Abstract
Objectives
Previously, based on 6 months of follow-up, we showed that HPV self-sampling improved participation in cervical screening compared to a reminder letter for Pap testing for never- and under-screened women. Here, we report follow-up and related screening outcomes for women who participated in the initial self-sampling over two screening rounds.
Setting
The randomised controlled trial was conducted in Australia.
Methods
Never- and under-screened women were randomly allocated to the HPV self-sampling or the reminder for Pap test arm and followed at 6 and 36 months since the kits were first mailed.
Results
The first round of HPV self-sampling kits were mailed from May–July 2014 to 12 572 women. After 36 months, 19% of never-screened and 9% of under-screened women returned a kit for HPV testing; 2.7% were HPV 16/18 and 5.8% non-16/18 HPV positive. Compliance with first round follow-up was 84% (95% CI: 77.1–89.5%). Non-compliant and cytology triage negative women were mailed another kit at 12 months. Compliance at 12-month follow-up was 59.3% (49.4 to 68.6%). Of 37 women with a 12-month repeat HPV, 70% were positive. Of women who tested negative for HPV in the first round (n = 1573), 25% attended regular screening in the next round and none had CIN2 + detected. The overall prevalence of CIN2 + was 8.5 per 1000 screened (4.8 to 13.9 per 1000).
Conclusion
While self-sampling can successfully engage women, compliance with repeat testing may require monitoring. The clinician-supported self-collection pathway now in use in Australia will likely improve women's engagement with follow-up.
Introduction
On December 1st 2017, the National Cervical Screening Program (NCSP) in Australia changed from 2-yearly Pap testing to 5-yearly primary human papillomavirus (HPV) testing with partial genotyping (genotyping for HPV16/HPV18 separately and a pooled result for 12 other oncogenic HPV types) and reflex liquid-based cytology (LBC) triage for women with HPV (not 16/18). The renewed program is for both HPV-vaccinated and unvaccinated asymptomatic women aged 25–69 years, followed by an exit HPV test between 70 and 74 years of age. The program also offers women who are 30 + years and overdue for a cervical screening test by 2 years or more, and have declined to have a practitioner-collected sample, the opportunity to collect their own sample for HPV testing in a supportive clinical setting (HPV self-sampling). 1
The sensitivity and specificity of HPV self-sampling for the detection of cervical intra-epithelial neoplasia (CIN) 2 + lesions is comparable to practitioner-collected samples provided a PCR-based test is used. 2 Trials conducted overseas, and in Australia (the iPap trial), have shown that HPV self-sampling increases participation in cervical screening when compared to a routine letter reminder to have a practitioner-collected sample amongst women who have never had a Pap test (never-screened) or had one a long time ago (under-screened).2,3 Involving these women in the screening program is vital to further reduce cervical cancer incidence and mortality rates, since non-participants account for more than half of all cervical cancer cases. High compliance with follow-up by women who test positive for HPV is also critical to making self-sampling both an effective and cost-effective intervention. Unlike practitioner-collected samples, reflex LBC cannot be done on self-collected samples. Therefore, pending the development of alternative forms of triage that can be conducted on self-collected specimens, in follow-up protocols involving cytology triage, women will need to make an appointment with a clinician more than once before being referred for colposcopy, which may be associated with greater loss to follow-up.
On the basis of earlier data suggesting self-sampling was associated with a small loss in sensitivity, and because of concerns about compliance and cost associated with additional health care visits needed for HPV non1618 positive women, the current aim of the renewed NCSP in Australia is to support women to join mainstream clinician-collected screening. 4 However, this is expected to change in the near future when self-collection will likely be made available to all people eligible for cervical screening in the renewed NCSP, giving them a choice between self- or practitioner-collected sampling, both accessed through health-care practitioners. Currently, there is no evidence on re-screening behaviour or compliance to clinical management pathways of never- and under-screened women who take up the offer of self-sampling.
In an earlier publication from the iPap trial we reported that 15.8% of never-screened and 7.3% of under-screened women returned a self-collected swab within six months of receiving the kit. Of those who tested positive for oncogenic HPV, 76% attended follow-up within 6 months of receiving their HPV positive test result. 3 Of note, in the iPap trial, the self-sampling kits were directly mailed and not offered in a clinical setting as is the case in the current NCSP. In this paper, we report HPV self-sampling participation and follow-up completion rates over 36 months from the start of the iPap trial in April 2014.
Methods
Study design
The design of the iPap trial has been reported previously. 5 In brief, this was a randomised controlled trial to determine whether HPV self-sampling increases participation in cervical screening by never- and under-screened women when compared with a reminder letter to attend for a Pap test Women were eligible if they had never had a Pap test (never-screened) or last had one between 5 and 15 years ago (under-screened) as recorded in the Victorian Cervical Screening Registry (VCSR; previously known as the Victorian Cervical Cytology Registry), and were aged 30–69 years, not pregnant and with no prior hysterectomy. Never-screened women were women who were recorded in the Victorian Electoral Roll (VER) and for whom no match was found in the VCSR, indicating that no cervical screening episode had been recorded. The two databases (VER and VCSR) were matched on name, address and date of birth to identify never-screened women. Randomisation was stratified by screening status (never- and under-screened). For each stratum, 8160 women were randomly allocated in a 7:1 ratio to the HPV self-sampling arm or to the Pap test reminder arm. For under-screened women, there was an additional stratification by time since last Pap test (5, 6, 7, 8, 9 and 10 + years). In the HPV self-sampling arm, women were initially mailed a pre-invitation letter and then a kit. The self-sampling kit comprised a nylon-tipped flocked swab (Copan Italia, Brescia, Italy) enclosed in a dry plastic tube. Women took a self-sample using the swab and returned the sample to VCS Pathology as per instructions in the kit. At VCS Pathology, the Roche cobas® 4800 test was used to measure the presence of clinically relevant levels of oncogenic HPV DNA as per manufacturer's instruction. The initial round of letters and kits were directly mailed between March and July 2014. The primary outcome of the trial was participation at 6 months, as indicated by the return of a swab or the notification of a Pap test result to the VCSR. The secondary outcome, for women in the self-sampling arm with a positive HPV test, was undergoing appropriate clinical investigation.
Clinical management
In the trial, women in the self-sampling arm who participated and had a valid HPV test result were sent their results letter, with a copy sent to their general practitioner (GP) if they nominated one. The letter to the GP explained the study, the HPV result and the recommendation for further clinical management. All women with positive results were followed up by the VCSR with the assistance of the VCS liaison physicians, as per iPap protocol, including more than one reminder to the woman. Women who tested positive for HPV 16/18 were directly referred for a follow-up colposcopy. For a non 16/18 HPV positive result, women were referred to a GP for a follow-up cytology test Further management was based on the result of this cytology test Women who did not have a follow-up test, or had one that was negative or a low-grade squamous intraepithelial lesion (LSIL), were mailed another self-sampling kit for a repeat HPV test at 12 months. Women who had a high-grade squamous intra-epithelial lesion (HSIL) or any glandular abnormality on the follow-up cytology were referred for colposcopy. The second round of kits were directly mailed between June and August 2015, and women were asked to return the kit within 4 weeks. The aim of the repeat self-sample test was to see if the HPV infection had cleared. If the 12-month repeat self-sample test was positive for oncogenic HPV (any type), women were referred for colposcopy. Women who tested negative for oncogenic HPV in the initial round of self-sampling were recommended to have a Pap test within 24 months (as per previous national screening policy; Figure 1).
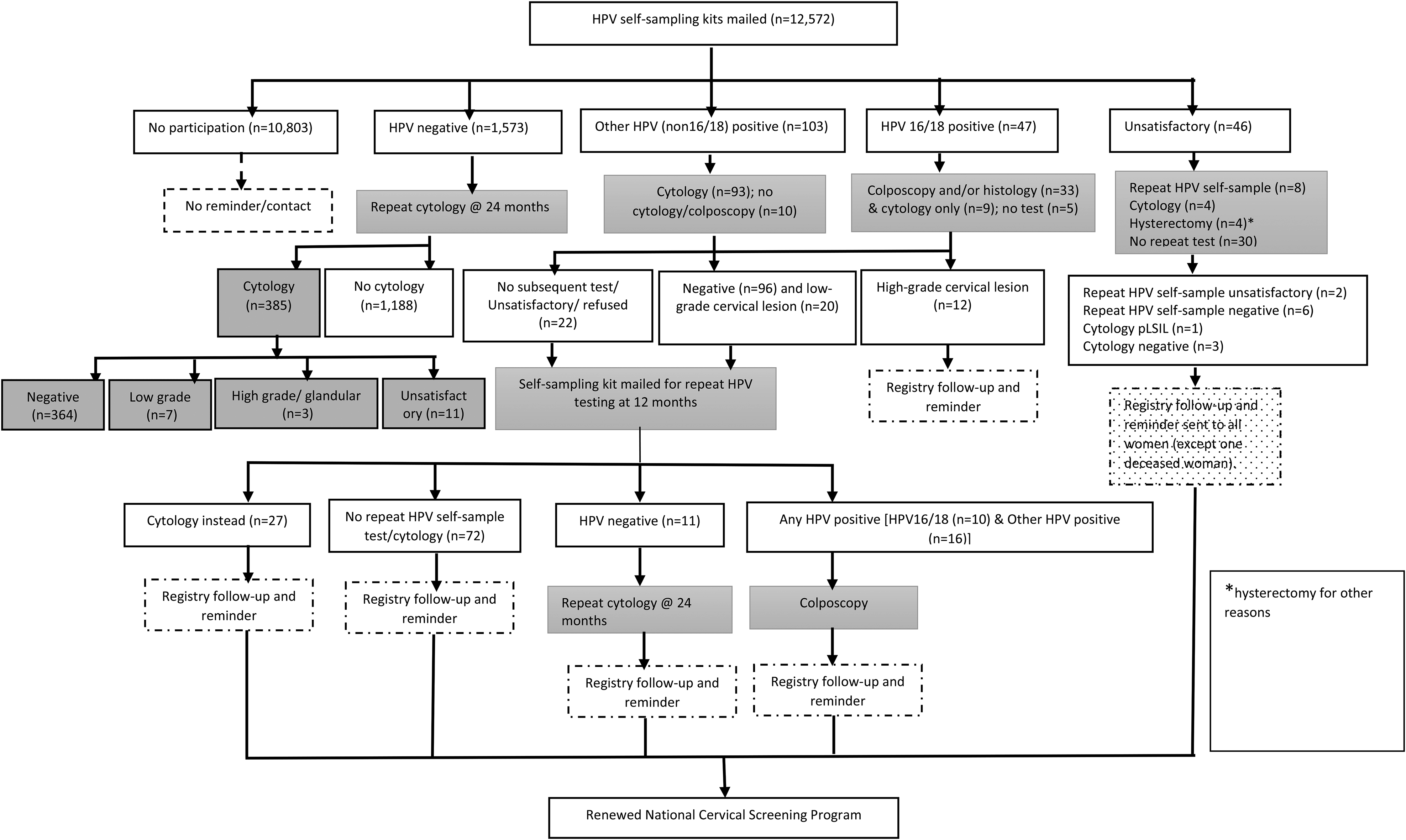
Clinical management flow chart and stages of follow-up of women in the HPV self-sampling arm in the iPap trial.
Outcome
Participation in this follow-up study was defined by the return of a completed self-sampling kit to VCS Pathology for HPV testing within 36 months of the mailout of the initial kits. In the main trial, to identify women who had one or more Pap tests 6 months after randomisation, a semi-automated match of all women in the trial with the Registry records of all Pap tests conducted in Victoria was undertaken in March 2015. In October 2015, all identifiable information for never-screened women who did not participate in the trial was destroyed, as per the requirements of the Victorian Electoral Commission, preventing any repeat of the matching process for never-screened women. Therefore, the current analysis included women who completed at least one HPV self-sampling test
Compliance with follow-up was defined as the proportion of women who had a positive HPV self-sample test who then went on to have further clinical investigation as per the recommendation in the iPap protocol. Follow-up was reported in two stages: 1) after the initial HPV self-sampling test (stage 1) and 2) after the 12-month repeat HPV self-sampling test (stage 2). We also report the proportion of women who had a negative HPV self-sampling result in the initial round who went on to have a Pap test after 24 months as per national policy. By the end of May 2017 (study end date), 90% of women who had tested negative for HPV in the iPap trial would have received a reminder letter for their next screening test and had six months to undertake subsequent screening (practitioner-collected). A final reminder was also sent to women who tested positive for oncogenic HPV and for whom a follow-up test was not recorded by the registry in the recommended time frame.
Statistical analysis
Kaplan-Meier analyses were performed to assess self-sampling participation proportions over time. Date of participation was the date that the self-collected sample was received by the laboratory. Time to participation was measured in months from the date the kits were mailed until the date that the self-collected sample was received by the laboratory. Women were censored at the date that the kits were returned to the sender, date of death, or date the study ended (31 May 2017), whichever was earlier. Participation proportions are presented separately for never- and under-screened women. For stage 1 follow-up, we estimated the proportion (and 95% confidence interval, CI) of women whose sample tested positive in the initial round of HPV self-sampling who completed follow-up investigation, which for HPV 16/18 was colposcopy and for other HPV (non-HPV 16/18) was cytology. For stage 2 follow-up, we estimated the proportion (95% CI) of women whose sample tested positive for any HPV in the 12-month repeat self-sampling who had a colposcopy. Median follow-up times were also reported for the different HPV-positive groups. All statistical analyses were performed using STATA version 12. The study was approved by the Human Research Ethics Committee of the Victorian Department of Health.
Results
Participation in the initial round
A total of 1218 [18.7% (95% CI: 17.8%–19.7%)] out of 6501 never-screened and 551 [9.1% (8.4%–9.8%)] out of 6071 under-screened women participated in the initial round of self-sampling by returning a self-collected swab to VCS Pathology for HPV testing within 36 months of the mailout of the kit. The majority of women, however, participated within 6 months [1155/1218 (95%) in never-screened and 520/551 (94%) in under-screened] with a small proportion participating thereafter (Figure 2).
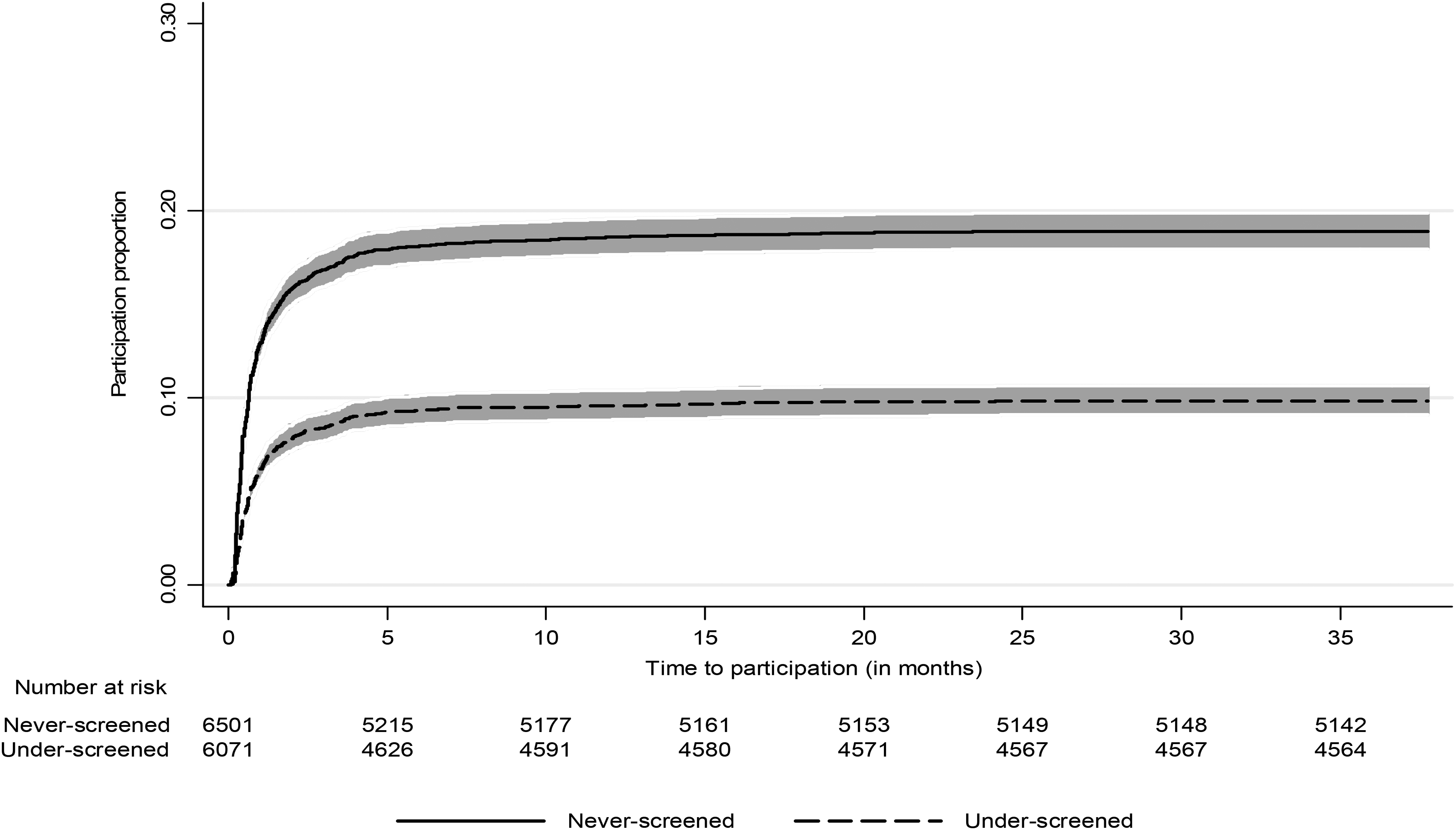
Kaplan-Meier curves demonstrating participation (shaded area – 95% CI) in the self-sampling HPV testing arm of the iPap trial. Participation is defined as return of the self-sampling kit to VCS Pathology within 36 months of follow-up. Number at risk here refers to number available to participate.
Participation in the next screening round
Of the 1769 samples returned in the initial round, 1573 tested negative for oncogenic HPV and 385 [24.5% (22.4%–26.7%)] of these women had a subsequent screening round cytology test (Figure 1). Median follow-up time (25th – 75th percentile) for cytology was 26 months (13 to 32 months). Women who were negative in their initial self-sampling who had subsequent cytology were likely to be younger (p < 0.001) and to have a GP (58% vs. 49%, p = 0.001) compared to their counterparts (Table 1).
Socio-demographic profile of women who had an initial HPV negative test on the self-collected sample who went on to have a Pap test at around 24 months.
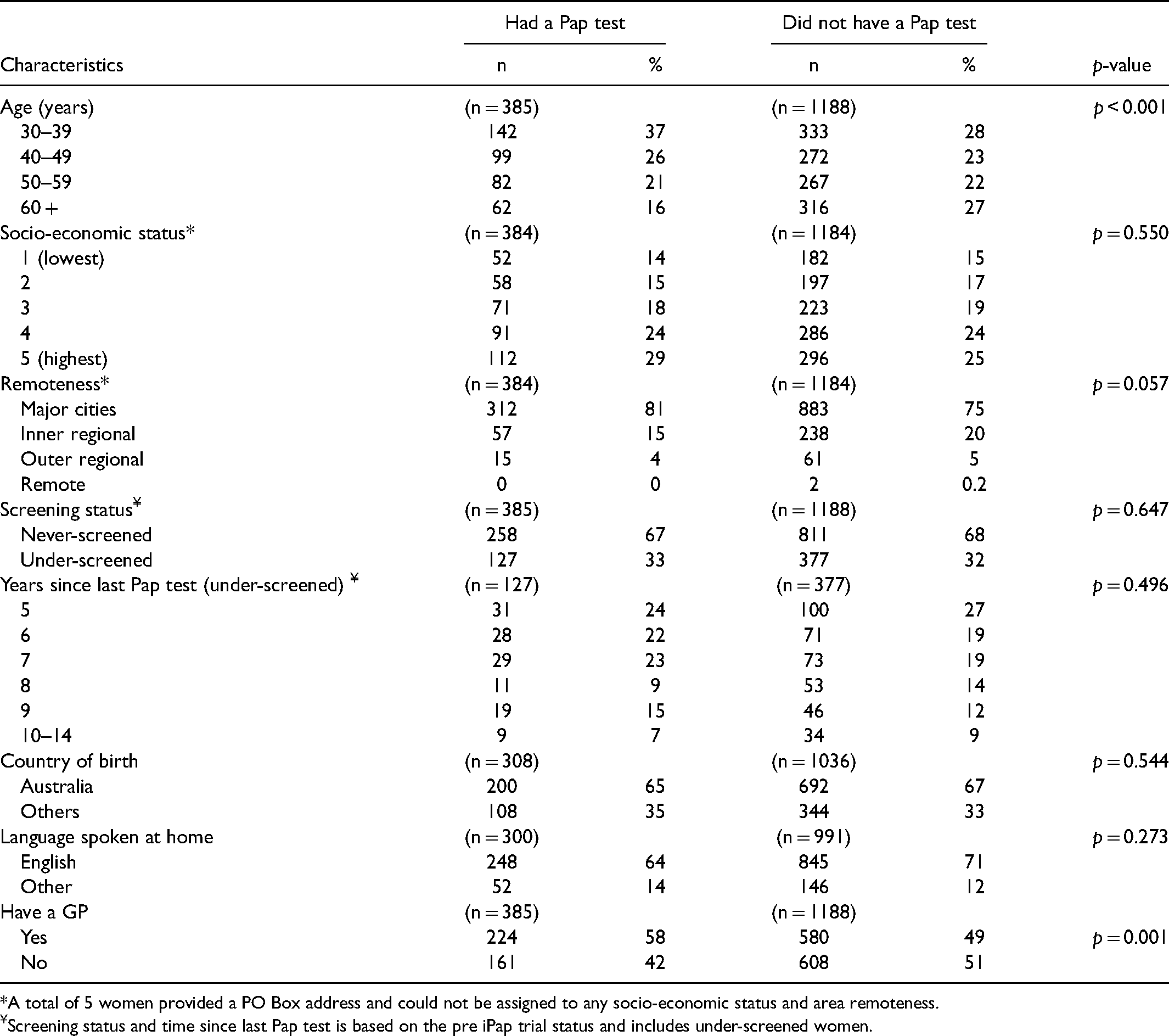
*A total of 5 women provided a PO Box address and could not be assigned to any socio-economic status and area remoteness.
Screening status and time since last Pap test is based on the pre iPap trial status and includes under-screened women.
HPV positivity
A total of 47 of the 1769 returned samples [2.7% (2.0%–3.5%)] tested positive for HPV 16/18, 103 [5.8% (4.8%–7.0%)] tested positive for HPV (not 16/18) and 46 [2.6% (1.9%–3.5%)] samples were unsatisfactory. (Figure 1).
Follow-up
Overall, 126 out of 150 [84% (77.1%–89.5%)] women whose sample tested positive for oncogenic HPV had a follow-up investigation as per protocol. Of the 47 women with HPV 16/18 positive result, 33 (70.2%) had colposcopy/biopsy and 9 (19.1%) had cytology only. Of the 103 women with another HPV (not 16/18) positive result, 93 (90.3%) had cytology as recommended (Figure 1). Median follow-up time to colposcopy/histology for HPV 16/18 positive women was 3 months (25th-75th percentile: 2.4–5.3 months) and that for other HPV (not 16/18) to cytology was 1.1 month (25th-75th percentile: 0.7 to 2.6 months).
A total of 108 were sent another self-sampling kit for a repeat HPV test at 12 months. Women were not mailed a kit at 12 months if they had a recent practitioner-collected sample for Pap or HPV test, or were under treatment for other reasons. Of the 108 sent another kit, 37 women [34.3% (25.4%–44.0%)] had a repeat HPV test and another 27 women [25% (17.2%–34.3%)] had a cytology test, giving an overall 2nd stage follow-up of 59.3% (49.4%–68.6%). Of the 37 women who had a repeat self-sample HPV test, 70% (26 out of 37) tested positive for any HPV [HPV 16/18 positive (n = 10) and other HPV (not 16/18) positive (n = 16)] on the repeat test (Figure 1).
CIN2 + lesions
Of the 47 women who were HPV 16/18 positive, 26 women had a biopsy in the first round with CIN2 + lesions identified in eight women. One additional HPV 16/18 positive woman was detected with a CIN3 lesion in the second round (Table 2). Of the 103 women positive for other HPV, 93 had a subsequent Pap test of whom five had HSIL detected and one had a cytological prediction of squamous carcinoma. All six women with HSIL + cytology had a biopsy and three had CIN2 + lesions and one had CIN NOS (not otherwise specified). An additional six women had a biopsy after a repeat HPV self-sample positive test, two of whom were detected with CIN2 + lesions (Table 2). A total of 15 CIN2 + lesions were detected over a follow-up period of 36 months from 1769 never- and under-screened women who were screened by HPV self-sampling, giving a prevalence of 8.5 (95% CI: 4.8–13.9) per 1000 women.
Follow-up test and results after an HPV self-sampling positive test result.
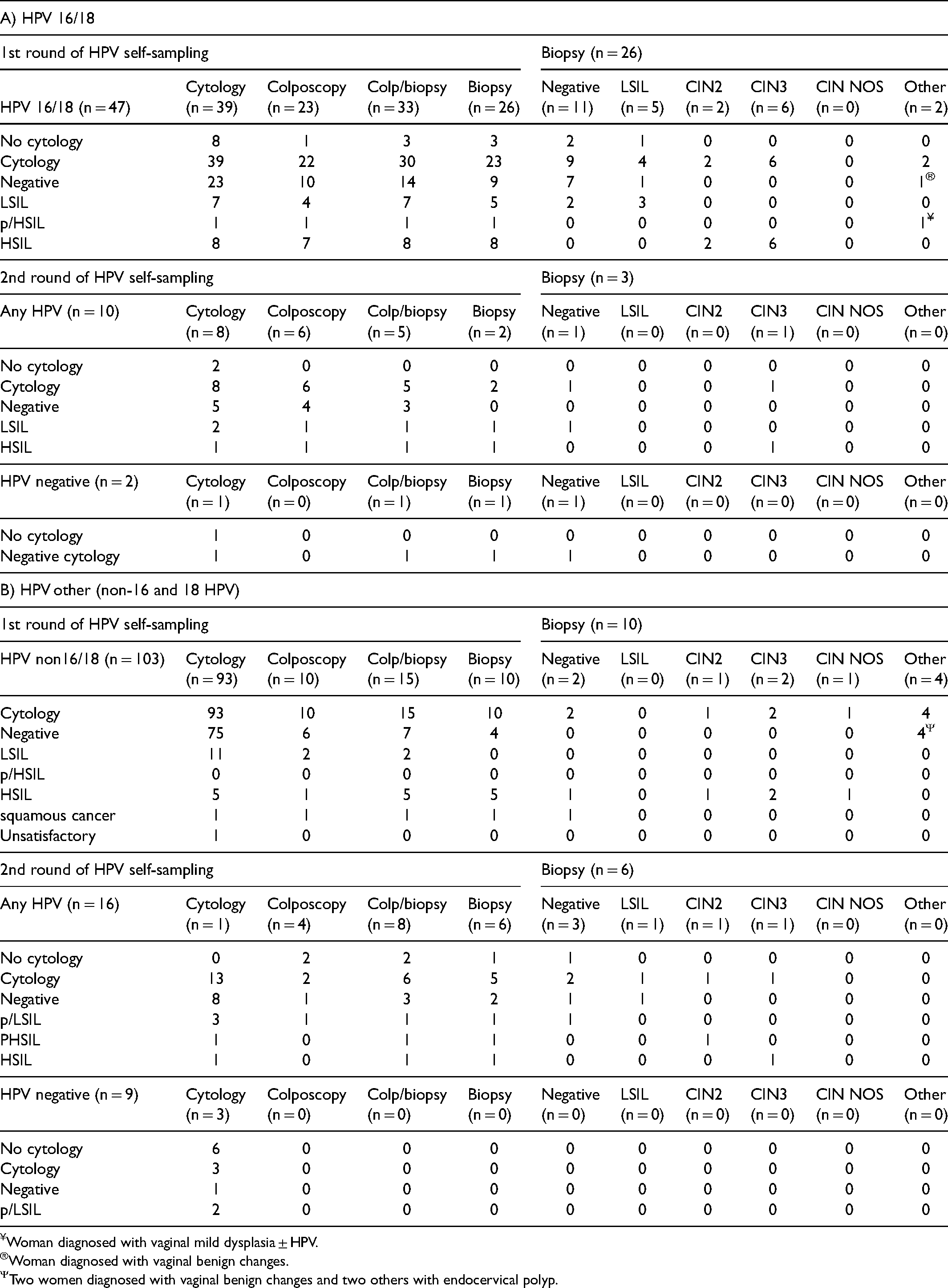
Woman diagnosed with vaginal mild dysplasia ± HPV.
Woman diagnosed with vaginal benign changes.
Two women diagnosed with vaginal benign changes and two others with endocervical polyp.
Discussion
This study reports compliance with follow-up and related screening outcomes at 36 months amongst never- and under-screened women who responded to a mailed invitation to self-collect a sample for cervical screening at home. Previously, we have shown that self-sampling for HPV testing resulted in a substantially greater participation in screening than an invitation or reminder letter for a Pap test in both never- and under-screened women. 3 In the current study we found that most never- and under-screened women who decide to self-sample do so in a timely manner. The proportion of never- and under-screened women in the self-sampling arm who participated by returning a swab within 6 months of mailout of the kit was 15.8% and 7.3% respectively. 3 At 36 months, participation had risen only slightly to 18.7% and 9.1%, respectively. However, there were no formal reminders if the kit was not returned. In a Swedish trial, a moderate amount of participation in the self-sampling arm (39%) was achieved for under-screened women where women were reminded in a second invitation if the kits were not returned. 6 In Victoria, around 30% women have a Pap test within three months of receiving their first reminder letter 7 and it is possible that a further reminder could have triggered participation by women who intended to complete and return the test In the iPap trial, the two main reasons for non-return reported by women in a survey completed six months after the mailout of the kits were that they didn't have the time or forgot about the test 8 This suggests that routine reminders are important for successful implementation of any new strategy to improve screening uptake.
We used a mail-to-all strategy in the iPap trial where the self-sampling kits were directly mailed to the woman's home address. 5 A recent systematic review, including 25 trials, found pooled participation in the self-sampling arm to be 19.2% when the kits were directly mailed to home and 7.8% when women had to both request the kit and then complete it (opt-in). 2 Participation in the self-sampling arm was excellent (ranging from 79.8% to 98.2%) when women were offered self-sampling by health-care workers through door-to-door visits as shown by four trials conducted in developing countries. 2 However, the door-to-door strategy may be too expensive and impractical in developed country settings where most women regularly attend primary care services.
The direct offer of a self-sampling kit by a health-care worker in a clinic setting can result in high uptake amongst under- and never-screened women, as shown by a pilot study in Australia where 79 out of 98 (81%) disadvantaged women, who refused a practitioner-collected conventional Pap smear, accepted the offer of self-sampling. 9 Offering self-sampling through clinics could overcome some of the issues with home-based self-sampling; in particular, concerns expressed by women around their ability to do the test adequately and about the accuracy of self-sampling.8,10 The health-care provider can engage the woman personally about such concerns as well as provide information on when and how the results will be obtained, the meaning of an HPV positive result, and the importance of further follow-up. Supporting the potential of utilising trusted provider relationships, within our study we identified that women who were supported by their nominated health-care provider were more likely to engage in timely follow-up when HPV positive 3 and also more likely to join mainstream screening thereafter. About 25% of never- and under-screened women who took up the offer of self-collection and tested negative attended regular screening in the next round, which is encouraging given that these women are hardest to reach. 11 From 1st of July 2022, the self-collection policy will be expanded to include all people eligible for a cervical screening test (and not restricted to those aged 30 years or older and never or under-screened), who through their nominated health-care provider will be able to choose to self-collect their sample in one or more subsequent screening rounds. This change in policy is likely to improve screening participation. 12
In the iPap trial, the overall follow-up rate amongst those who required it was substantial at 84%. However, when stratified by recommendation, follow-up was higher in the cytology triage group than in the direct colposcopy group (90% vs. 70%), which is in contrast to the finding of the systematic review, where most studies with direct referral had a higher follow-up rate than those with a triage policy. 2 This may be explained by the fact that a substantial proportion of HPV 16/18 positive women with negative cytology (9/23; 40%) in our study were not referred by their practitioners for colposcopy. One potential reason for this could be that the laboratory reporting the triage cytology may have recommended a 2-year repeat cytology (as was standard practice at the time) without taking into account the HPV result. This was despite communication about the recommendation for colposcopy to both general practitioners and the affected woman by letter and phone from liaison physicians at VCS Pathology. Direct referral for colposcopy following a positive 16/18 HPV test was a major change for general practice but one which is now becoming understood with the renewed HPV-based program. 13
Cross-sectional and longitudinal follow-up studies in the past have found HPV 16 to be associated with a higher risk of developing CIN2 + /CIN3 + lesions compared to other oncogenic types. 14 The histologically confirmed high-grade lesion detection was much higher for the 16/18 positive women than for those with non-16/18 HPV types (24% [8/33 women who attended colposcopy] vs. 4% [4/93 who attended for cytology and follow-up]) in our study and well above the typical threshold for colposcopy referral. In Australia, the HPV-based screening program routinely uses partial genotyping to differentiate those with HPV16/18 from other oncogenic HPV types, with more intensive pathways for those who are 16/18 positive (direct colposcopy) than others (cytology triage). In the renewed program, women who are HPV positive for non-16/18 type with negative or low-grade cytology are asked to have a repeat HPV test at 12 months to confirm viral clearance. Very few studies to date have examined 12 months’ follow-up after a negative/low-grade triage test In two studies from the Netherlands,11,15 57–58% women complied with 12 months’ follow-up, which is similar to the follow-up rate found in our study (59%) but markedly lower (>80%) than amongst women routinely attending for a follow-up test following a low-grade result as part of the previous cytology-based program in Victoria (unpublished data, VCSR 2015). The low follow-up rate could be due to most women having had a negative cytology on a smear taken by a practitioner which would result in the usual laboratory recommendation of a 2-year repeat. This suggests that appropriate education and communication strategies for women, practitioners and laboratories are required to ensure the recommended clinical pathways for women utilising the self-collection pathway are understood. Because women in the mainstream HPV testing program also require a 1-year repeat for non-16/18 infection in the context of a negative/low grade cytology result, this change in routine practice should reduce the aforementioned barriers.
Notably, about 70% of HPV + women tested positive for HPV again at 12 months, suggesting that never and under-screened women may be, as expected, more likely to have long-standing persistent HPV infections already associated with lesions that do not resolve over the 12-month period. The overall detection rate of CIN2 + for never- and under-screened women aged 30 to 69 years in this study was 8.5 (95% CI: 4.8–13.9) per 1000 women screened, which is higher than the estimate of 5.4 per 1000 women screened (5.2–5.7) for all Victorian women aged 30–69 years (unpublished data, VCSR 2015). However, loss to follow-up for the 12 months’ repeat test was substantial and the yield of CIN2 + lesions in this study is likely an underestimate. A higher yield of CIN2 + was also found in under-screened women who participated in a trial of self-sampling when compared to regular screeners in the Netherlands [relative risk of CIN2 + was 1.6 (95% CI:1.4–1.9)]. Cervical carcinomas were also more frequently detected in self-sampling responders than regular screening participants in this Dutch study (0.09% vs. 0.03%; p = 0.002) indicating that these women are at increased risk compared to regular screeners. 16 It is, however, encouraging that none of the women who tested negative in the initial round were diagnosed with high-grade disease on subsequent cytology, indicating the expected very low risk following an HPV negative result.
Strengths
The iPap trial was conducted in the context of over two decades of organised national screening and was managed using the VCSR, which has near complete coverage of cervical cytology, histology and self- and practitioner-initiated HPV tests, as required by the renewed program, thereby enabling us to capture the relevant outcomes in the trial accurately and efficiently in the 36 months’ follow-up. 17 Specimens were processed by VCS Pathology, which provides half of cervical screening tests in Victoria and is currently one of the two laboratories in Australia accredited to test self-collected samples. We used the dry flocked swab for self-collection and the cobas® 4800 HPV test, a test that is clinically validated and Therapeutic Goods Administration (TGA) approved (a requirement in Australia). 18 We were also able to look at participation over two rounds of screening and describe the characteristics of women who would rescreen. The long duration of follow-up time also allowed us to determine secondary end points, especially women with a positive other HPV result with negative or low-grade cytology.
Limitations
A major limitation of this study is that we could not further match and update cytology results for our never-screened group who did not participate (in a year's time) and were therefore unable to make comparisons with the control arm or report updated Pap test results (post 6 months) in the self-sampling arm in the never-screened group.
Conclusion
Never-screened women were more likely to participate in self-sampling than under-screened women. Overall compliance to follow-up at baseline was also high for these women. However, loss to follow-up of HPV-positive women without abnormal cytology who were advised to repeat an HPV test at one year was substantial. As self-collection becomes a routinely used cervical screening approach, monitoring of compliance with recommended follow-up will be important to ensure its full potential for disease prevention is realised. A key advantage of the clinician-supported approach to self-sampling being used in the Australian NCSP, as opposed to the mailed kit approach, is the opportunity for clinicians to directly support women, encouraging their ongoing engagement with the program including follow-up tests where necessary.
Footnotes
Acknowledgements
FS acknowledges Cancer Council Victoria for the Postdoctoral Research Fellowship during which time the study was conceived. We thank the VCS Digital Health team for linkages and database management; VCS Pathology staff, particularly Dr David Hawkes, for managing laboratory processes; VCS data processing officers for handling incoming forms and phone calls; and the VCS Liaison Physicians for follow-up of HPV positive women. JAS is funded by a NHMRC Senior Research Fellowship (1104975).
Author Contributions
DG was the principal investigator of the study and was responsible for the overall conduct of the study. JMLB had supervising oversight of the conduct of the follow-up stage of the study presented in this paper. FS developed the first draft of the study protocol that was further developed by DG, DE, FS, JAS, JMLB and MS. DG, DE, MS, JMLB, JAS and FS were primarily responsible for the design of the study, with input from all authors. FS was a PhD student doing her PhD on the topic and was responsible for scientific coordination of the trial, statistical analysis and manuscript preparation, with oversight from DE, DG, JAS, JMLB and MS. KD managed the operational coordination of the study. MS oversaw the laboratory testing for self-sampled HPV, reporting of results and laboratory quality assurance. DW and SH provided clinical advice on follow-up of women with positive results. All authors read and approved the final manuscript.
Ethics Approval and Consent to Participate
The study was approved by the Human Research Ethics Committee (HREC) of the Victorian Department of Health, approval number 05/13. Informed consent was waived for the study because it was primarily a trial of participation in screening, and we wanted the results to be directly translatable to the screening program at the time of the study.
The study was conducted in accordance with the ethical standards of the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Data Availability
Study data are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
JMLB and MS are investigators on a trial of cytology and primary HPV screening in Australia (Compass) that have received equipment and funding contribution for Compass from Roche Molecular Systems and Ventana Inc USA. No funding from Roche was received for the purpose of the iPap trial.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The trial was funded by the National Health and Medical Research Council (Grant Number: 1045346).
