Abstract
Objective
Lung cancer is the leading cancer killer in women, resulting in more deaths than breast, cervical and ovarian cancer combined. Screening for lung cancer has been shown to significantly reduce mortality, with some evidence that women may have a greater benefit. This study demonstrates that a population of women being screened for breast cancer may greatly benefit from screening for lung cancer.
Methods
Data from 18,040 women who were screened for breast cancer in 2015 at two imaging facilities that also performed lung screening were reviewed. A natural language-processing algorithm followed by a manual chart review identified women eligible for lung cancer screening by U.S. Preventive Services Task Force (USPSTF) criteria. A chart review of these eligible women was performed to determine subsequent enrollment in a lung screening program (2016–2019), current screening eligibility, cancer diagnoses and cancer-related outcomes.
Results
Natural language processing identified 685 women undergoing screening mammography who were also potentially eligible for lung screening based on age and smoking history. Manual chart review confirmed 251 were eligible under USPSTF criteria. By June 2019, 63 (25%) had enrolled in lung screening, of which three were diagnosed with screening-detected lung cancer resulting in zero deaths. Of 188 not screened, seven were diagnosed with lung cancer resulting in five deaths by study end. Four women received a diagnosis of breast cancer with no deaths.
Conclusion
Women screened for breast cancer are dying from lung cancer. We must capitalize on reducing barriers to improve screening for lung cancer among high-risk women.
Introduction
More women die from lung cancer annually than breast, cervical, and ovarian cancer combined. 1 The number of lung cancer deaths in women increased between 1999 and 2012 while the rate plateaued in men. 2 Recently reported randomized-controlled trials have not only demonstrated the efficacy of screening for lung cancer to reduce mortality, but several, including the National Lung Screening Trial, demonstrated a disproportionately higher benefit among women. 3 Preliminary results of the Nederlands–Leuvens Longkanker Screenings Onderzoek (NELSON) trial show a 26% reduction in mortality from lung cancer in men (rate ratio of lung cancer-related death of 0.74) with even more significant benefit in women (rate ratio of 0.39–0.61), though women comprised a minority of study participants. 4 In 2019 the German Lung Cancer Screening Intervention echoed NELSON’s results in their smaller study, with a 69% reduction in mortality in lung cancer in women compared to 6% in men. 5 The estimated 5-year survival for early-stage lung cancer is well over 50%, but is less than 5% when the disease has spread to other parts of the body. Without lung screening, a mere 16% of lung cancers are diagnosed at an early stage. 1
Despite the availability of lung screening within the United States, which is covered in full by Medicare and all Affordable Care Act compliant insurers, and has been since its approval in 2015, fewer than 5% of estimated eligible participants have enrolled in a screening program.6–8 The underutilization of lung screening is in stark contrast to the uptake of mammography among eligible women. The CDC (Centers for Disease Control and Prevention) has reported that up to 70% of women over age 40 have had a mammogram in the past two years. 9 One of the greatest obstacles to cancer screening is access to care, 10 and a specific obstacle to lung screening is easy access to an accredited screening facility. 11 Women who are being screened for breast cancer at imaging facilities that offer both lung and breast screening are therefore an ideal population for targeted enrollment efforts as they are currently engaged in the healthcare system, have a history of willingness to be screened and are physically present to be screened for lung cancer.
Our previous work has shown that over 80% of women in our lung screening program have been screened for breast cancer. Yet, those having mammograms who are eligible for lung cancer screening are unlikely to have enrolled in a lung screening program. 12 We, therefore, sought to create a method of identifying women within our institutional electronic health record who were actively undergoing mammographic screening and were eligible for lung cancer screening, and to retrospectively compare lung cancer screening enrollment and cancer outcomes between those dual-eligible women over three years.
Materials and methods
This study was performed in accordance with the Health Insurance Portability and Accountability Act (HIPAA) with approval from our Institutional Review Board (IRB# 180060 and 181156). Data from 18,040 women who underwent screening mammography in 2015 at an accredited site where lung cancer screening was also available were reviewed. All mammograms were performed at one of two outpatient imaging centers capable of performing low-dose computed tomography (LDCT) lung screening and affiliated with a large academic medical center.
A natural language-processing (NLP) selection algorithm named SHAPES (Smoking History and Pack-year Extraction System) 13 was utilized within the electronic health record (EHR) to extract and quantify each patient’s smoking history, and predict women eligible for lung screening who had undergone screening mammography at our institution in 2015. SHAPES can identify smoking histories of tobacco exposure and quit time, based on information found in the EHR. SHAPES identified patients eligible for lung screening based on original U.S. Preventive Services Task Force (USPSTF) criteria, including age 55–80 with a 30 pack-year history and having smoked within the past 15 years. 14 Once women were identified as eligible by the algorithm, manual chart review was performed to confirm eligibility status. Algorithm development and sensitivity analysis were performed using those currently enrolled in the lung screening program to assess algorithm accuracy.
Women identified as having undergone screening mammography in 2015 at a site accredited for both lung and breast screening and who were confirmed to be eligible for lung screening by subsequent chart review were included. Baseline cohort demographics, clinical characteristics, outcomes and cancer screening status for both breast and lung from January 2016 through June 2019 were abstracted. Patients within the 2015 eligible cohort were no longer eligible for screening during the outcome review period if: (1) age was greater than 80, (2) they had quit smoking for more than 15 years, or (3) because of a new cancer diagnosis. All new cancer diagnoses and available details of that diagnosis were recorded.
We compared characteristics of women enrolled in lung cancer screening and those who did not enroll, including use of chi-square test for categorical variables, t-test for parametric continuous variables, and Wilcoxon rank-sum for nonparametric continuous variables.
Results
Of the 18,040 women who underwent breast screening at either of our two imaging facilities in 2015, 685 were identified as eligible for lung screening by the SHAPES algorithm (Figure 1). Of these, 251 were confirmed as eligible for lung screening by chart review and 30 (12%) had previously enrolled in a lung screening program. Patients were determined to be ineligible and were subsequently excluded if documented smoking history totaled less than 30 pack-years (n = 378), if smoking history could not be confirmed with clinical data in the medical record (n = 56), or if a patient had received a new cancer diagnosis (excluding non-melanoma skin cancer) within the past five years and was therefore considered ineligible for lung cancer screening (n = 1).
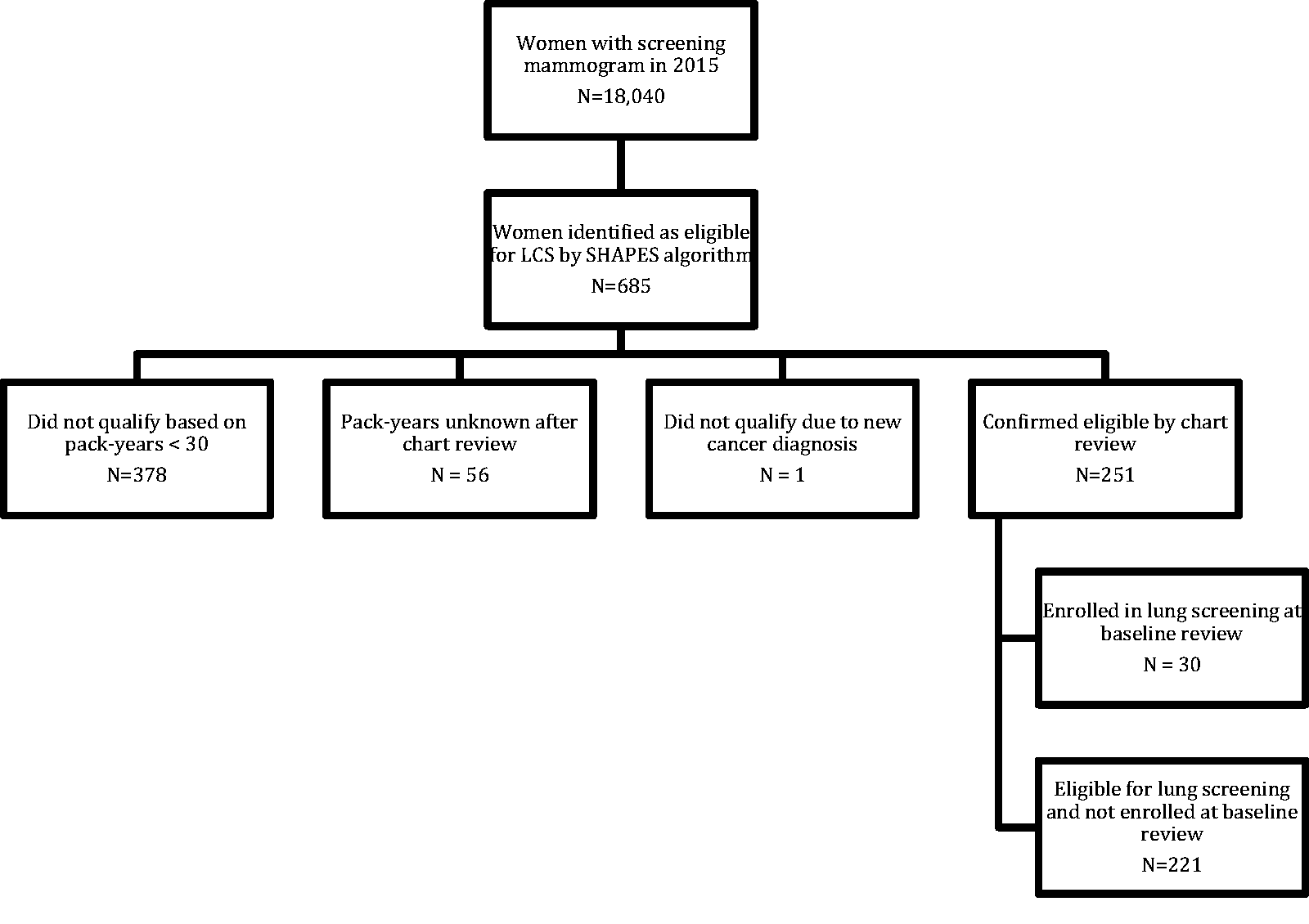
Flowchart of eligibility for study population.
The SHAPES algorithm was evaluated for sensitivity by implementation within the medical records of women previously enrolled in a lung screening program. At the time of initial chart review in 2016, 187 women were known to be enrolled in lung screening. Of these, 32 women were identified as screening-eligible by the algorithm (17%). Of the 155 not identified by the algorithm, 133 did not have a smoking history documented within the medical record and 22 had pack-years that were below the eligible threshold of 30 pack-years (recorded 0 to 26.25 pack-years). 13
At the time of their screening mammogram in 2015, 30 women out of the 251 confirmed eligible for lung screening had participated in a lung cancer screening program with at least one screening LDCT scan. Over the next three years, an additional 33 women self-enrolled into our lung cancer screening program for a total of 63 (25%). The median age in this cohort of women was 64 (interquartile range, IQR: 59, 69), and no mammogram performed in 2015 reported a BIRADS (Breast Imaging-Reporting and Data System) result of 4 (Table 1). Two-thirds of the women (42/63) who enrolled in lung cancer screening were current smokers (Table 2) as compared to only 41% (77/188) of those who did not enroll in lung cancer screening (p < 0.001).
Baseline characteristics of 251 women undergoing screening mammography in 2015 and identified by NLP as eligible for lung cancer screening (LCS).
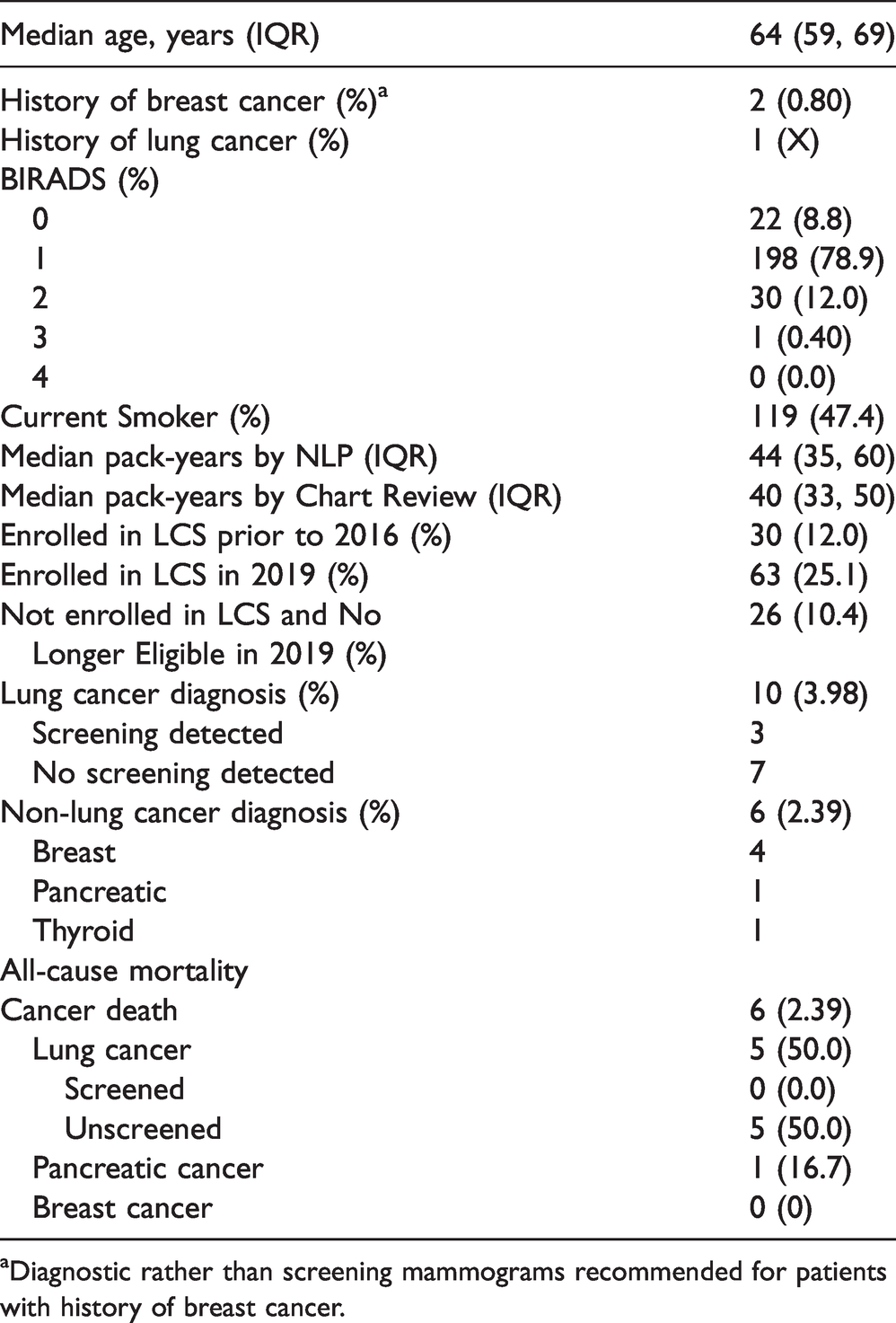
aDiagnostic rather than screening mammograms recommended for patients with history of breast cancer.
Characteristics of 251 women undergoing screening mammography in 2015 and eligible for lung cancer screening who did and did not screen, by 2019.

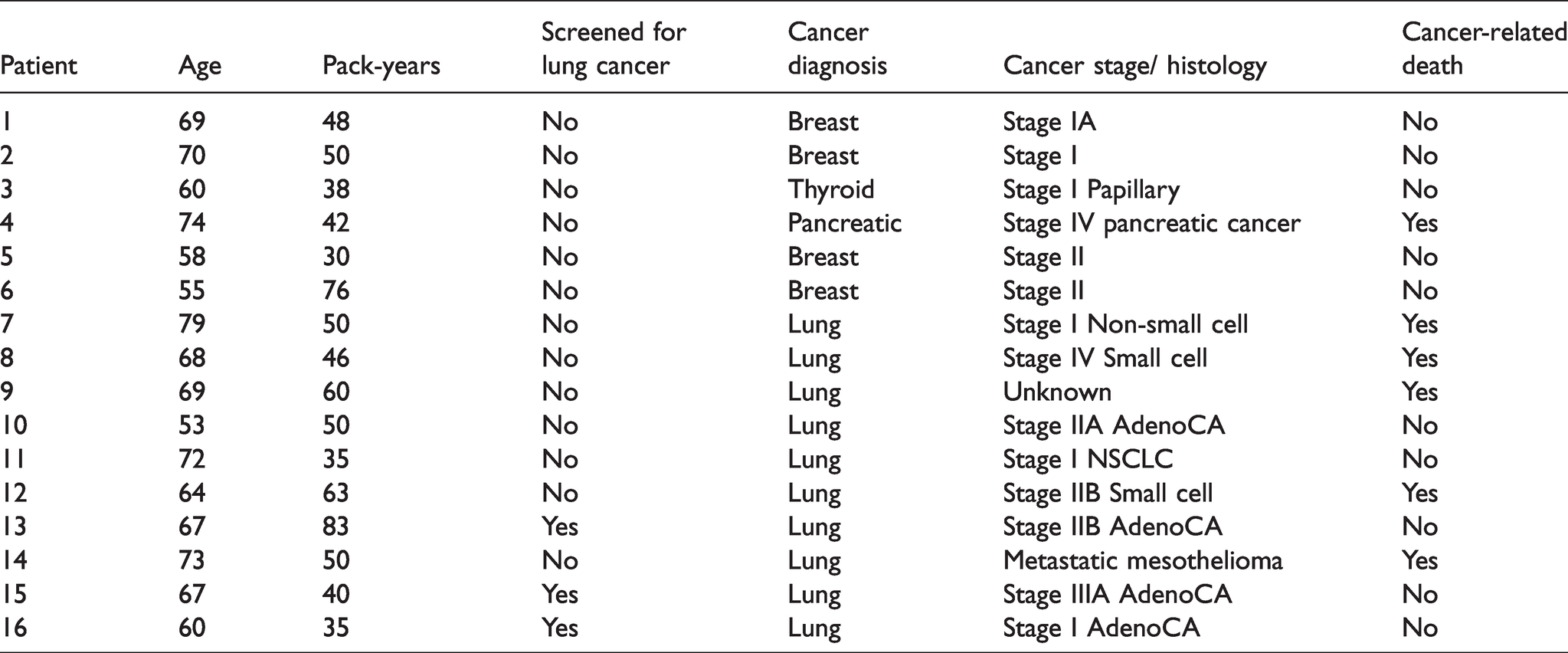
During our three-year study period, 16 new cancer diagnoses were reported in the cohort of dual screening eligible women. These included 10 lung cancers, four breast cancers, one pancreatic cancer and one thyroid cancer. Six cancer-related deaths occurred, including one from pancreatic cancer and five from lung cancer, all in the population not enrolled in lung cancer screening (Table 3). No deaths occurred among women with breast cancer in this population during the three-year follow-up period. Additionally, there were no deaths among women with screening-detected lung cancers (n = 3). Mean follow-up time was 46.3 months for breast cancers and 40.1 months for the ten lung cancers. Lung cancers occurred at a rate of 11.4 per 1000 person-years compared to breast cancers at 4.6 per 1000 person-years (p = 0.68).
Cancers diagnosed from 2016 through 2019 in women at high risk for lung cancer and being screened for breast cancer.
Discussion
More lung than breast cancers have been discovered at our institution among a subgroup of women (1.4%) undergoing mammography who were eligible to be screened for lung cancer. More lung cancer-related deaths occurred among those eligible for but who did not participate in lung cancer screening. Numerous randomized-controlled trials have demonstrated a significant mortality benefit to lung cancer screening.3–5, 15 Women included in our cohort had a mammogram at a facility where breast and lung screening were available on the same day. All Affordable Care Act compliant insurers covered both screening modalities in full. Within this group of women, there have been more than twice as many lung cancer diagnoses among the lung screening unenrolled compared to breast cancer (3.3 per year as compared to 1.3 per year). Recently published randomized controlled trials of lung cancer screening have suggested a disproportionate mortality benefit in women, with up to a 69% reduction in lung cancer deaths among female subjects. 5
We believe that a greater number of women will enroll in lung screening if it is made concurrently available with their annual mammogram. Reducing the barriers to lung screening results not only from the convenience of having both studies performed in the same imaging facility and during the same visit, but also by utilizing the screening mammogram as a “teachable moment” to promote other screening examinations. 16 We expect improved lung screening enrollment and outcomes in this population specifically because (1) these patients are already being screened for breast cancer and (2) women have been shown to have a potentially greater benefit from screening than men. Without targeted intervention, 25% of the women in our mammography cohort enrolled in lung screening, which was five times greater than the national average of eligible screening participants enrolled at that time.
The SHAPES algorithm identified 3.8% of women having a screening mammogram at a site offering both breast and lung screening as eligible to be screened for lung cancer based on original USPSTF guidelines. Chart review confirmed that 37% of those identified by the algorithm were in fact eligible (false positive rate of 63%). However, NLP only identified 17% of women who had previously enrolled in our lung screening program and had a mammogram in 2015. We can assume it failed to identify a significant number of women undergoing mammography who were eligible for lung cancer screening.
A recently published study utilizing nationally representative cross-sectional survey data estimated that 7.1% of women who underwent screening mammography in the past two years were eligible for lung cancer screening and 9.8% were current smokers. Among women who had a mammogram and were eligible for lung screening, 7.9% reported having a low-dose CT to screen for lung cancer. 17 The experience at our institution, where the majority of women eligible for lung screening undergoing mammography have not been screened for lung cancer, is in accordance with the national survey results. Additionally, a statistically significant proportion of women enrolled in lung screening are current rather than former smokers, emphasizing the need for additional patient and provider education of screening eligibility among those with a more complex smoking history.
This study suggests that women who are at high risk for lung cancer and are currently actively participating in breast cancer screening would benefit from also participating in lung cancer screening. Over three years, 10 women screened for breast cancer were diagnosed with lung cancer while four were diagnosed with breast cancer. There were no deaths from breast cancer in this group and 50% mortality among those with lung cancer, testimony to the lethality of lung cancer. Among the women who were screened for lung cancer, two early-stage (Stage I adenocarcinoma and Stage IIB adenocarcinoma) curable tumors were found in three participants and no cancer-related deaths were observed within the limited follow-up period.
Limitations
This study is limited by the small sample size and the limited follow-up period of 46.3 months for those with breast cancer and 40.1 months for those with lung cancer. Though over 18,000 women were evaluated for screening eligibility, only 251 of 685 identified by NLP were confirmed eligible for lung cancer screening by manual chart review. The number of women having mammograms who were subsequently eligible to be screened for lung cancer was underestimated as the SHAPES algorithm failed to identify the majority of women in the EHR who had previously enrolled in the lung screening program. Our future outreach initiatives will include surveys of women at the time of mammography to better determine lung screening eligibility based on self-reported smoking history. This study design differentially enrolled those women who underwent screening mammography in 2015 and therefore may have a lower risk for lung cancer compared to the unscreened population. Cancer incidence was determined based on clinical data in the EHR; however, we do not have records for patients who received care elsewhere or had a recent diagnosis outside the follow-up review period, as evidenced by one unstaged cancer patient.
Eligibility and screening practices included in this manuscript are specific to the United States. While there have been clinical trials demonstrating the benefits of lung screening in other countries, 4 , 18 , 19 routine screening has not yet been implemented. Pilot programs within the UK have demonstrated that lung screening can be successful and outreach initiatives will likely be designed around future guidelines. 20
Conclusion
Efforts should be made to reduce barriers to lung cancer screening by identifying those currently engaged in other cancer screening modalities and by creating the opportunity to perform multiple screening examinations during one visit.
Footnotes
Authors’ contribution
KLS and ABP conceived the work; KLS, DH, ABP, TJO, CCS, EAP and SAD took part in the design of the work; KLS, DH, and SAD wrote the manuscript; KLS, DH, ABP, TJO, CCS, EAP and SAD Were involved in the revision of manuscript; KLS, DH, ABP, TJO, CCS, EAP and SAD approved the manuscript. All authors are accountable for manuscript contents.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Vanderbilt-Ingram Cancer Center Cancer Early Detection and Prevention Pilot Project, Vanderbilt-Ingram Cancer Center Young Ambassadors Discovery Award.
Statement of data access and integrity
The authors declare that they had full access to all of the data in this study and the authors take complete responsibility for the integrity of the data and the accuracy of the data analysis.
