Abstract
Objective
In the context of a mature mammographic screening programme, the aim of this population-based study was to estimate rates of breast-cancer mortality among participants versus non-participants in Queensland, Australia.
Methods
The Queensland Electoral Roll was used to identify women aged 50–65 in the year 2000 (n = 269,198). Women with a prior history of invasive or in situ breast cancer were excluded (n = 6,848). The study population was then linked to mammography records from BreastScreen Queensland together with the Wesley Breast Screening Clinic (the largest provider of private screening in Queensland) to establish a screened cohort (n = 187,558) and an unscreened cohort (n = 74,792). Cohort members were matched and linked to cancer notifications and deaths through the state-based Queensland Oncology Repository. Differences in breast-cancer mortality between the two cohorts were measured using Cox proportional hazards regression.
Results
After 16 years of follow-up, women in the screened cohort showed a 39% reduction in breast-cancer mortality compared to the unscreened cohort (HR = 0.61, 95%CI = 0.55–0.68). Cumulative mortality over the same period was 0.47% and 0.77% in the screened and unscreened cohorts, respectively.
Conclusions
This study found a significant reduction in breast-cancer mortality for women who participated in mammographic screening compared to unscreened women. Our findings of a breast-cancer mortality benefit for women who have mammographic screening are in line with other observational studies.
Introduction
More than 30 years after the first randomised-controlled trials (RCTs) were reported,1,2 there is still some uncertainty about the benefits and outcomes of mammographic screening. Estimates of a reduction in breast-cancer mortality attributable to screening range from 20% in the RCTs,2,3 to 40% in more recent observational studies. 4 Improved imaging, additional views and the application of breast ultrasound may have contributed to these recent results. Other variables including attitudes to screening and demographics may also have an influence.
RCTs to assess the efficacy of mammographic screening on breast-cancer mortality began in the late 1970s. The first population-based trials showed mammography screening resulted in an approximate 30% reduction in breast-cancer mortality for women invited to screening versus uninvited women.2,3 More recently, a review by the Independent United Kingdom Panel on Breast Cancer Screening 5 concluded that there was a 20% relative risk reduction in breast-cancer mortality, comparing women invited to screening versus control women. The results were based on a meta-analysis of nine RCTs. While the RCTs were conducted many years ago, and there have been substantial improvements in the technology of screening, the Panel concluded the risk reduction of 20% remains a realistic estimate of the benefit of screening. Other reviews including one by the International Agency for Research on Cancer (IARC) Breast Screening Working Group have also reported an approximate 20% reduction in breast-cancer mortality.6–10
More recently, observational studies across several countries have examined the effect of mammography screening on breast-cancer mortality comparing screened with non-screened populations.11–18 While these studies used a variety of methodologies, and despite the biases inherent in these types of studies, results overall indicate a breast-cancer mortality benefit for screened compared to unscreened cohorts of a similar if not higher magnitude to that found by RCTs.
A national programme (BreastScreen Australia) began in Australia in 1991 and was fully implemented by 1994. This free programme is offered to all Australian women aged 50–74 (extended from 50–69 in 2013) within each Australian State and Territory. Participation in the programme is about 55% of eligible women and has remained relatively stable over several decades. 19 Within the programme, BreastScreen Queensland (BSQ) provides biennial mammography screening free of charge to women within the target age range. At the age of 50, all Queensland women registered on the Electoral Roll (voting is compulsory in Australia) who have not previously attended breast screening are invited to attend BSQ for an initial (prevalent) mammogram. After age 50, women are encouraged to attend for a mammographic screen every 2 years until age 74.
Breast screening using mammography is also conducted within private fee-for-service clinics. In Queensland, the Wesley Breast Screening Clinic (WBSC), a fully accredited facility and the largest source of private screening in Queensland, has provided breast screening for more than 30 years. WBSC offers asymptomatic women mammography and clinical examination. Each visit includes advice regarding further screening examination, breast self-examination, and information about breast cancer and its detection, with most patients receiving their results on the day of their clinic visit. If an abnormality is detected, further investigations (such as ultrasound, core biopsy, etc.) are undertaken on the same day. A blind second read is done later by a radiologist, and the woman is recalled if necessary. Reminders are routinely sent to women prior to their next mammogram appointment.
The aim of this population-based study was to compare breast-cancer mortality among women who have participated in breast-cancer screening within an organised service or through a private screening facility compared to those who have not.
Methods
The study was conducted in Queensland, Australia’s second largest state in area, with a population of approximately 5.0 million. The study population comprised 269,198 women aged 50–65 recorded on the Queensland Electoral Roll in the year 2000. From this population, 6,848 women with a prior history of invasive or in situ breast cancer were excluded (Figure 1), giving a final study population of 262,350. The accrual period for identifying mammography screening began on 1 January 2000 and continued to 31 December 2005. The upper age limit for entry (65 years) was selected to ensure all women would remain within the screening-eligible age group (50–69 in the year 2000), for the duration of the accrual period.
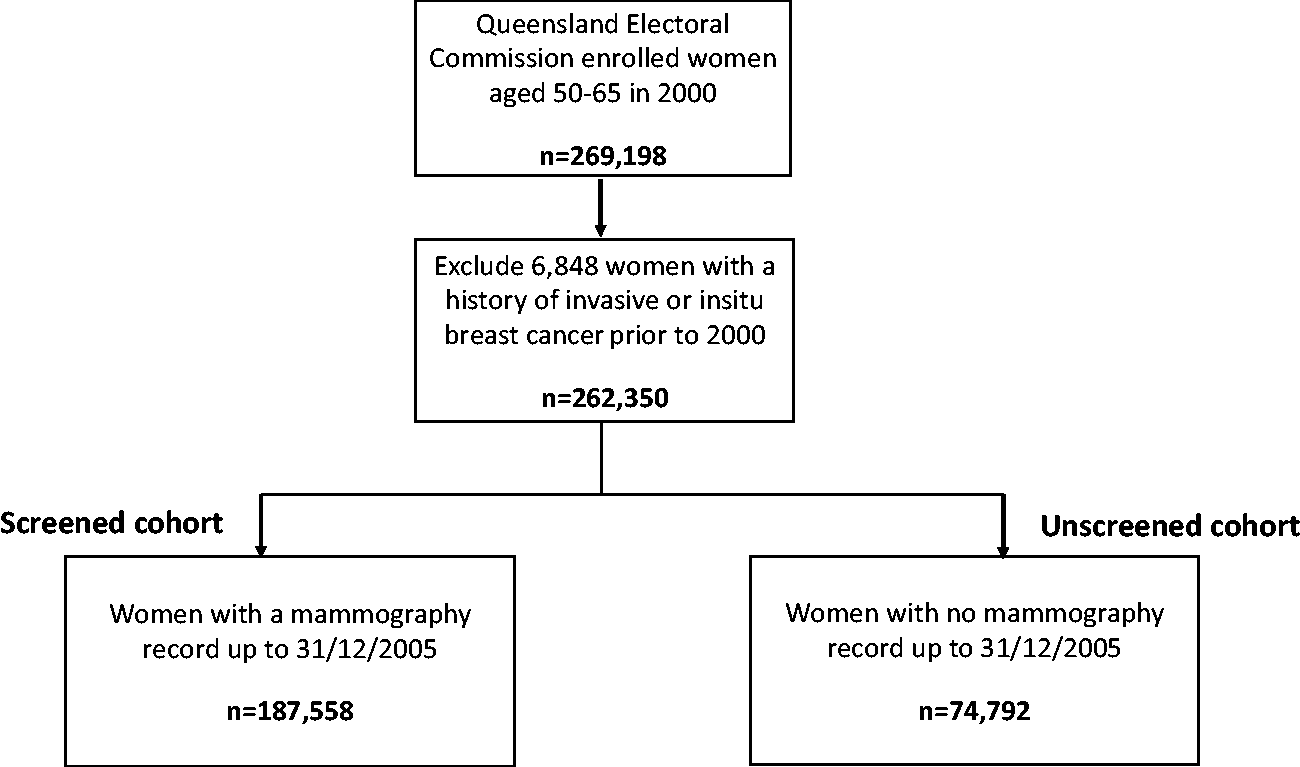
Flow diagram showing screened and unscreened cohorts.
During the accrual period, mammograms performed by BSQ or the WBSC were identified and the screening records were linked with the study population. While women with a history of breast cancer, or a family history of breast cancer, can have a mammogram in other private radiology practices where Medicare (Australia’s free public healthcare system) will pay a benefit, these numbers are relatively few and are more likely to be diagnostic mammograms.
Women with no record of screening prior to the beginning of the accrual period (n = 100,043) contributed person-years in the non-screened cohort until the date of first attendance for screening during the accrual period. Women with a record of screening prior to 2000 (n = 162,307) were allocated to the screened group from the commencement of the accrual period. This approach has been used to assess differences in mortality in other mammography screening programmes. 20 At the end of the accrual period, the screened cohort included 187,558 women and the unscreened cohort included 74,792 women (Figure 1). Both cohorts were then followed up until 31 December 2015. The study design was used after consideration of previous studies that suggest measurable effects on breast-cancer mortality may not rise for 10 years or more.15,21
Data linkage
Cohort members were matched and linked to records in the Queensland Oncology Repository (QOR) from 2000 to 2015, a state-wide clinical cancer database managed by the Queensland Cancer Control Analysis Team. QOR uses deterministic matching to link data from various sources including the Queensland Cancer Register and the Register of Births, Deaths and Marriages. In Queensland, notification of cancer is a statutory requirement.
Variables included
We included age, socioeconomic status (SES) and residential location. SES was assigned according to the Australian Bureau of Statistics Socio-Economic Index for Areas, 22 a census-based measure of social and economic well-being. Residence was classified into urban or rural, based on the Australian Standard Geographical Classification, 23 aggregated at the level of Statistical Area 2 utilised by the Australian Bureau of Statistics.
Statistical analysis
Differences in breast-cancer mortality between the two cohorts were measured using time-dependent Cox proportional hazards regression. Screening status was treated as a time-dependent covariate in the model by splitting time-span records into two separate observations for before and after the commencement of screening. Age was included as a categorical variable in 5-year groups. Proportional hazards assumptions were tested by using the Schoenfeld residuals via the
Cumulative mortality from breast cancer for both cohorts was calculated across the 16 years of follow-up. Censoring was performed for non-breast cancer deaths occurring during the follow-up period. Hazard ratios (HR) obtained from the Cox regression were used to estimate the difference in the rate of death observed in the screened cohort compared to the unscreened cohort, adjusting for age, socio-economic status and residence.
In addition, we performed a sensitivity analysis of the risk of breast-cancer mortality using a population of women with no record of screening prior to 1 January 2000 (n = 100,043). This provided a screened cohort of 36,864 and unscreened cohort of 63,179.
Ethics
Ethical approval for our study was granted by the Metro South Health Human Research Ethics Committee and the Uniting Care Health HREC (with respect to WBSC data).
Results
Of the 262,350 women in the study population, 71.5% (n = 187,558) had a record of at least one mammogram during the accrual period. Table 1 provides a description of the sociodemographic characteristics of the screened and unscreened cohorts. There were modest differences in screening participation according to age, SES and residential location.
Sociodemographic characteristics of 262,350 women aged 50–65.
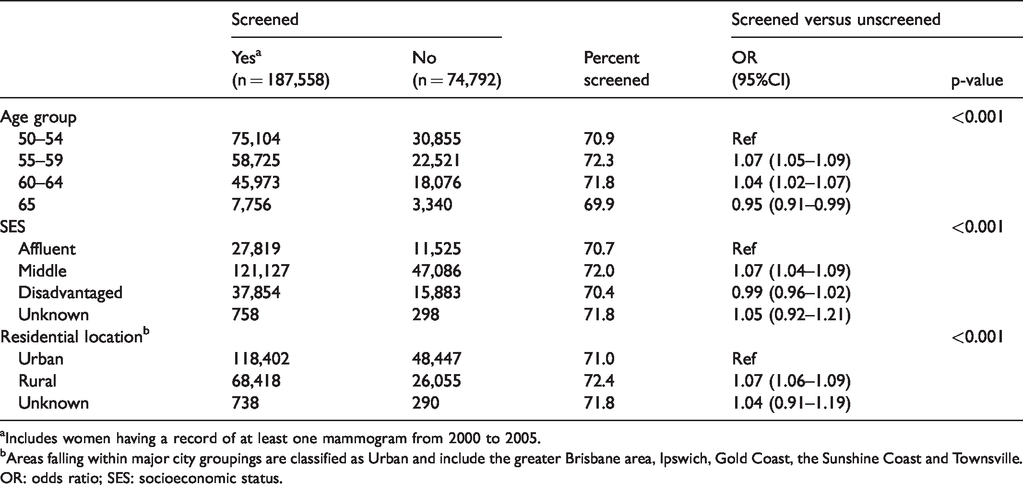
aIncludes women having a record of at least one mammogram from 2000 to 2005.
bAreas falling within major city groupings are classified as Urban and include the greater Brisbane area, Ipswich, Gold Coast, the Sunshine Coast and Townsville.
OR: odds ratio; SES: socioeconomic status.
From the start of follow-up on 1 January 2000 to 31 December 2014, 11,143 cases of invasive breast cancer were diagnosed in the screened cohort and 3,219 in the unscreened cohort.
Breast-cancer mortality
In adjusted analysis, after 16 years of follow-up, the HR for death from breast cancer in the screened versus unscreened cohort was 0.61 (95%CI = 0.55–0.68, p <0.001) (Table 2). This equates to a reduction of 39% in the risk of death from breast cancer among screened compared to unscreened women.
Risk of breast-cancer mortality in screened versus unscreened women.
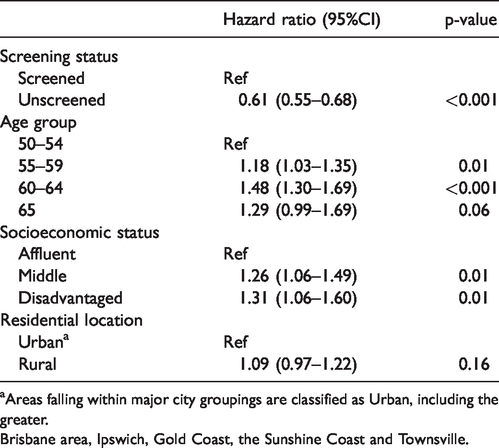
aAreas falling within major city groupings are classified as Urban, including the greater.
Brisbane area, Ipswich, Gold Coast, the Sunshine Coast and Townsville.
We also examined the risk of breast-cancer mortality by age group and SES, irrespective of screening status. Compared to women aged 50–54 , those aged 60–64 were about 50% more likely to die from breast cancer (HR = 1.48, 95%CI = 1.30–1.69, p < 0.001) (Table 2). Further, the risk of death from breast cancer was about 25% and 30% higher for middle or disadvantaged SES women compared to affluent SES women (HR = 1.26, 95%CI = 1.06–1.49 and HR = 1.31, 95%CI = 1.06–1.60, respectively). No difference in breast-cancer mortality was observed according to residential location (p = 0.16).
The cumulative mortality from 1 January 2000 to 31 December 2015 is shown in Figure 2. During the 16 years of follow-up, 873 breast-cancer deaths occurred in the screened cohort giving a cumulative mortality rate of 0.47%. In the unscreened cohort, there were 479 deaths for a cumulative mortality of 0.77%.
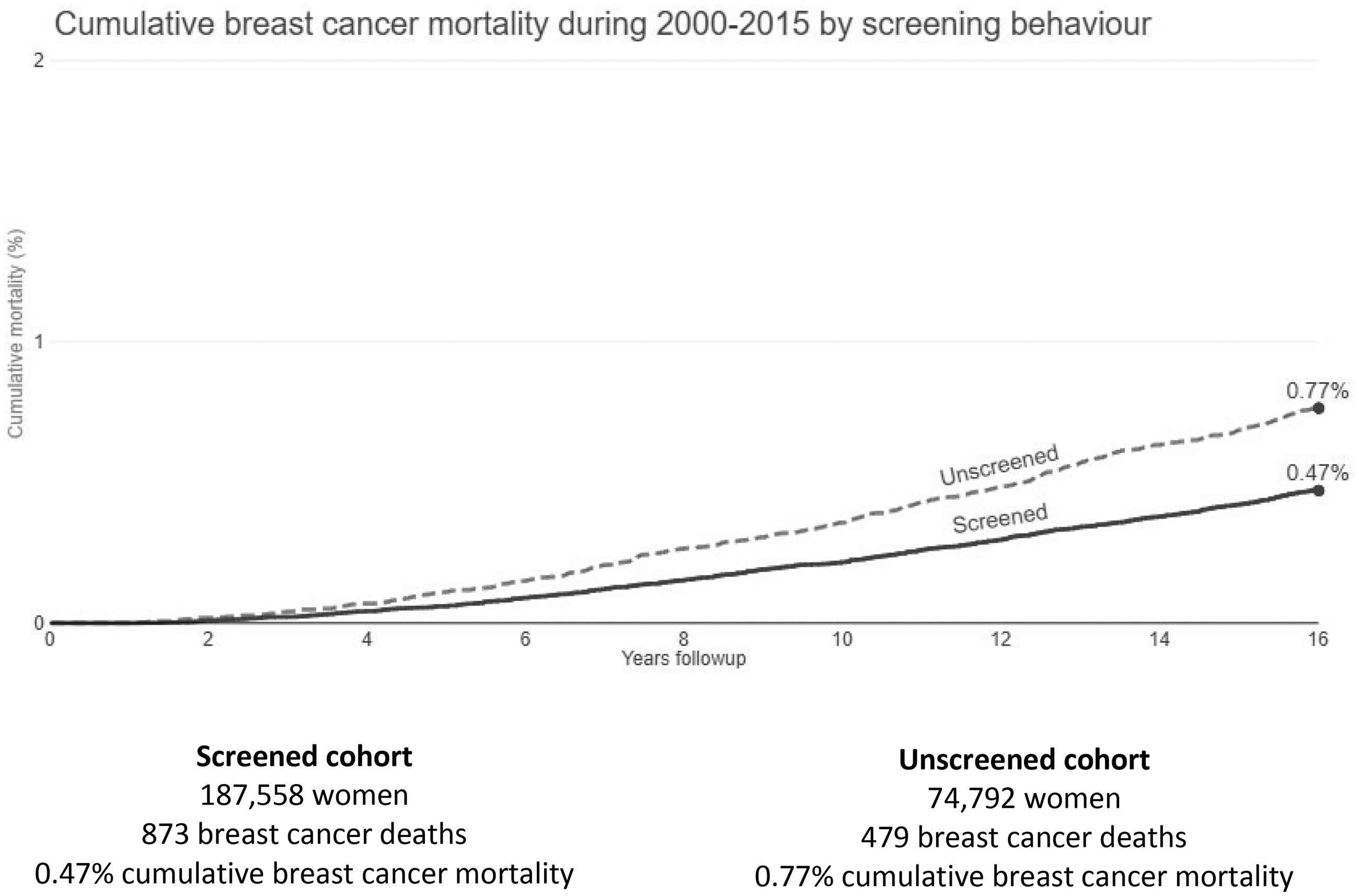
Cumulative breast-cancer mortality by screening behaviour.
Additional analysis
In an additional analysis which included only women with no history of screening prior to 1 January 2000, the risk of breast-cancer mortality in the screened compared to the unscreened group was 0.72 (OR = 0.60–0.87, p = 0.001). This equated to a 28% reduction in breast-cancer mortality for women with a history of mammography screening.
Discussion
The risk of death from breast cancer in this study was 39% lower for women who participated in mammography screening compared to non-participants (OR = 0.61, 95%CI = 0.55–0.68). The adjusted HR we observed is similar in magnitude to the adjusted relative risk reported in a recent New Zealand study using data from a linked population-based screening programme. 24 After adjustment for screening bias and a target screening participation rate of 70%, they found a mortality reduction of 34% for screened versus never-screened women. An earlier case-control study conducted in a South Australian population reported an odds ratio for breast-cancer mortality of 0.59 for BreastScreen participants compared to non-participants. 15 Further, the breast-cancer mortality benefit in our study is similar in magnitude to the 33%–43% reduction in breast-cancer mortality among observational studies reported by the IARC Working Group 10 and in a recent systematic review of European studies. 4 In that review, the authors noted the reductions in breast-cancer mortality were greater for studies examining screened versus unscreened women, rather than in those comparing women invited to screening versus an uninvited population. For ethical reasons, it is no longer possible to conduct an RCT.
To account for the potential misclassification of women having a mammogram prior to the beginning of our accrual period, we elected to include women whose only record of a mammogram was prior to 1 January 2000 in the screening group. This reduced the risk of biasing any mortality benefit in screened women towards the null. When we re-analysed the data using only women who had no record of screening prior to 1 January 2000, our results again showed a significant but reduced breast-cancer mortality benefit for screened versus unscreened women (28% versus 39%).
The addition of an adjustment factor to address screening self-selection bias has been used in several cohort16,25,26 and case-control studies.27–29 In our study, all women within the target age range were offered screening through the BreastScreen programme, and general public screening awareness campaigns are run both nationally and on a state-by-state basis from time to time. That said, using a similar approach to Roder et al., 15 and also Taylor et al., 17 the application of the formula proposed by Duffy et al. 30 for the adjustment factor resulted in a corrected HR of 0.77 – a 23% reduction in breast-cancer mortality.
Women who choose to participate in breast-cancer screening may have different risk profiles to those who do not screen. In this study, we were unable to measure common risk factors associated with screening participation. However, during the follow-up period, BSQ records showed that 16% of women attending for screening had a family history of breast cancer and 15% had a history of benign breast disease; in the same age groups the use of hormone replacement therapy (HRT) ranged from 8.2% to 10.8%. 31 Although these data give some perspective of risk factors in Queensland women who participate in screening, no data are available on the prevalence of these or other risk factors in the unscreened cohort. To address potential differences in breast-cancer risk profiles, Roder et al. 15 utilised a population-based survey that included questions related to breast-cancer risk and participation in breast-cancer screening, as a complement to their case-control study. Their results suggested a slightly higher age-adjusted risk profile for women who screened compared to unscreened women.
We also considered the possibility that differences in treatment could have influenced these results. While most studies examining outcomes from screening services have been conducted since the introduction of effective systemic treatment such as anti-oestrogens, aromatase inhibitors and chemotherapy, earlier studies conducted in Sweden and the Netherlands prior to the use of such therapies found equivalent mortality reductions.32,33 Estimates of the magnitude of the reduction in breast-cancer mortality attributable to systemic treatment range from 7% 34 to about 35% in a large meta-analysis of polychemotherapy trials, 35 and further advances in breast-cancer mortality reductions have been observed following the introduction of aromatase inhibitors. 36
However, the follow-up period for our study was 16 years, and it is extremely unlikely that there would have been differences in treatment between the cohorts during that period. Most medical oncologists in Queensland practise in both the public and private sectors, and the post-surgical management of women with breast cancer in Queensland is routinely determined in multi-disciplinary clinics. A recent Australian study reported 82% compliance with adjuvant treatments recommended by multi-disciplinary clinics and 92% for chemotherapy recommendations. 37
Perhaps, the most surprising finding in this study is that although the screening participation rates for the three SES subsets were virtually identical (Table 1), significantly higher breast-cancer mortality rates were observed in women from middle and disadvantaged areas compared to those in affluent areas (Table 2). If any benefit from screening was due exclusively to mammography, it would be expected that with similar participation there would be similar mortality across SES groups.
We have considered two possible explanations: first, the interval from diagnosis to initial treatment might be longer for women from middle and disadvantaged areas who tend to use the free public hospital system compared with those in affluent areas who typically use private hospitals. Examination of our unpublished data, which monitor breast-cancer treatment in all Queensland hospitals, shows a difference of 14% in the average interval from pathological diagnosis to initial surgical treatment, indicating that there is a relative but small increase in waiting times for public compared to private hospitals. We were unable to assess whether this could influence treatment outcomes but believe any effect would be relatively small.
A second possible explanation for the difference in mortality rates between SES subsets could be related to differences in health behaviours and ‘health awareness’ across SES strata. Compared to those living in more advantaged areas, individuals from more disadvantaged areas are more likely to have higher rates of illness, less healthy lifestyles and potentially reduced access to medical services.38,39 While in our analysis we adjusted for screening behaviours, we have previously reported that women in the screened cohort were observed to have a lower risk of dying from other cancers (including lung, cervix, ovary and bowel) and from non-cancer causes, 40 indicating that differences also exist between those who participate or do not participate in a health-related programme (such as breast screening).
The breast screening RCTs that found a breast-cancer mortality benefit in screened populations were conducted several decades ago, and several observational studies have since followed. The question arises about the need to continue conducting such studies into the future. While our findings are similar to the majority of other observational studies, using a more contemporary cohort does enable comparisons over time. This is of particular importance to service providers and policy makers given the ongoing developments in breast screening techniques (such as digital mammography).
Limitations
While our study was population-based, as in all observational studies, some limitations should be considered. We did not have any information on the individual risk profiles of screened and unscreened women, and we elected not to add an adjustment factor into the mortality analysis. However, we felt that including an adjustment factor to account for self-selection bias was not appropriate for our study design.
Additionally, some women who attended a screening examination outside either BSQ or WBSC would have been allocated to the unscreened group. We believe this number was likely to be small and any effect would bias the results towards the null.
This population-based study is strengthened by the inclusion of women who attended the largest provider of private breast-cancer screening in Queensland, particularly given it is estimated that about 20% of women attend screening outside the BreastScreen programme. 15
Conclusions
We found a 39% reduction in breast-cancer mortality among screened compared to unscreened women. Our results are in line with previous observational studies and earlier RCTs showing mortality benefit for women who participate in breast-cancer screening. The observed substantial difference in mortality between SES subsets is unexplained and requires further investigation.
Footnotes
Acknowledgements
We are grateful to Cancer Alliance Queensland, Metro South Hospital and Health Service, Princess Alexandra Hospital for ongoing statistical and administrative support. We thank Dr Jeanette Young, Chief Medical Officer, Queensland Health for her support in progressing this study. We also thank Dr Lisa Erzetich, Director, for allowing access to relevant data from the Wesley Breast Screening Service.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We thank the Australian Centre for Health Services Innovation (AusHSI) and Queensland Government Metro South SERTA Trust Fund who provided funding to conduct this study.
