Abstract
Objective
Digital health care offers an opportunity to scale and personalize cancer screening programs, such as mailed outreach for colorectal cancer (CRC) screening. However, studies that describe the patient selection strategy and process for CRC screening are limited. Our objective was to evaluate implementation strategies for selecting patients for CRC screening programs in large health care systems.
Methods
We conducted a systematic review of 30 studies along with key informant surveys and interviews to describe programmatic implementation strategies for selecting patients for CRC screening. PubMed and Embase were searched since inception through December 2018, and hand searches were performed of the retrieved reference lists but none were incorporated (n = 0). No language exclusions were applied.
Results
Common criteria for outreach exclusion included: being up-to-date with routine CRC screening (n = 22), comorbidities (n = 20), and personal history (n = 22) or family history of cancer (n = 9). Key informant surveys and interviews were performed (n = 28) to understand data sources and practices for patient outreach selection, and found that 13 studies leveraged electronic medical care records, 10 studies leveraged a population registry (national, municipal, community, health), 4 studies required patient opt-in, and 1 study required primary care provider referral. Broad ranges in fecal immunochemical test completion were observed in community clinic (n = 8, 31.0–59.6%), integrated health system (n = 5, 21.2–82.7%), and national regional CRC screening programs (n = 17, 23.0–64.7%). Six studies used technical codes, and four studies required patient self-reporting from a questionnaire to participate.
Conclusion
This systematic review provides health systems with the diverse outreach practices and technical tools to support efforts to automate patient selection for CRC screening outreach.
Keywords
Introduction
Colorectal cancer (CRC) is the second leading cause of cancer deaths in the United States. 1 Despite the increase in CRC screening programs around the world and evidence that the fecal immunochemical test (FIT) is a highly effective and commonly used screening method, 2 population-level CRC screening could still be greatly improved through increased efforts in population reach, personalization of testing, and integration of interventional research outreach.3,4
With concerns over screening rates 5 and the digitization of health records leading to accountable precision population health, 6 there remain opportunities for large health systems that have not yet established a CRC screening program, and as a checklist for those where CRC screening is already established, to assess the comprehensiveness of their system by responding to standardized quality metrics to improve strategies for CRC screening. 7
Several clinical trials, systematic reviews, and meta-analyses have identified organized outreach and FIT kit mailing as the most effective strategy.8–16 However, there is limited data on how patients are selected, including the implementation strategy applied and technical procedural codes used.17,18 A systematic review evaluating the patient selection process with key informant interviews may help improve organized CRC screening.
Our objective was to evaluate implementation strategies for selecting patients for CRC screening programs in large health care systems. To examine this issue, we performed a systematic review and key informant interviews to describe the factors used to exclude patients from a population-based CRC screening program.
Methods
We performed a systematic review according to the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines.
Data source and literature searches
We developed our search strategy with a medical librarian (EW) using keywords for immunochemical based fecal tests and cancer screening (Supplemental Table 1). We searched PubMed and Embase until 31 December 2018. This systematic review was conducted according to the methods described in the Cochrane Handbook for Systematic Review of Interventions and the PRISMA standard. A review protocol was registered a priori through PROSPERO (CRD42018114370). This study was registered immediately after preliminary searches were conducted but before the main search was conducted.
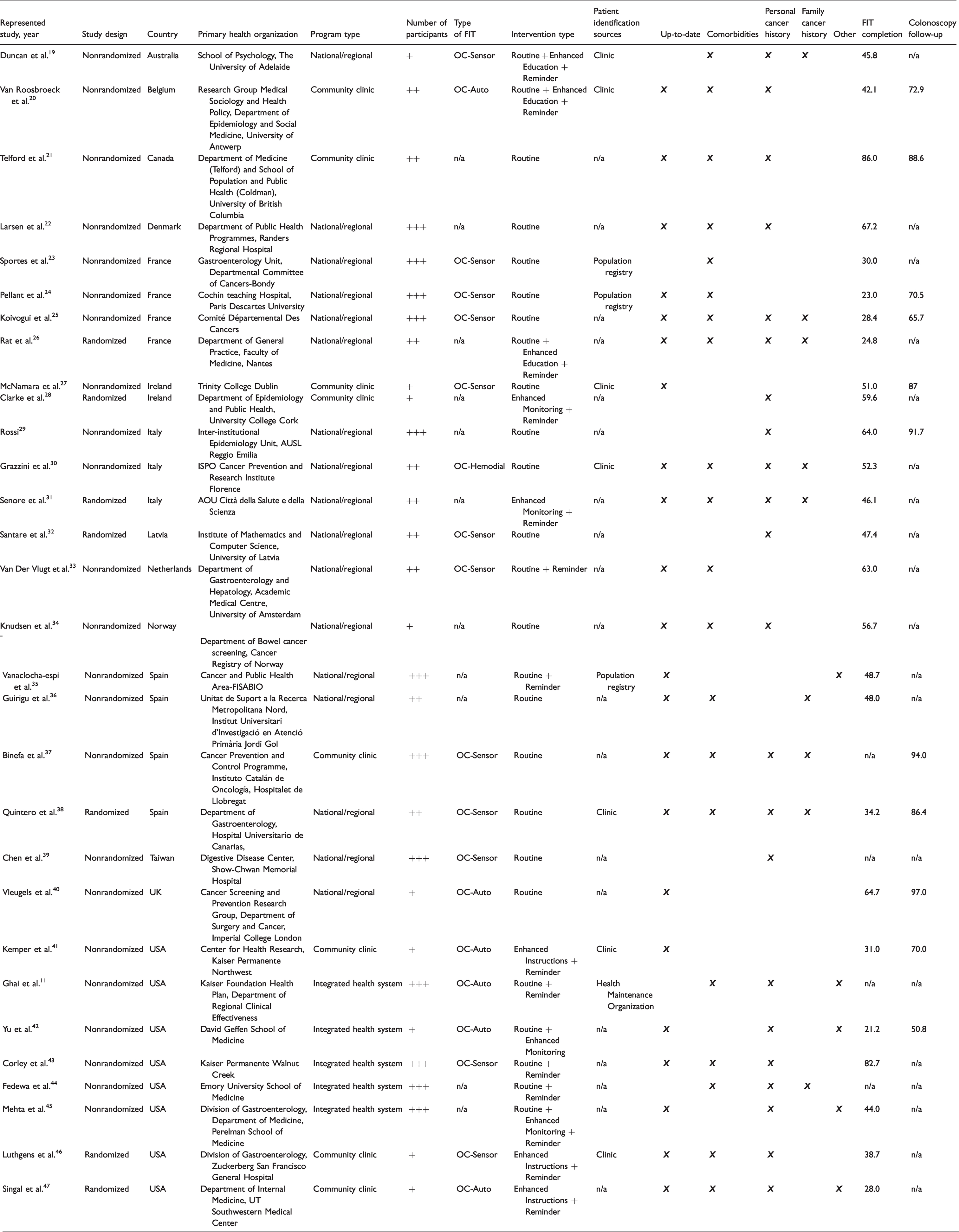
Characteristics of the included studies performing mailed FIT outreach.
Study eligibility and selection
We sought to evaluate studies with details on how patients were selected for mailed outreach CRC screening programs in community-based health care systems. The reviewers (AW, BL) appraised the pertinent studies to determine eligibility and studies were included if they: (1) used mailed FIT or immunochemical fecal occult blood test, (2) reported > 5000 patients (large CRC screening program). To reduce heterogeneity, large CRC screening programs were selected for the purposes of population screening automation as compared to the individualized selection process in smaller CRC screening programs. Included articles were grouped by the corresponding author’s affiliate health organization. The most recent article to date of each institute was selected for descriptive analysis. We included randomized controlled trials and nonrandomized controlled trials. Non-English language articles were translated through the publisher’s website or Google translator but none were included. We excluded studies using guaiac fecal occult blood test 2 (i.e. a different stool test using three samples with a lower compliance rate 48 ), out-of-scope review articles, population surveys, simulation model, and conference abstracts without accompanying full manuscripts.
Data abstraction
Our search strategy is shown in Supplemental Table 1. Titles and abstracts were evaluated for initial screening. Full text articles were assessed for eligibility and subsequent abstracted for study inclusion. At all stages, the reviewers (AW, BL) independently reviewed and abstracted data from the included studies into a Microsoft Excel Spreadsheet (version 2016; Microsoft, Redmond, WA, USA). Information was abstracted on article information, country, primary health organization of corresponding author, program type, number of study patients, FIT brand, intervention type, patient identification sources, patient exclusion criteria, FIT completion rate, and colonoscopy follow-up after positive FIT completion rate. Any disagreements in eligibility and abstraction were resolved through discussion (κ = 0.68, 83% of agreement, substantial agreement).
Data synthesis
Mailed FIT articles were summarized according to study design, program type (national or regional involvement, community clinic local involvement, integrated health system/integrated managed care consortium involvement), country, number of participants (+ = 5000–9999, ++ = 10,000–50,000, +++ = >50,001), type of FIT, outreach intervention type (routine screening, enhanced instructions, enhanced monitoring, enhanced education, added reminder communication), and source of patient selection. Program types were considered to improve study generalizability and utilization applicability. Standard mailed FIT kits included notification to participate, brief education pamphlet, FIT device, and manufacturer FIT instructions. Routine screening was defined as annual or biennial testing depending on the accepted practice standards in that country. Enhanced instruction was defined as tutorials, pictorials, low-literacy wordings. Enhanced monitoring was defined as additional navigators and tracking systems for patients. Enhanced education was defined as low-literacy wordings, and psychosocial and racial ethnic modifications. Added reminder communication was defined as the addition of mail, email, text message, or phone call reminders to patients to complete screening. Mailed FIT studies with additional interventional components did not lead to exclusion of the study.
Patient exclusion criteria were abstracted from each article. They were determined a priori (established by a preliminary search) and categorized into the following groups: comorbidities, personal or family history of CRC-related conditions, or uncategorized. Comorbidities included CRC-related symptoms (blood in stool, bowel obstructions), inflammatory bowel disease, institutionalization, and terminal diseases. Being up to date with routine CRC screening included colonoscopy in the prior 5–10 years, sigmoidoscopy in the prior 5 years, FIT in prior year, positive FIT, and colectomy. Personal or family history of related CRC conditions included familial adenomatous polyposis, hereditary nonpolyposis cancer, and other cancers.
Risk of bias was not assessed as the outcomes would not be affected by the study’s quality.
Key informant surveys and interviews
Key informant surveys and interviews were performed by emailing the corresponding authors of included articles (Supplemental Tables 5 and 6). The online email survey included questions on program type, location, patient identification source, patient inclusion and exclusion criteria, and technical selection codes (if any) (Supplemental Table 7). A standard e-mail template was followed, which invited corresponding authors to participate. Authors had two weeks to respond before a final reminder email was sent (Supplemental Table 8).
Analytical plan
A mixed methods approach was taken to gather study data. The review focused on summarizing study characteristics, patient identification and selection, FIT completion and CRC follow-up participation, and key informant surveys from articles with patient exclusions. Characteristics, the selection process, and outreach surveys were described as counts and proportions. Participation was described in ranges. Technical procedure codes (International Classification of Disease (ICD), Current Procedural Terminology (CPT), Healthcare Common Procedure Coding System (HCPCS), Logical Observation Identifiers Names and Codes (LOINC)) were summarized by patient exclusion categories. Due to the high risk of bias over differences in study characteristics, a meta-analysis was not performed. Our analysis approach was to focus on implementation strategies in mailed FIT outreach that accounted for individual patients, and contextualizing the risks (exclusion criteria) of the screening process. Any disagreements in synthesis and analysis were resolved through discussion and expressed as Cohen’s kappa (κ).
Compliance with ethical standards
All authors had access to the study data and reviewed and approved the final article. This study was exempt. The authors have no conflicts of interest to declare.
Results
Summary of literature search and study selection
After removal of duplicate records, the search identified 2081 articles, of which 434 full-text articles were evaluated (Figure 1). A total of 72 reports remained after stand-alone abstracts, commentaries or guideline articles, and duplicates were excluded. Articles that did not distribute FIT by mail, were not relevant, or were simulation models, systematic or meta-analysis reviews, or proposal articles were also excluded. To prevent duplication of results from the same CRC screening program, we included the most recent article from centers with multiple publications on the same cohort (Supplemental Table 3). After limiting articles to those with more than 5000 participants, 43 articles remained. Thirty articles contained documentation of patient exclusion criteria (Table 1) and 13 articles did not (Supplemental Table 2). Of the articles with no patient exclusion, 11 studies were from national and regional programs, and 8 studies used population registries as the source of patient outreach.
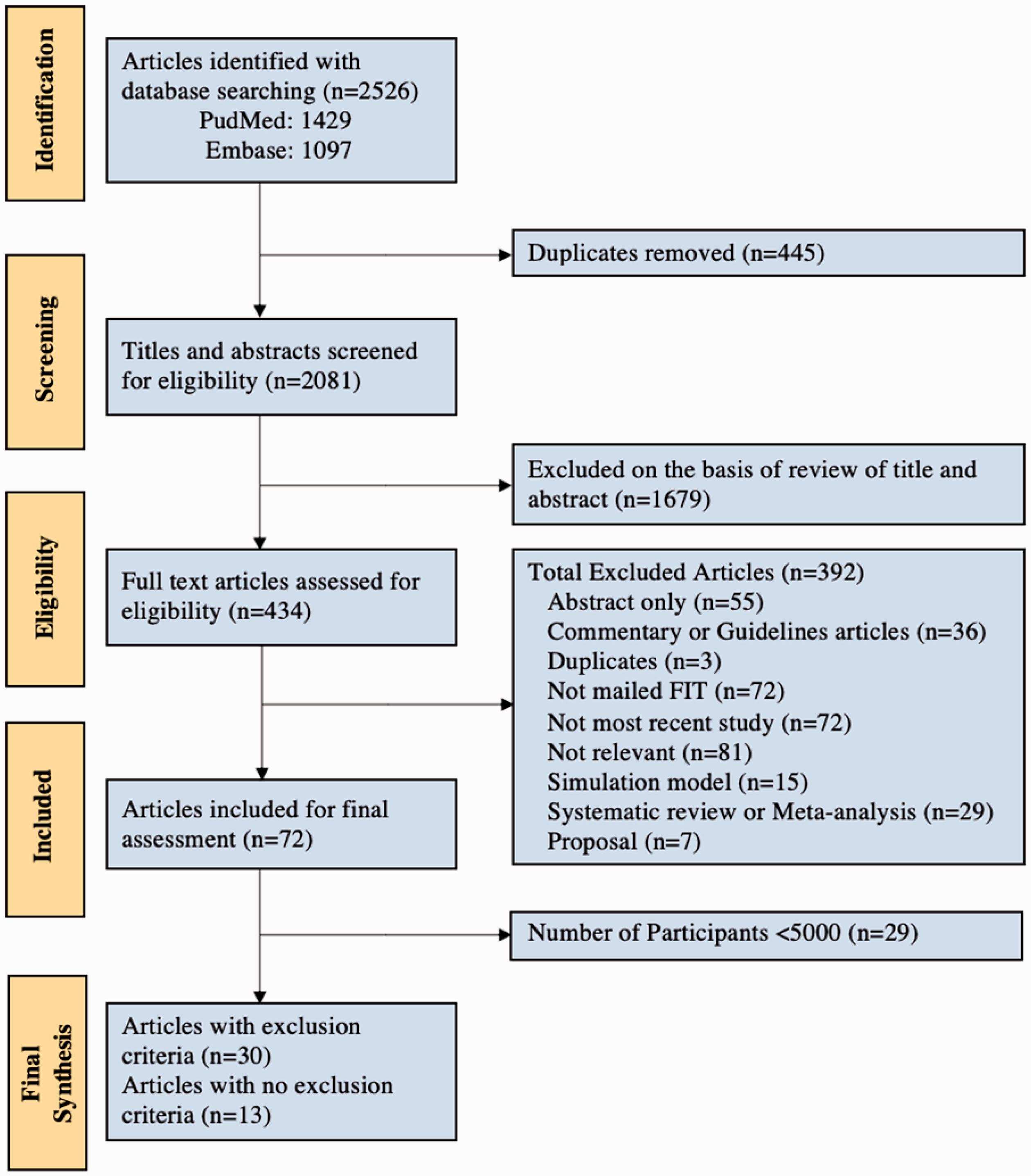
Flow chart of study selection to evaluate implementation strategies that select patients for colorectal cancer screening program in community-based health care systems.
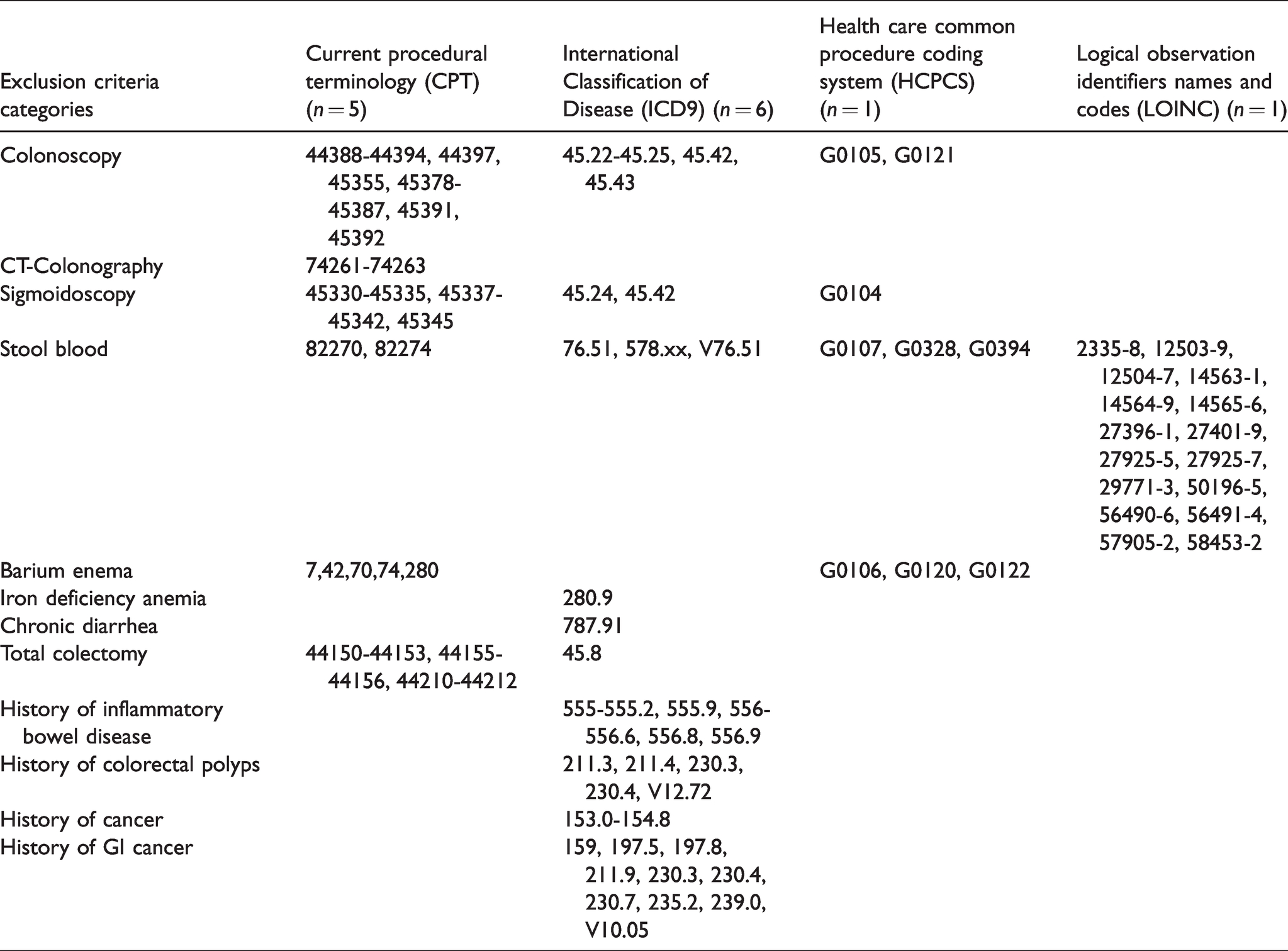
Technical codes identified and used to optimize patient selection for colorectal cancer screening.
Characteristics of mailed FIT programs with patient exclusion
Of the 30 articles (22% missing data, “n/a”) that contained documentation describing patient exclusion (Table 1), 7 were randomized studies. Fourteen countries were represented and the majority of studies were from the United States (n = 8), France (n = 4), Spain (n = 4), and Italy (n = 3). Some countries despite having well-established CRC programs were not included if selection criteria was not defined (Supplemental Tables 2 and 3). Other excluded studies did not meet study inclusion criteria (Figure 1) or had no published reports.
In the subset of large CRC screening programs that utilized a mailed FIT approach with patient exclusions, 17 studies were at the national or regional level, 8 studies at the community clinic level, and 5 studies at the integrated health organization level. The number of patients in the screening program ranged in size: 5000 to 9999 (n = 9), 10,000 to 49,999 (n = 9), 50,000+ patients (n = 12). The cohort used a variety of FIT kits: OC-Sensor (n = 12), OC-Auto (n = 8), OC-Hemodial (n = 1), or not reported (n = 11).
Study outreach interventions were diverse. Integrated health organizations and community clinics were more likely to incorporate other interventions in addition to mailed FIT outreach (Table 1). Among community clinics (n = 8), mailed FIT interventions varied with each program using one or a combination of the following: no additional intervention (n = 4), reminder (n = 5), enhanced monitoring (n = 1), enhanced education (n = 1), enhanced instructions (n = 3). Among integrated health organizations (n = 5), interventions included one or a combination of the following: routine (n = 5), reminder (n = 4), enhanced monitoring (n = 2). Finally, among national or regional CRC programs (n = 17), interventions included: routine (n = 16), reminder (n = 5), enhanced monitoring (n = 1), enhanced education (n = 2).
Patient identification and selection in programs with patient exclusion
Community-based programs (n = 8) used various methods for patient outreach and selection (Table 1). Four studies removed patients up to date with CRC screening, and six studies utilized a personal cancer history, five studies incorporated patient comorbidities, and one study used family history of cancer as reasons to exclude individuals from receiving FIT mailing. Generally, smaller community-based programs (n = 4) relied on their own electronic medical care records (EMR) or individual or primary care provider/general practitioner (PCP/GP) selection (n = 2) as the source for patient identification, to which they applied subsequent exclusionary criteria.
Integrated health systems (n = 5) used similar methods for patient outreach and selection (Table 1). Three studies removed patients up to date with CRC screening, and five studies utilized a personal cancer history, three studies incorporated patient comorbidities, and one study used family history of cancer as reasons to exclude individuals from receiving FIT mailing. Integrated health organizations often had a large number of patients participating (>50,000, n = 4), and only one study reported relying on internally linked shared EMR as the source for patient identification across the consortium of health clinics in the area.
National or regional programs (n = 17) used similar methods for patient outreach and selection (Table 1). Twelve studies removed patients up to date with CRC screening, and 11 studies utilized a personal cancer history, 12 studies incorporated patient comorbidities, and 7 studies used family history of cancer as reasons to exclude individuals from receiving FIT mailing. The vast majority of these programs had a higher number of patients participating (>10,000, n = 14). While national or regional programs included three studies that utilized population registries and four studies that utilized local clinics for indiscriminant mass screening, patients were often sent informative leaflets (n = 4) asking them to “self opt-out” if they did not qualify. After inclusion, few programs (n = 2) verified a patient’s eligibility through EMR or surveys.
Participation in programs with patient exclusion
From the 30 studies included, broad ranges in FIT completion (n = 26) and subsequent (n = 11) colonoscopy follow-up were observed. National and regional programs were more likely to have higher median participation rates (Table 1) and reported FIT completion also varied. Completion of mailed FIT in community-based programs (n = 7) ranged from 31.0% to 59.6%, in integrated health systems (n = 3) from 21.2% to 82.7%, and in national or regional programs (n = 16) from 23.0% to 64.7%. In studies with reported colonoscopy follow-up after abnormal FIT, the results were: community-based programs (n = 5, 70.0–94.0%), integrated health systems (n = 1, 50.8%), and national or regional programs (n = 5, 65.7–97.0%).
Key informant surveys and interviews
Of the 28 studies (65% response rate) that responded to the survey and interview inquiry (Supplemental Table 4), responses were from individuals representing national (n = 11) and integrated health systems (n = 17). Corresponding authors identified patients for FIT outreach based on data obtained from the following sources: EMR (n = 13), population registries (n = 10), patient opt-in (n = 4), and PCP referral (n = 1). In total, six studies reported utilizing technical codes (all integrated health systems), seven studies required self-reporting from a questionnaire to participate (n = 3 national, n = 4 integrated health systems), and fifteen studies did not further elaborate on the selection process.
The common technical codes gathered from key informant outreach used to identify patients for outreach are outlined (and defined) in Table 2: colonoscopy (CPT 44388–44394, 44397, 45355, 45378–45387, 45391, 45392; ICD9 45.22–45.25, 45.42, 45.43; HCPCS G0105, G012), CT colonoscopy (CPT 74261–74263), sigmoidoscopy (CPT 45330–45335, 45337–45342, 45345; ICD9 45.24, 45.42; HCPCS G0104), stool blood (CPT 8,22,70,82,274; ICD9 76.51, 578.xx, V76.51; HCPCS G0107, G0328, G0394; LOINC 2335–8, 12503–9, 12504–7, 14563–1, 14564–9, 14565–6, 27396–1, 27401–9, 27925–5, 27925–7, 29771–3, 50196–5, 56490–6, 56491–4, 57905–2, 58453–2), barium enema (CPT 7,42,70,74,280; HCPCS G0106, G0120, G0122), iron deficiency anemia (ICD9 280.9), chronic diarrhea (ICD9 787.91), total colectomy (CPT 44150–44153, 44155–44156, 44210–44212; ICD9 45.8), history of inflammatory bowel disease (ICD9 211.3, 211.4, 230.3, 230.4, V12.72), history of colorectal polyps (ICD9 153.0–154.8), and history of gastrointestinal cancer (ICD9 159, 197.5, 197.8, 211.9, 230.3, 230.4, 230.7, 235.2, 239.0, V10.05). While two studies reported utilizing ICD10, codes were unspecified and not incorporated.
Discussion
While CRC screening rates have improved globally, they still remain suboptimal and the COVID-19 pandemic has now stalled in-person screening efforts. This review, describing the use of population registries and electronic records, offers an opportunity for health systems to transform from opportunistic screening to population-level screening, which has the potential to reduce CRC incidence and mortality. As health records become increasingly digitized, using algorithmic metrics to identify patients for CRC screening is an important first step to improving precision population health.6,7 To our knowledge, our systematic review is the first to describe the methods by which screening programs identify and select patients for mailed FIT outreach programs. We show that while national or regional CRC screening programs typically rely on population registries for patient self-reported exclusion or direct GP/PCP recruitment, community clinics and integrated health organizations use internal electronic health records to select patients for screening. In addition, there are many large CRC screening programs around the world that use cancer history and comorbidities as exclusionary criteria.
Organized screening programs in large health care systems have been shown to increase participation, improve patient handling of FIT, reduce disparities, reduce potential harms of screening, and reduce the overall care costs.49–51 And multiple studies have demonstrated the effectiveness and acceptable cost of mailed FIT outreach.8–16,49 In the United States, population health entities within integrated health systems have arisen due to the adoption of electronic health records. To date, they often serve to report on the quality of care in order to obtain payment incentives. However, through the data infrastructure, population health entities should also transition to provide clinical services that improve the health of populations. In this review, we also identified multiple publications from Kaiser Permanente in different regions of the United States; they have previously described a centrally organized CRC screening model that includes mailed FIT kits.11,12 As digitization of health and centrally managed mailed FIT programs become more widespread, these population health entities can enhance overall health care maintenance and cancer prevention. Therefore, a concerted effort should be made to improve tailored prevention with the goal of refining patient selection criteria for a more personalized and cost-effective outcome. 50 Specifically, to ensure trust between health systems, providers, and patients, organized outreach should offer screening to patients whose provider would have also intended to screen. In this review, while we identified 30 articles and ascertained each of their patient eligibilities with variable cohort definitions, the implementation strategies used for mailed FIT outreach were lacking. For example, while we suspect most studies used algorithmic code to identify patients for CRC screening, only one study specified ICD/common procedural codes.
While implemented CRC screening practices have historically limited patients by age, risk, and lack of symptoms, 2 this study shows that health organizations often implement different methods for identifying patient comorbidities or history of cancer-related conditions, leading to inconsistent CRC screening participation. 51 Moreover, the implementation of screening is markedly different across regions and countries. While not covered in this review, health care systems can proactively incorporate data elements used for Healthcare Effectiveness Data and Information Set (HEDIS) performance measures for CRC screening to develop population registries for targeted screening. As an example, the most recent HEDIS measure and technical resource provides guidance on excluding patients from the CRC report when there is use of palliative care or the medication donepezil for dementia. This review summarizes the patient selection criteria and the technical codes that exist in different health care settings, to inform health systems considering implementing mailed FIT outreach.
There are several limitations to this study. First, few studies report patient selection criteria or systems used to identify patients for outreach. Different health organizations may have had internal practices; however, the specified metrics of patient identification and acquisition used in varying clinic practices were not articulated in the manuscript or through contact with the authors. Second, there is a potential for section bias as we only described a subgroup of studies that utilized mailed FIT and published their data. We are aware that multiple national programs exist but published data, along with the specified processes around selection of patients for screening, were not publicly available. We may have also missed other patient selection processes because we excluded smaller studies (<5000 participants) from our review. From a systems implementation perspective, we excluded small studies because these were more likely to involve patient selection processes that were not economical (e.g. consent of patients, chart review, permission from provider) for population health screening automation. Finally, we cannot directly compare or perform a meta-analysis on screening outcomes with respect to selection criteria due to the heterogeneity in targeting populations for screening. Difference in screening outcomes may be of interest but cannot be meaningfully compared given the differences between the individual CRC screening programs.
In conclusion, our systematic review with key informant interviews describes the patient selection criteria and implementation strategies of 30 studies. We found that large CRC screening programs may use heterogenous methods for excluding patients for FIT outreach. This systematic review sought to provide health systems with the technical tools to support efforts to automate patient selection for CRC screening outreach. These efforts are particularly timely given the COVID-19 pandemic, which has increased concern for in-person visits and has accelerated the adoption of telehealth and organized outreach services. Optimizing the patient identification process and selection criteria can strengthen preventive care services, improve patient outcomes, and reduce cost.
Supplemental Material
sj-pdf-1-msc-10.1177_0969141321997482 - Supplemental material for Selection of patients for large mailed fecal immunochemical test colorectal cancer screening outreach programs: A systematic review
Supplemental material, sj-pdf-1-msc-10.1177_0969141321997482 for Selection of patients for large mailed fecal immunochemical test colorectal cancer screening outreach programs: A systematic review by Andrew Wang, Briton Lee, Shreya Patel, Evans Whitaker, Rachel B Issaka and Ma Somsouk in Journal of Medical Screening
Footnotes
Authors’ contribution
Andrew Wang – drafting of the article; acquisition of data; interview outreach; analysis and interpretation of data; and critical revision of the article. Briton Lee – acquisition of data. Shreya Patel – acquisition of data; interview outreach; analysis and interpretation of data; and critical revision of the article. Evans Whitaker – technical or material support; study concept and design (search terms). Rachel B Issaka – study concept and design; critical revision of the article. Ma Somsouk – principal investigator; study concept and design; interview outreach; analysis and interpretation of data; critical revision of the article; and study supervisor.
Acknowledgments
The authors are grateful to Robert Hiatt, MD, PhD and David Glidden, PhD (Department of Epidemiology and Biostatistics, University of California, San Francisco, CA, USA) for their insightful feedback and continued support of the study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the San Francisco Cancer (SF CAN) Initiative, a collaborative community effort initiated by the UCSF HDFCCC to reduce the cancer burden across the city and beyond [![]() ]. Dr Issaka was supported by the National Institutes of Health/National Cancer Institute award number K08 CA241296.
]. Dr Issaka was supported by the National Institutes of Health/National Cancer Institute award number K08 CA241296.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
