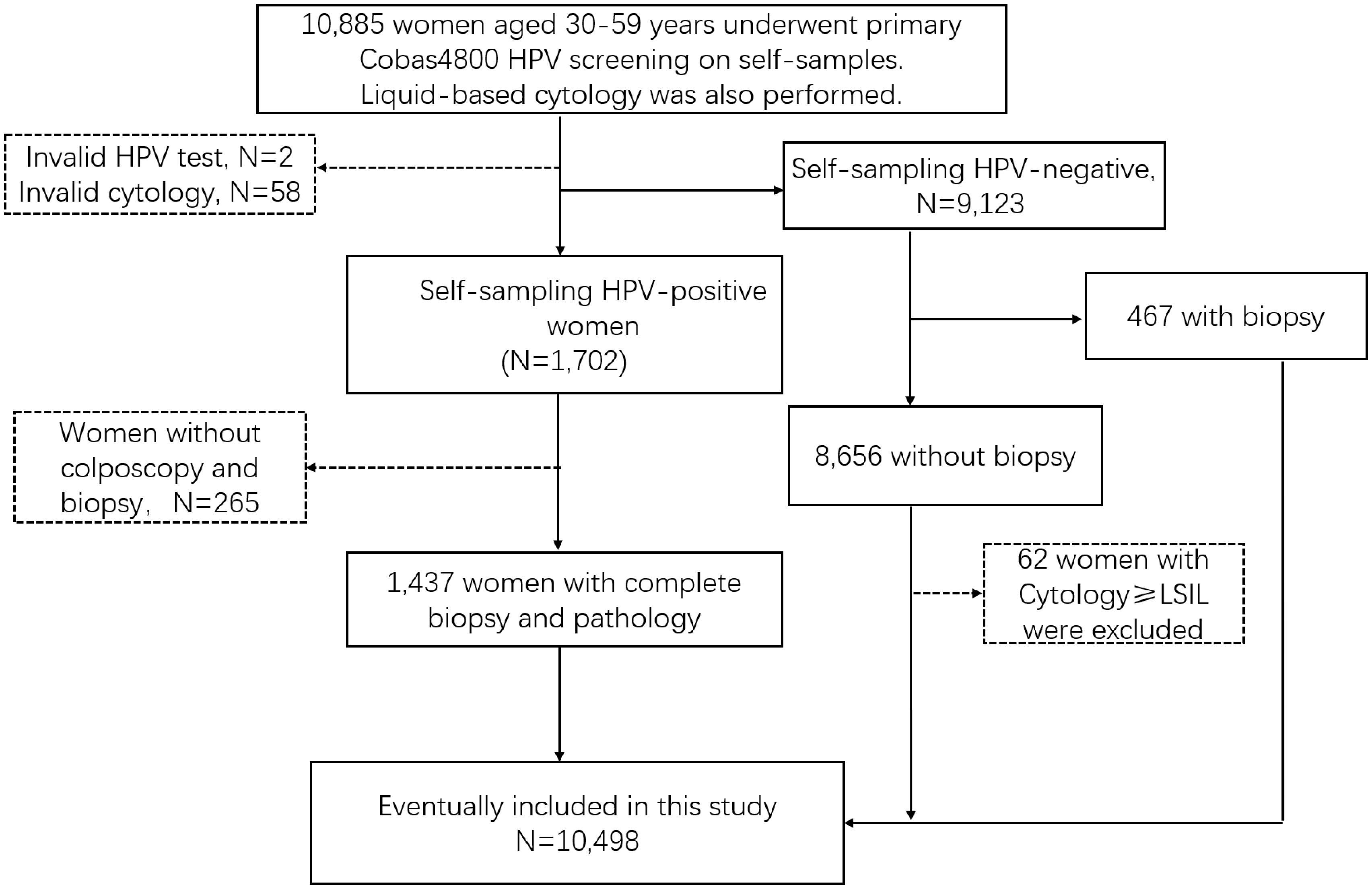
Flowchart of the study population inclusion.
The Ct-value of HPV16 and Other hrHPV significantly decreased along with the upgrading of the cervical histological severities (HPV16, r = −0.31, p < 0.001; Other hrHPV, r = −0.17, p < 0.0001, Figure 2(a)), and the cytological severities (HPV16, r = −0.29, p < 0.0001; Other hrHPV, r = −0.23, p < 0.0001, Figure 2(b)). The Ct-values of HPV18 showed no significant correlation with the histological severities (r = 0.06, p = 0.620, Figure 2(a)) or the cytological severities (r = −0.14, p = 0.169, Figure 2(b)).
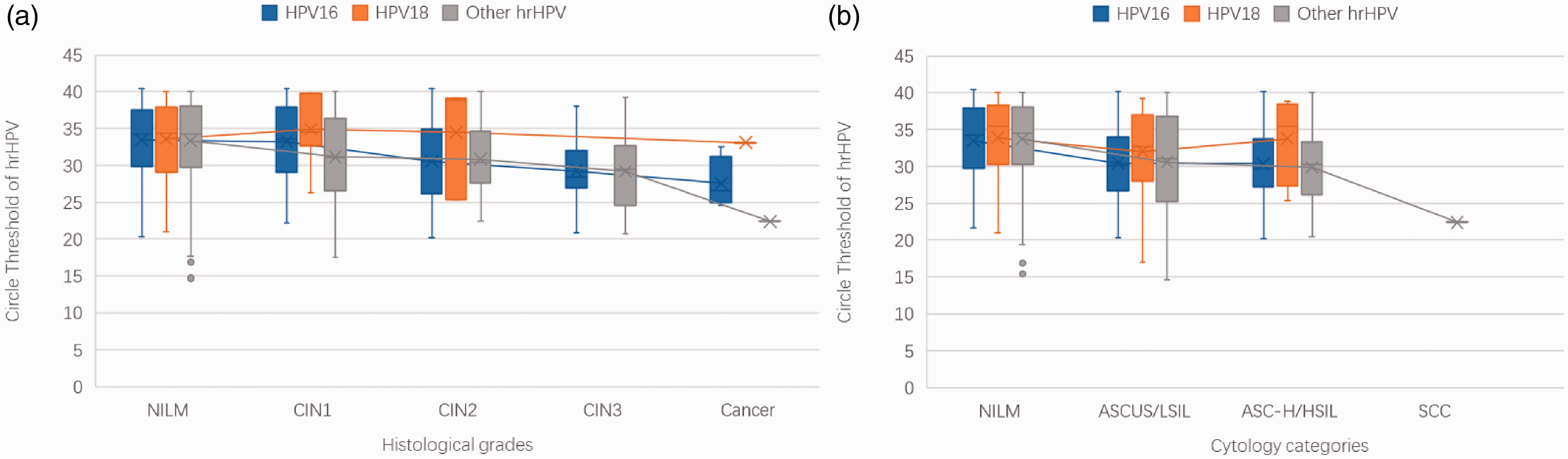
Boxplots of Ct-values and severity of cervical histological grades and cytologic categories. Boxes depict 25% to 75% percentile; middle line depicts the median; Whisker: min. or max. Outliers are shown (gray circle). The blue box indicates the Ct-values of HPV16,the yellow box indicates the Ct-values of HPV18, the gray box indicates the Ct-values of Other hrHPV. Spearman’s rank correlation coefficient (r) was used to test for linear trends between Ct-values and the severity of the cervical abnormality. (a) Ct-values of HPV16 (n = 255, r = −0.31, p < 0.001), HPV18 (n = 80, r=0.06, p = 0.620) and Other hrHPV (n = 1102, r = −0.17, p < 0.0001) among various histological grades; (b) Ct-values of HPV16 (n = 254, r = −0.29, p < 0.0001), HPV18 (n = 80, r = −0.14, p = 0.169) and Other hrHPV(n = 1099, r = −0.23, p < 0.0001) among different cytology categories. Four cases of atypical glandular cells were excluded.
As Table 1 shows, the RRs of HPV16/18 vs. Other hrHPV for CIN2+/CIN3+ were 4.4 (95%CI, 3.0–6.3) and 9.0 (95%CI, 4.9–16.7) respectively, which were statistically significant (p < 0.001). With the decrease of Ct-values, the RRs for CIN2+/CIN3+ were significantly increased among women positive for HPV16 (ptrend <0.0001 for CIN2+/CIN3+), similar results were found among women positive for Other hrHPV (ptrend <0.0001 for CIN2+/CIN3+), while there was no such significance among women positive for HPV18.
Relative risk (RR) of each HPV type and related Ct-values for CIN2+/CIN3+.
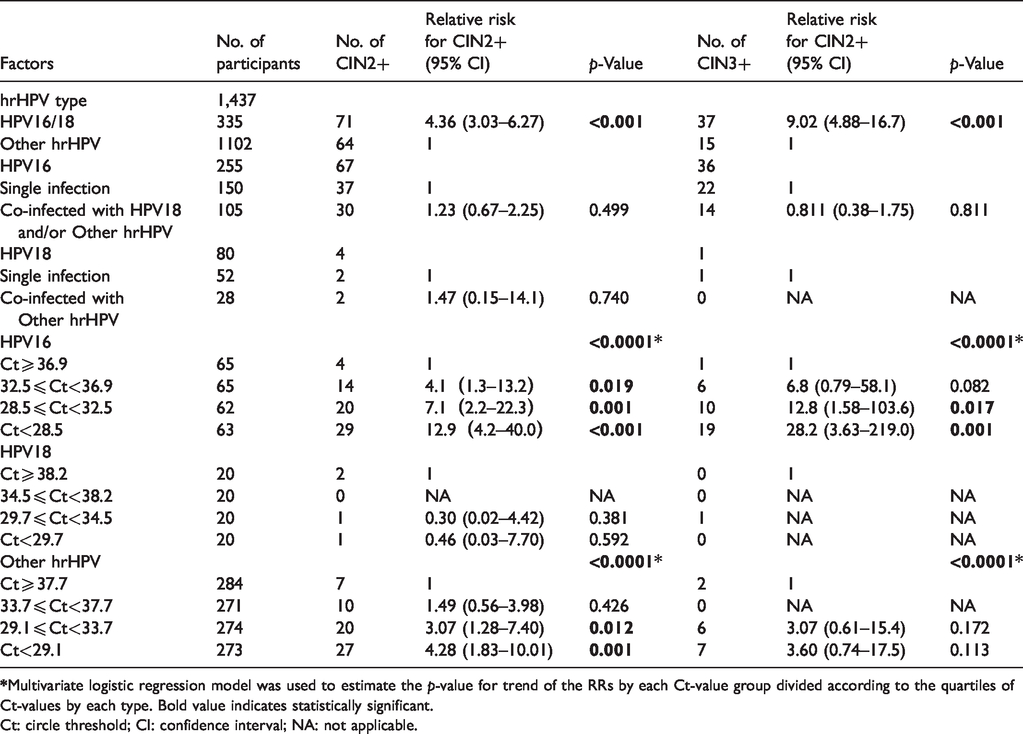
Ct: circle threshold; CI: confidence interval; NA: not applicable.
Detailed results of the eight strategies are presented in Table 2. HPV screening triage with cytology ≥ASCUS (C) achieved a sensitivity of 96.3% and a specificity of 97.0% for CIN3+, with 7.1 colposcopies needed to detect one case of CIN3+. The strategy of “genotyping HPV16/18 and reflex cytology ≥ASCUS” (E) showed favorable sensitivities compared with algorithm C (81.3% vs. 70.1% for CIN2+, p <0.001; 96.3% vs. 96.3% for CIN3+, p = 1.000), while resulted in slightly lower specificities than algorithm C.
Performance of different screening algorithms for CIN2+/CIN3+ detection (n = 10,498).
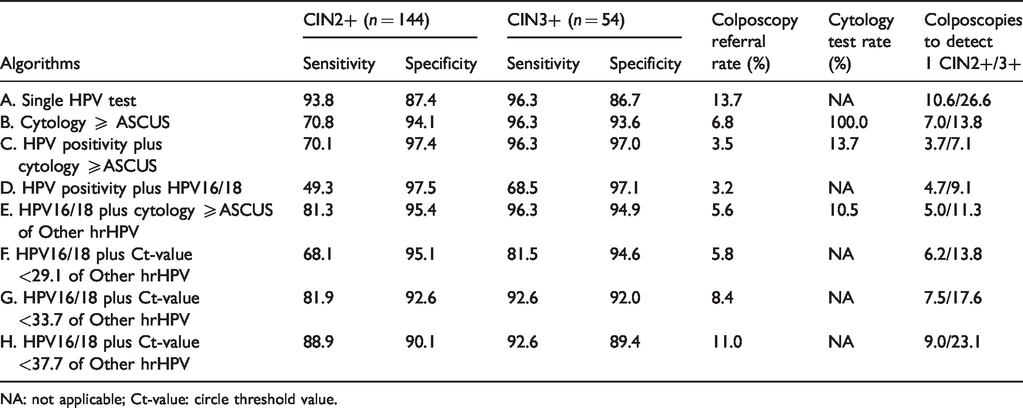
NA: not applicable; Ct-value: circle threshold value.
Algorithm G refers HPV16/18 positives with reflex Ct-values <33.7 of Other hrHPV positives to colposcopy, yielding a favorable sensitivity to algorithm C and a comparable sensitivity to algorithm E, while leading to a slightly lower specificity than algorithm C and D. However, Algorithm G shows a comparable sensitivity for CIN3+, and reduces the referral rate to 8.4% compared to that of 13.7% for HPV testing alone. HPV16/18 with reflex Ct-values <29.1 leads to a worse sensitivity/specificity than algorithm E. HPV16/18 with reflex Ct-values <37.7 (algorithm H) had a favorable sensitivity, but at the expense of lower specificity compared with 33.7 as the cut-off, leading to a high colposcopy referral rate of 11.0%.
Discussion
Large population-based cervical screening in low-resource settings could be achieved by promoting self-collection and centralized high-throughput, low-cost testing by PCR-based HPV tests.5,18 However, high positive rates of HPV on self-samples (13.7% in this group) not only lead to overtreatment, as most infections are transient and only a subset of HPV-positive women harbor clinically relevant disease, but also cause mental burden for the women involved, 19 so proper triage is urgently needed to overcome the challenge. 6 Given that cytology cannot reliably be performed on self-collected samples, triage in this case requires an additional hospital visit for taking a pap smear, which may result in significant loss to follow-up.
The Cobas4800 HPV assay is the first FDA-approved HPV test for primary cervical cancer screening, 20 and it is worth discussing how to extend its application value through providing type-specific viral load reflected by Ct-value. When it comes to individual genotypes, the Ct-value of HPV16 and Other hrHPV significantly decreased with the severity of cervical abnormalities, indicating that viral load reflected by Ct-values is an important factor of cervical carcinogenesis. Notably, the trend was not seen for HPV18, possibly due to the low number of cases, and further studies involving more HPV18-positive women are warranted to address this issue. A recent study confirmed that reverse Ct-values for the three channels in Xpert HPV assay were significantly predictive of CIN2+. 12
Risk stratification based on HPV genotyping and viral load has important implications for personalizing triage plans, particularly HPV-based screening on self-samples. 7 By using the quartiles of Ct-values, Ct-values of HPV16 and Other hrHPV add significant risk stratification for CIN2+/CIN3+, indicating their potential role in triage after primary HPV screening. Since 71% of cervical cancers are caused by HPV16/18, 21 and noting the significantly higher risk of HPV16/18 vs. Other hrHPV for high-grade lesions, our findings support the guidelines in referring all HPV16/18-positive women to colposcopy. 4 However, HPV16/18 cannot be used as sole triage because of the low sensitivity (49.3% and 68.5% respectively for CIN2+/CIN3+). The optimal management of HPV-positive women, particularly those infected with Other hrHPV, to maximize disease detection and minimize colposcopy rates, however, is still unclear. 10
With all screening tests, there is an inevitable trade-off between sensitivity and specificity. Specificity can be improved if reductions in sensitivity can be tolerated. 12 Cytology (≥ASCUS) triage and the FDA-approved protocol (HPV16/18 and reflex cytology ≥ASCUS) are the two most common triages for HPV-positive women.4,6 In this study, the “HPV16/18 with reflex Ct-value <33.7” triage algorithm (G) significantly improved the specificity and reduced the referral rate compared with HPV screening alone, and achieved a similar sensitivity to the FDA-approved algorithm as well as a favorable sensitivity compared with cytological triage, thus resulting in a satisfactory clinical accuracy; but algorithm G also simplifies the screening process as does not require an additional test. In a recent cohort study, the 10-year cumulative incidence rate of CIN2+ among Other hrHPV-positive women was significantly stratified by cytology ≥ASCUS (2.0% vs. 34.6%), viral load cutoffs at 10 relative light units/cutoff (RLU/CO, 5.1% vs. 27.2%), and at 100 RLU/CO (11.0% vs. 35.5%), but not by visual inspection with acetic acid (VIA, 19.1% vs. 19.0%). 10 From these results and the current study, we conclude that in fully developed settings where high-quality cytology can be guaranteed cytology as a triage should be retained. However, in low-resource areas where high-quality cytology cannot be guaranteed or cytology screening is lacking, the Ct-value application is a potential alternative. That is, HPV16/18 with reflex Ct value <33.7 could achieve a good balance between disease detection and colposcopy referral, and be an alternative triage for hrHPV-positive women.
There are many advantages to molecular tests either as primary screening or triage: they are objective, highly reproducible, suitable for self-samples, and permit high-throughput testing. 22 Moreover, failure of screening programs is largely due to the multistep process required by the traditional model. If amenable to self-sampling, the molecular triage test can be used for HPV-positive women without requiring a second visit for a clinician-sample. Information on quantitative Ct-values could be used to improve the specificity of HPV testing while also avoiding additional screening tests at the second stage of HPV screening, 12 further decreasing expenditure and the rate of loss to follow-up compared with cytological triage. With a combination of HPV primary screening and triage of viral load reflected by Ct-values, an entirely molecular screening approach may be achieved. Therefore, the present finding might widen utilization of the Cobas4800 HPV assay and benefit countries with comparable settings to China.
To the best of our knowledge, this is the first study to evaluate the clinical use of Cobas4800 Ct-values in the management of HPV-positive patients using self-samples. A major strength lies in its large size, from six diverse provinces reflecting the demography of China, which rendered the results applicable to the general population in various settings. In addition, women found positive in any HPV assay, whether from self-samples or clinician-samples, were referred to colposcopy according to standard POI protocol, 18 which reduced the risk of missed diagnosis and confirmed accuracy. Several limitations of this study should also be noted. Firstly, as a cross-sectional study, viral load might have limited ability to infer etiology of cervical lesions and capture disease missed by colposcopy. However, a longitudinal study revealed a similar role of viral load in stratifying risk of HPV-positive women. 10 Secondly, Pap cytological testing at CHUMUST may be more sensitive and specific than that performed at other health facilities. Consequently, the performance comparisons between Ct-value and Pap cytology may differ in other settings. In addition, the results above only apply to Cobas4800 assay; other PCR-based platforms would still need to validate this association between their Ct-values and lesion severity, as well as their clinical performance by adjusting corresponding Ct-values. In the future, more large-scale long-term prospective cohort studies or population trials from various settings or regions are required to confirm our results and promote the potential utility of Ct-values of different PCR-based HPV assays in clinical practice.
Conclusion
Type-specific viral loads (reflected by the corresponding Ct-values) are associated with the severity of cervical lesions and useful for risk stratification. Moreover, the use of Ct-value with an appropriate cut-off of 33.7 for triage of “Other hrHPV”-positive women, in combination with HPV16/18 genotyping, serves as an attractive molecular triage approach in HPV screening, particularly for self-collected samples, and may be an alternative triage for hrHPV-positive women where high-quality cytology is inaccessible. Our findings indicate that a transition towards full molecular self-screening in HPV-based cervical screening programs is feasible.
CHUMUST team
Lead author, Ruifang Wu, E-mail: wurf100@126.com, Department of Obstetrics and Gynecology, Peking University Shenzhen Hospital, and Shenzhen Key Laboratory on Technology for Early Diagnosis of Major Gynecological Diseases, PR China. Jerome L. Belinson, Preventive Oncology International, Inc. and the Cleveland Clinic, Cleveland, USA. Lihui Wei, Department of Obstetrics and Gynecology, Peking University People's Hospital, PR China. Yan Liu, The Northern Division of Huashan Hospital Affiliated to Fudan University, PR China. Xianzhi Duan, Hua Jin, Mongolia Wushenqi People’s Hospital, PR China. Wei Zhang, Zhongnan Hospital Affiliated to Wuhan University. Bin Shi, The Second Hospital of Hebei Medical University, PR China. Bin Yang, The Cleveland Clinic, Cleveland, USA. Hui Du, Ruizhen Li, Guixiang Wang, Lijie Zhang, Chun Wang, Hongxue Luo, Xia Huang, Danhong Huang, Department of Obstetrics and Gynecology, Peking University Shenzhen Hospital, and Shenzhen Key Laboratory on Technology for Early Diagnosis of Major Gynecological Diseases, PR China. Xinfeng Qu, Sanming Project of Medicine in Shenzhen, Peking University Shenzhen Hospital, PR China. Jing Mei, Medical Records and Statistics Room, Peking University Shenzhen Hospital, PR China.
Footnotes
Acknowledgements
We really appreciate Ms Duan LF for her advice on editing this article and Ms Chen ZP for the data collection of Ct-values. They sincerely thank all members of the CHUMUST team for their meticulous work and all the women participated in this study.
Authors’ contribution
Song FB, Du H, Huang X, Wang C collected and interpreted the data and drafted the article, Wu RF, Belinson JL, Wei LH, Song FB, Qu XF contributed to design the study and revised the article. All authors read and approved the final article.
Availability of data and materials
The data sets used and analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval and consent to participate
All the authors state that manuscripts reporting studies involving human participants got the ethics approval and consent from the Ethics Committee, Peking University Shenzhen Hospital, Shenzhen, Guangdong, China (No. 2016001). All the participants provided informed consent to take part in our study, and the consent obtained from the participants was written.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported by Shenzhen Health Family Planning Commission, Shenzhen, PR China (No. SZLY2017005); Sanming Project of Medicine in Shenzhen (SZSM201412010); The Governmental Funds for Shenzhen Leading Gynecological Subject (Science and Education of Shenzhen Health [2018]61); China International Exchange and Promotion Association for medical and Healthcare.
