Abstract
Objectives
Cervical cancer incidence and mortality is increasing in Japanese women under age 50. Screening uptake is low and proactive recommendations for human papillomavirus vaccination have been suspended. Other cervical cancer prevention initiatives are urgently needed. We assessed whether human papillomavirus self-sampling might be an acceptable alternative to physician-led screening, particularly in women with limited experience of tampon use. We also sought to identify any practical, logistical, or safety issues in women already attending for screening, before carrying out further large-scale studies in non-responders.
Methods
In total, 203 women aged 20–49 attending their annual workplace healthcheck in Sapporo, northern Japan, performed unsupervised human papillomavirus self-sampling before undergoing a physician-led cervical smear and human papillomavirus test, and completing a measure of acceptability for both tests.
Results
Ninety per cent of participants stated they would use self-sampling again. They found instructions easy to follow and reported no issues with the usability of the self-sampling device. Compared with physician-led testing, women found self-sampling significantly less painful, less embarrassing and could relax more (p < 0.001), regardless of history of tampon use, which was associated with negative experiences in physician sampling (p = 0.034). Women lacked confidence the test had been performed correctly, despite no unsatisfactory samples. No safety issues were reported.
Conclusions
Self-sampling was highly acceptable in this population of women. They could perform the test safely unsupervised, but lacked confidence the test has been carried out correctly. Japanese women need to be educated about the accuracy of human papillomavirus self-sampling and further large-scale studies are necessary in non-responders.
Introduction
Cervical cancer, caused by persistent infection with a high-risk oncogenic human papillomavirus (HPV) type, is the fourth most common cancer globally. 1 Unlike other countries with organized screening programmes, age-standardized incidence of cervical cancer in Japan is relatively high, 12.1 per 100,000 in 2011, compared with only 7.7 and 5.5 per 100,000 in the US and Australia, respectively.2–4 Mortality rates in Japanese women aged <50 are also increasing. 5 Regardless of country of origin, most women who develop cervical cancer are either under-screened or never-screened.6–8 Organized cervical screening coverage in Japanese women is <30%. 2 When opportunistic screening is included, participation is still low, at around 40%. 5 Reasons why women do not attend for screening include: psychological factors, such as embarrassment, discomfort, and fear (both of the procedure and the results)9–12; cognitive factors, particularly risk factors for cervical cancer10,13,14; and practical factors such as time, cost and access to screening facilities or a female practitioner.9,15
Recent Japanese government initiatives targeting cervical cancer have included free screening coupons for women aged 20, 25, 30, 35, and 40. However uptake was poor at <25% for all age groups. 16 Free HPV vaccination was also introduced for girls aged 12–16 in 2010. However, after media reports of adverse events following immunization, the government suspended proactive recommendation of the vaccine in 2013. Consequently, uptake has plummeted from 75 to <1%. 17 Given this situation, other cervical cancer prevention initiatives are needed. One such initiative may be HPV self-sampling.
HPV self-sampling for high-risk oncogenic HPV types (hereafter self-sampling) is a simple alternative to clinician-led speculum examination, which allows women to take their sample using a brush, lavage, or other collection device, in the comfort and convenience of their own home. A review of the clinical accuracy of HPV self-sampling versus physician-led screening found that self-sampling was ‘at least (as), if not more sensitive’ for cervical intraepithelial neoplasia grade 2 or more (CIN2+) than conventional cytology. It also indicated that self-sampling may improve screening participation in non-attendees. 18 Similarly, a meta-analysis revealed that PCR-based HPV tests tended to show similar sensitivity on both self-sampling and clinician-led samples. 19 A systematic review by Schmeink et al., which included women of various ages and from different cultural backgrounds, found that acceptance of self-sampling by the women themselves was moderate to high, between 75 and 93%, with most women finding the sampling device easy to use, and reporting less embarrassment, less discomfort, and higher levels of relaxation during the test. 20 However, no study has investigated acceptance of HPV self-sampling in Japanese women. Unlike their Western counterparts, Japanese women have limited experience with tampons or vaginal medications, and may not feel comfortable with the act of inserting a foreign object into their vagina.
The primary aim of this feasibility study was, therefore, to assess whether HPV self-sampling might be an acceptable alternative to physician-led screening for Japanese women. A secondary aim was to identify any practical, logistical, or safety issues in women already attending for screening, before carrying out large-scale studies in under- or never-screened women. This included investigating perceptions of the self-sampling device (size, length, etc.) and women’s ability to follow the instructions (written and pictorial) in an unsupervised setting. Finally, we also investigated sociodemographic barriers to HPV self-sampling acceptance, with a focus on history of tampon use.
Methods
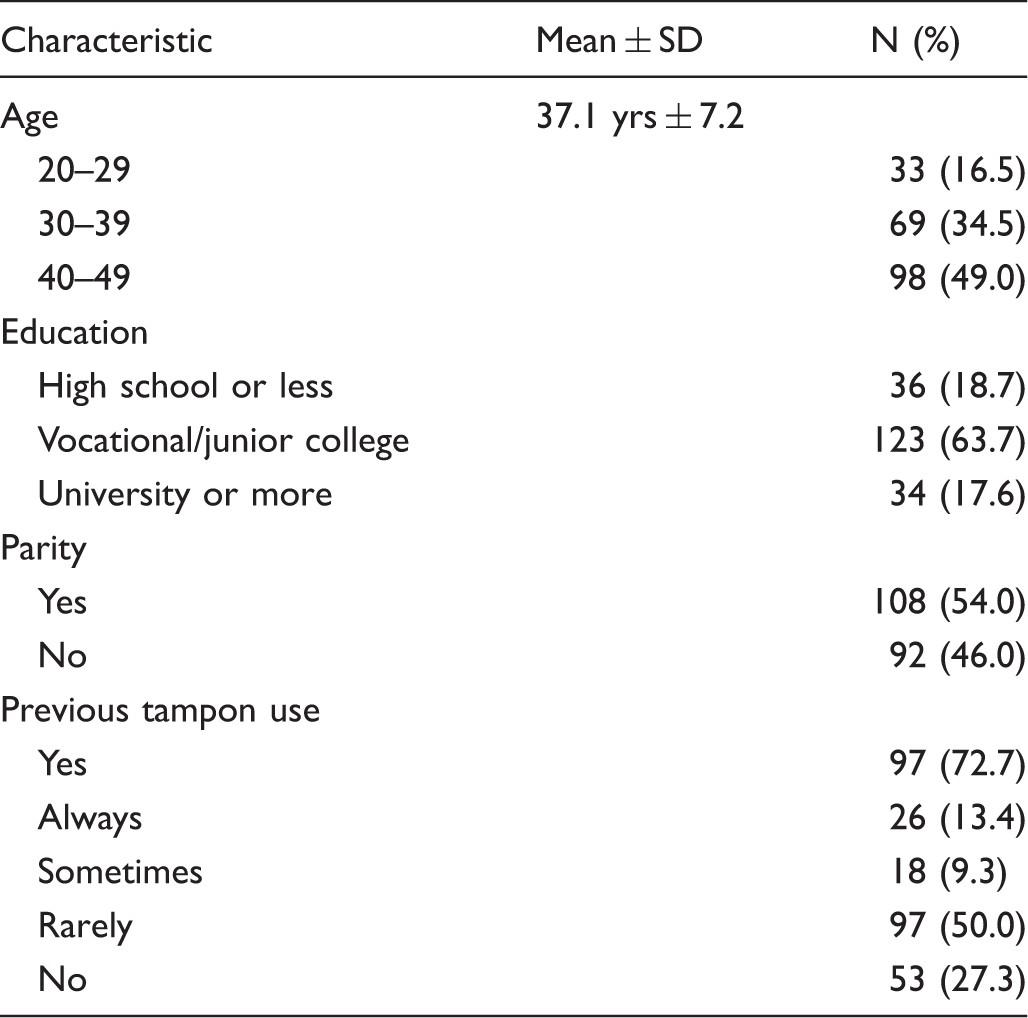
A total of 203 working women aged between 20 and 49 attending for their annual workplace health check-up at Sapporo Industrial Health Management Screening Center between September 2013 and March 2014 took part in the study. No information was available on participants’ previous screening history. The women were approached by a public health nurse (SK) who explained the purpose and content of the study and obtained written and oral informed consent. Participants received the self-sampling kit and written and pictorial instructions detailing how to perform the test. Self-sampling was performed in the screening centre toilet, with no other instructions or guidance provided. After self-sampling, women underwent a physician-led HPV test, and Pap smear using liquid-based cytology. Immediately after completion of both tests, women were asked to fill out an acceptability questionnaire.
The 25-item questionnaire was developed based on previous research 21 and adapted for a Japanese population. It assessed sociodemographic characteristics, history of tampon use, comprehension of self-sampling instructions, perceptions of the size of the self-sampling device, attitudes towards self-sampling and the physician-led test, ease of use, and acceptance of HPV self-sampling. Sociodemographic factors included participants’ age, educational background, and parity. For the question, ‘How often do you use tampons?’ women chose from: ‘never’, ‘sometimes’, ‘often’, or ‘always’. For logistic regression analyses, responses were dichotomized into ‘never’ and ‘ever’ used. Attitudes towards each test were assessed using a four-point scale ranging from 1 = ‘not at all’ to 4 = ‘extremely’ that investigated embarrassment, pain (physical discomfort), unpleasantness (mental discomfort), and confidence that the test had been conducted correctly. A total attitude score for each test (possible range between 5 and 20) was also generated by adding together the scores for embarrassment, pain, unpleasantness, confidence, and relaxation, to investigate attitudinal differences among the different sociodemographic groups. A higher score indicated a more negative attitude. Acceptance of self-sampling was assessed by participants’ stated willingness to use the test again in the future.
The self-sampler used was the Evalyn Brush (Rovers Medical Devices BV, Netherlands) and the HPV test was Qiagen’s Hybrid Capture II (HC-II) hr-HPV DNA Test (Qiagen, Mitsubishi Chemical Medience Corporation, Japan). HC-II confirms infection with one or more of 13 oncogenic HPV types (types 16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59, 68). Results were recorded in relative light units compared with a 1.0 pg/ml standard. A positive result was defined as a value greater than or equal to the standard threshold of 1.0 pg/ml.
Data were analysed using IBM SPSS Statistics Version 22.0 (SPSS Inc., Chicago, USA). Paired samples t-tests were used to investigate differences in attitudes between physician-led screening and self-sampling. A one-way between-group analysis of variance with post hoc comparisons using Tukey’s Honestly Significant Difference test was conducted to explore demographic differences in attitudes towards self-sampling and physician sampling. Finally, logistic regression analysis was performed to investigate the association between self-sampling acceptability and predictor variables. Two models were constructed: unadjusted, and model 1 adjusted for age, education, parity, and tampon use. Statistical significance was defined as a two-tailed p-value of < 0.05.
The study was approved by the Ethics Review Board for Epidemiological Studies at Hokkaido University Graduate School of Medicine (Ref: 13-055) and the Ethics Review Board of Hokkaido Cancer Society (Ref: 12-01-001). Pap smears were performed as part of the women’s annual workplace health check-up and HPV tests were provided for free. No other incentive was given for participation in the study.
Results
Of 203 (100%) questionnaires returned, three (1.5%) were excluded due to incomplete data on attitudes towards self-sampling. Data on 200 women were used in the final analysis.
Sociodemographic characteristics of participants.
Usability of self-sampling device and self-sampling test.

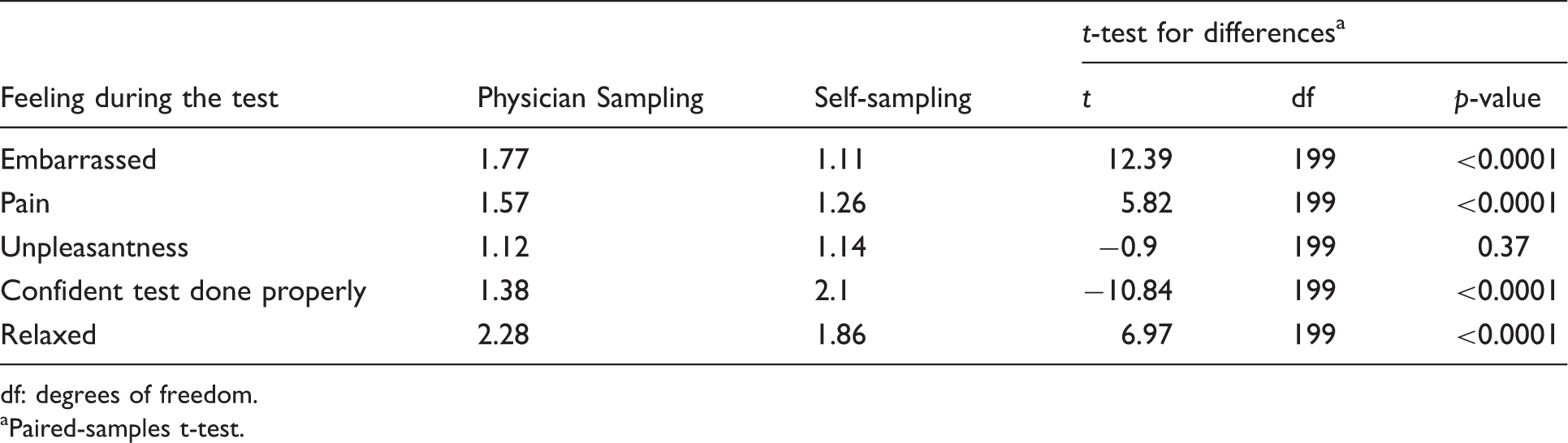
A mean score was calculated for each attitudinal question for both self-sampling and the physician-led test. The results are shown in Table 3. Participants found self-sampling to be significantly less embarrassing (p < 0.001) and less painful (p < 0.001), and were able to relax more (p < 0.001). However, as with many previous studies, they had more confidence that the physician-led test had been performed correctly (p < 0.001). No significant difference was found for unpleasantness. Difference scores between both tests were also generated for each participant for each attitude variable, so the number of participants who rated the two tests differently or the same could be calculated (Figure 1). Fewer than 10% of women rated physician sampling better for embarrassment, pain, unpleasantness, and degree of relaxation. However, only 3.0% rated confidence higher for self-sampling, with 55.5% being more confident with the results of physician-led sampling.
Comparison of participants’ experiences of self-sampling and physician-led screening. Differences in perceptions of the two tests (mean scores on a scale of 1–4). df: degrees of freedom. Paired-samples t-test.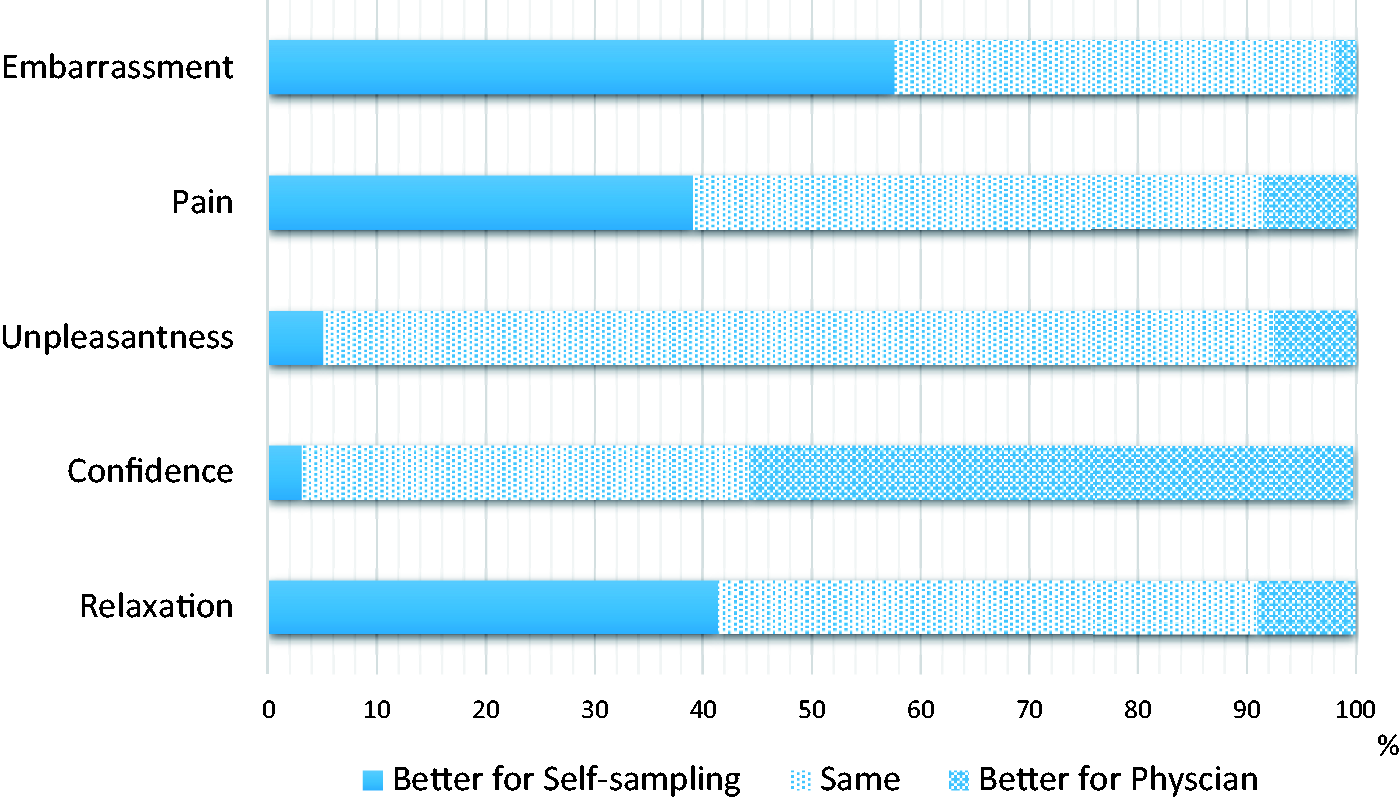
No significant within-group differences were found by age, education, parity, or tampon use for self-sampling. However, for physician-led testing, statistically significant differences within groups were obtained for frequency of tampon use (F3, 189 = 3.49; p = 0.017) and education (F2, 189 = 4.43; p = 0.013). Post hoc comparison with Tukey’s Honestly Significant Difference test revealed that women who had never used tampons had significantly more negative attitudes towards physician-led screening compared with those who always used tampons (mean difference = 1.23 ± 0.45, p = 0.034). Women with the highest education (university or more) had significantly more negative attitudes towards physician-led screening compared with those who had attended junior/vocational collage (mean difference = 0.92 ± 0.36, p = 0.033) or with those who attended high school or less (mean difference = 1.27 ± 0.45, p = 0.015). For both tampon use and education, statistical significance remained after controlling for all other sociodemographic factors. The scales showed acceptable internal consistency with a Cronbach’s α of 0.67 and 0.66 for physician-led testing and self-sampling, respectively.
Sociodemographic factors associated with acceptance of HPV self-sampling and ease of use.
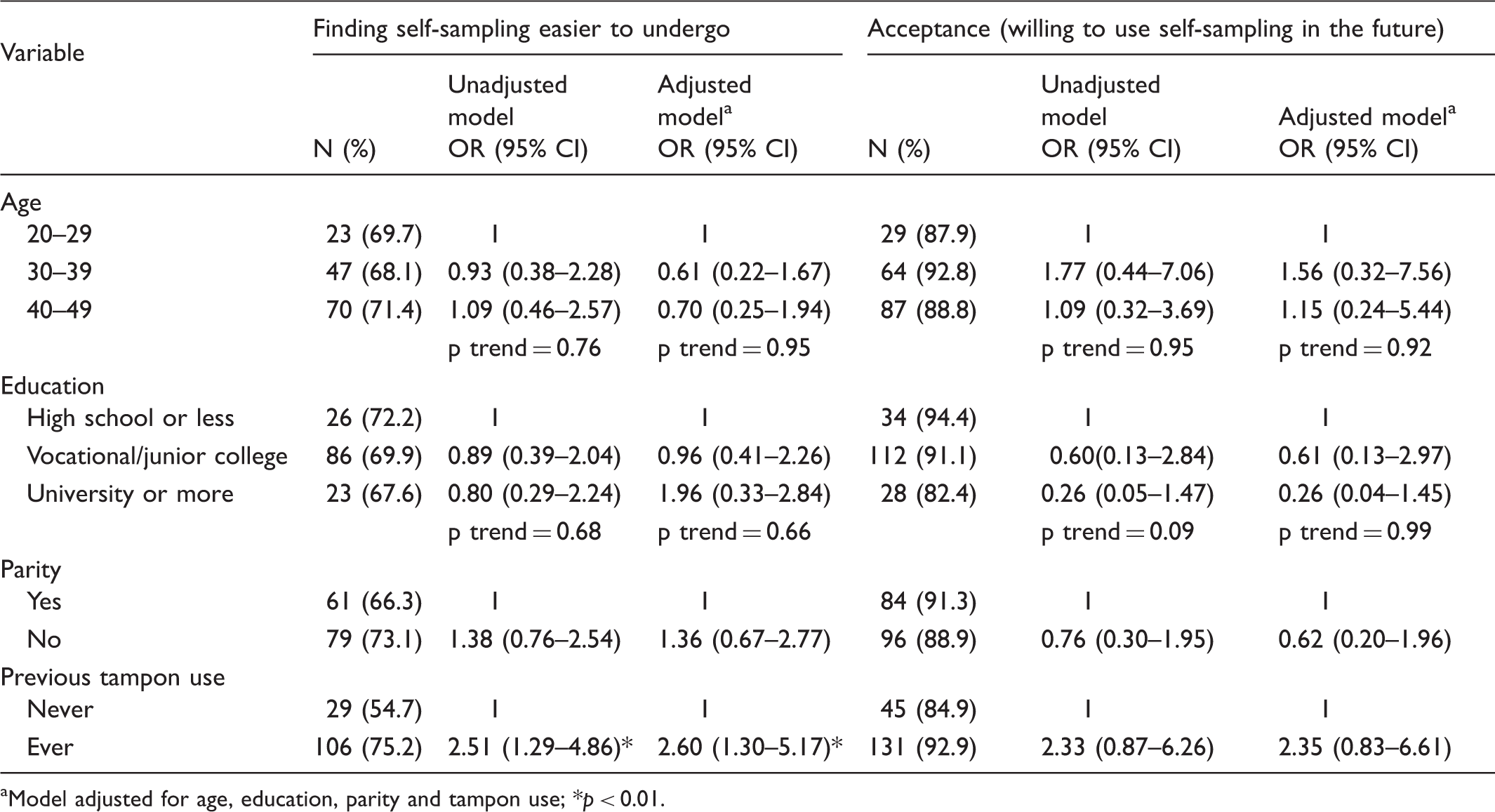
Model adjusted for age, education, parity and tampon use; *
Discussion
To assess whether HPV self-sampling might be an acceptable alternative means of cervical cancer screening in Japanese women, and whether they would be able to perform the test at home using written instructions alone, we investigated attitudes towards and acceptance of self-sampling in 200 women who underwent both self-sampling and physician-led screening.
As in previous studies,21–25 acceptability was high, with 90% of women willing to use the test again. Furthermore, as with similar studies conducted in the UK and Cameroon,21,26 women found self-sampling to be significantly less painful, less embarrassing, and were able to relax more compared with physician sampling. Although no significant difference was found for unpleasantness, only 7.5% of women reported having a more positive experience with physician testing. These results are encouraging because a previous study of never-screened young Japanese women reported embarrassment and fear of pain to be main barriers to attendance. 10 However, as with many previous studies on self-sampling,12,14,21,27 participants in this study felt significantly less confident that the test had been performed correctly, and consequently less confident with the test results. Despite these concerns, there were no unsatisfactory samples submitted in our study, suggesting women had used the correct technique. Similar results were also reported in a study of medically underserved women in the US. 22
Although no women presented with CIN2+, high-risk HPV DNA was detected by self-sampling in six out of the seven cases (86%) of low-grade squamous intraepithelial lesions, compared with seven cases (100%) of physician sampling, giving a high Kappa value of 0.82. 28 Similar agreement between test results for the two approaches has been found in several other studies,29–31 suggesting that women need to be educated more about the accuracy of self-sampling. In the previously mentioned Cameroon study, 90% of women who took part in an educational intervention about HPV self-sampling felt confident that the test had been performed correctly, regardless of previous knowledge about HPV and cervical cancer. 26
The 10% of women in our study unwilling to undergo self-sampling again gave reasons including not only worries about accuracy of the tests result, but also missing the opportunity to talk with a gynaecologist about other concerns or symptoms they may have. Similar views have been expressed by women in countries such as the US25,32 where, like Japan, Pap smears are performed by gynaecologists and not by nurses, and are often part of a more extensive annual medical check-up, during which other investigations, such as ultrasound or blood tests, can be performed. Such concerns, however, are more likely to be expressed by regular attenders, rather than under- or never-screened women.
As with previous studies conducted both in economically developed and developing countries, acceptance of self-sampling did not decrease with lower age or lower educational background.15,21,26,33 The instructions being both written and pictorial may contribute to this. In one study of Somali immigrants in the US, 45% of whom had no formal education and a further 29% of whom had only been educated to the eighth grade, all participants indicated that the instructions were easy to follow, and they experienced no difficulties collecting the samples. 34 Our results are promising because women of younger age, and with lower educational background, tend to be those who are particularly under-screened in Japan. 35 If self-sampling is to be successful, it must be acceptable to women across different sociodemographic groups, to ensure existing health disparities do not widen further.
Demographic variations were found for physician sampling. Women with a higher education were significantly less likely to accept physician sampling, even after adjustment for other sociodemographic factors. This could be because women with higher education tend to be in higher powered jobs and are used to being more in control of what happens to them. Fortunately, differences in attitudes to physician sampling do not seem to be translated into practice, as more educated women are the ones being screened in Japan. This may be because having more correct knowledge about cervical cancer prevention takes precedence over the experience of undergoing an unpleasant test.
Finally, we investigated whether previous history of tampon use, i.e. feeling comfortable with the act of inserting a foreign object into the vagina might be a barrier to acceptance of self-sampling. Tampon use, while common among European women and European Americans, is less common in African American, Latino, and Asian women.12,36 Studies from North America have shown that cervical cancer screening uptake rates in the latter three racial/ethnic groups are also lower than in Caucasian women and, consequently, women from these racial/ethnic groups are at increased risk of developing and dying from cervical cancer.37–39 If limited experience with tampons were to be a barrier to self-sampling acceptance, racial/ethnic differences in the burden of cervical cancer could be exasperated further. Despite this, to our knowledge, no study has investigated the association between tampon use and self-sampling acceptance in these racial/ethnic groups. Encouragingly, we found history of tampon use was only significant in relation to more negative perceptions of physician-led screening but not for self-sampling. Additionally, it was not a barrier to willingness to use self-sampling again. Our findings also concur with previous studies on self-sampling usability in African American and Hispanic women. One study of African American women from the Mississippi Delta reported that 100% of participants found self-sampling easy to perform, 23 while a study on Hispanic women living in California found 99.3% of women rated ease of use of the self-sampling kit as good to excellent. 24
While this is the first study to show high acceptance of HPV self-sampling in Japanese women, several limitations must be mentioned. Firstly, we investigated attitudes to HPV self-sampling in Japanese women already attending for conventional screening and, therefore, may not represent the attitudes of non-responders. Furthermore, it took place in a clinical setting, so while women performed self-sampling unassisted, medical professionals were nearby, which may have made them feel more relaxed. Additionally, it only took place in one area of Japan, and thus the results may not be applicable to the Japanese population as a whole. However, one of the main aims of this feasibility study was to assess the usability, safety, and acceptability of the self-sampling device, before undertaking larger scale studies with actual non-responders within the Japanese screening system. We have shown that the device was usable and accepted, regardless of sociodemographic status, and no safety issues were reported.
Conclusion
Our results indicate that HPV self-sampling might be an acceptable alternative to conventional physician-led cervical screening, if women can be assured about the accuracy of the test. Women had no issues with the size of the sampling device and found the instructions easy to follow in an unsupervised setting. We have also shown for the first time that history of tampon use does not seem to be a barrier to self-sampling in women with limited experience of these products. However, to assess whether self-sampling may be an effective tool to increase cervical screening rates in Japan, further large-scale studies are needed in under- or never-screened women in a non-clinical setting.
Footnotes
Acknowledgements
We thank Louise Cadman and the late Anne Szarewski of the Wolfson Institute of Preventive Medicine, Queen Mary University of London, and Jo Waller of the Department of Epidemiology & Public Health, University College London, for their invaluable help and cooperation with the questionnaire. We also express our sincere gratitude to all the women who took part in the study.
Declaration of conflicting interests
SJBH declares receiving no personal fees. HF and NS declare receiving grants from Roche Diagnostics and Qiagen Japan, and personal fees from Roche Diagnostics. NS is the Director and SJBH and HF are Executive Board Members of the non-profit organization, the People’s Campaign against Female Cancers (PCAF). PCAF has received unrestricted funding from Roche Diagnostics and Qiagen. SY, SK, AT, PD, NK, HW, and MK declare no conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Qiagen Japan provided the self-sampling kits and Mitsubishi Chemical Medience Corporation Japan performed HPV testing. Both were free of charge. Neither company had any role in the design, data analysis or writing up of the study.
