Abstract
Objectives
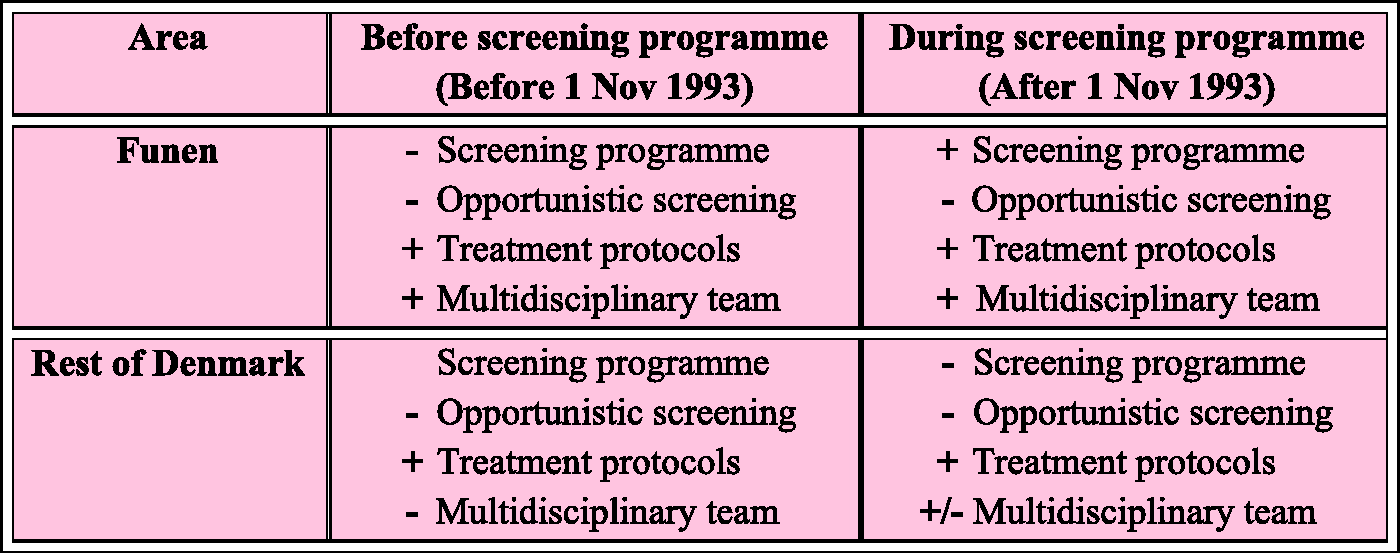
When estimating the decline in breast cancer mortality attributable to screening, the challenge is to provide valid comparison groups and to distinguish the screening effect from other effects. In Funen, Denmark, multidisciplinary breast cancer management teams started before screening was introduced; both activities came later in the rest of Denmark. Because Denmark had national protocols for breast cancer treatment, but hardly any opportunistic screening, Funen formed a “natural experiment”, providing valid comparison groups and enabling the separation of the effect of screening from other factors.
Methods
Using Poisson regression we compared the observed breast cancer mortality rate in Funen after implementation of screening with the expected rate without screening. The latter was estimated from breast cancer mortality in the rest of Denmark controlled for historical differences between Funen/rest of Denmark. As multidisciplinary teams were introduced gradually in the rest of Denmark from 1994, the screening effect was slightly underestimated.
Results
Over 14 years, women targeted by screening in Funen experienced a 22% (95% confidence interval 11%–32%) reduction in breast cancer mortality associated with screening (a reduction in breast cancer mortality rate from 61 to 47 per 100,000). The estimated reduction for participants corrected for selection bias was 28% (13%–41%). Excluding deaths in breast cancer cases diagnosed after end of screening, these numbers became 26% and 31%, respectively.
Conclusions
There is additional benefit in reducing breast cancer mortality from the early detection of breast cancer through mammographic screening over and above the benefits arising from improvements in treatment alone.
Introduction
The purpose of mammography screening is to reduce breast cancer mortality. An early summary of randomized controlled trials data from Sweden indicated a 25% reduction in breast cancer mortality for women targeted by screening at ages 50–69, 1 although reduced mortality in randomized controlled trials does not necessarily mean that screening also works in routine health care.
An observed reduction in breast cancer mortality can be attributable to both treatment and screening effects. 2 The introduction of mammography screening is often closely connected to establishment of multidisciplinary breast cancer management teams, making it impossible to distinguish one effect from the other. To determine how much of the observed reduction in breast cancer mortality is attributable to screening, the challenge is to distinguish the screening effect from effects of advances in treatment.
In the county of Funen, Denmark, an organized biennial screening programme for women aged 50–69 was introduced in November 1993. The programme was assessed as satisfactory on the European short-term indicators.
3
While a few other areas in Denmark started screening at about the same time as Funen, the rest of Denmark started screening in 2008 (see Figure 1).
4
From August 1979 Funen introduced multidisciplinary teams, consisting of specialized surgeons, breast radiologists, specialized pathologists, and oncologists, whereas this was implemented gradually from 1994 in the rest of Denmark. In addition, from 1977 all Danish breast cancer patients were treated according to national protocols issued by the Danish Breast Cancer Cooperative Group (DBCG).
5
The use of opportunistic screening has always been limited in Denmark.
6
Denmark therefore performed a “natural experiment” which allows the comparison of breast cancer mortality in Funen with the expected breast cancer mortality in the absence of screening, thereby making it possible to distinguish the effect of screening from the effect of improvements in treatment (see Figure 2). We report here on this “natural experiment”.
Time line for introduction of national breast cancer treatment protocols, multidisciplinary teams, and screening programmes in Denmark. The presence/absence of screening programme, opportunistic screening, breast cancer treatment protocols, and multidisciplinary breast cancer management teams in Funen before and during screening, and in the rest of Denmark in the same calendar periods.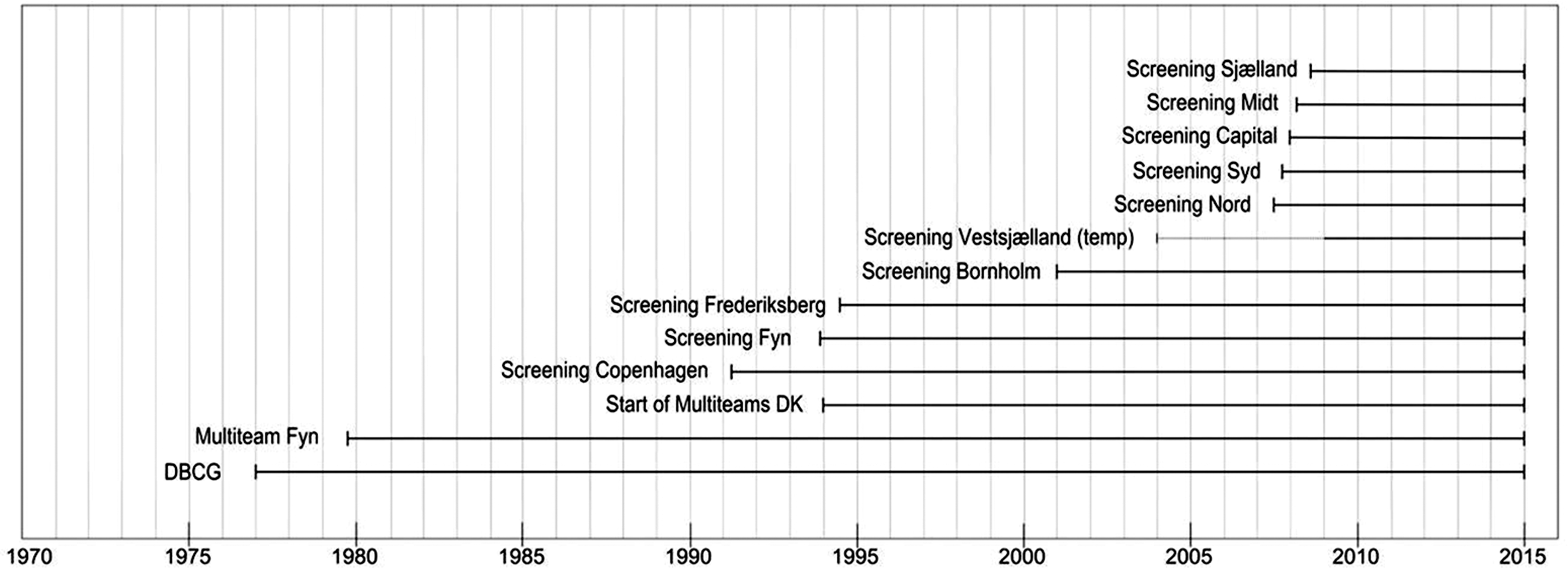
Methods
Setting and screening organization
The DBCG, formed in 1977 and including all medical specialties involved in the diagnostics and treatment of Danish breast cancer patients, has issued national, evidence-based, and updated protocols for breast cancer treatment, and the activity has been monitored via a common database and reported in numerous scientific publications. Multidisciplinary teams, where the surgeon, radiologist, pathologist, and oncologist form the core diagnostic group, 7 were implemented in Funen from August 1979, but only introduced gradually in the rest of Denmark from 1994. 8
The Funen screening programme started on 1 November 1993, offering biennial screening to women aged 50–69 at the date of invitation. All targeted women were personally invited to participate in the first round. In subsequent invitation rounds, targeted women were invited only if they had participated in the previous round, were newcomers to the programme, or had themselves contacted the programme. All screens were taken by radiographers and specialized nurses, and attending women did not see a radiologist. At first screen, all women had two views (craniocaudal and oblique) of each breast. Up until 2004 women with mixed/dense breast tissue continued to have two views at subsequent screens, while women with fatty breast tissue had one view oblique mammography. From 2004 onwards, all women had two-view mammography. All mammograms were centrally evaluated independently by two radiologists, and were compared at subsequent screens with those taken earlier. During the first invitation round the participation rate was 84%. 3
Regional, organized programmes with biennial mammography screening of women aged 50–69 were implemented in the municipalities of Copenhagen in 1991, Frederiksberg in 1994, Bornholm in 2001, and in part of the County of Vestsjælland in 2004. The national roll out of organized mammography screening in Denmark commenced in 2008. 4
Construction of study group Funen1993–2007/09 and regional control group rest-DK1993–2007/09
The study group Funen1993–2007/09 included women invited to the Funen mammography screening programme without prior invitation to any of the other organized programmes (see Figure 3). A woman was considered at risk from the date of her first invitation until date of death, emigration, or end of follow-up.
Study design illustrated in Lexis’ diagram.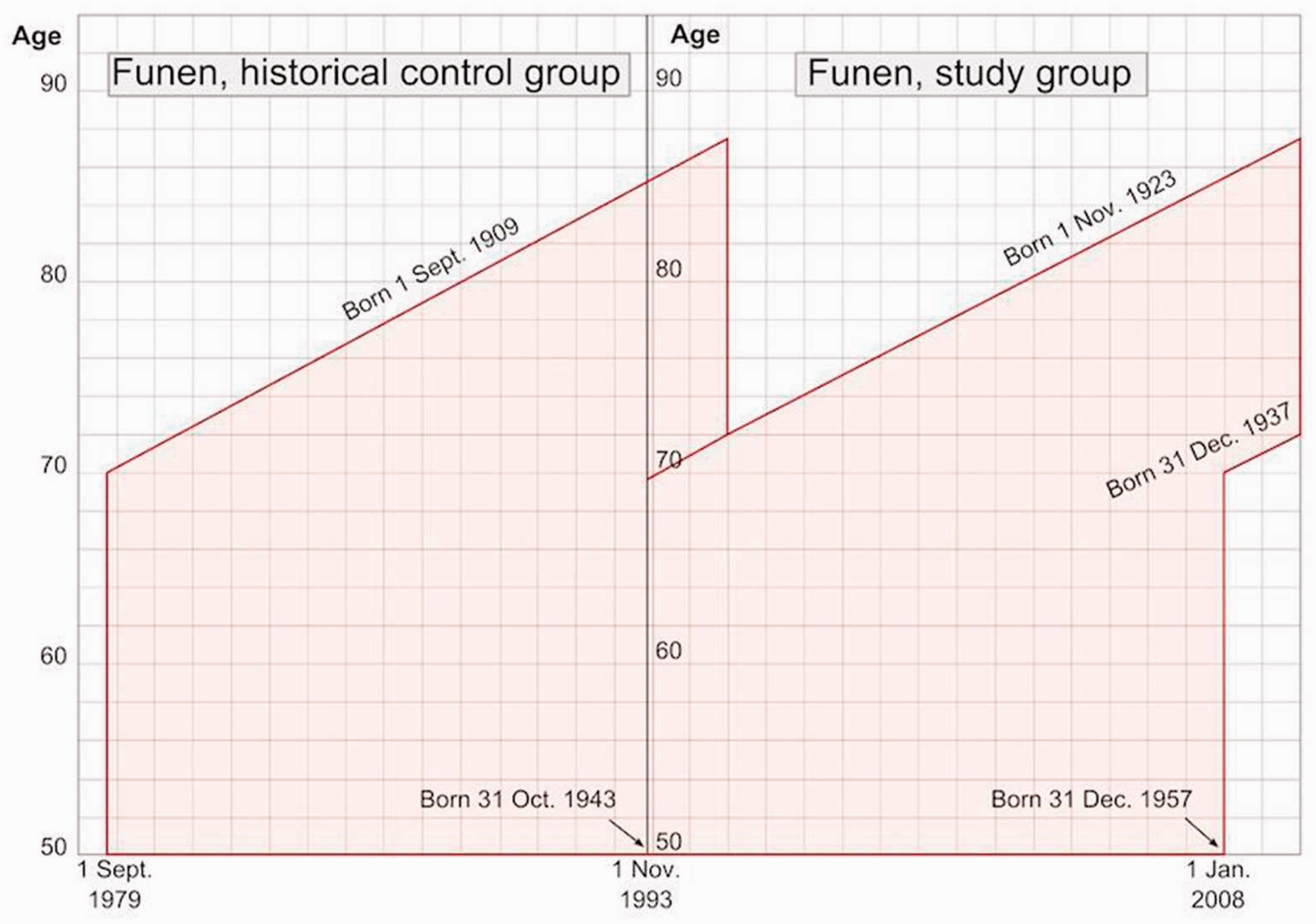
The Danish Central Population Register holds information on current and historical addresses for all citizens living in Denmark since 1968. Information from the Central Register and in all other registers used in the present study can be linked by personal identification number. Date of first invitation and participation was retrieved from the Funen County IT-Centre mammography screening database, however, in this database an invitation date could be overwritten at re-invitation. For women who participated continuously in the programme this does not happen. It was, however, a problem to find the first invitation date for some subgroups, such as initial non-attenders, who later contacted the programme to obtain a new invitation date. For these women, date of first invitation was defined based on stored invitation dates, date of birth, and addresses in the Population Register. Women living in Copenhagen, Frederiksberg, Vestsjælland, or Bornholm at screening age and operational periods for these programmes prior to their first invitation to the Funen programme, were excluded from Funen1993–2007/09.
The study group Rest-DK1993–2007/09 was constructed to resemble Funen1993–2007/09 and included women from the rest of Denmark except Copenhagen, Frederiksberg, Vestsjælland, or Bornholm, where screening programmes had been in place. For simplicity, we call this area rest of Denmark, shortened to rest-DK. Data from the Population Register were used for this purpose. Women in rest-DK1993–2007/09 were allocated pseudo first invitation dates on a random basis, based on the invitation schedule used in Funen1993–2007/09.
Because women born after 1 January 1938 could have been invited to the national screening programme from 1 January 2008 onwards, these women were followed-up only until 31 December 2007. Women born before 1 January 1938 could not have been invited to the national screening programme, and were therefore followed-up until 31 December 2009. Person years at risk were accumulated from the woman’s entry into Funen1993–2007/09 or rest-DK1993–2007/09 until death, emigration, or end of follow-up (31 December 2007 or 31 December 2009), whichever came first.
Construction of historical control group Funen1979–1993/95 and historical, regional control group rest-DK1979–1993/95
In order to check and possibly adjust for regional differences between Funen and the rest of Denmark, we constructed two groups, Funen1979–1993/95 and rest-DK1979–1993/95. Funen1979–1993/95 resembled Funen1993–2007/09, including women living in Funen at screening age in the 14 years and 2 months preceding the start of screening, but excluding women living in Copenhagen, Frederiksberg, Vestsjælland, or Bornholm in this period (see Figure 3). Rest-DK1979–1993/95 resembled rest-DK1993–2007/09, including women living in the rest of Denmark in the 14 years and 2 months preceding the start of screening in Funen, who had not moved to Copenhagen, Frederiksberg, Vestsjælland, or Bornholm during this period. Women in these two control groups were allocated pseudo first invitation dates 14 years and 2 months earlier than the first invitation dates in the Funen programme.
To allow equal follow-up time for all groups, we followed women in Funen1979–1993/95 and rest-DK1979–1993/95 born after 1 November 1923 until 31 October 1993, and women born before 1 November 1923 until 31 October 1995. Person years at risk were accumulated from the woman’s entry into Funen1979–1993/95 or rest-DK1979–1993/95 until death, emigration, or end of follow-up (31 October 1993 or 31 October 1995), whichever came first.
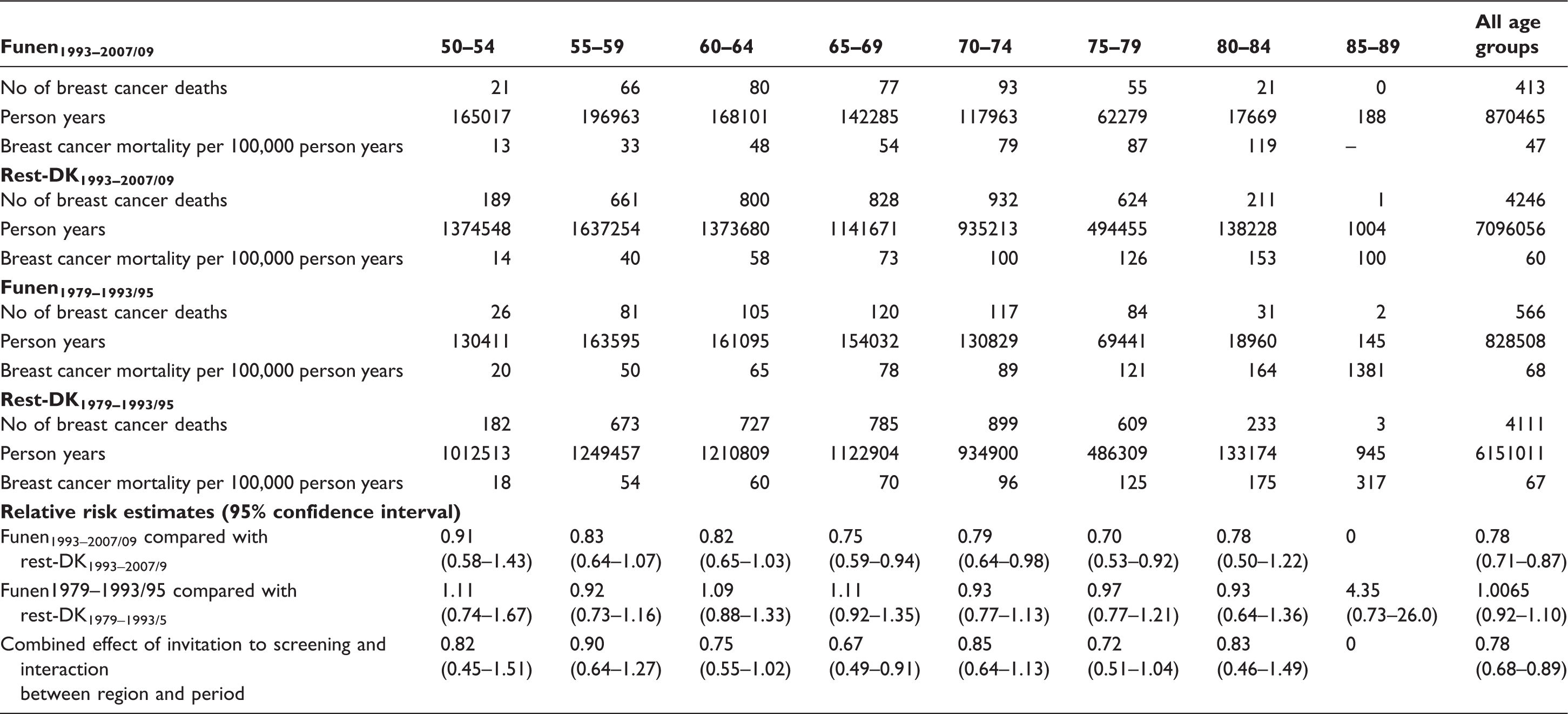
Breast cancer deaths and person years (in women diagnosed after first invitation to screening) in Funen1993–2007/09, Rest-DK1993–2007/09, Funen1979–1993/95 and Rest-DK1979–1993/95, during the follow up from 1 November 1993 to 31 December 2007 (women born after 1 January 1938) or 31 December 2009 (women born before 1 January 1938), as well as breast cancer deaths and person years for the three control groups (see text for details).
All groups
Incident cases of breast cancer and ductal carcinoma in situ were identified by linkage to the Danish Cancer Register, which holds nationwide data on breast cancer cases since 1943, and the database of the DBCG, which holds nationwide data on invasive breast cancer and ductal carcinoma in situ dating back to 1977. Because breast cancers diagnosed before invitation to mammography screening cannot benefit from screening, women with breast cancer diagnoses prior to the date (or pseudo-date) of first invitation to mammography screening were excluded from all four groups, following the incidence-based-mortality method. 9
Analysis
The expected breast cancer mortality in the absence of screening was estimated based on the breast cancer mortality in rest-DK1993–2007/09 controlled for possible historical differences in breast cancer mortality between Funen1979–1993/5 and rest-DK1979–1993/95. This will only give a reliable estimate of the expected breast cancer mortality if breast cancer mortality in the absence of screening developed equally in Funen and rest of Denmark, ie. no interaction between region and period. When interpreting the results, we therefore have to take this possible interaction into consideration.
Poisson regression was used to analyze the breast cancer mortality rate as a function of exposure (women invited to screening, women not invited to screening), period (1979–1993/5, 1993–2007/9), region (rest-DK, Funen) and 5-year birth cohorts. The results were reported as relative risks (RR), with 2-sided 95% confidence intervals (CI).
We performed a corresponding analysis for women participating in at least one of the screenings offered to them. To correct RR among participants for selection bias we used the method of Duffy et al. 10
Breast cancer deaths in cases diagnosed above the age of 70 cannot benefit from screening, because the women are no longer targeted by the screening programme, however, excluding these breast cancer deaths from the analysis would create a bias, due to the lead time. All breast cancer deaths were therefore included in the analysis. Additionally, to estimate in an unbiased way the effect of screening on breast cancer mortality among cases diagnosed while the women were still eligible for screening, we used a counterfactual exclusion method described in Appendix.
To study the screening effect by age at first invitation, we calculated cumulative breast cancer mortality in Funen1993–2007/09 and in the rest-DK1993–2007/09 for women aged 50–59 and 60–69, respectively, at first invitation.
Poisson regression analyses were conducted using SAS version 9.2 (SAS version 9.2, SAS Institute Inc, Cary, NC).
The study was approved by the Danish Data Inspection Board, which according to Danish legislation serves as ethical approval of register-based research projects (notification number 2008-41-2191).
Results
Between 1979 and 1993/95, the breast cancer mortality rate was remarkably similar in Funen and in rest-DK, with a RR of 1.0065 (95% CI 0.92–1.10, see Table 1). From 1993 to 2007/09, however, the breast cancer mortality rate was significantly lower in Funen than in rest-DK, with a RR of 0.78 (95% CI 0.71–0.87). Taking the very small historical difference between Funen and the rest of Denmark into account gave a RR of 0.78 (95% CI 0.68–0.89).
Among screening participants in Funen1993–2007/09, the RR of breast cancer mortality was 0.68 (95% CI 0.59–0.79) compared with what would be expected without screening. Non-participants had a RR of 1.51 (95% CI 1.22–1.86). When this selection bias was controlled for, the RR for participants became 0.72 (95% CI 0.59–0.87).
A subsequent analysis, excluding breast cancer deaths in cases diagnosed in women aged over 70 (76 women), gave an estimated effect of screening in invited women of RR 0.74, and an estimated effect in participants of RR 0.69.
Among women aged 60–69 at first invitation, the reduction in breast cancer mortality was already visible after 2–3 years (Figure 4). Among women aged 50–59 at first invitation, the reduction in breast cancer mortality was visible after 6 years.
Cumulative breast cancer mortality per 100,000 by time since first invitation for women aged 50–59 and 60–69 years at invitation. Dotted line: Study group, Full line: Regional control group.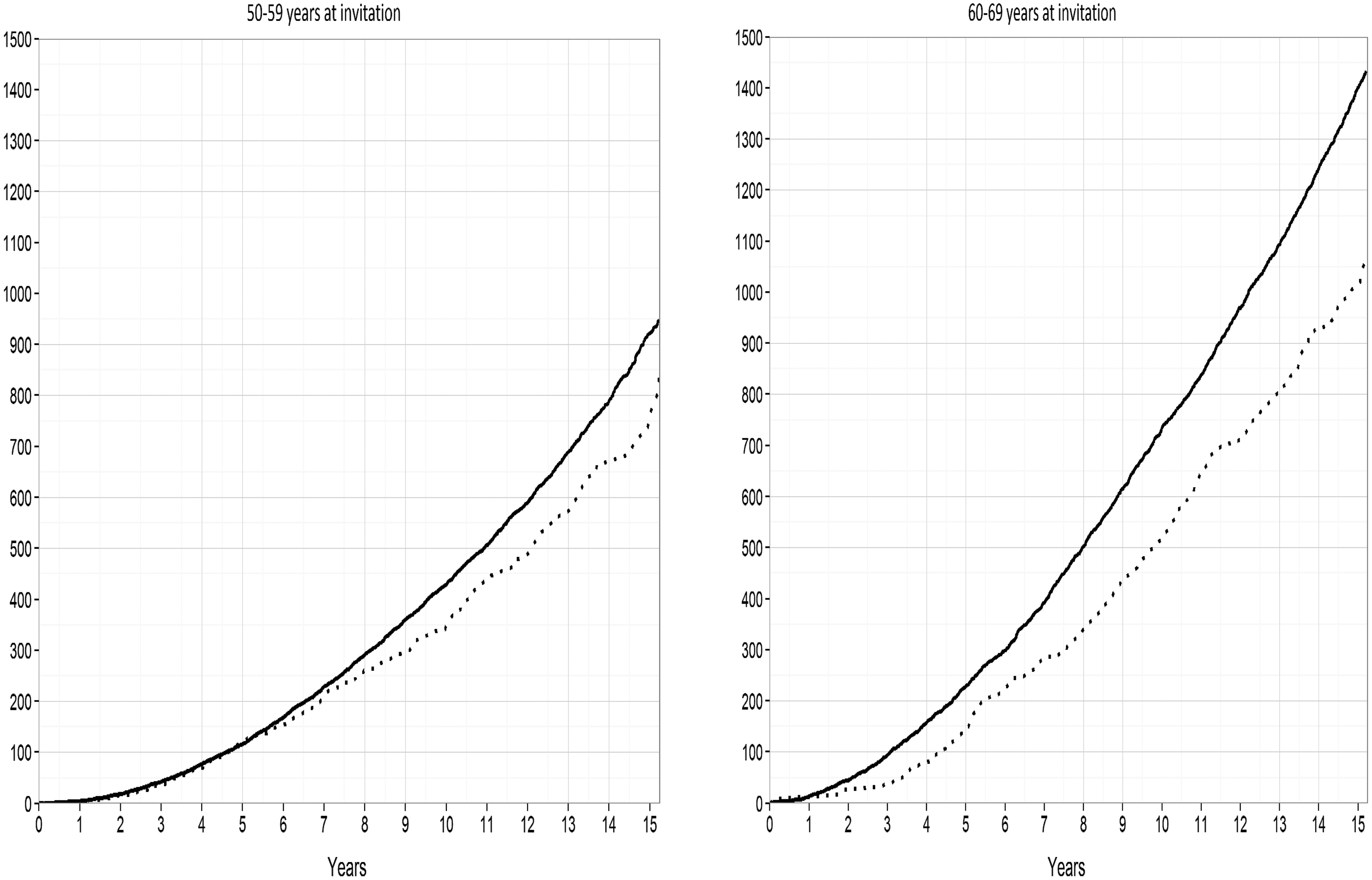
Discussion
Principal findings
This incidence-based mortality study found that women targeted by the organized mammography screening programme in Funen, Denmark, experienced a 22% lower breast cancer mortality than expected in the absence of screening. When interpreting this result, we have to take the possible interaction between region and period into consideration. Such an interaction would occur if breast cancer incidence had developed differently over time in Funen and Rest-DK, or if Funen had, as well as screening, implemented improvements in treatment not implemented in the rest of Denmark, but this was not the case. We have previously shown that the breast cancer incidence developed similarly over time in Funen and in the Rest-DK. 11 Furthermore, since 1977 all hospital departments have used uniform DBCG protocols for histopathology, surgery, radiotherapy, and systemic therapy, and the multidisciplinary teams existed in Funen long before the screening started. This means that the use of systemic therapy, a key driver in the decrease of breast cancer mortality, was controlled for in this study. Based on this, it is reasonable to expect interaction between period and region to be very small.
Participating women experienced a 28% lower breast cancer mortality (corrected for selection bias). When deaths in breast cancer cases diagnosed after the women left the screening programme were excluded, the reduced risk became 26% and 31%, respectively.
Funen had multidisciplinary teams in both study periods (1979–1993/95 and 1993–2007/9), but if multidisciplinary teams had not been introduced in the rest of Denmark before the end of 2009, we could have estimated the expected breast cancer mortality in the absence of screening without interference from the effect of multidisciplinary teams. However multidisciplinary teams were implemented gradually in the rest of Denmark from 1994, thus yielding slightly lower breast cancer mortality than would have been expected without the implementation of multidisciplinary teams. Our estimates of screening effect on breast cancer mortality are therefore slightly underestimated.
A 22% reduction in the breast cancer mortality rate for Funen translates into a reduction from 61 per 100,000 to 47 per 100,000. In absolute numbers these relative reductions equal 413 breast cancer deaths among screened women, and 531 breast cancer deaths without screening, giving 118 lives saved among the 86,726 women invited to mammography screening in Funen.
Strengths and weaknesses of the study
The fact that for part of the study population we had to define missing first invitations from Population Register data on date of birth and addresses could, in theory, have led to over- or under-estimation of individual risk periods. However, first invitation dates were estimated for only 2 out of 413 breast cancer deaths in Funen1993–2007/09.
In the incidence-based mortality analysis we excluded women diagnosed with invasive breast cancer or ductal carcinoma in situ prior to their first invitation from both study and control groups. However, from 1997 onwards, the Funen programme did not invite women with prior excisions from non-specified neoplasms. As this diagnosis is very rare, this exclusion is unlikely to have affected the result.
Comparison with other studies
Recently, the Independent UK panel on Breast Cancer Screening estimated the effect on all age groups included in the trials to be 20%. 12 A recent review of incidence-based mortality studies from European population-based mammography screening programmes gave a pooled estimate of a 26% breast cancer mortality reduction among invited women. 13
Using simulation models, Berry et al 14 showed that screening and adjuvant therapy, compared with adjuvant therapy alone, reduced breast cancer mortality just as much as screening and no adjuvant therapy compared with no screening and no adjuvant therapy. This suggests that screening reduces breast cancer mortality equally in a population with and without adjuvant therapy. We found that screening reduces breast cancer mortality by at least 22% in a population where multidisciplinary teams were already implemented, where previous incidence-based studies have found that screening reduces breast cancer mortality by approximately 26% in a population where multidisciplinary teams were not implemented. 13
In other studies, time trends in routine, age-specific rates have been used to estimate the impact of screening (eg. Jørgensen et al. 15 and Burton et al. 16 ). Moss et al. 17 considered that estimating screening impact on breast cancer mortality by analyzing mortality trends was a method of limited value.
Jørgensen et al. analyzed time trends in breast cancer mortality for the period 1971–2006 in Denmark. This study merged data from Copenhagen, Funen, and Frederiksberg, used age-groups instead of birth cohorts, and population instead of individual data. 15 The study therefore did not exclude deaths in breast cancer cases diagnosed prior to start of screening. Jørgensen et al. furthermore focused on the slopes of mortality trends before and after screening, disregarding changes in a transition period. The conclusion was that the researchers were unable to find an effect of the Danish screening programme on breast cancer mortality. A recalculation of their data, taking the change in level of lines within the transition period into account, is compatible with a 13% decline in breast cancer mortality following introduction of screening. 18 It is noteworthy that no decline was seen in the age-group 35–54, nor in the age-group 75–84. This estimate, based on the Jørgensen et al. data, which are crude and contaminated by deaths in women diagnosed with breast cancer before screening, is well in line with the reductions of 25% 19 and 22%, respectively, found in the incidence-based mortality data.
Kalager et al. used data from the Norwegian screening programme to try to distinguish the mortality reduction caused by screening from the mortality reduction caused by advances in breast cancer awareness and treatment. 20 Unfortunately, the implementation of screening and multidisciplinary teams happened almost simultaneously in Norway, 20 making it very hard to distinguish between the two effects/strategies. Kalager et al. quantified the reduction in mortality caused by the establishment of multidisciplinary teams according to the relative reduction observed among women aged 70 or older at diagnosis, but the relative reduction among these women is affected by a lead time bias. Because breast cancer cases among screened women are diagnosed earlier, there will be fewer cases in screened women older than 70, yielding a lower breast cancer mortality, independently of changes in treatment.
The estimate of 22% in the present study corresponds well with the 26% (95% CI 13%–36%) from other incidence-based mortality studies, 13 especially considering that our 22% is probably an underestimate. It is probably due to underestimation, or chance, that our estimated effect for participating women of 31% in Funen is lower than the 37% previously found for the Copenhagen programme. 19
For women first invited at age 60–69, the effect of screening was visible 2–3 years after the start of screening; for women first invited at age 50–59, the effect was visible after 6 years. This is comparable with the results from the Swedish randomized controlled trials for women in the same age-groups. 21
Conclusions
There is additional benefit in reducing breast cancer mortality from the early detection of breast cancer through mammographic screening over and above the benefits arising from improvements in treatment alone. In the organized, population-based service mammography screening programme in Funen, Denmark, we observed a 22% decrease in breast cancer mortality in targeted women. This decrease can, at most, only have been slightly affected by advances in breast cancer awareness and treatment. Correcting for self-selection bias resulted in a 28% decrease in breast cancer mortality among participants.
Footnotes
Funding
This study was financially supported by the Esper and Olga Boel Foundation. The funding source had no role in the study design, the analysis, interpretation of data, writing of the manuscript, or the decision to submit it for publication.
Acknowledgements
We are indebted to Miguel Vazquez-Prada Baillet for graphical presentations.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
