Abstract
Objective
A decrease in advanced breast cancer incidence is considered an early indicator of breast cancer mortality reduction in a screening programme. We describe trends in breast cancer incidence according to tumour size and age in three French administrative areas, where an organized screening programme was implemented during the 1990s.
Methods
Our study included all 28,092 invasive breast cancers diagnosed from 2000 to 2010 in women living in three areas (Hérault, Isère, Loire-Atlantique). Age, year of diagnosis, and size of tumour at diagnosis was provided by the three area cancer registries. Poisson regression models were fitted to estimate changes in incidence over time, after adjustment for age and administrative area.
Results
From 2000 to 2010, the incidence rate of large (tumour size >20 mm) breast cancer linearly decreased in women aged 50–74 (target age of the screening programme) from 108.4 to 84.1/100,000 (annual percent change = −1.9%, p < 0.001). No change in large breast cancer incidence rate was found in women aged 20–49, or older than 74.
Conclusions
A decreasing trend in incidence of large tumour size breast cancer in the target age of the screening programme is demonstrated for the first time in France. The overall 20.9% linear decrease over 11 years in these three areas is encouraging and should be closely monitored and extended to other areas of France, where the screening programme was generally implemented only in 2004.
Introduction
The balance between harms and benefits of screening for breast cancer (BC) is still under debate.1–4 Determining the impact of mammographic screening on BC mortality in general populations is a challenge, because of methodological issues. Screening is assumed to allow BC diagnosis at an earlier stage and, consequently, to lower BC mortality. An approximate proportional decrease in risk of advanced BC (either TNM stage II+ or tumour size >20 mm) and in BC mortality has been shown in several randomized trials. A decrease in advanced BC incidence in the target population is considered an early indicator of BC mortality reduction.5–9 In several countries, results are contrasted regarding the evolution of advanced-stage BC incidence.10–18 However, the effectiveness of a screening programme at national level will be determined not only by its design and ability to detect small tumours but also by the cultural, medical, and economic context determining programme attendance and also timeliness and accurate treatment.
In France, the BC screening programme was gradually implemented from 1989 in pilot screening areas, until implementation across the entire country in 2004. 19 After the introduction of the screening programme in the pilot areas, a decreasing trend of advanced BC incidence was expected in the target age group of women (50–74). No information has been available in France regarding the assessment of the screening programme based on advanced-stage BC.
We aimed to describe trends in BC incidence, according to tumour size, in women aged 50–74 compared with women in other age groups in the general population, in three pilot screening areas covered by a population-based cancer registry in France.
Methods
This observational population-based study was conducted in three screening pilot areas (Hérault, Isère, Loire-Atlantique) with 3.6 million inhabitants, covered by cancer registries, and where the screening programme was implemented before 2000. All first invasive breast carcinomas diagnosed from 2000 to 2010 in women living in these areas were recorded by the three cancer registries and included in the study.
French cancer registries routinely collect data on cases from pathological laboratories, departments of medical data processing and clinical services of public and private hospitals, health insurance funds, and medical practitioners. Data are recorded according to the European Network of Cancer Registries recommendations. The quality and completeness of these population-based registries are certified every four years in an audit by the French National Institute of Health and Medical Research and the French Institute for Public Health Surveillance.
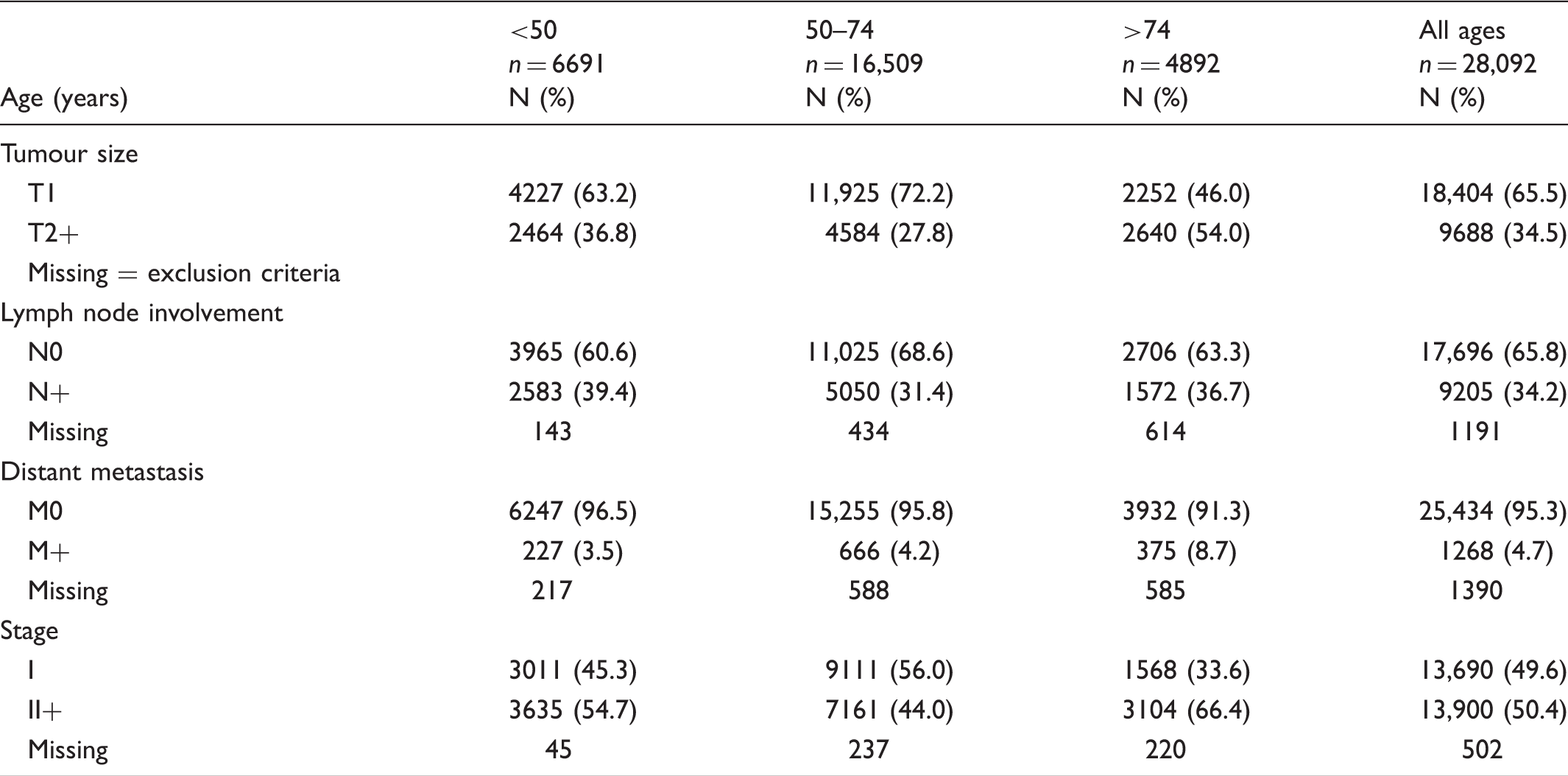
Because mammographic screening aims to detect breast tumours while they are small, the size of invasive cancer was chosen as the indicator of cancer stage. The size for distinguishing early and advanced BC was 20 mm, corresponding to the threshold between T1 and T2 classification (Union for International Cancer Control TNM classification) recorded in cancer registries. 20 We defined T2+ BC as BC with a tumour size >20 mm and T1 BC as BC with a tumour size ≤20 mm, whatever lymph node involvement or distant metastasis. In situ carcinoma, lymphoma, and sarcoma were excluded. Women with no available tumour size (n = 991) were also excluded from analyses.
The analyses were performed in three age-groups: the target age group for organized screening (50–74), and the youngest (20–49) and oldest women (75 and over). Incidence rates were calculated using the French female population from official census data of the three areas for each age group, and standardized using the world standard population, which was truncated according to the corresponding age group. Poisson regression models were fitted to estimate changes in incidence over time, after adjustment for age and administrative area, from which we derived annual percent changes (APCs) and 95% confidence intervals (CIs). Stata SE11 was used. 21
Results
Breast cancer characteristics according to age group (2000–2010).
Breast cancer (BC) incidence trend according to tumour size at diagnosis and age group (2000–2010).
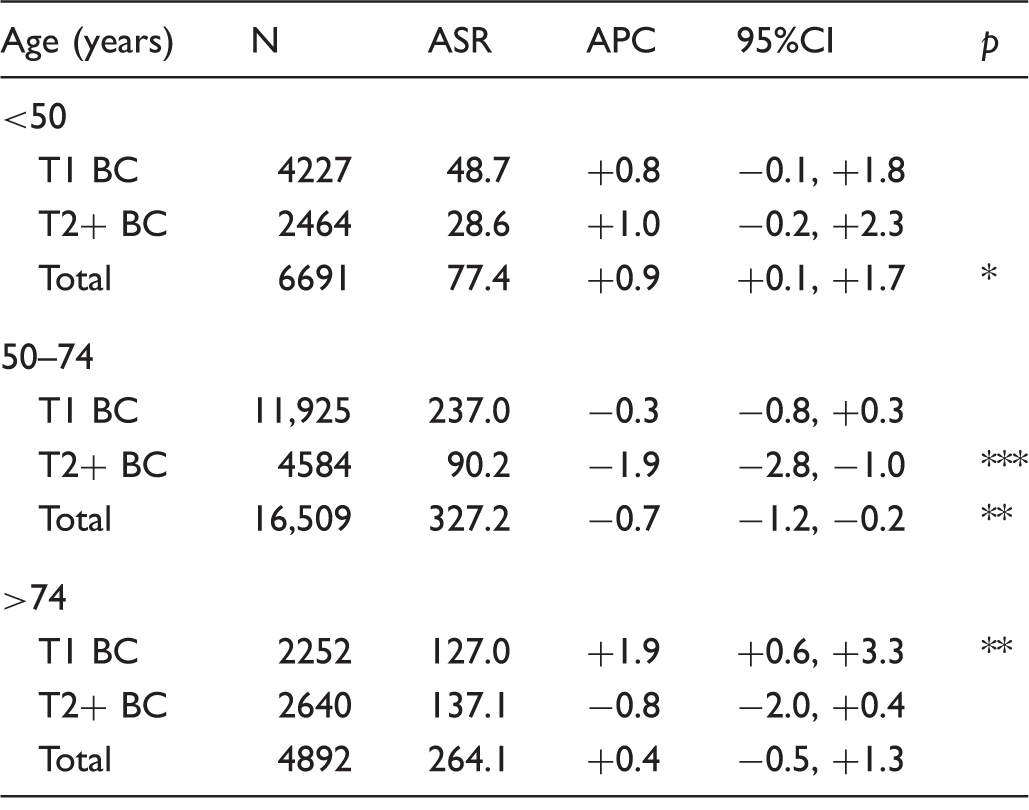
ASR: age standardized rate per 100,000; APC: annual percent change (%), with adjustment for age and administrative area.
p ≤ 0.05, **p ≤ 0.01, ***p ≤ 0.001; otherwise not significant.
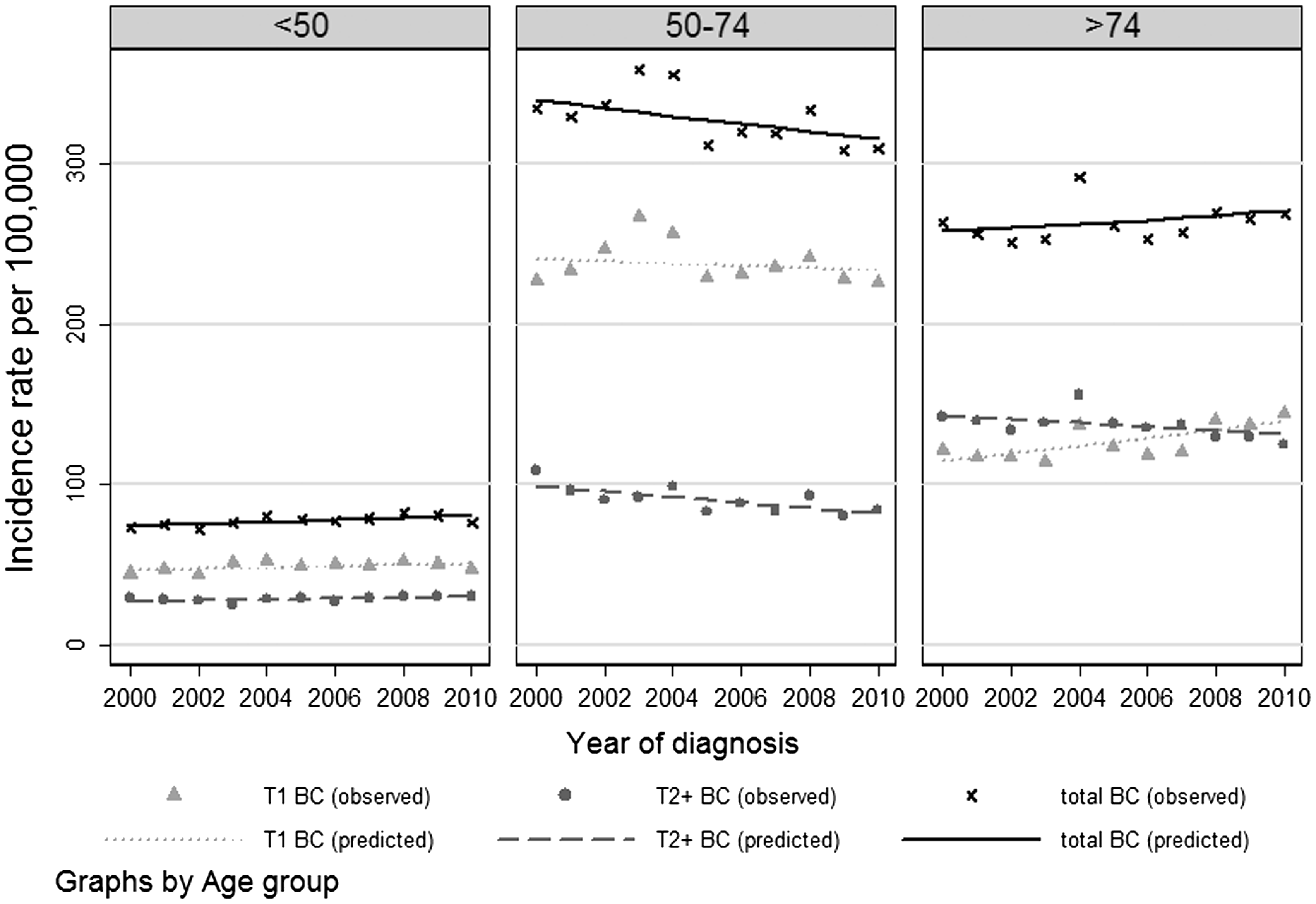
Breast cancer (BC) incidence trend according to tumour size at diagnosis (2000–2010).
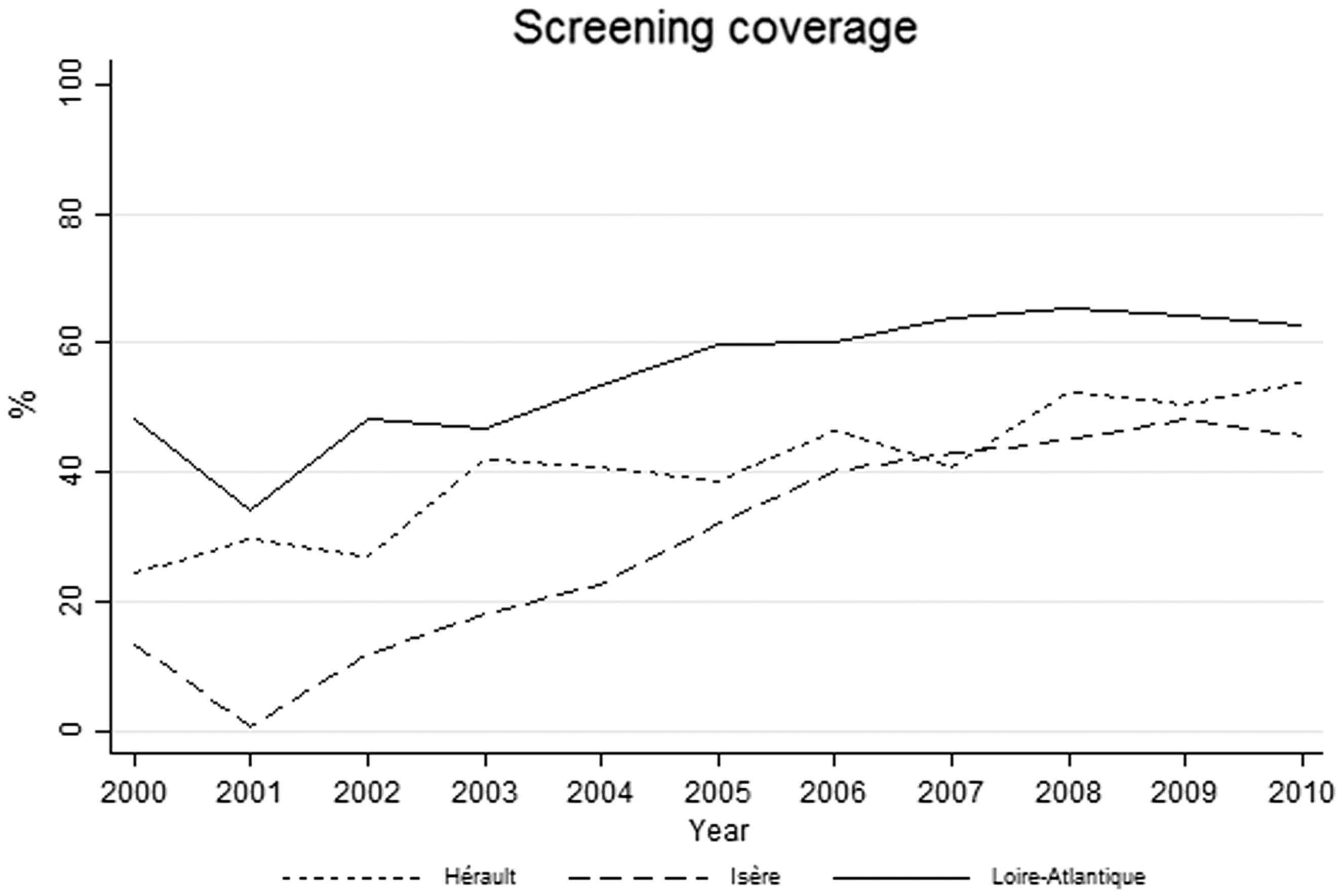
Breast organized screening coverage in the studied area (2000–2010).
Contrary to the steady and linear 20.9% decrease of T2+ BC over the 11 years of the study, the evolution of T1 BC in women aged 50–74 followed an increasing trend until 2003, a decreasing trend until 2006, and stabilized thereafter (Figure 1). In further contrast, in women aged under 50, the overall incidence rate steadily rose by 0.9% a year for all-stage BC (Table 2). In women older than 74, no increase in incidence rates of T2 + BC was observed, whereas early stage BCs were still in progression (+1.9%).
Breast cancer (BC) incidence trend according to stage at diagnosis and age group (2000–2010).
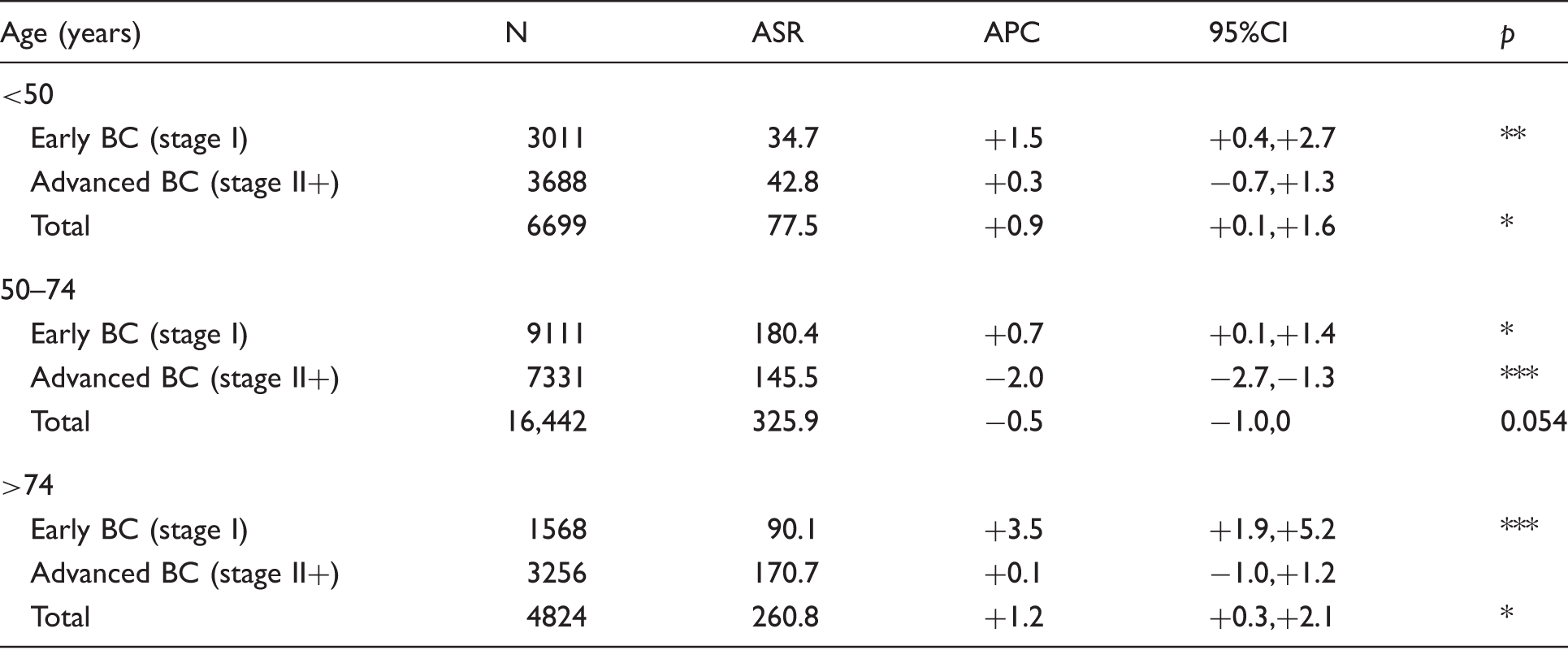
ASR: age standardized rate per 100,000; APC: annual percent change (%), with adjustment for age and administrative area.
p ≤ 0.05, **p ≤ 0.01, ***p ≤ 0.001; otherwise not significant.
Discussion
This study demonstrates a 20.9% steady decrease of T2+ BC incidence from 2000 to 2010 in French women aged 50–74, in areas where a screening programme was implemented before 2000. This is the first time that a diminishing trend has been highlighted in France. The reduction of advanced BC, defined as T2+ as well as stage II+, has been proven to be correlated to the reduction of BC mortality in randomized trials.6–8 The size of invasive cancer is strongly correlated to nodal status, and its measurement has remained stable over time (contrary to nodal involvement determination). We used complementary analyses to address potential limitations of this definition. Decreasing trend of advanced BC was confirmed if advanced BC was defined as stage II+ (>20 mm or N+ or M+).
Differences in design, study period, and definition of late-stage BC make it difficult to compare studies; however, other studies have also found a reduction in advanced BC incidence after the introduction of organized mammography screening. In Italy, a strong decrease was observed, with the incidence rate ratio around 0.8 between the observed rate and the expected rate of advanced T2+ BC eight years after organized screening implementation. 10 In Finland, East Anglia, US, The Netherlands, and Sweden, decreasing trends in late-stage BC have also been observed.11–15 Other studies failed to evidence a reduction in advanced BC incidence, sustaining controversy on the efficacy of organized screening.16–18 These discrepant study results highlight the methodological difficulties to take into account possible bias in the choice of the period, the reference population, and particularly the hypothesis regarding the unknown underlying trend of BC incidence which, if increasing, may mask a potential decrease in advanced BC incidence. However, evolution of advanced stage incidence is not the only indicator of efficacy of an organized screening programme that may show favourable outcomes, even without reduction in advanced BC incidence. 22
Organized screening was introduced during the 1990s in the three areas of this study, but participation rates were low before 2000 and were still in progression during the study period. Although the results of this observational study must be interpreted with caution, some facts could be consistent with a possible impact of the screening programme on the reduction of BC diagnosed at an advanced stage in women aged 50–74. The drop in T2+ BC in women aged 50–74 contrasted with the stability of T2+ BC in the oldest and youngest groups of women. In older women, the stability of T2+ BC while T1 BC incidence was still in progression would be concordant with an impact of the screening programme in a context of growing underlying trend of incidence. It can also be argued that the higher the rate of participation in the screening programme in one area, the greater the decrease of T2+ BC incidence. Contrary to the nonlinear evolution of T1 BC incidence rate in the 50–74 age group, the changes in the T2+ BC incidence rate were linear throughout the study period. Most particularly, the evolution of T1 BC in women aged 50–74 followed a well-known pattern described in many countries, including France, which has already been discussed in relation to the decreasing prescription of hormone replacement therapy.23,24 The decrease in T1 BC incidence was restricted to the 2003–2006 period; a stabilization occurred after 2006 when hormone replacement therapy use stabilized at a lower rate. The extent to which overdiagnosis could explain the changes in T1 BC remains under debate.25–27 In this study, the advanced T2+ BC incidence reduction is possibly underestimated for two main reasons. First, rather than remaining steady, the underlying BC incidence may have increased. The increasing BC incidence observed in younger women in our study (+0.9%/year), as well as in other studies during a longer period (1990–2010), would be consistent with this hypothesis.28,29 Second, many women delay or skip a screening session (the re-attendance rate for the next screening round was estimated at around 80% in Isère in 2003), losing the benefit of screening every two years, and thus exposing themselves to a BC diagnosed at a more advanced stage. 30 Only three areas were included in this study, and these may not be representative of the whole of France. However, they were pilot areas for screening programme implementation before 2000, whereas the screening programme was introduced in most other areas only in 2004, too recently for an adequate assessment of trends.
This observational study does not allow causal inference, and it cannot ascertain whether, or which part of, the overall linear decrease of T2+ BC incidence in the target age group of the screening programme was related to the screening programme itself. However, this result is encouraging, and further studies are needed in other geographic areas where organized screening was introduced after 2004.
Footnotes
Acknowledgements
We thank Jacques Estève for assistance in methodology and interpretation of the results. We are grateful for the generous assistance of the pathologists, oncologists, Departments of Medical Data Processing of public and private hospitals, and medical practitioners as well as the medical services of the national health insurance programme that routinely provide data to the cancer registries. The authors also thank Linda Northrup (English Solutions) for translation and editorial assistance.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Grant sponsors for routine collection of data: National Cancer Institute (INCa), French Institute for Public Health Surveillance (InVS), local public institutions (Conseil Régional, Conseil Départemental), and Ligue contre le cancer.
