Abstract
Objective
To present performance indicators from the second round of the Vallecamonica-Sebino HPV screening programme in women who had tested negative about four years earlier (mean 45 months).
Methods
From 2010 to 2012, the target female population (aged 25–64) was invited to the first HPV screening round. In 2013–2017, women were rescreened for the second round. HPV-negative women at the first round were initially rescreened after three years. The interval was gradually increased to five years. HPV-positive women underwent cytology triage: positives were referred to colposcopy and negatives to repeat testing after one year. If HPV was persistently positive, women were referred to colposcopy, if negative, to normal interval rescreening.
Results
In the second round, of 13,824 previously HPV-negative women, 598 were HPV-positive (4.3%), of whom 297 were positive at cytology triage. Of those referred to one-year HPV test, 291 complied (98.0%), 133 (50.2%) of whom were persistently positive. Total referral was 3.1% compared with 6.6% in the first round (age-adjusted relative referral 0.59, 95% CI: 0.53–0.65). There were 24 cervical intraepithelial neoplasia 2+ (three cervical intraepithelial neoplasia 3+). Detection was 0.17%, compared with 0.9% in the first round. Age-adjusted relative detections were 0.25 (95% CI: 0.16–0.39) and 0.18 (95% CI: 0.05–0.61) for cervical intraepithelial neoplasia 2+ and cervical intraepithelial neoplasia 3+, respectively. Positive predictive value was 5.7%, compared with 14.6% in the first round.
Conclusions
At second round, referral was half that at first round, while cervical intraepithelial neoplasia 2+ detection decreased nine-fold. Consequently, positive predictive value decreased dramatically. Rescreening four years after an HPV-negative test makes the process inefficient due to the low prevalence of lesions.
Keywords
Introduction
In 2012, an Italian Health Technology Assessment report recommended the use of HPV DNA testing for screening cervical cancer precursors. 1 In 2013, the Italian Ministry of Health asked Regional Governments to begin implementation. This decision followed the publication of several trials showing that HPV-based testing is more effective than Pap testing in the early diagnosis of cervical intraepithelial neoplasia 3 (CIN3).2–5 The evidence was then corroborated by the pooled analysis of European trials, showing that HPV screening was more effective than Pap test screening in preventing cervical cancer. 6 In 2015, European guidelines recommended the implementation of HPV-based screening, followed by cytological triage to identify women to be referred to colposcopy, and those to be referred to early rescreening. 7 After the publication of the European guidelines, the Italian Ministry of Health and the Regional Governments set the goal of complete conversion of Italian screening programmes from Pap test to HPV test by the end of 2018. 8
From 2007, when evidence from randomized trials began to emerge,2,3 the Italian Ministry of Health funded several pilot studies to test organizational impact, acceptability by women and operators, performance of cytology triage, impact on midwifes and the laboratory, cytology and colposcopy workload, and costs of HPV-based screening.9–15 The Vallecamonica-Sebino pilot study started in 2010 16 and has now completed the second-round follow-up for the target population. Given the relatively remote and isolated area, it represents a unique opportunity to study a cohort of women in a European country who have been screened using HPV testing over a long period.
The aim of this paper was to report the results of the second round of an HPV-based screening programme and to compare them with those of the first round.
Methods
Vallecamonica-Sebino is a community in Northern Italy (99,776 inhabitants in 2010). In 2010, public health services were provided by a single Local Health Authority, but in 2015 that Authority merged with others, forming in a much larger provincial Health Authority that included the province of Brescia, with about 1.3 million inhabitants. Vallecamonica-Sebino is located in the pre-Alpine and Alpine zone in the region of Lombardy. A screening programme that invites the entire female population aged 25–64 every three years for a Pap test was introduced in 2002. All the tests, ascertainment and treatment are free.
The pilot study using HPV as primary screening test started in 2010, completing its first round in 2012. The entire eligible female resident population was invited to participate (n. 27,581 in 2010). 16 We here report the main performance and early outcome indicators in women at their second screening round (screened from 2013 to 2017) who had previously tested HPV-negative.
The pilot study adopted the experimental HPV-based protocol proposed by the Italian Group for cervical cancer screening, 17 described in the report of the first screening round. 16 Eligible women aged 25–64 were invited for a new screening round with HPV testing. A double sampling (conventional smear and Specimen Transport Medium (Qiagen, Hilden Germany) for HPV) was performed on all the participating women. After the results of the HPV test, slides from positive tests were dyed and interpreted by a single cytologist. Women with cytology showing atypical squamous cells of undetermined significance (ASC-US) or more severe, and those with unsatisfactory slides were referred to colposcopy. Women with negative cytology were referred to one-year HPV re-testing. HPV-negative women were initially referred to three-year rescreening, but based on the Italian Health Technology Assessment report, 1 the Ministry of Health in January 2013 recommended an interval of at least five years. The interval was gradually increased, and women screened in 2012 were invited to second-round screening after more than 4.5 years.
HPV testing was performed using the Hybrid Capture 2 Assay (HC2; Qiagen, Hilden, Germany). Only the group of probes designed to detect 13 high-risk HPV types was used (12 included in IARC group 1: 16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59; one included in IARC group 2a: 68). 18 All samples with a relative light unit/positive control ≥ ratio 1 were considered positive. The laboratory participated in the Gruppo Italiano Screening Cervicale inter-laboratory quality programme. 19 Cytology was classified according to the 2001 Bethesda System. 20 For the external quality control, all positive slides and a 10% random sample of negative slide were reviewed by an external supervisor (MC).
Colposcopy was performed according to Italian recommendations. 21 Colposcopy-guided biopsies were taken in all positive colposcopies, and almost always in cases of atypical squamous cell (ASC-H), high-grade squamous intraepithelial lesion (HSIL), and glandular lesions. Histology was performed in the pathology laboratory of the local hospital by a single pathologist with haematoxylin and eosin staining. P16 immunostaining was used as diagnostic help in equivocal cases to distinguish between CIN2 and CIN1 or CIN2 and atypical immature metaplasia.
We compared the values of the main performance and early outcome screening indicators7,22 in women in the second round who tested negative at baseline HPV test in the previous round, with those in the first round:
participation in screening programme; positivity to HPV test; rate of abnormal cytology among HPV-positives; compliance with one-year follow-up referral; compliance with colposcopy referral (immediate and at one-year follow-up); PPV for CIN2+ and CIN3+ (overall, at baseline and at one-year follow-up); CIN2+ and CIN3+ detection rate (overall, at baseline and at one-year follow-up).
Overall detection rate includes all CIN2+ or CIN3+ detected in women referred to colposcopy within 24 months of the first screening test (i.e. up to 2018 for women screened in 2016). It also includes some lesions found in the follow-up after a first colposcopy negative for CIN2+ and is therefore equal to or greater than the sum of baseline and one-year follow-up detection. Comparisons are expressed as age-adjusted relative measures and difference for 1000 screened women with 95% CI.
The study was approved by the Vallecamonica-Sebino Ethics Committee.
Results
In the first round of the programme (March 2010–December 2012), 27,581 women were invited to the HPV-based screening programme, and 18,728 participated. Of these, 17,095 tested negative. After a median time of 42 months (mean 45), 15,677 women were still eligible for screening and were invited to the second round (1418 exited the target age or moved out of the programme catchment area). Participation in the second round was 88.2% (Figure 1).
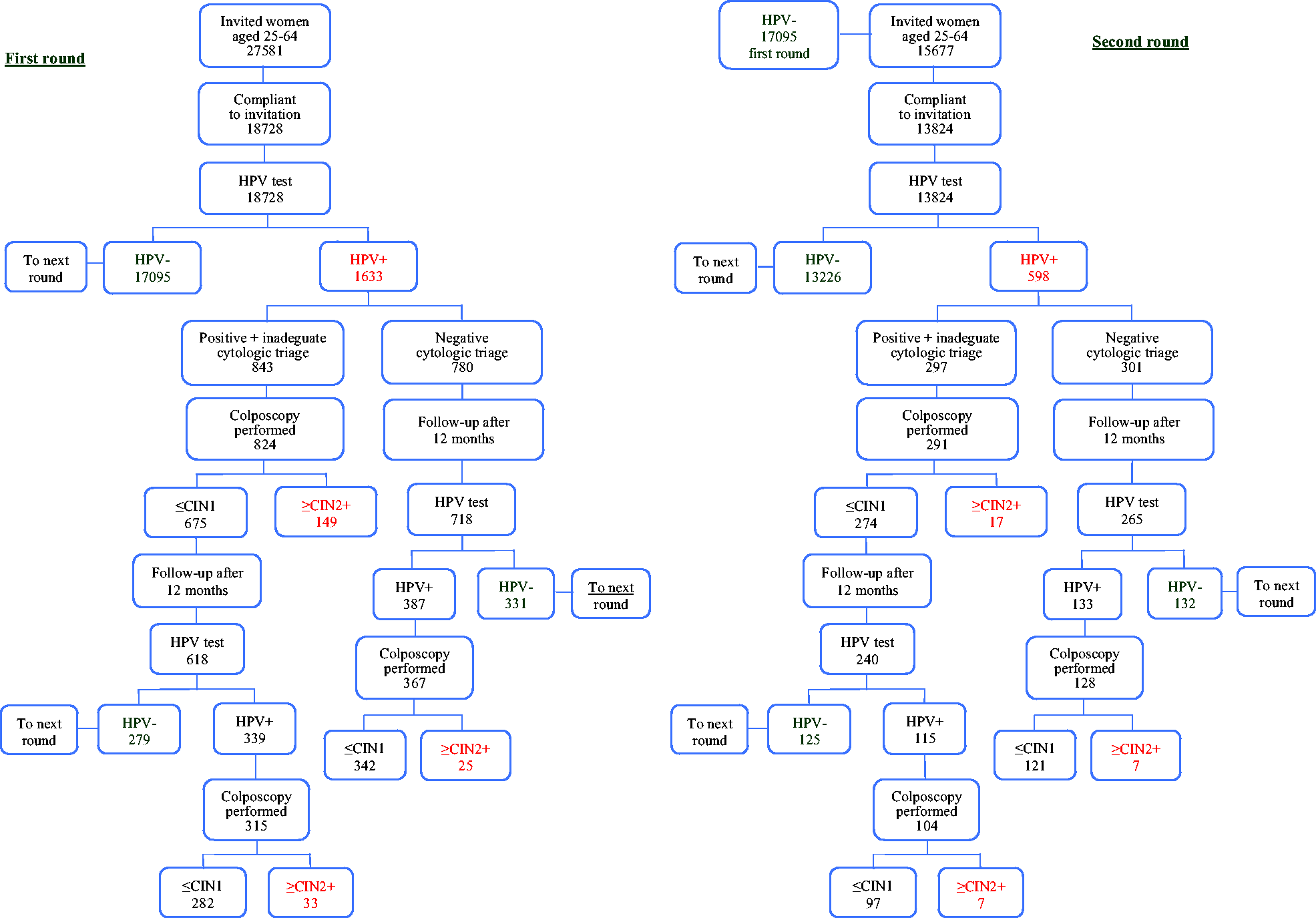
Flowchart and main data of the second round of the Vallecamonica-Sebino HPV test-based screening programme, Italy 2013–2018.
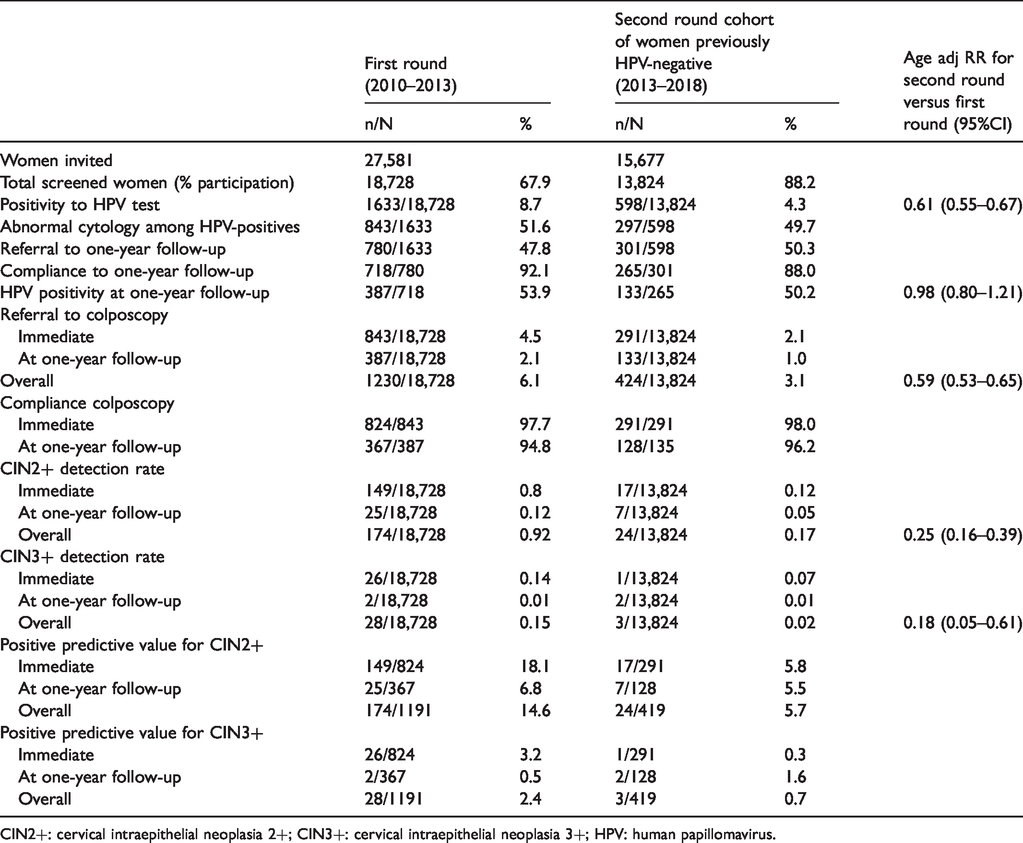
The HPV positivity rate was 4.3% (598/13824), compared with 8.7% in the first round (age-adjusted prevalence ratio 0.61, 95% CI 0.55–0.67) (Table 1). The curve of HPV prevalence by age at first round and at second round (where only newly detected infections were present) decreased proportionally, with prevalence in the second round equal to almost one-half the prevalence in the first round. The rate of newly detected infections did not fall to zero, even in women aged over 60 (Figure 2(a)).
Main performance and early outcome indicators of the HPV test-based screening programme. First round (women screened from March 2010 to December 2012 and followed up to 2013) and second round (women screened in 2013–2017 and followed up to 2018).
CIN2+: cervical intraepithelial neoplasia 2+; CIN3+: cervical intraepithelial neoplasia 3+; HPV: human papillomavirus.
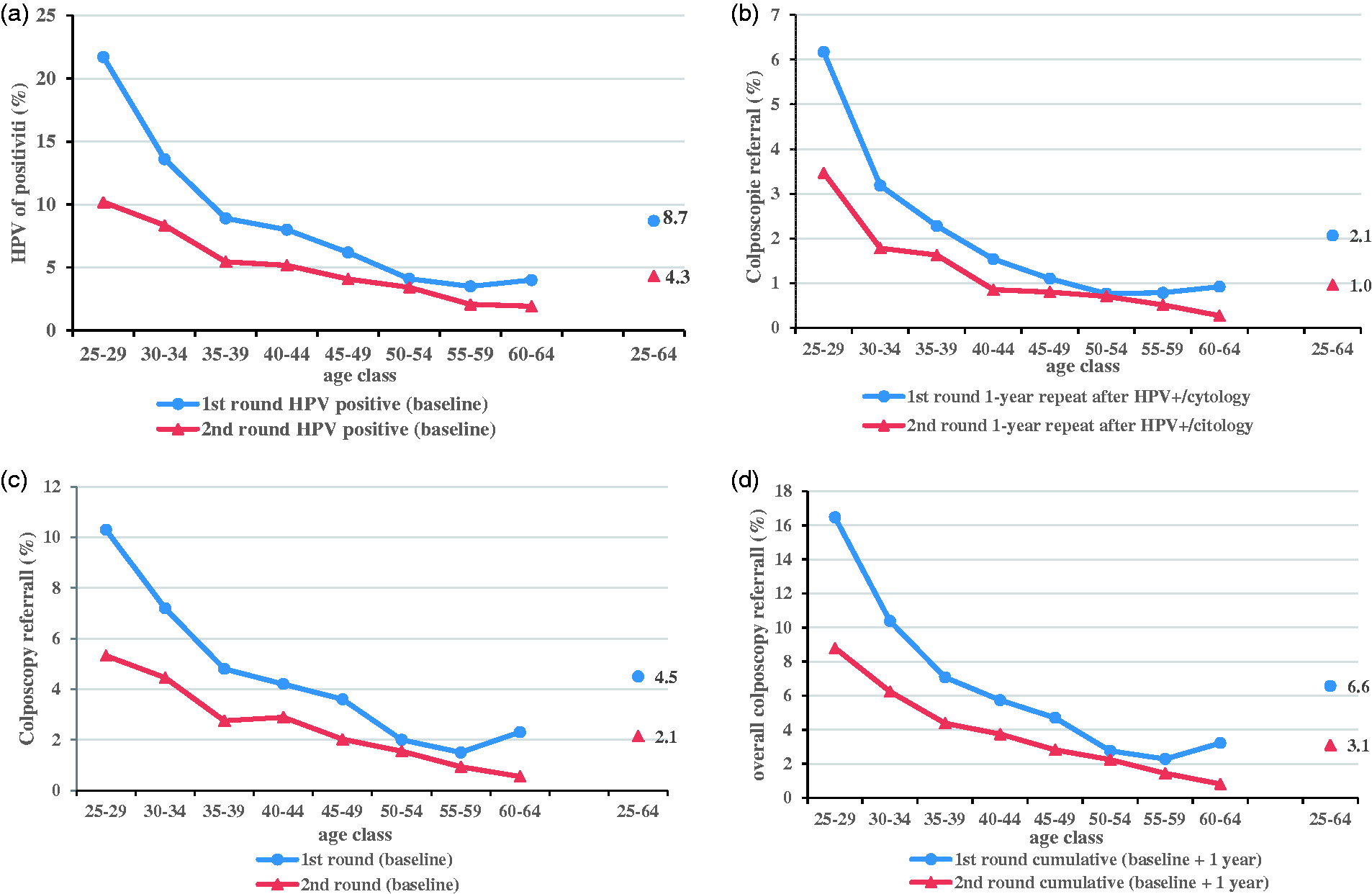
HPV positivity (a), colposcopy referral at baseline (b), at one-year follow up (c), and overall colposcopy referral (d), by age and screening round.
Among the 598 HPV-positive women, 49.7% (297) had a non-negative cytology. In the first round, the proportion of non-negative cytology was 50.3% (Table 2). In the second round, ASC-US and low-grade squamous intraepithelial lesion (LSIL) represented about 94% of all epithelial cell abnormalities, both with a very low PPV (4.8 and 6.1%, respectively). ASC-H and HSIL classes maintained a sufficient PPV, but they accounted only for 3% of all the abnormalities. As both HPV positivity and cytology triage positivity decrease with age, immediate colposcopy referral also rapidly decreases with age. In the first round, the positivity to cytology triage did not change with age (Figure 2(b)).
Proportion of epithelial cell abnormalities in cytology triage.
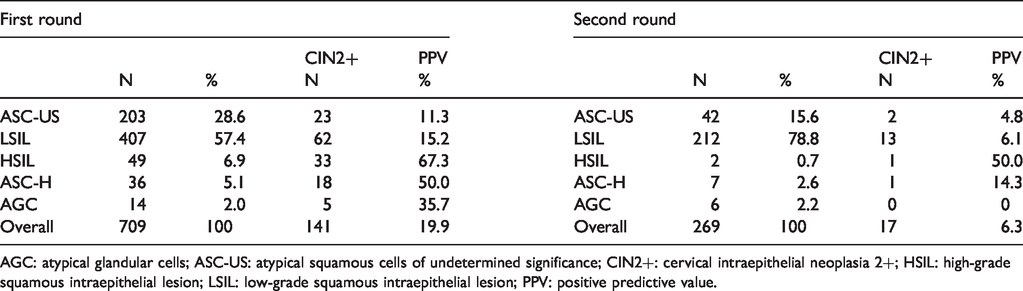
AGC: atypical glandular cells; ASC-US: atypical squamous cells of undetermined significance; CIN2+: cervical intraepithelial neoplasia 2+; HSIL: high-grade squamous intraepithelial lesion; LSIL: low-grade squamous intraepithelial lesion; PPV: positive predictive value.
Of the 301 women with negative cytology, 265 (88.0%) had a new HPV test about 12 months later; 133 (50.2%) resulted positive. Persistence was 54% in the first round (age-adjusted relative persistence 0.98, 95% CI 0.80–1.21). The age curves of one-year persistence show a slightly decreasing trend in both rounds (Figure 2(c)).
The overall second-round colposcopy referral was 3.1%: 2.1% (291) immediately and 1.0% (133) after one year. In the first round, by comparison, it was 6.6% overall, 4.5% immediately and 2.1% after one year. Compliance to one-year repeat was similar in the two rounds, 96.2 and 94.8%, in the second and first round, respectively. The age-adjusted relative colposcopy referral of the second versus first round was 0.59 (95% CI 0.53–0.65). The curves by age decreased proportionally with age. Although compliance to colposcopy was over 96.2% in both rounds, in the first round it was slightly lower in the one-year repeat (94.8%). As compliance to one-year follow-up was also slightly lower in the first round, the overall ascertainment in women HPV-positive/cytology-negative during the first round was lower than in the second round.
We found 21 CIN2, two CIN3, and one adenocarcinoma in situ (AIS), for an overall detection rate of 0.17%. In the first round, the detection rate was 0.93% (age-adjusted relative detection 0.25, 95% CI 0.16–0.39 and 0.18, 95% CI 0.05–0.61, for CIN2+ and CIN3+, respectively); 17 lesions (16 CIN2 and 1 CIN3) were found at baseline, while seven (five CIN2, one CIN3, and one AIS) were found at one-year follow-up (Figure 3). The proportion of lesions found at baseline was higher in the first than in the second round (87% versus 73%, p = 0.085). The curve by age decreased rapidly in the first round, while in the second round, even with small numbers, it seemed to be flat up to age 45; no lesion was detected after this age.
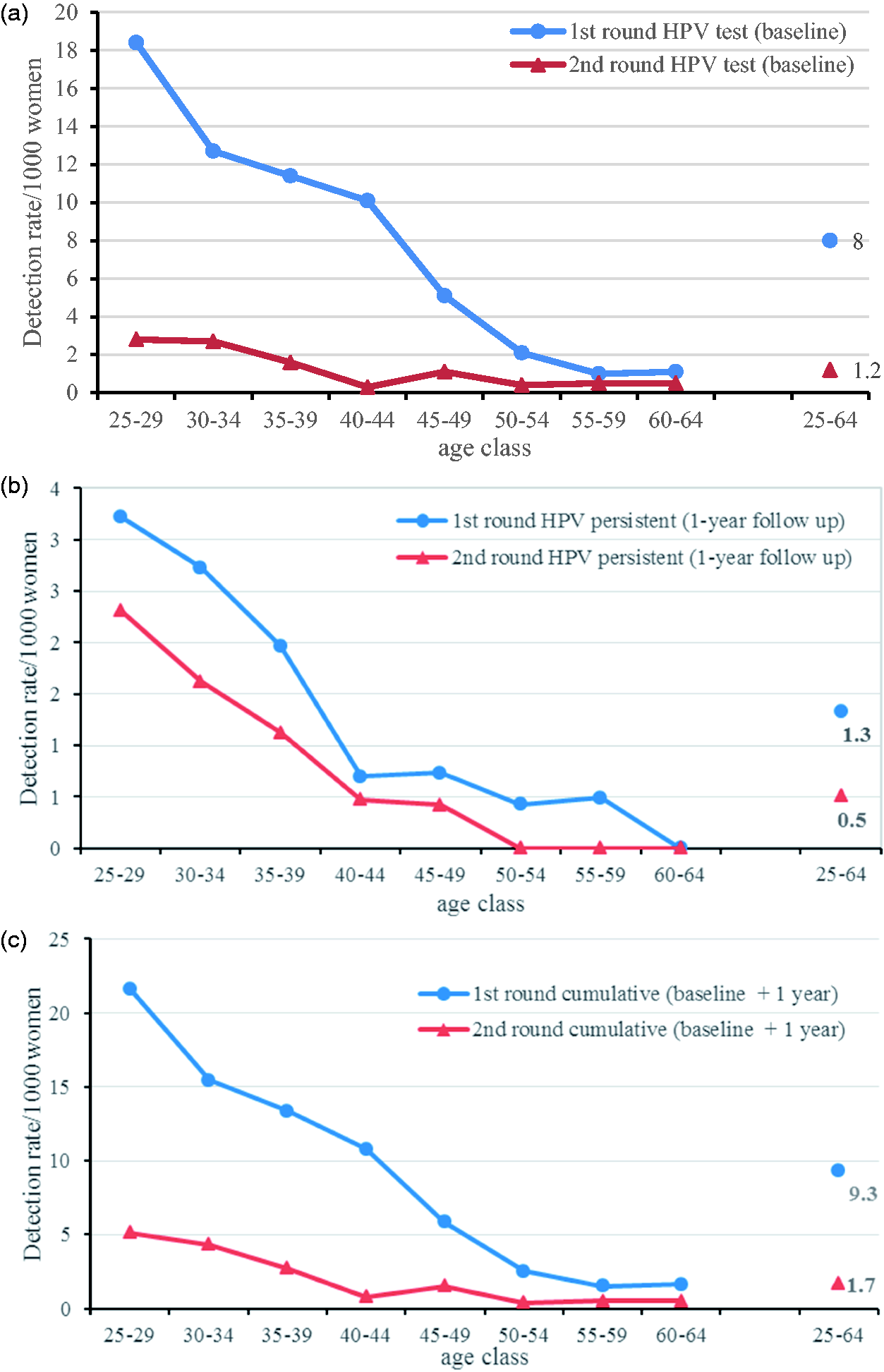
CIN2+ detection rate at baseline (a), at one-year follow up (b), and overall colposcopy referral (c), by age and screening round.
The overall PPV in the second round was 5.7, 5.8, and 5.5% in HPV− positive/cytology-positive and HPV+ persistent women, respectively. During the first round, the overall PPV was 14.6, 18.1, and 6.8% in HPV-positive, cytology-positive, and HPV-persistent women, respectively.
Discussion
Our results show that in screening a cohort of 10,000 women aged 25–60, 4.5 years after a previous negative HPV DNA test, 430 will test HPV-positive and 310 will be referred to colposcopy (210 immediately because cytology-positive and 100 after one year because HPV-persistent). A CIN2+ lesion will be found in only 13 women, two of which will be CIN3+. The same numbers for 10,000 women screened for the first time with HPV were 870 HPV+, 640 referred to colposcopy (450 immediately and 190 after one year) and 92 CIN2+, of which 15 CIN3+.
This is not the first report of a second round in a routine screening programme, but most of the previous published studies had three-year intervals12,15 or were based on a retrospective study design.22,23 This study thus allows us to observe the impact of a strategy recommended by the new European guidelines. 7 Our results are very similar to those observed in other Italian pilot studies,12,15 to those of the New Technology in Cervical Cancer trial 5 and to the Kaiser Permanente analyses, 22 where CIN3 detection or cumulative incidence at three or five years was below 1 per 1000 women. By contrast, detection was higher in the second round of the POBASCAM trial 4 and in the Swedish trial, 3 although the data are not fully comparable.
Previously participating women’s participation in the screening programme after invitation was high, as expected, but we do not have a comparable indicator for the first round, i.e. participation among those who had undergone preceding Pap test-based screening. Second-round compliance to one-year early rescreen was higher than in the first round; compliance to colposcopy for immediate colposcopy and after one-year referral was similar, while in the first round it was slightly lower for colposcopy referral after one year. The two observations suggest that one-year follow-up in women HPV-positive/cytology-negative is becoming a consolidated management strategy and that the problems initially faced9–12 have gradually been overcome.
Overall, the colposcopy referral decreased from the first round; at 3.1%, it was similar to that in the last round of the Pap test-based screening (3.7%) in the Vallecamonica-Sebino programme. 16 Colposcopy referral in a programme in its steady state would be higher than 3.1%, because the overall target population would always include some young women at their first screening round. Because the mix of women at first HPV round and women at following HPV rounds varies from programme to programme according to the strategy adopted to shift from Pap-based screening to HPV-based screening, to the participation and fidelity of women in the programme, and to migration into the area, we present data separately by HPV round. Readers can infer how the workload may change in their setting by averaging the parameters according to the different case mix in their target population.
Surprisingly, clearance after one year was very similar in the second and first rounds. As infections in the second round should be relatively recent, we expected a higher clearance compared with the first round. Our findings do not confirm the results observed in the Guanacaste cohort, where recent infections (i.e. less than one year) showed a higher rate of clearance compared with prevalent infections of unknown duration. 24 The difference could be due to the longer interval since previous negative test – 4.5 years in our cohort and 1 year in the Guanacaste cohort – but does not seem to be due to age. Although the Guanacaste cohort had lower average age, clearance in our study was only slightly negatively associated with age. Also surprising was that the proportion of HPV-positive women who were cytology-positive was only slightly lower in the second round. Only the proportion of high-grade cytological findings was, as expected, much lower in the second round than in the first. The low rate of high-grade cytology also resulted in a much lower prevalence of CIN2+ in HPV-positive/cytology-positive women in the second round than in the first, and in almost no CIN3+. Our interpretation is that new infections occur frequently and are an important part of the prevalence even at the first screening round. As long-term persistent infections, which have a very small probability of clearing in year 1, are a small part of the overall prevalence, the clearance rate is similar in the first and second rounds. On the other hand, the small proportion of long-term persistent infections is very important in terms of the number of underlying CIN2+; detection thus dramatically drops at the second round.
The prevalence of CIN2+ in women with persistent HPV showed a smaller decrease in the second round. Furthermore, we found one CIN3 and one AIS out of six lesions in this group. These data are consistent with those observed by Polman et al. 25 in the follow-up of the POBASCAM study, and suggest the presence of a small subset of fast growing, cytology-negative lesions that could challenge those screening algorithms according to which women are never referred to colposcopy if cytology-negative 26 ; such algorithms are among the alternatives suggested by the European Guidelines. 7
A strength of this pilot study is that it was implemented within the real practice of a screening programme, making it possible to observe the performance indicators of a second round of an HPV-based screening that adopts the recommendations of the new European guidelines. The only difference with the European recommendations is that our study included women from age 25, while the guidelines recommend not screening with HPV below age 30.
As the screening interval after HPV-negative test increased gradually from three to five years over the course of the study, our results may slightly underestimate the prevalence of lesions at the second round. Unfortunately, the population is too small to detect any trend of screening indicators with increasing intervals.
We did not have an independent review of pathology results. Therefore, the absolute risk of CIN2+ and CIN3+ may have been influenced by the interpretation being given by only one pathology laboratory, and we know that the inter-reader reproducibility of these diagnoses is not very high. 27 Nevertheless, as the diagnosing pathologists were the same in the first and in the second round, the comparison should be unbiased.
Conclusion
At the second round of screening, HPV prevalence and referral were almost 50% lower than at first round, while CIN2+ detection decreased eight-fold. As a result, PPV fell from 18.1 to 5.8%; CIN3+ detection was 2 per 10,000 screened women. Rescreening HPV-negative women after 4.5 years makes the process very inefficient, due to the very low prevalence of lesions. Further research should define the best way to implement HPV screening and should determine how to adopt the European guidelines recommendation that HPV-negative women should be rescreened after 5–10 years.
Footnotes
Acknowledgements
We thank Jacqueline M. Costa for the English language editing. We thank all the screening programme operators and all the women who participated in the screening.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project started as part of a multicentre feasibility pilot study sponsored by the Italian Ministry of Health (Finanziamento CCM 2009) and the Agenzia Sanitaria Abruzzo (Lgs 138/09) that included five other screening programmes (Brescia, Ferrara, Florence, Monselice, Roma G); the Vallecamonica-Sebino pilot project was also funded by the Regione Lombardia (DRG N.10813 del 16 December 2009).
