Abstract
Aim
To determine the diagnostic accuracy of colon capsule endoscopy for colorectal cancer screening.
Methods
Studies that compared the diagnostic performance of colonoscopy and second-generation colon capsule endoscopy (CCE-2) for screening of asymptomatic patients aged 50–75 years were included. The primary outcomes were sensitivity, specificity, and positive and negative likelihood ratios for polyps and adenomas measuring at least 6 mm or 10 mm.
Results
Eight full-text studies that evaluated 1602 patients were included for systematic review. Of these, 840 (52.43%) patients participated in an opportunistic screening program. The pooled outcomes of CCE-2 for polyps at least 6 mm / 10 mm were (CI = confidence interval): sensitivity: 88% (95% CI: 0.84–0.91) / 88% (95% CI: 0.82–0.93), specificity: 94% (95% CI: 0.92–0.95) / 95.5% (95% CI: 0.94–0.97); positive likelihood ratio: 11.86 (95% CI: 5.53–25.46) / 23.07 (95% CI: 6.163–86.36); negative likelihood ratio: 0.14 (95% CI: 0.1–0.21) / 0.14 (95% CI: 0.09–0.21). The area under the summary receiver operating characteristic curve for polyps at least 6 and 10 mm was 96.3% and 96.7%, respectively. The only cancer missed by complete CCE-2 was shown at multiple frames in the unblinded review. In total, 125 (7.8%) patients presented mild adverse events mostly related to bowel preparation.
Conclusion
CCE-2 is demonstrated to be an effective and safe alternative method for colorectal cancer screening. Diagnostic performance of CCE-2 for polyps of at least 6 and 10 mm was similar. Completion rates still need to be improved.
Introduction
Compliance to colorectal cancer screening remains suboptimal. Even though screening is feasible for colorectal neoplasia, it represents the second leading cause of cancer death globally, and its incidence continues to increase in developing nations. 1 Several studies have shown that colorectal cancer increasingly affects younger patients in the USA, the UK and Europe,2,3 suggesting the need for a lower age to start screening, of 45 years old in a recent guideline. 4 This recommendation enlarges the proportion of patients who require a screening investigation, potentially increasing the unmet demands on endoscopy resources.
Conventional colonoscopy (CC) represents the gold standard method for detection and resection of early colorectal neoplasia. However, several barriers limit patient adherence: the perceived invasive nature of the method, fear of discomfort, need for sedation and risk of complications. 5 Other technologies have emerged as potential screening and diagnostic approaches. 6 Colon capsule endoscopy (CCE) was first introduced in 2006 as a promising non-invasive, painless, patient-friendly technology that allows colon inspection without sedation or insufflation. 7 Technological modifications were applied to create a second-generation capsule (CCE-2) that has double-headed lenses, longer recording capacity, and an adaptive frame rate that allows acquisition of images at between 4 and 35 frames per second according to the speed of travel of the capsule.8,9 This device has demonstrated improved sensitivity and accuracy for the detection of colorectal neoplasia and feasibility as an out-of-clinic test. 10
The years 2020–21 have been dramatically marked by the Covid-19 pandemic. 11 This has limited endoscopic screening and elective diagnostic procedures, thus increasing the risk for several gastrointestinal disorders including colorectal cancer.12,13 While over 3.9 million deaths related to Covid-19 have been reported, 10 million cancer deaths occurred in 2018, with colorectal and gastric cancer representing 17% of mortality.14,15
European guidelines have suggested the use of CCE-2 within organized screening programs in patients with positive fecal occult blood test (FOBT) or fecal immunochemical test (FIT) in whom colonoscopy is not feasible. 16 Although previous meta-analysis has shown that the accuracy of CCE-2 for detection of colorectal lesions is comparable to that of colonoscopy, 17 the former is not widely accepted as an alternative screening method. Taking into consideration that endoscopic resources for screening are limited, the current difficulty of access to colonoscopy, the potential reluctance of patients to attend medical facilities, and the feasibility of CCE-2 as an out-of-clinic procedure, we question whether CCE-2 could serve as an additional method to effectively facilitate early detection of colorectal lesions. The aim of this study was to determine the diagnostic accuracy of CCE for colorectal cancer screening.
Patients and Methods
This study was structured according to the PRISMA 18 recommendations. 18 It was registered in the PROSPERO international database https://www.crd.york.ac.uk/prospero/ 19 with the study number CRD42020202376.
Literature search
A comprehensive search was conducted in Medline and Embase from 2009 until December 2020 following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines. Peer-reviewed articles published in English that compared the accuracy of second-generation colon capsule and colonoscopy for screening of colorectal cancer were identified. The following search terms were used: “Capsule endoscopy and colon”, “Capsule endoscopy and colorectal”, “Capsule endoscopy and polyp”, “Capsule endoscopy and polyps”. Two authors (MS and WMB) screened the titles and abstracts identified based on predetermined inclusion and exclusion criteria. Any discrepancies were solved by consensus and further analysis by a senior author (EGHM).
Selection criteria
Full-length cross-sectional studies that included average-risk patients who underwent both CCE-2 and colonoscopy for colorectal cancer screening, and with data available for the construction of a 2 × 2 contingence table, were analyzed. Studies involving patients with high risk for colorectal cancer, such as patients with positive family history of colorectal cancer or polyposis syndromes, were excluded. Series of cases with 10 patients or less were not included. We studied the diagnostic accuracy of CCE-2 for screening patients aged 50–75 years with average risk for colorectal cancer.
Data items
Population: asymptomatic patients aged 50–75 and submitted to CCE-2 and colonoscopy for colorectal cancer screening. Index test: second-generation colon capsule endoscopy performed for screening indication. Reference test: optical colonoscopy. Outcomes: sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV). Additionally, polyp detection rate (PDR) was analyzed when available.
Quality of included studies
The quality of included studies was assessed using the QUADAS-2 (Quality Assessment of Diagnostic Accuracy Studies) questionnaire. 20
Summary measures
The primary outcome measures were sensitivity, specificity, and positive and negative likelihood ratios for polyps and adenomas measuring at least 6 mm or 10 mm. Additionally, polyp detection rate was analyzed when available.
Planned methods of analysis
Considering colonoscopy as the reference standard and CCE-2 as the index test, 2 × 2 statistical tables were constructed for each study. Where 0 counts occurred in at least 1 cell of study data, a continuity correction of 0.5 was added to every value for that study to make the calculation of sensitivity and specificity. Based on these data, the sensitivity, specificity, positive likelihood ratio (PLR), and negative likelihood ratio (NLR) (with corresponding 95% confidence intervals [CIs]) of CCE-2 were calculated for polyps and adenomas of at least 6 or 10 mm. Pooled results with corresponding 95%CIs were derived by using the random effects model. Summary receiver operating characteristic curves (SROC) were constructed based on the Moses-Shapiro-Littenberg method. 21 The area under the curve (AUC) was computed as a measure of the overall performance of CCE-2 to accurately identify polyps and adenomas when screening average risk patients for colorectal cancer. A preferred test has an AUC close to 1, and a poor test has an AUC close to 0.5. Cochrane's Q2 test was used to assess heterogeneity, and the I2 statistic was used to measure inconsistency. A value of I2 below 30 % was not considered to be statistically significant. I 2 values of 30 % to 60 % were considered to represent moderate heterogeneity, between 50 % and 90 % substantial heterogeneity, and between 75 % and 100 % considerable heterogeneity. 22 The analysis was performed with Meta-Disc version 1.4 statistical software (Unit of Clinical Biostatistics at the Ramon y Cajal Hospital, Madrid, Spain).
Results
A total of 818 citations were retrieved in the initial search. After removal of duplicates and preliminary review, 51 full-length articles were evaluated for eligibility. Finally, 8 articles were included for systematic review and 7 for meta-analysis. Figure 1
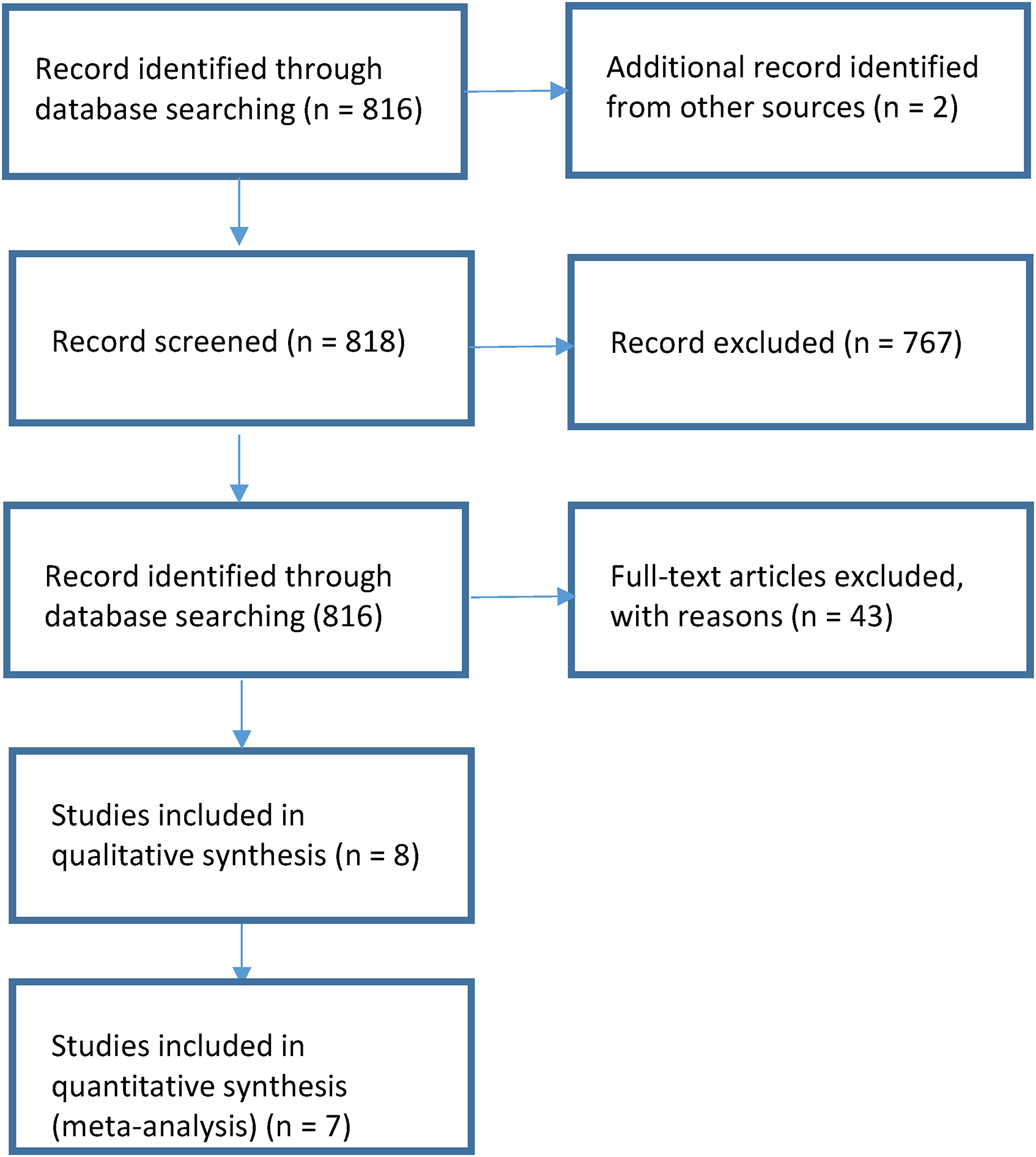
Flow diagram of the process for selecting eligible studies.
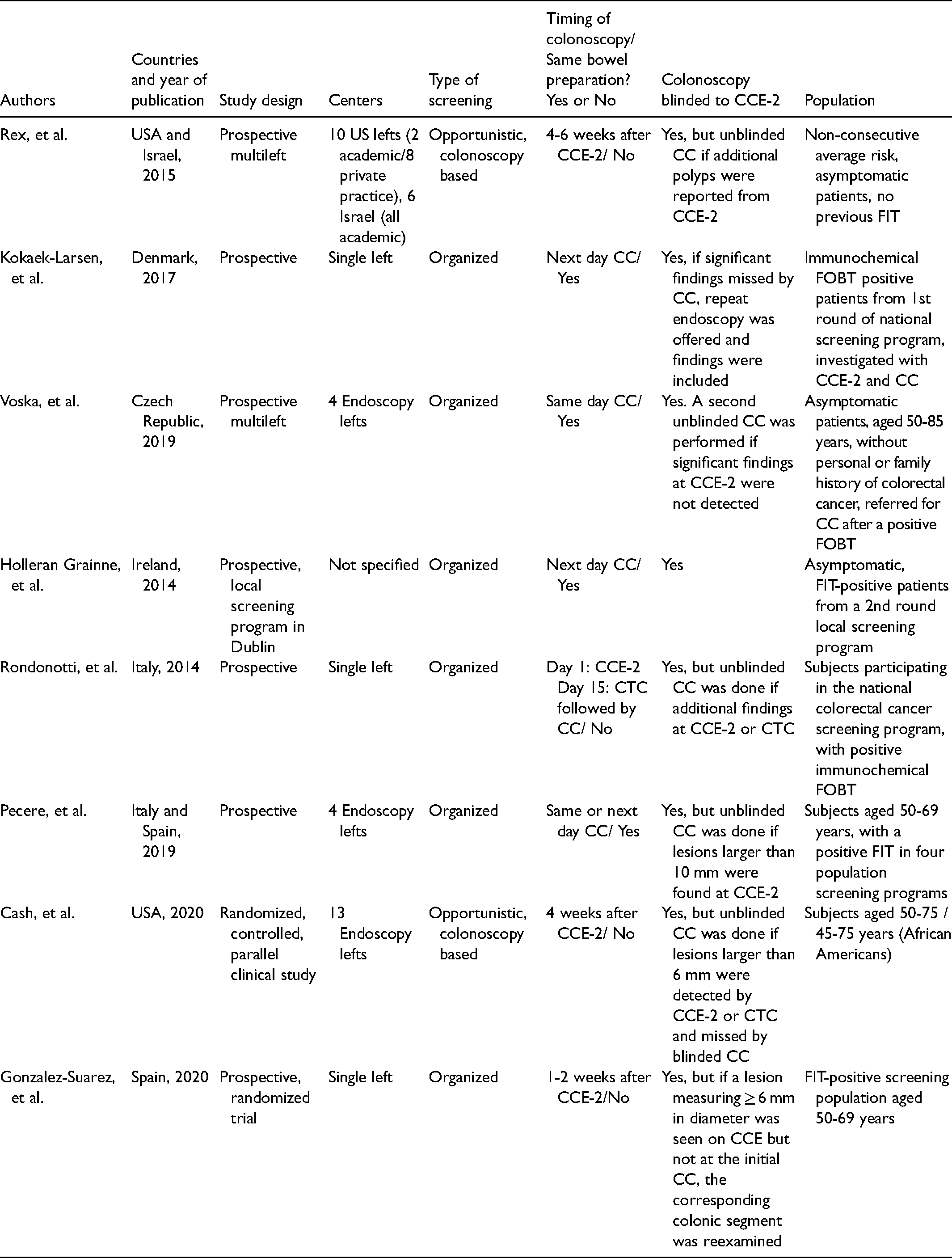
A total of 1602 patients, with age range from 55 to 64 years, underwent CCE-2 and CC for screening. Of these, 840 (52.43%) were from the USA and Israel, evaluated as part of an opportunistic colorectal screening program.23,24 There were 762 patients (47.57%) analyzed in six studies of organized screening in European countries: Denmark, Czech Republic, Ireland, Italy and Spain.25–30 The included studies were published between 2014 and 2020. Four studies performed CCE-2 and CC with a single bowel preparation, either the same day or on two consecutive days.25–27,29 Initially all colonoscopies were blinded to the CCE-2 results. However, if additional pathology was suggested at CCE-2, a second unblinded colonoscopy was performed in 7/8 studies and findings were included for analysis. Timing and matching rule between procedures, blinded colonoscopy results and true positive criteria are further described in Table 1. All but one study used polyethylene glycol solution in variable volumes for bowel preparation with an additional booster.
Patients and methods of included studies.
Overall, the rate of incomplete CCE-2 was 11.58%, ranging from 2% to 46%. Quality indicator of colonoscopies mainly expressed as adenoma detection rate (ADR) was provided in 7/8 studies, range 17.6% to 54.9%. PDR range was 31.6% to 74% for CCE-2 and 27.6% to 64% for colonoscopy. Range of adequate bowel preparation was 54–92% for CCE-2 and 90–100% for CC. No serious adverse events occurred during CCE-2 or diagnostic colonoscopy. Regarding diagnostic performance of CCE-2 for cancer, there was one overlooked case of cancer in complete, blinded CCE-2 that was shown in unblinded exam in multiple frames. 24 In incomplete CCE-2, there were four cancers missed in anatomic regions not shown by CCE-2, in the study by KobaekKobaek-Larsen, et al.. 25 Table 2
Results of systematic review of included studies.
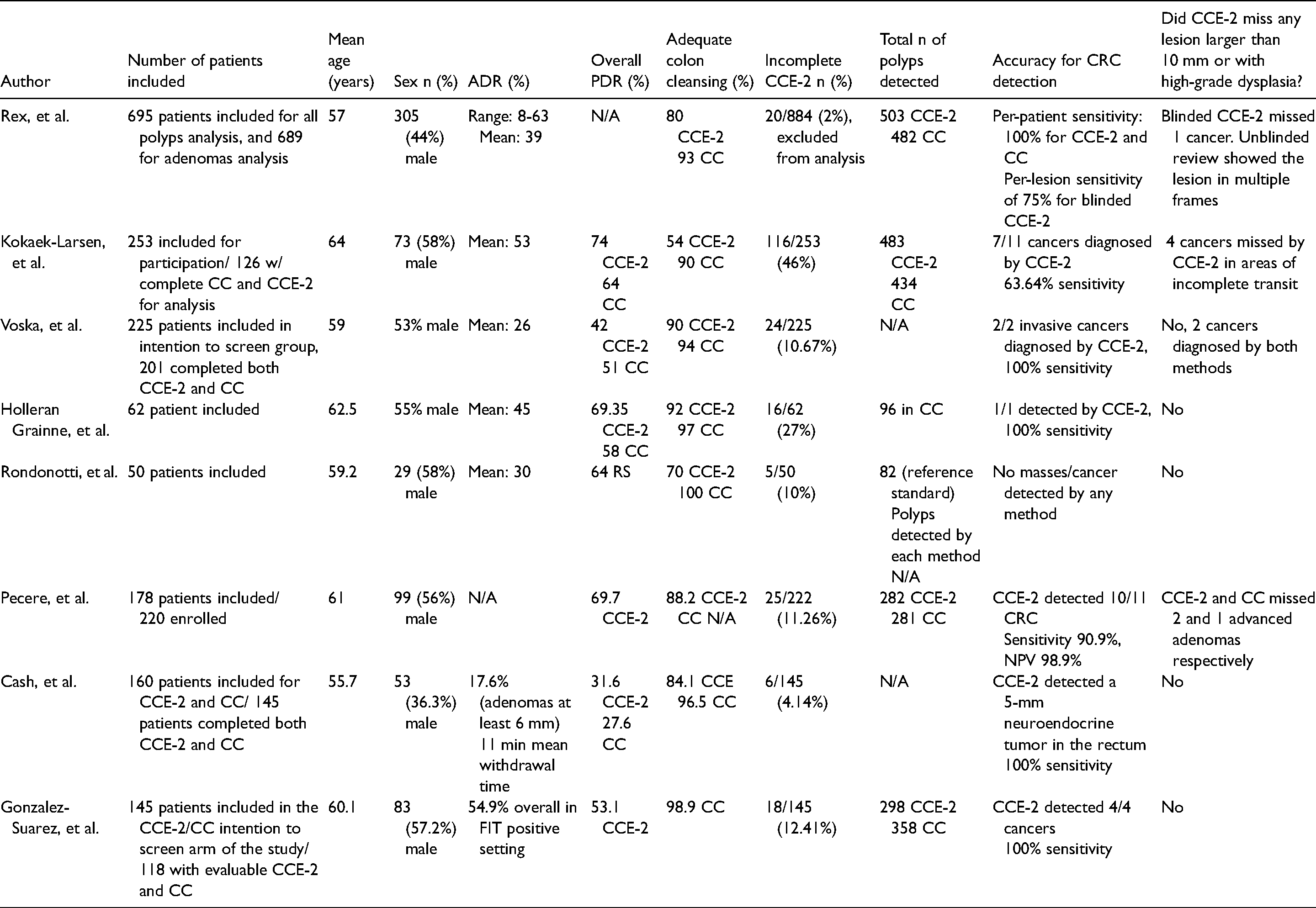
Mild adverse events occurred in 125/1602 (7.8%) patients during CCE-2, mostly related to bowel preparation. Serious adverse events did not occur after diagnostic CC. There were 5 cases of abdominal pain, 7 of post-polypectomy bleeding, 2 of rectal bleeding and 2 of perforation after CC. One of the perforation cases required surgical management. Table 2
Meta-analysis
The pooled outcomes for polyps measuring at least 6 mm / 10 mm were:
Sensitivity: 88%/88% (95% CI: 0.84–0.91/0.82–0.93), with heterogeneity 15.27/4.71 (p = 0.009/p = 0.32), inconsistency = 67.3%/15%; Figure 2 Specificity: 94%/95.5% (95% CI: 0.92–0.95/0.94–0.97), with heterogeneity 26.26/54.06 (p = 0.001/ p = 0.0001), inconsistency = 81%/92.6%; Figure 2 Positive likelihood ratio: 11.86/23.07 (95% CI: 5.53–25.46/6.163–86.36), with heterogeneity 35.82/63.45 (p = 0.001/ p = 0.001), inconsistency = 86%/93.7%; Figure 2 Negative likelihood ratio: 0.14/0.14 (95% CI: 0.1–0.21/0.09–0.21), with heterogeneity 9.32/3.43 (p = 0.097/ p = 0.49), inconsistency = 46.4%/0.1%; Figure 2 The area under the SROC for polyps at least 6 and 10 mm was 96.3% and 96.7% respectively; Figure 3.
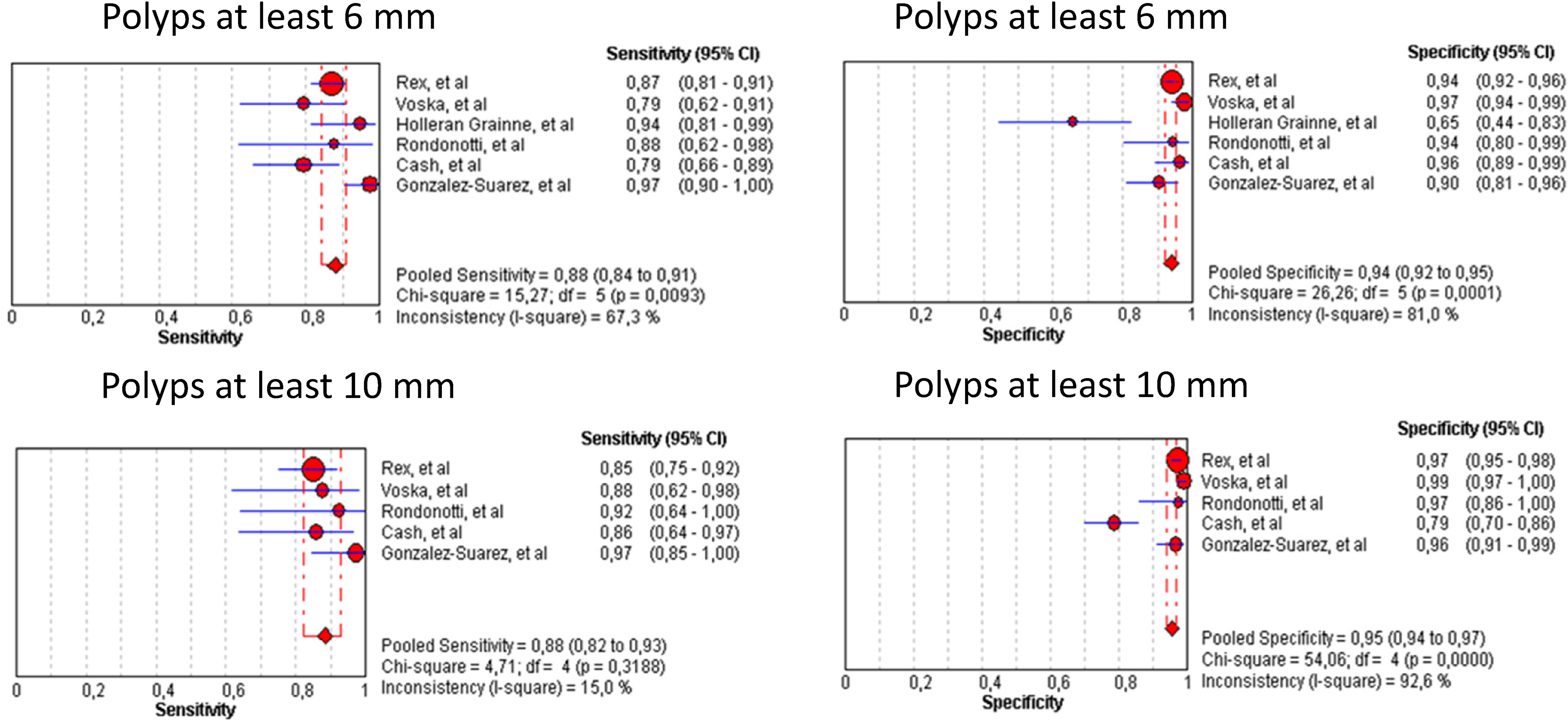
Forest plots of CCE-2 sensitivity and specificity for diagnosis of polyps at least 6 mm and 10 mm.
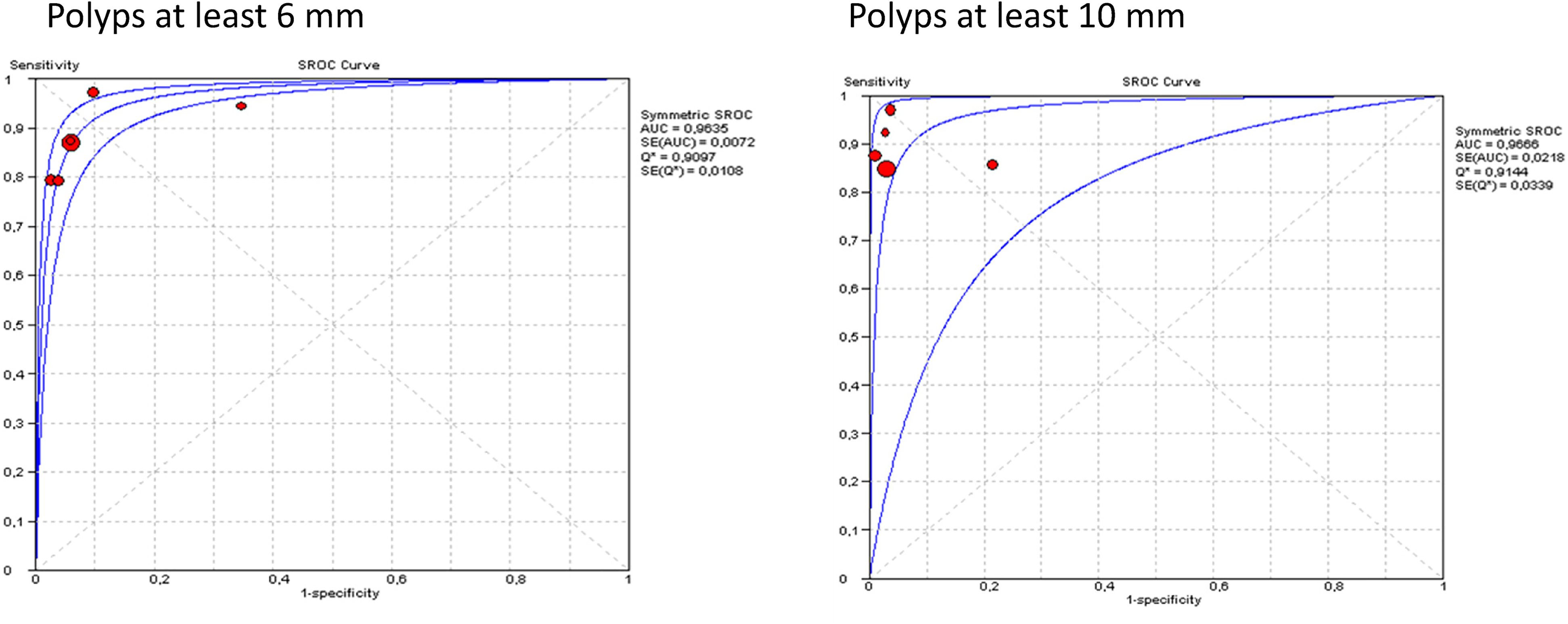
Summary receiver operating characteristic curve (SROC) for the diagnosis of polyps measuring at least 6 mm and 10 mm. AUC, area under the curve; Q*, Q index; SE, standard.
The pooled outcomes for adenomas measuring at least 6 mm / 10 mm were:
Sensitivity: 90%/85% (95% CI: 0.85–0.94/0.77–0.91), with heterogeneity 0.02/6.85 (p = 0.891/0.032), inconsistency = 0.1%/70.8. Specificity: 80%/95% (95% CI: 0.77–0.83/0.94–0.96), with heterogeneity 15.82/9.41 (p = 0.001/ 0.009), inconsistency = 93.7%/78.7%. Positive likelihood ratio: 3.79/18.55 (95% CI: 1.91–7.52/ 7.57–45.47), with heterogeneity 17.68/13.75 (p = 0.001), inconsistency = 94.3%/85.5%. Negative likelihood ratio: 0.13/0.12 (95% CI: 0.08–0.2/ 0.03- 0.46), with heterogeneity 0.37/7.87(p = 0.55/ 0.02), inconsistency = 0.1%/74.6%.
Risk of bias within studies
The risk of bias in the selected studies regarding patient selection and index time was qualified as low. Reference test risk for bias was qualified as high in 6/8 studies, mainly because second-look colonoscopy results were validated as reference standard. Same or next day examinations were performed in 4/8 studies and these were considered at low risk for time and flow bias.
Discussion
This meta-analysis demonstrated a comparable diagnostic performance by CCE for polyps measuring at least 6 and 10 mm (AUC: 96.3% and 97% respectively) in a screening population at average risk for colorectal cancer when compared to the gold standard method: colonoscopy. This result is in line with a recent multicenter, randomized trial that evaluated CCE-2 versus computed tomography colonography showing a comparable diagnostic yield between CCE-2 and colonoscopy for polyps at least 6 mm (31.6% vs. 27.6%) and 10 mm (13.5% vs. 10.3%). 23 Our study showed that a negative likelihood ratio of 0.14 was consistent for polyps of at least 6 and 10 mm. This is a valuable indicator of the usefulness of CCE-2 as a filter screening test for better selecting patients that could benefit from a colonoscopy.
The projected short-term effect of disruption to colorectal cancer screening during the Covid-19 pandemic is modest However, there is likely to be a marked impact with regard to colorectal cancer incidence and deaths between 2020 and 2050 attributable to missed screening. This should be mitigated by catch-up strategies to limit the mid- and long-term impact.12,31,32 A means to overcome the low screening activity during the Covid-19 pandemic could be the introduction of CCE-2 as an alternative screening modality. 33 In the pandemic context, capsule endoscopy has unique advantages over other gastrointestinal endoscopy modalities,34,35 including single use with low risk of cross-infection, low risk of aerosol generation, minimal medical staff requirement, separation of examination and reading/reporting, and good tolerance of the test. 36 Additionally, CCE-2 offers the advantage of being feasible as an out-of-hospital exam. 10 Interestingly, a Danish study included in the present analysis performed CCE-2 as a home-delivered service by trained nurses who supported patients throughout the procedure. 25 This was achieved before the Covid-19 pandemic, and the implications of this modality of care applied to the current limited scenario for screening are encouraging. Additionally, the present systematic review shows that colonoscopy was performed with the same bowel preparation in 6/8 included studies. CCE-2 has been recommended by recent studies as an elective test for colorectal cancer screening during the Covid-19 pandemic. 36
The PDR range was 31.6% to 74% and 27.6% to 64% for CCE-2 and colonoscopy, respectively. When significant pathology found at CCE-2 was not detected by CC, a second unblinded colonoscopy was performed and additional polyps initially missed by CC were designated as true positive capsule detections in 7/8 studies. These results show that colonoscopy is not a perfect gold standard. Studies featuring tandem colonoscopies have shown a miss rate of small polyps of between 11% and 27% and a miss rate of large polyps (≥ 10 mm) of between 2% and 6%.37,38 Outcomes of colonoscopy depend on the quality of the procedure measured by standard quality indicators such as ADR, withdrawal time, cecal intubation rate and bowel cleansing quality. If CCE-2 were to be introduced as an alternative screening method in patients unwilling or unable to undergo CC, standard quality metrics would be needed in order to measure its performance as well. A clear PDR benchmark for CCE-2 should be established. Application of colonoscopy quality metrics to CCE-2 shows that there is still room for improvement. In the present systematic review, the rate of incomplete CC2–2 ranged from 2% to 46%. Additionally, the range of adequate bowel preparation was 54–92% for CCE-2 in contrast to 90–100% for CC. Taking into consideration that even studies that performed both tests using a single bowel preparation presented a more favorable cleansing level for colonoscopy, it should be recognized that these differences may reflect intrinsic technical limitations of CCE-2.
The only study that reported significant pathology missed by CCE-2 outlined that these lesions were missed in anatomic areas that were not reached by the capsule due to incomplete exams. The authors suggest that all incomplete CCE-2 should be followed by CC. On the other hand, when only complete CCE-2 and CC were compared, no advanced lesions or cancers were missed, and CCE-2 outperformed CC regarding PDR. 25 Although variable rates of incomplete CCE-2 were reported, with an overall rate of 11.58%, this drawback could be outweighed by the potential decrease in number of colonoscopies required, which has been estimated in up to one third of cases, 27 and its high diagnostic yield for polyps measuring at least 6 and 10 mm. CCE-2 could serve as an additional tool for screening to alleviate the unmet demands for screening colonoscopies. There could also be selection of patients who would benefit the most from a colonoscopy with the intrinsic therapeutic advantages of that method, while CCE-2 could be used for those who can safely perform another test or prefer a less invasive choice.
Regarding the screening approach of the included studies, it is worth mentioning that 840 (52.43%) patients were evaluated as part of an opportunistic program23,24 while 762 (47.57%) underwent organized screening.25–30 Although we did not aim to compare CCE-2 performance between screening modalities, it is interesting to note that the proportion of screened patients and the outcomes were similar, suggesting that CCE-2 may be used either in FIT/FOBT-positive patients from average-risk organized screening programs or an average-risk population submitted to opportunistic evaluation to better select patients who deserve a colonoscopy.
We acknowledge several limitations of this study. Lessons learned from the Paris 39 and Kudo classifications 40 have made gastroenterologists around the world aware of the potential risk of advanced lesions even in adenomas smaller than 5 mm with depressed morphology. Detection of lesions smaller than 6 mm and morphology characterization were not defined as outcomes of the present study. However, we do reinforce the fact that the diagnostic accuracy of CCE-2 was comparable in lesions sized at least 6 mm and 10 mm. This remains an advantage over other imaging modalities 23 for patients in whom colonoscopy might not be feasible. Although CCE-2 demonstrated a high diagnostic accuracy in the present study, an up-to-date economic analysis is needed in order to assess the practical introduction of the method as an alternative colorectal screening choice.
Conclusions
CCE-2 is demonstrated to be an effective and safe method in the setting of colorectal cancer screening. The diagnostic performance of CCE-2 was similar for polyps measuring at least 6 and 10 mm. Likelihood ratios obtained suggest that CCE-2 may have a strong impact on screening, adequately filtering patients who would benefit the most from colonoscopy. Completion rates still need to be improved.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
