Abstract
Background
Although lung cancer screening (LCS) has been proven effective in reducing lung cancer mortality, it is associated with some potential harms, such as false positives and invasive follow-up procedures. Determining the time to next screen based on individual risk could reduce harms while maintaining health gains. Here, we evaluate the benefits and harms of LCS strategies with adaptive schedules, and compare these with those from non-adaptive strategies.
Methods
We extended the Lee and Zelen risk threshold method to select screening schedules based on individual's lung cancer risk and life expectancy (adaptive schedules). We compared the health benefits and harms of these adaptive schedules with regular (non-adaptive) schedules (annual, biennial and triennial) using a validated lung cancer microsimulation model. Outcomes include lung cancer deaths (LCD) averted, life years gained (LYG), discounted quality adjusted life years (QALYs) gained, and false positives per LCD averted. We also explored the impact of varying screening-related disutilities.
Results
In comparison to standard regular screening recommendations, risk-dependent adaptive screening reduced screening harms while maintaining a similar level of health benefits. The net gains and the balance of benefits and harms from LCS with efficient adaptive schedules were improved compared to those from regular screening, especially when the screening-related disutilities are high.
Conclusions
Adaptive screening schedules can reduce the associated harms of screening while maintaining its associated lung cancer mortality reductions and years of life gained. Our study identifies individually tailored schedules that optimize the screening benefit/harm trade-offs.
Introduction
Lung cancer continues to be the leading cause of cancer-related mortality in the US, leading to more deaths than the three most common cancers in the US combined (breast, prostate and colon cancer). 1 This high mortality burden is likely due to the fact that lung cancer is an aggressive disease, with a 5-year survival of about 20%. 2 This can be improved to about 60% if lung cancer is diagnosed when it is still localized; however, currently only about 17% of lung cancers are diagnosed at early stages. 2 Thus, early detection and treatment interventions are still needed to further reduce the lung cancer burden in the US.
Previous lung cancer screening (LCS) trials have concluded that low-dose computed tomography (LDCT) screening is effective in reducing lung cancer mortality.3,4 Both the United States Preventive Services Task Force (USPSTF) and the Centers for Medicare and Medicaid Services (CMS) recommend annual LCS for eligible smokers.5,6 However, LDCT screening is associated with some potential harms, like many other cancer screening interventions. For instance, individuals may have to lose a day of work to attend screening, and experience pre- and post-screen psychological burdens. 7 In addition, the observed false positive rate of LCS varied widely from 5% to 50% across different studies and implementations, 8 and is expected to be in the order of 12% under current management protocol.9,10 In any case, false positive results not only induce a psychological burden, 7 but also lead to unnecessary and potentially invasive follow-up tests, such as biopsies.3,8 Furthermore, complications or even diagnostic deaths from invasive procedures might occur. 3 Hence, when assessing the overall impact from screening, it is necessary to consider both the benefits and harms.
Although USPSTF and CMS both recommend conducting LCS annually, earlier studies have found that individuals with previous negative screens are at a considerably lower risk of lung cancer,11,12 suggesting a longer interval to the next screen, which could potentially reduce the false-positive rate.13,14 These findings provide an opportunity to reduce the costs and potential harms of screening without greatly compromising its benefits, by moving low-risk individuals from the recommended annual program to a less intensive dynamic schedule (adaptive scheduling). In this study, we evaluate the impact of such adaptive protocols selected by a “risk threshold method” on screening benefits and harms, considering not only past screening results, but also an individual's smoking history and other risk factors, including sex and age. Furthermore, as life expectancy at the time of screening affects the potential net benefits of LCS,15,16 we also consider life expectancy when identifying screening schedules, and assessing their performance.
Methods
We adapted the threshold method, initially developed by Lee and Zelen for breast cancer screening, 17 to select LCS schedules for the whole screen-eligible population, specific groups (by sex or by pack-year), and each individual in the study population. We then compared the screening-related health outcomes from adaptive schedules to those from regular (non-adaptive) schedules (triennial, biennial and annual) using the Cancer Intervention and Surveillance Modeling Network (CISNET) Michigan Lung Cancer Natural History and Screening model (MichiganLung).15,16,18,19 This research was conducted using publicly available data with deidentified human subject information and thus exempted by the University of Michigan Institutional Review Board.
The threshold method
The threshold method is a deterministic approach that calculates the probability of being in a preclinical disease state conditioned on past negative screens, utilizing a pre-specific risk threshold to determine when to screen: whenever the probability becomes higher than the pre-specified risk threshold, a screen exam is recommended/given. 17 It was originally developed and applied for breast cancer screening, which we now extended to LCS, with details of the adaptation presented in Appendix Text S1 and model parameters summarized in Table S1.
We used the threshold method to generate LCS schedules under various assumptions. We varied the risk threshold from 0.15% to 0.85%, corresponding to the probability of being in a preclinical state around age 45 and 60 respectively, with 0.05% increments. We first allowed for screening to start at any time, and then constrained it by the 2021 USPSTF minimum age to screen (50 years old) so that starting age would be 50 or later. We then conducted separate analyses where we fixed the starting age of screening at 50 and let the threshold method decide when the next screen would be. We further incorporated life expectancy into the threshold method by adjusting the age-specific lung cancer incidence by life expectancy at each age. We adapted the threshold method to result in increments of one year or more between screens. Detailed description of different extensions and the life expectancy adjustment are provided in the Appendix Text S1 and S2 respectively.
We hereon refer to each scenario using the following acronym: population (Whole, Bysex, Bypky or Individual)_life expectancy (Adjusted or Unadjusted)_starting age (50: fixed or 0: non-fixed)_risk threshold (0.15% to 0.85%). For example, “Individual_Unadjusted_50_0.15%” refers to the individualized scenario without adjusting the individual lung cancer risk for life expectancy, fixing the starting age at 50 and with the risk threshold set as 0.15%.
CISNET Michigan lung cancer natural history and screening model
To obtain and compare screening-related health outcomes for different screening schedules, we used a previously developed and validated lung cancer microsimulation model—the CISNET Michigan Lung Cancer Natural History and Screening (MichiganLung) Model—to simulate lung cancer natural history and clinical and survival outcomes of LDCT screening.15,16,18,19 This model takes discrete screening ages as an input, and conducts screening based on screening eligibility and schedule using sensitivities and specificities consistent with the National Lung Screening Trial (NLST) and Prostate, Lung, Colorectal and Ovarian cancer screening trial (PLCO), and the Lung-RADS nodule assessment and management protocols.9,20,21 Consistent with previous studies, a negative screening result in our model corresponds to Lung-RADS score 1 or 2, whereas a positive screening result refers to Lung-RADS score 3 or 4.9,21,22 Furthermore, we utilized the 2021 USPSTF screening eligibility criteria, being smokers aged 50 to 80 with a smoking history of 20 pack-years or more, who have not quit or quit smoking no more than 15 years ago. To reflect clinical guidelines, as described above, when the resulting screening schedules fall outside of screening eligible ages, screening is given only at the scheduled ages within the eligible age range. Model parameters (description and references) are presented in Appendix Table S2.
Population
We used the CISNET Smoking History Generator (SHG)23–25 to generate smoking histories of 1 million men and 1 million women from the 1960 birth cohort, which are representative of smoking patterns of the current screen-eligible population. The simulated individual smoking histories contain information including smoking starting age, smoking quitting age, and the number of daily cigaretes smoked per age, which allows us to determine screening eligibility for each individual.
Utilities and disutilities
We conducted analyzes adjusting for the changes in quality of life from clinical procedures and lung cancer stages. The age-based quality of life utilities, varied by age, sex and lung cancer status, were obtained from a recent cost-effectiveness study of LCS. 19
We also quantify the screening burdens using disutilities—the decrements in quality of life from anxiety and discomfort. The MichiganLung model simulates detailed follow-up procedures after a positive screen, using probabilities obtained from the NLST. 26 We quantified the burdens from screening and follow-up tests by multiplying the number of each procedure with its disutility. We further quantified the burdens of follow-up test complications by multiplying the number of each procedure with its corresponding complication rate and disutility. The screening disutilities were derived from published decision analysis, 15 and probabilities of complications by procedure type and screening result (false/true positive) were obtained from the NLST published data 3,26 (Appendix Table S3).
Study outcomes
Screening-related health outcomes included lung cancer deaths (LCD) averted, life years gained (LYG) and quality adjusted life years (QALYs) gained compared with no screening. We further quantified the benefits and harms together using one measure: the discounted QALYs, which is the total QALYs minus the total harms (disutilities) from screening procedures. We also evaluated the balance of harms and benefits using the number of false positives per lung cancer death averted.
We further varied screening-related disutilities by multiplying the baseline values with a factor ranging from 2 to 6 to examine changes in patterns of discounted QALYs due to increased screening burdens, consistent with the previous modeling study evaluating the impact of higher disutilities on the effectiveness of LCS. 15
Data envelopment analysis
Efficiency of all scenarios was assessed using data envelopment analysis (DEA), a nonparametric linear programing method for assessing the efficiency of different strategies, 27 with LCD averted and LYG as the two outputs, and the number of screens as the input.5,21 Higher efficiency scores indicate higher gains in the outputs given a unit of input. DEA was conducted using the “dea” function in the “nonparaeff” package in R (version 4.0.4). 28
Results
The schedules selected by the threshold method were less frequent at younger ages and became annual screening after age 70 across assumptions. However, after adjusting for life expectancy, the screening schedules may become less frequent than annual for older ages. Adaptive schedules were more frequent for males (vs. females), for the 40+ pack-year groups (vs. 20–29 or 30–39 pack-year groups), without adjustment for life expectancy, and with the starting age fixed at age 50 (Tables S5–S7). In addition, individualized schedules generally had fewer screens than other schedules: the most intensive individualized schedule “Individual_Unadjusted_50_0.015%” had 356,646 screens per 100,000 population, which was 68,743 fewer screens per 100,000 population than annual screening.
Figures 1 and 2 show the LCD averted (LCDs averted) and the LYG per 100,000 total population by number of LDCT screens per 100,000 total population respectively. The LCDs averted and LYG increased with the number of screens with diminishing returns. Individualized adaptive schedules (marked as pink circle dots) achieved higher LCDs averted and LYG than any other screening schedules with similar numbers of screens. Schedules selected by pack-year performed slightly better than schedules selected by sex or for the whole screen-eligible population. Biennial and triennial screenings had lower LCD averted than all adaptive screening schedules with similar numbers of screens. In terms of LYG, biennial and triennial screenings had similar LYG as those adaptive schedules selected by either sex or pack-year, but performed worse than individualized schedules.
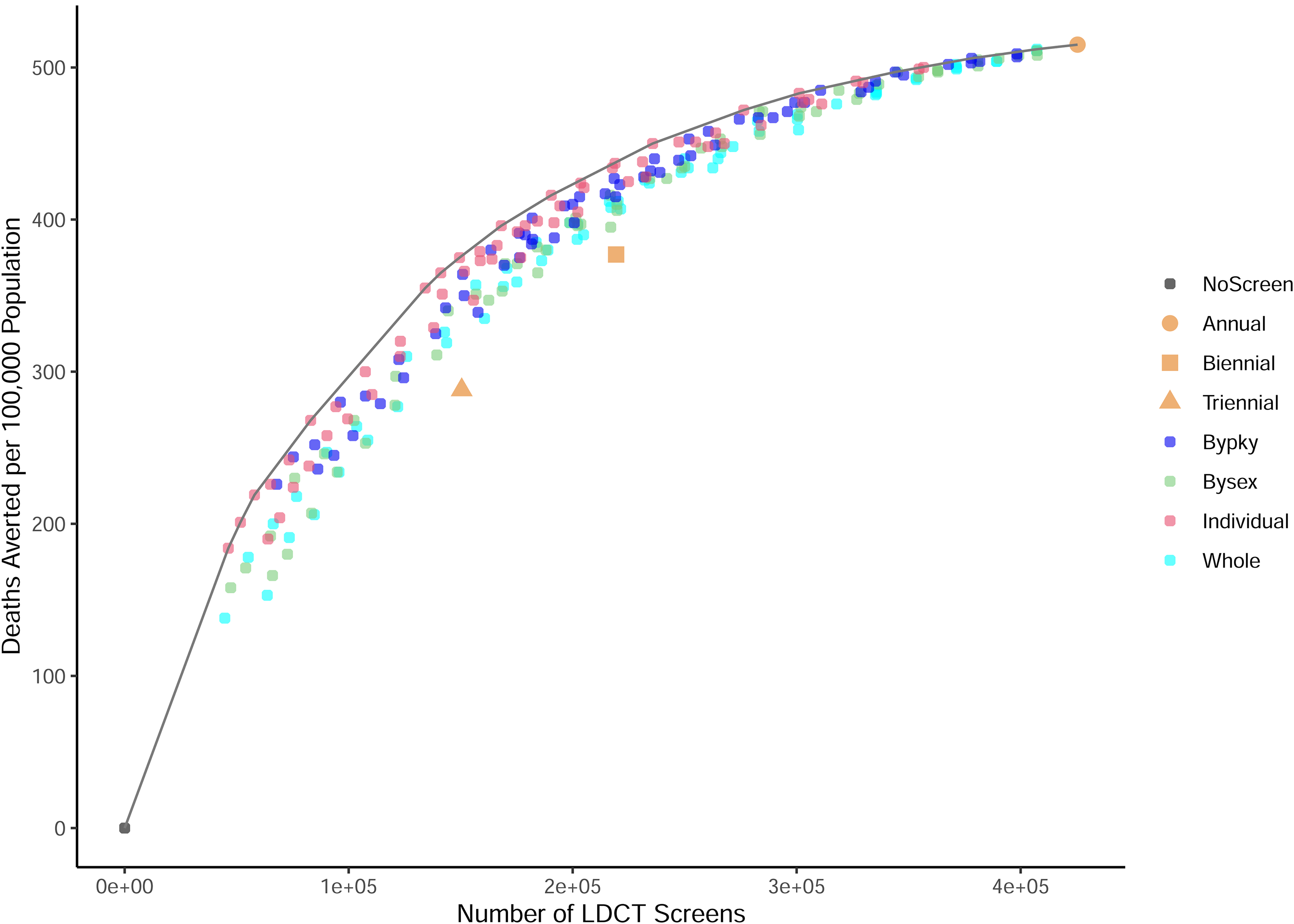
Low-dose computed tomography (LDCT) screening vs. lung cancer deaths averted compared with no screening for the 240 adaptive and 3 regular screening scenarios for the 1960 US birth cohort. The gray curve is the efficient frontier, connecting the dots with the highest lung cancer deaths averted given numbers of screens. Symbols represent regular screening: annual (circle), biennial (square), and triennial (triangle). Smaller circles represent: individualized schedules (pink), by-pack-year schedules (dark blue), by-sex schedules (light green), schedules selected for the whole screen-eligible 1960 birth cohort (light blue).
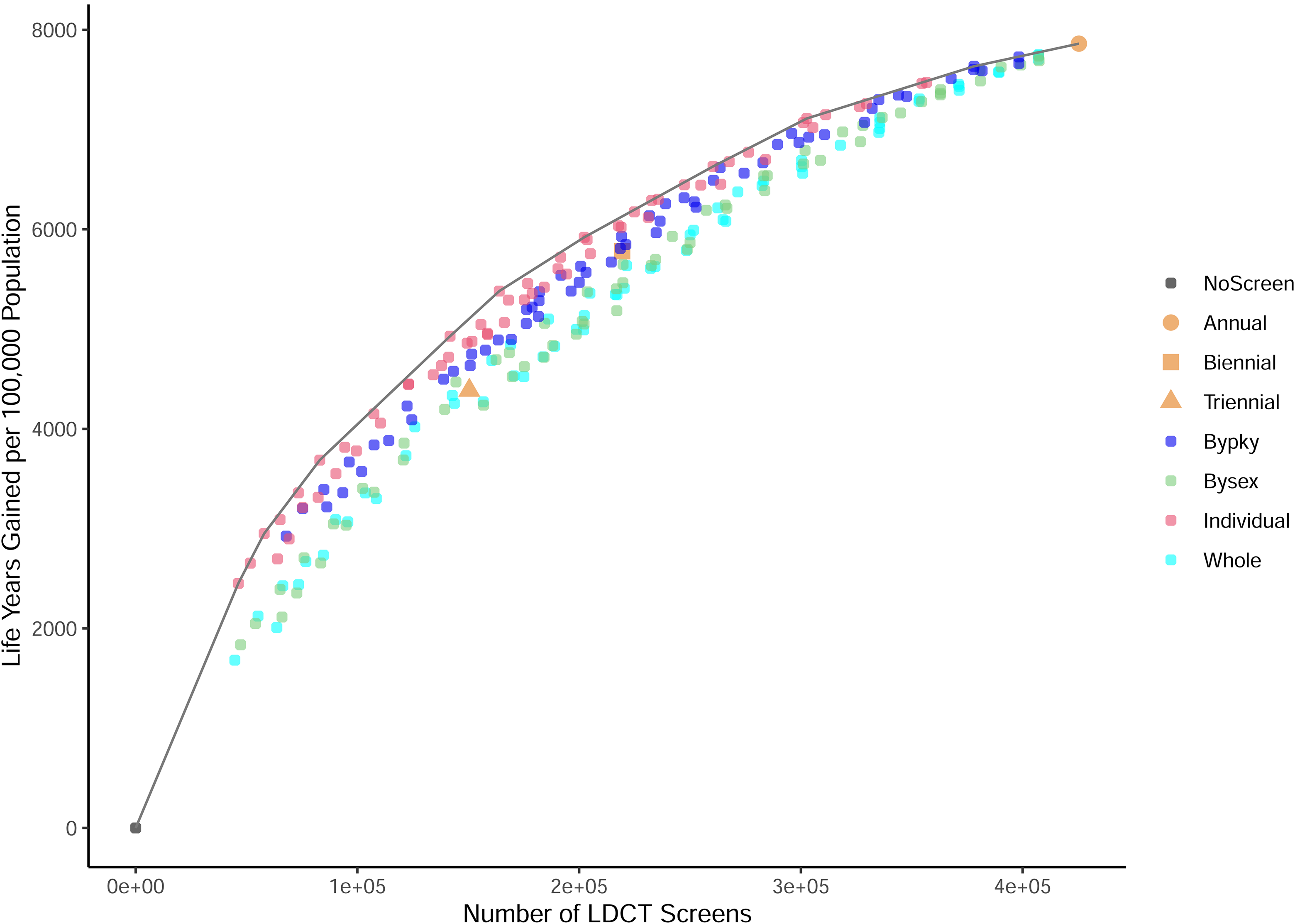
Low-dose computed tomography (LDCT) screening vs. life years gained compared with no screening for the 240 adaptive and 3 regular screening scenarios for the 1960 US birth cohort. The gray curve is the efficient frontier, connecting the dots with the highest life years gained given numbers of screens. Symbols represent regular screening: annual (circle), biennial (square), and triennial (triangle). Smaller circles represent: individualized schedules (pink), by-pack-year schedules (dark blue), by-sex schedules (light green), schedules selected for the whole screen-eligible 1960 birth cohort (light blue).
Table 1 shows the screening characteristics and health outcomes for the 25 scenarios with the highest efficiency scores, i.e., the largest benefits (LCDs averted and LYG) for a given number of screens. Annual screening is the most intensive schedule, also resulting in the most LCDs averted and LYG. Nineteen out of 25 efficient scenarios had individualized adaptive screening schedules, and three scenarios had adaptive schedules differing by pack-year category. The number of false positives per 100,000 population decreased with the number of screens, whereas the number of overdiagnoses per 100,000 population in general decreased and at a slower rate. Furthermore, the percent reduction in the number of screens for the efficient adaptive schedules vs. annual screening ranged from 4% to 67%, resulting in 4% to 65% reduction in false positives. The percent reductions in LCDs averted and LYG of adaptive schedules vs. annual screening were much lower, ranging from 1%–40%. For example, the Individual_Adjusted_0_0.015% scenario had 29% fewer screens and 28% fewer false positives than annual screening, but its LCDs averted and LYG were only reduced by 7% and 10%, respectively. The number of false-positive screens over LCDs averted, a harm-to-benefit ratio, generally decreased as the number of screens decreased for the top 25 most efficient scenarios.
Benefits and harms of the top 25 efficient screening schedules ordered by the number of low-dose computed tomography (LDCT) screens for the 1960 US birth cohort.
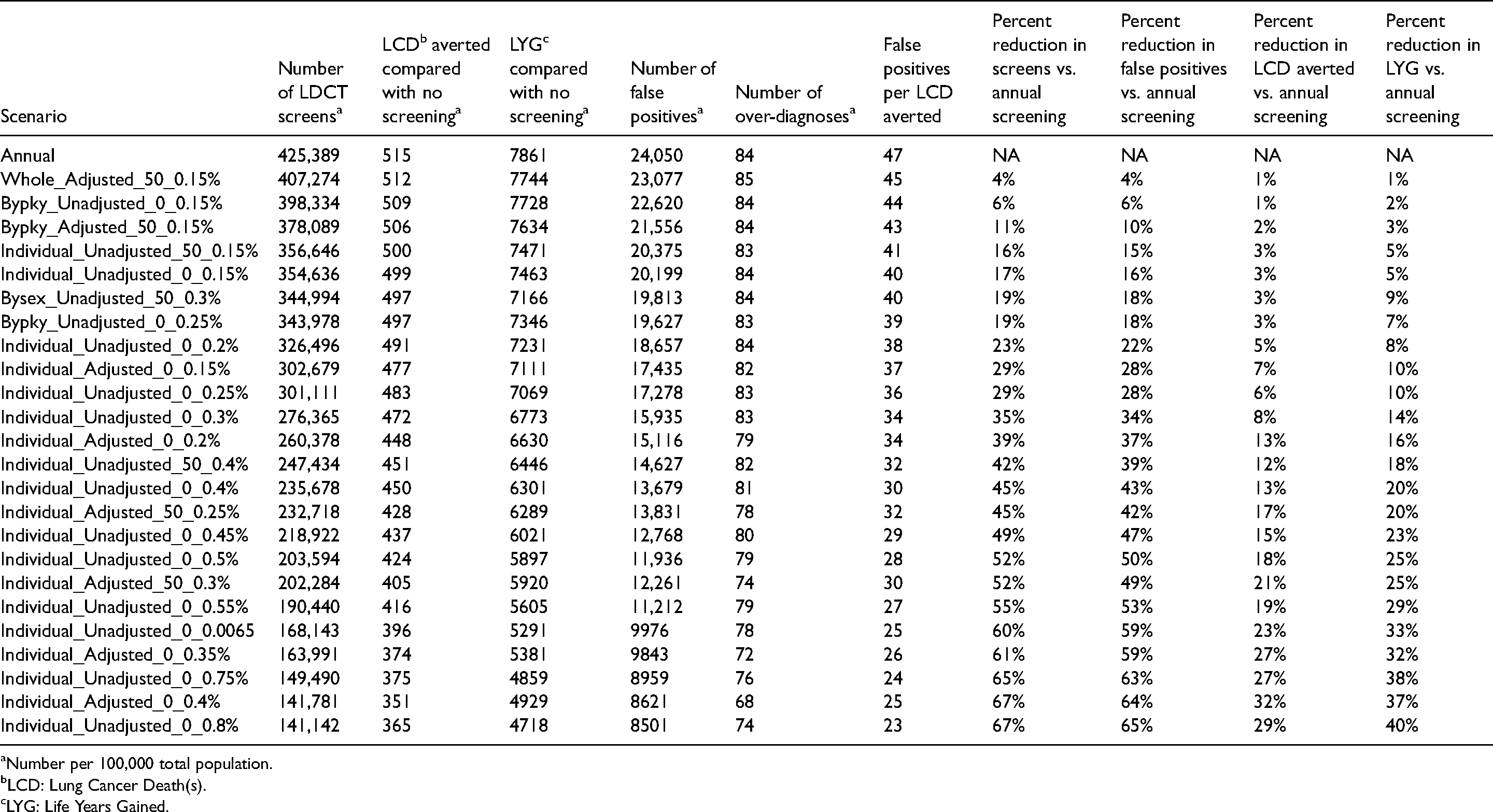
Number per 100,000 total population.
LCD: Lung Cancer Death(s).
LYG: Life Years Gained.
Figure 3 shows the discounted QALYs gained compared with no screening per 100,000 total population by disutility level. After applying baseline disutilities from screening on the lifetime QALYs, annual screening still has the highest discounted QALYs gained (Figure 3(a)). When the disutilities increased 4-fold, Bypky_Adjusted_50_0.15% had the highest discounted QALYs gained (Figure 3(b)), whereas if the disutilities increased 6-fold, Individual_Adjusted_0_0.15% had the highest discounted QALYs gained (Figure 3(c)). Additionally, schedules controlling for life expectancy had higher discounted QALYs gained than schedules without control on life expectancy when the disutility level was high (Figure S2(c)). Fixing starting age at 55 in the threshold method did not affect the discounted QALYs gained (Figure S3). The discounted QALYs gained by the disutility level for the top 25 scenarios with the highest efficient scores were shown in Table S4. We could identify a scenario with equally high discounted QALYs as annual screening when the disutilities increased by at least 3.2 times or higher.
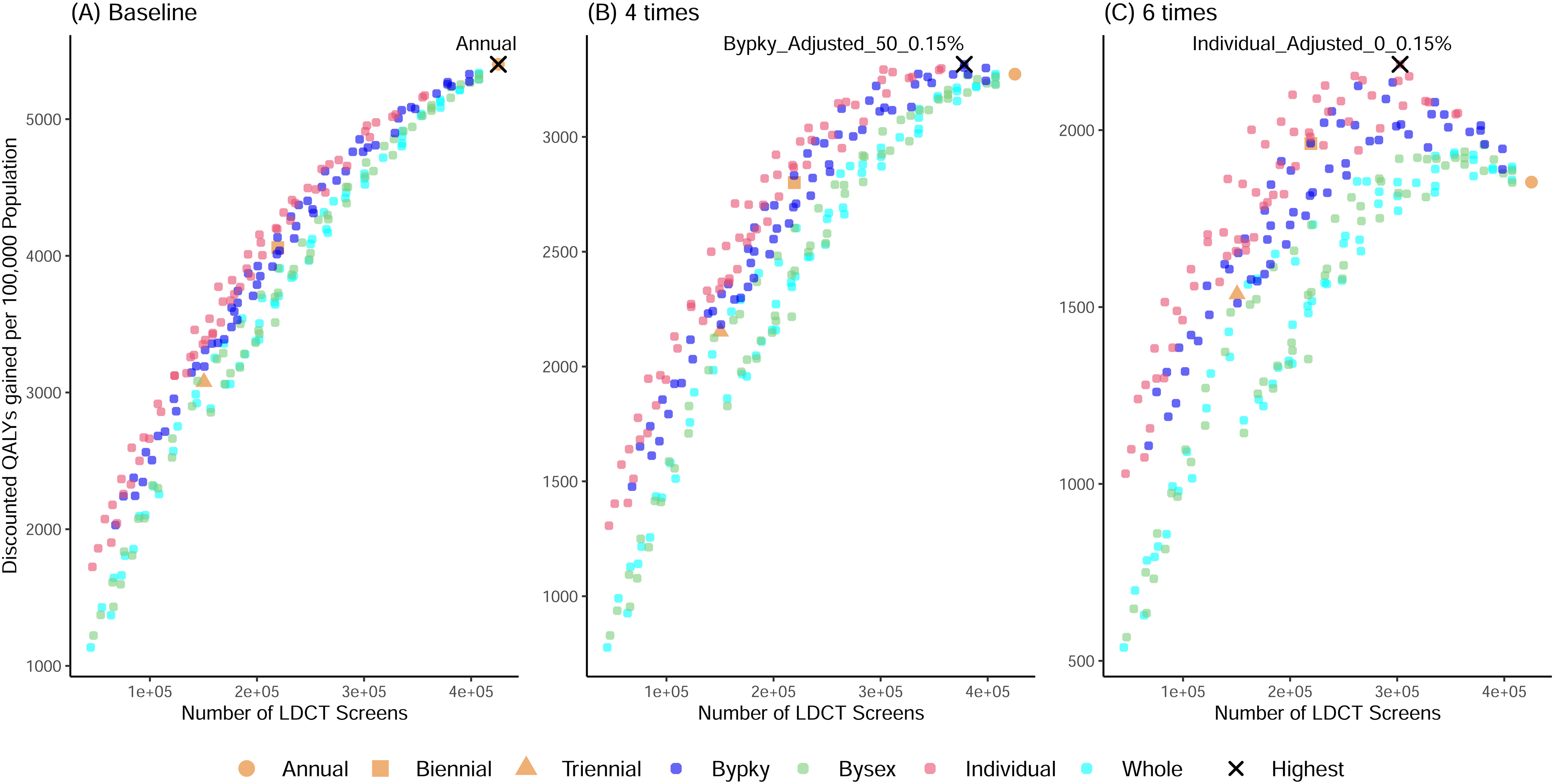
Discounted quality adjusted life years (QALYs) gained compared with no screening by the disutility level for the 240 adaptive and 3 regular screening scenarios for the 1960 US birth cohort. (a) Baseline disutilities; (b) 4-times disutilities; (c) 6-times disutilities. (a)–(c) have different y-axis limits. Symbols represent regular screening: annual (circle), biennial (square), and triennial (triangle). Smaller circles represent: individualized schedules (pink), by-pack-year schedules (dark blue), by-sex schedules (light green), schedules selected for the whole screen-eligible 1960 birth cohort (light blue). Black crosses highlight the scenario(s) with the highest discounted QALYs, being (a) annual screening, (b) Bypky_Adjusted_50_0.15%, and (c) Individual_Adjusted_0_0.15%.
Discussion
In this study, we found that in comparison to standard regular screening recommendations, risk-dependent adaptive screening reduced screening harms while maintaining similar level of health benefits. The net gains and the balance of benefits and harms from LCS with efficient adaptive schedules were improved compared to those from regular annual/biennial/triennial screening, especially when the disutilities from screening procedures were more severe.
Most efficient scenarios (those on the efficient frontiers for both LCDs averted and LYG) were individualized schedules. Furthermore, individualized schedules had fewer screens than annual screening, with less than 9% of screen-eligible individuals receiving annual screening in the most intensive individualized scenario. This suggests that annual screening is not necessary for all screen-eligible individuals, and the determination of the screening interval should vary according to each person's risk and smoking history. In addition, strategies where the schedule varied according to pack-year level resulted in slightly more benefits than adaptive schedules varying only by sex, given the same number of screenings. Different pack-year groups (20–29, 30–39 or 40+ pack-years) had different lung cancer risks, leading to distinct and potentially more suitable schedules for each group. On the other hand, males’ and females’ lung cancer risks may not be sufficiently different to justify different schedules. This finding indicates that identifying groups with distinct lung cancer risks in the screen-eligible population may lead to more suitable schedules, and therefore result in better health gains.
Patients’ preferences towards screening may greatly impact the potential benefits from screening. Adaptive schedules had the highest benefits if screen-related disutilities were medium to high (around 3 times or higher than baseline). This observation indicates that when patients’ perceived burdens on screening and related procedures were medium to high (modeled by the disutilities), less intensive schedules, such as the Individual_Adjusted_0_0.15% scenario, may be preferable to one-size-fits-all approaches. Other studies have also found that patients’ preferences towards screening may significantly affect the potential benefits from screening, and thus a shared decision-making session prior to screening is needed to better understand the patient's attitudes.15,29,30
In addition to our work, other studies have started exploring the potential of adaptive screening for LCS. Researchers in Europe are conducting a multi-country clinical trial (4-IN-THE-LUNG-RUN) to study the effectiveness and cost-effectiveness of the risk-based less intensive LCS after a negative CT screen. 31 Robbins et al., developed a tool based on a risk-prediction model to identify candidates for longer LCS intervals following a negative lung screening result. 13 Their analysis found that while many screen-negatives would benefit from extending the screening interval, the decision should be a function of individual risk and preferences. Similarly, Tammemagi et al. developed a lung cancer risk prediction model that incorporates past screening results. 12 The authors concluded that the model can be used to improve stratification of patients being screened for lung cancer into high- and low-risk strata, which could help guide decision making regarding screening interval. 12 A recent study by Toumazis et al. presented a framework based on Partially Observed Markov Processes that could determine optimal schedules and evaluate their performance simultaneously. 14 Even though our methods are fundamentally different, our results are comparable to those from Toumazis et al. in that the screening schedules recommended by both methods are less frequent in younger ages, and that the adaptive screening is more efficient—with lower harm-to-benefit ratio—than annual screening.
Our study has several strengths. While we await the results from clinical trials and observational studies, we have applied a modeling framework to simulate and project the potential impact of implementing lung cancer risk-oriented screening schedules on short- and long-term benefits and harms from screening. We adapted the threshold method 17 to obtain LCS schedules. The threshold method is a deterministic approach, and thus timesaving as it does not require any simulation. Instead of randomly selecting screening ages, the threshold method provides a solid analytical foundation that guides the decision of when to screen. We further extended the threshold method to adjust the age-specific cancer risk for competing causes of death. This life expectancy adjustment approach effectively reduces screening frequency in older ages, resulting in schedules with the highest discounted QALYs when the disutility level is high. We then used a well-established CISNET lung cancer natural history and screening model to compare the effectiveness of adaptive and regular (non-adaptive) schedules. This microsimulation model has been previously used to study the long-term benefits and harms of various LCS eligibility criteria, 21 the cost-effectiveness of strategies varying the age at stopping screening, 19 and the effectiveness of risk-based screening strategies for the US population. 16
Despite the number of strengths, our study also has a few caveats. First, the risk threshold method may be sensitive to the assumed sojourn time distribution and parameter values. For example, if we set the preclinical sojourn time (PST) distribution to be Weibull-distributed with previously published shape and scale parameters, some of the transition rates output from the threshold method may become negative. However, the threshold method is more robust to an exponentially distributed PST, and therefore we re-fitted the PST (one of the natural history components from the MichiganLung model) using an exponential distribution. It is unclear how the selection of a different PST distribution may affect the schedules selected by the threshold method, so further methodological studies are needed in this area.
Second, in our approach we used the threshold method to determine schedules, and then independently assessed their performance and identified the most efficient schedule using a microsimulation model. Alternatively, one could jointly determine optimal schedules and evaluate their performance simultaneously,14,32 but this approach is usually more computationally intensive.
Third, the threshold method treats each negative screen as an independent event. However, individuals with a negative screen may be more likely to test negative in the subsequent screens (interaction), and thereon at an even lower risk of lung cancer.12,13 The threshold method could be extended to incorporate this information by including an additional term in the formula of the probability of being in preclinical state to further deflate the probability if the individual has more than one negative screen.
Fourth, we used an ad hoc approach to adjust for life expectancy to account for competing causes of death. This effectively reduces the frequency of screening at older ages. Future studies are needed to explore other approaches to account for competing causes of death and life expectancy in the threshold method.
Finally, in this study, we only considered 100% screening adherence rate. However, recent studies found that the adherence rate to annual LCS varies widely from 12% to 91%, depending on smoking status, age, education, and race. 33 For example, older individuals aged 65 to 73 are more likely to adhere than younger individuals aged 50 to 64. 33 If younger individuals are more likely to comply with less intensive screening than annual screening, the adaptive schedules selected by the threshold method will be preferable. Future studies are needed to understand how adherence plays this role.
In summary, our findings illustrate that lung cancer risk-oriented adaptive schedules can provide a better balance of screening benefits and harms than the currently implemented annual screening. Our study provides a possible solution to optimize LCS, by reducing LCS burdens while preserving the majority of its benefits. Both individual lung cancer risk and individual preferences play an important role in the potential net gain from LCS, which calls for a patient-centered decision-making process and tailored screening intervals.
Supplemental Material
sj-pdf-1-msc-10.1177_09691413221118194 - Supplemental material for Evaluation of benefits and harms of adaptive screening schedules for lung cancer: A microsimulation study
Supplemental material, sj-pdf-1-msc-10.1177_09691413221118194 for Evaluation of benefits and harms of adaptive screening schedules for lung cancer: A microsimulation study by Pianpian Cao, Jihyoun Jeon and Rafael Meza in Journal of Medical Screening
Footnotes
Acknowledgements
We would like to acknowledge Dr Sandra J. Lee for helping us understand the threshold method and providing technical support in our replication of the original method. We also thank Dr Marisa Eisenberg and Dr Jeremy Taylor for their comments and suggestions to this manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Cancer Institute (grant number U01CA199284, U01CA253858)
Data availability
The data underlying this article will be shared on reasonable request to the corresponding author.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
