Abstract
Objective
To present the results of the first and second round human papilloma virus (HPV)-based screening programme in the Umbria region after three years.
Methods
From August 2010 to November 2011, the entire female population aged 35–64 in a local health district was invited for HPV testing (HPV-DNA cobas4800 on a liquid-based cytology sample). HPV-negative women were re-invited after three years. For HPV-positive women, a slide was prepared and interpreted. Positive cytologies were referred to colposcopy; negatives were referred to repeat HPV after one year. If HPV was persistently positive, women were referred to colposcopy; if negative, to normal screening. Indicators of the first and second round are compared with those of cytology screening in the same area in the preceding three years.
Results
Participation was 56.5%, the same as cytology (56.6%). HPV-positivity was 6.4% (396/6272), cytology triage positivity was 35.6%; 251 cytology negative women were referred to one-year HPV retesting, 84.1% complied, and 55.5% were positive. Total colposcopy referral was 4.1%, and for cytology 1%. The detection rate for cervical intraepithelial neoplasia grade 2 or more severe was 10‰, compared with 3.7‰ using cytology. After three years, HPV-positivity was 3.4% (129/3831), overall colposcopy referral was 2.3% (most at one-year follow-up), and detection rate was 0.5/1000.
Conclusions
The first round detection rate was more than twice that of cytology screening, while colposcopy referral increased fourfold. At the second round, the detection rate decreased dramatically, showing that longer interval and more conservative protocols are needed.
Introduction
The detection of human papilloma virus (HPV) DNA as the primary screening test has been shown to be more effective than cytology in detecting clinically relevant cervical intraepithelial neoplasia (CIN) and to prevent cancer.1–5 Molecular assays capable of detecting the DNA of “high-risk” HPV (HR-HPV) genotypes are more sensitive, although less specific, than traditional cytology (Pap test) in the detection of CIN. 6 A possible drawback is that the use of an HR-HPV molecular assay may lead to an increase in colposcopy referrals, and to unneeded testing.7,8 Triage tests may increase the specificity of the screening algorithm, but to date, the only widely recommended test for triaging HPV-positive women and reducing the number of colposcopies in HPV-based screening is cytology.9–12 It is therefore important to integrate the HPV-DNA test in a well-designed and organized screening programme, with well defined intervals, target age, and management for positive women.10,13
Although there is evidence of the efficacy of HPV-based screening, there is a need for pilot projects with results on the effectiveness of screening programmes in real practice. 14 In particular, the open questions are: participation of women, adherence to screening protocols,15,16 specifically in the management of HPV positive cytology negative women, 17 and performance of cytological triage (as in most of the included trials cytology was blind to HPV results,1,3,8,18–21 while in practice the cytologist is aware of the HPV positivity status).22–24 Only a few studies have reported the performance of routine screening programmes with HPV as the primary test.17,23–26
The study aimed to test the practical feasibility of an HPV-based screening programme. Here, we present the performance indicators of the HPV-based screening programme and compare the performance with the Pap test-based screening conducted in the previous years in the same area.
Methods
Setting and population
The study was conducted from 1 August 2010 to 30 October 2011 in a district of the Umbrian Local Health Unit 1 (Perugia, Italy). A well-organized screening programme, in place since 1999, invites the target population every three years for a Pap test. The test, colposcopy, all second level testing, and treatment are free for these women. The programme also includes women who present spontaneously for screening and have not had a Pap test in the previous 2.5 years; most of these women are new residents, or women who anticipated the invitation letter by a maximum of six months. In 2013, following Ministry of Health guidelines, the programme shifted to HPV-DNA screening, re-screening all the women previously recruited in this pilot study for HPV.
Study design
In this prospective, population-based study, all women participating in the screening programme were offered screening with HPV DNA for 14 high-risk types (16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59, 66, and 68). Women in the target population (aged 35–64) eligible for a new screening round received an invitation letter describing HPV-based screening, and enclosing a leaflet about test characteristics and efficacy. Women presenting for screening were asked to sign informed consent forms to participate in the pilot project; conventional Pap testing was offered to those who refused. The standard protocol recommended by the Italian Group for Cervical Screening
27
was adopted, using a cytology triage for HPV-positive women, referring women with positive cytology to colposcopy, inviting women with unsatisfactory cytology to repeat cytology, and referring women with negative cytology to HPV retest after one year (see Figure 1). If HPV positivity persisted at retesting, women were referred to colposcopy regardless of the cytology results. Appointments for colposcopy and one year retesting were proposed by phone. All women who tested HPV negative were invited again for HPV testing after three years (in 2013–2014). The Ministry of Health in 2013 recommended a five-year interval; thus all the women with a negative HPV test recruited in this study received a response letter reporting the three year interval. During the second round, a new trial (NCT01837693) started recruitment, in which women HPV positive and cytology negative at baseline could be randomized to receive a colposcopy, instead of one-year follow-up.
Flow chart – screening results for first (2010–2011) and second round (2013–2014).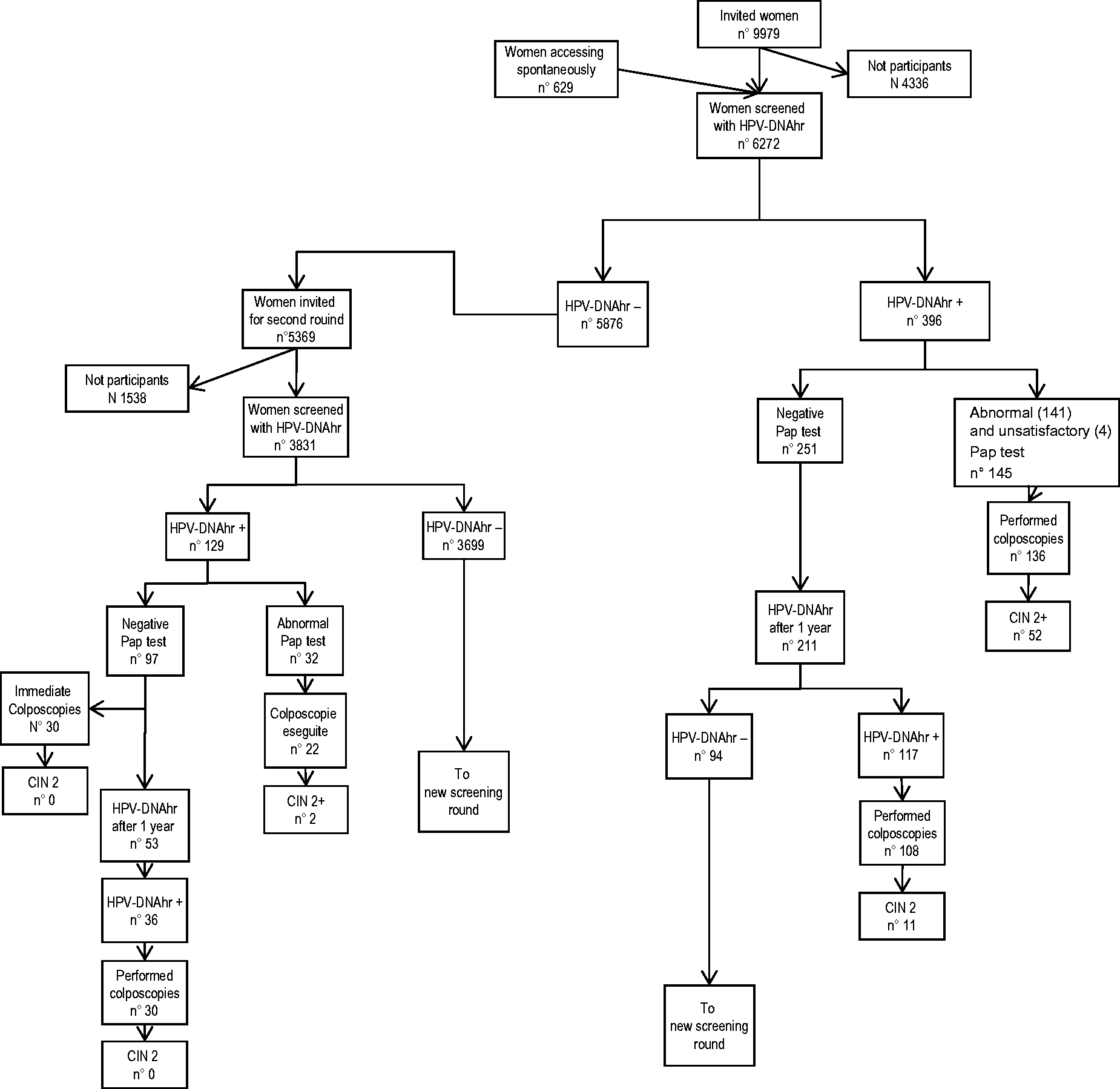
The feasibility and the impact of the HPV-based programme was measured with the following indicators: participation of women after invitation, compliance to colposcopy and one year retesting, cytology triage results, colposcopy referral rate, detection rate and positive predictive value, wait time for negative response and for colposcopy when results were positive. Indicators for the first round of HPV-based screening are presented for baseline, one year recall, and overall. For the second round, only baseline results are available. Results are compared with those from cytology-based screening in women aged 35–64 in the same area during the period 2008–2010. Cytology screening protocol was as follows: for low-grade abnormal cytology ASC-US o L-SIL, women are invited to undergo HR-HPV DNA testing; if HPV is positive women are referred to colposcopy, if negative, women are invited after one year to a repeat Pap test. For high-grade cytology, ASC-H and H-SIL, women are immediately referred to colposcopy. Women with a negative Pap test are invited to a new screening after three years.
This study is part of a Health Technology Assessment programme at national and regional level. 10 Material stored from liquid-based cytology medium was also used for testing new biomarkers. 28
Cervical sample collection
Cervical samples were collected by trained midwives in 12 clinics in the area; the same midwives were involved in the cytology screening. Samples were taken by cervex-brush or cervex-brush-combi stored in 20 ml of cobas PCR cell collection media (a methanol-based medium produced by Roche Molecular Systems, Inc-Branchburg, NJ 08876 USA, under licence of Hologic, which has the same composition of PreservCyt) at 4℃ until tested. The sample allowed for HPV-DNA testing, for the setting up of a cytology slide, and for stocking the remaining liquid for other biomarkers.
HPV-DNA laboratory methods
All molecular tests have been centralized to the UNICO Screening Laboratory. High-risk type HPV-DNA was detected through real time PCR with cobas® 4800 HPV Test (Roche Molecular Systems, Pleasanton, CA, USA). DNA extraction was performed on the cobas® x 480 platform and the amplification on the cobas® z 480 platform. This test is a four-channel real-time PCR-based assay allowing for the detection of HPV genotypes 16 and, 18, and of several other HR-HPV genotypes as a pool (HPV31, 33, 35, 39, 45, 51, 52, 56, 58, 59, 66, 68). The fourth channel is used for the detection of human β-globin as an internal control. Samples were considered positive according to critical threshold values suggested by the manufacturer, ≤40.5 cycles for HPV16, and ≤40.0 for HPV18 and other HR genotypes.
Cytology
LBC was prepared using the ThinPrep® 2000 System following the manufacturer’s instructions (Cytyc Corporation Marlborough-MA 01752-USA). Cytology triage was classified according to 2001 Bethesda reporting system. 29 Cytology interpretation was carried out by the six cytologists dedicated to routine cytology screening. All slides underwent collegial reading (at least three readers); final diagnosis was obtained after discussion. Cytology was classified according to the Bethesda 2001 System: Negative for intraepithelial lesion or malignancy (NILM), atypical squamous cells of undetermined significance (ASC-US), low-grade squamous intraepithelial lesion (LSIL), atypical squamous cells with possible high-grade lesions (ASC-H), high-grade squamous intraepithelial lesion (H-SIL, encompassing cervical intraepithelial moderate neoplasia CIN2, cervical intraepithelial severe neoplasia CIN3), squamous cell carcinoma, atypical glandular cells favour neoplastic (AGC), adenocarcinoma.
Colposcopy
Colposcopies were performed according to Italian recommendations. 30 Biopsies were taken only if a lesion was suspected. Endocervical sampling, through hysteroscopy, was performed if cytology was suggestive of glandular lesion or an endocervical lesion. CIN2 or more severe lesions were referred to treatment (almost always LEEP). See and treat was not an option. Treatment of CIN1 was allowed only in cases of high-grade cytology (never at first colposcopy) and after 24 months of persistence. The follow-up after a negative colposcopy or a histological CIN1 depended on triage cytology: women with L-SIL or less severe cytology were referred to one-year HPV testing; women with H-SIL or ASC-H were referred to repeat colposcopy at six months.
Histology
The end point used to calculate detection rate and positive predictive value is histologically confirmed CIN2 or more severe lesion. We only give results at first colposcopy, consistent with the definition of screening performance indicators in the European Guidelines. 31 Therefore, follow-up for this study closed: (1) with a diagnosis of CIN2 or a more severe lesion; (2) at the first colposcopy negative for CIN2; (3) at the first HPV-negative test. Follow-up for all other women in the study was considered inconclusive. When more than one biopsy or cone was present, the more severe diagnosis was used. Haematoxylin-eosin slides were interpreted according to the current WHO classification. The diagnoses of moderate dysplasia/CIN2 severe dysplasia/CIN3/carcinoma in situ, invasive Squamous Cell Carcinoma, adenocarcinoma in situ and invasive are hereafter referred to as CIN2+. P16 staining was routinely used on histological slides as diagnostic assistance in distinguishing CIN1 from CIN2 and immature atypical metaplasia from CIN2.
Data collection and analysis
All the clinics, the central screening laboratory, and the gynaecology unit are connected with the screening management software, which collates all information on invitations, sampling, first level tests, colposcopies, and histologies. A cancer registry also exists in the region. We present the screening performance indicators with relative 95% confidence intervals. For proportions these intervals are estimated with binomial exact distribution; for continuous variables they are estimated with normal or t-Student distribution approximation, according to the number of observations (>30 or ≤30, respectively). During the second round, some women with HPV positive and cytology negative results, both included in the original cohort and presenting for the first screening with HPV, were referred to immediate colposcopy under an experimental protocol. The total referral rate for second round has been calculated applying the proportion of HPV positivity at one-year follow-up to the women who complied to referral. Overall positive predictive value has been calculated as a weighted average of baseline and one-year follow-up positive predictive value.
Ethics
The study was conducted by mandate of the Umbria regional government (DGR 249/09). The pilot project protocol was evaluated and approved by the Healthcare Administration of the Local Health Authority, with the administrative decision of the Director General n.1011/2010 which assessed it for ethical committee approval.
Results
First round results
Screening performance indicators with hrHPV-DNA testing: baseline and one year follow-up of first and second round screening. For the second round results are presented for women previously HPV negative and for all women who have been screened.
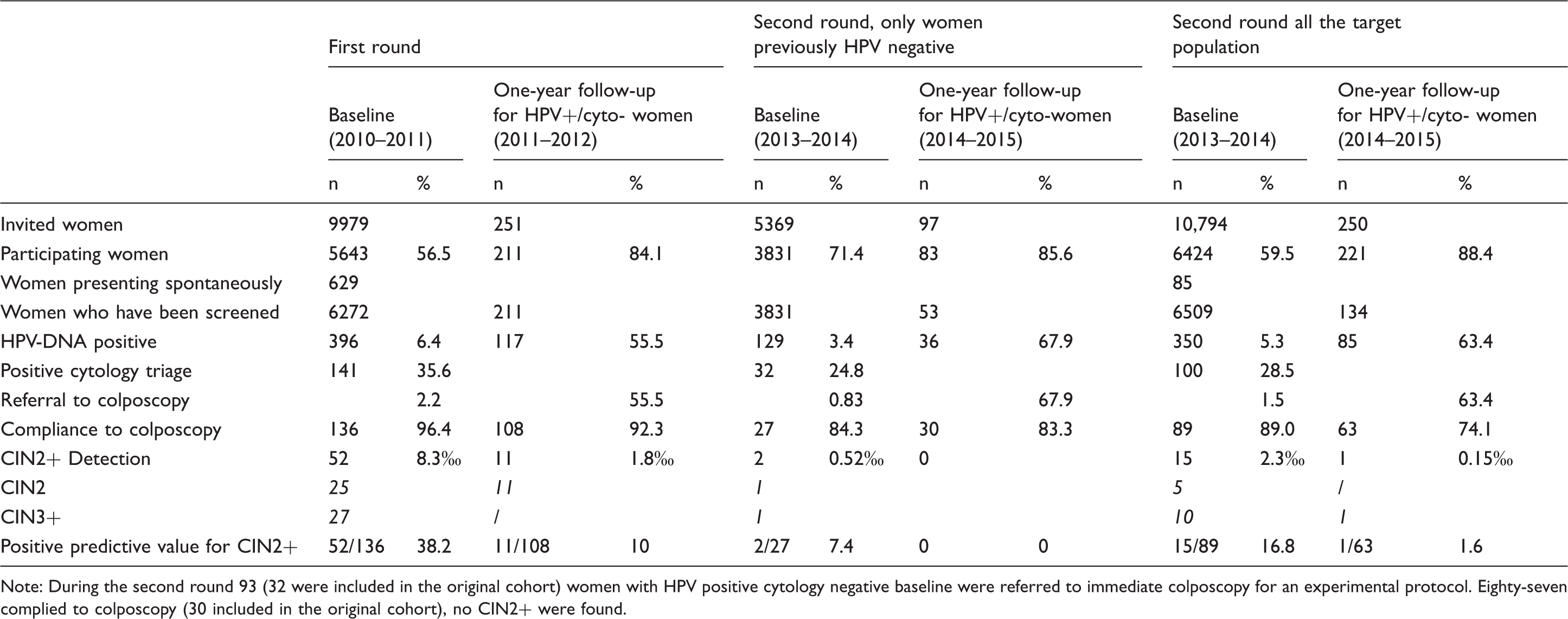
Note: During the second round 93 (32 were included in the original cohort) women with HPV positive cytology negative baseline were referred to immediate colposcopy for an experimental protocol. Eighty-seven complied to colposcopy (30 included in the original cohort), no CIN2+ were found.
Cytology and histology concordance at first round baseline colposcopies.

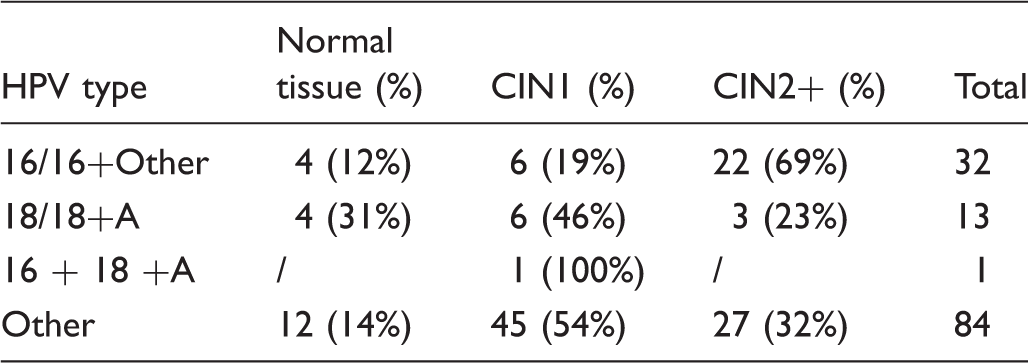
Distribution of HPV types by histological diagnosis in women HPV+/cytology+ at baseline and with a colposcopy-guided biopsy.
The 251 HPV-DNAhr positive cytology negative women were referred to repeat HPV after one year. In the 211 who complied (84.1%), HPV was still positive in 117 (55.5%). These women were referred to colposcopy, 108 (92.3%) complied, with 11 CIN2 found. The CIN2+ detection rate was 1.8‰ and the PPV 10% (Table 1).
Comparison with cytology screening
Comparison of Pap test-based screening and hrHPV-DNA screening performance indicators.
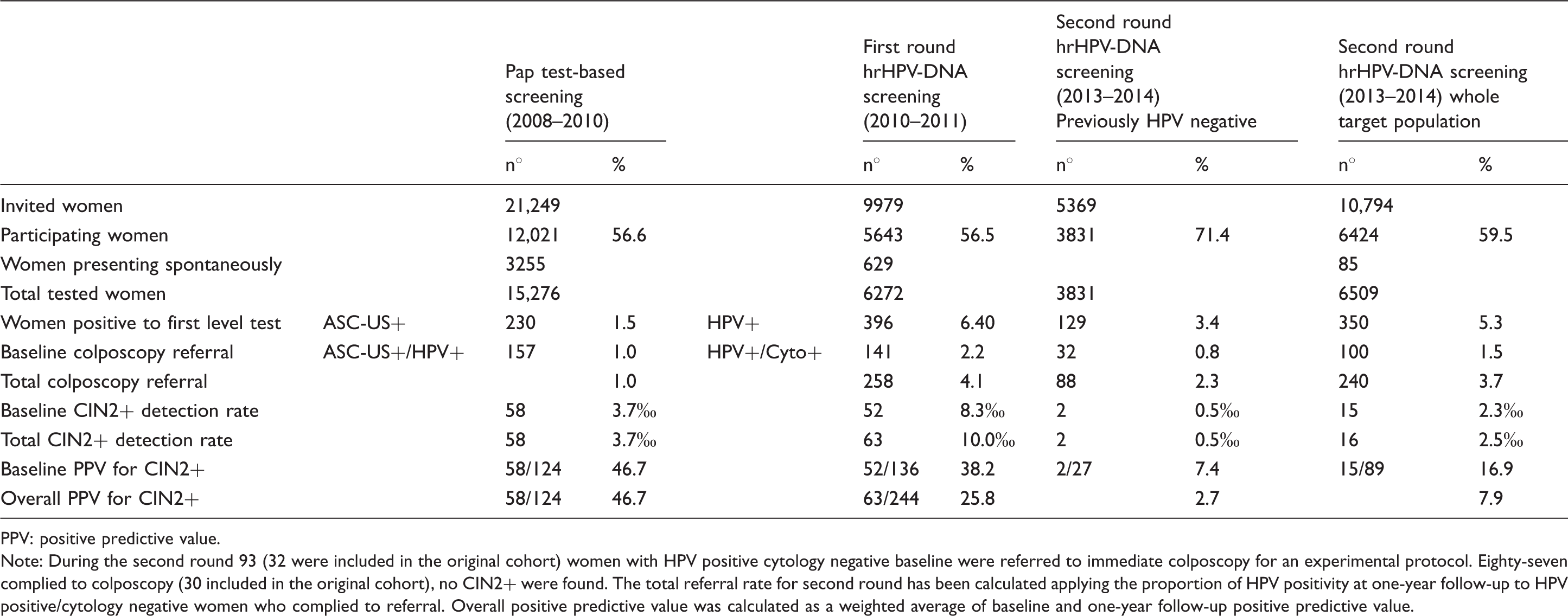
PPV: positive predictive value.
Note: During the second round 93 (32 were included in the original cohort) women with HPV positive cytology negative baseline were referred to immediate colposcopy for an experimental protocol. Eighty-seven complied to colposcopy (30 included in the original cohort), no CIN2+ were found. The total referral rate for second round has been calculated applying the proportion of HPV positivity at one-year follow-up to HPV positive/cytology negative women who complied to referral. Overall positive predictive value was calculated as a weighted average of baseline and one-year follow-up positive predictive value.
Second screening round
Of 5876 women who tested HPV-negative at first round, 5369 were re-invited before 31 December 2014, 3831 (71.4%) participated and completed the baseline screening. There were 3699 (96.5%) negative HPV-DNAhr tests; these women were invited for a new screening after five years. Of 13 unsatisfactory samples, 10 women repeated the test, and 3 were lost. There were 129 (3.4%) women positive for HPV-DNAhr, among whom 35 (27.1%) were HPV 16 or 18 positive. Triage cytology was negative in 97 women (75%) and 32 (25%) (25 L-SIL, 5 ASC-H and 2 CIN2) were referred to colposcopy, a 0.8% base-line referral rate. Two Pap tests were unsatisfactory and women repeated cytology sampling. Twenty-seven women (84.3%) complied to colposcopy, and 2 CIN2+ were found, resulting in a CIN2+ detection rate for the baseline testing alone of 0.52‰ and a 7.4% PPV for CIN2+ of the baseline colposcopy referral. Of the 97 cytology negative women, 30 received an immediate colposcopy because they were included in another study. No CIN2+ were found. The other 67 women were invited for the one year follow-up, and 57 complied. No CIN2+ have been found in this group, but 67.9% of the women continued to have HPV positive results, and the overall referral rate increased to 2.3% with a PPV for CIN2+ of the overall colposcopy referral of 2.7%. The overall CIN2+ detection rate remained 0.52‰ (Table 4). Even when considering not only those previously negative, but the whole second round target population (from 1 August 2013 to 31 December 2014, 10,749 invited women, 6509 screened women), the positivity rate, the detection rate, and the positive predictive value showed a relevant reduction compared with the first round, while the referral to colposcopy only slightly decreased (Table 1).
Discussion
Consistent with previous studies,17,23,24,32 our results confirm that HPV screening is well accepted by women who have participated in previous cytological screening. Participation in HPV screening was higher in younger women, a group that in Italy usually responds less to screening programme invitations than older women. 33 While the invitation letter made clear that a new test had been adopted, and included a leaflet explaining the rationale for using HPV tests, no mass campaign or community level communication was possible to inform the target population of the new screening. As the health district has no natural borders, and women move for work from one district to another, it would be very difficult and unpopular to explain why a new test has been introduced in only some municipalities.
Using a sampling medium allowing for both HPV testing and cytology does not require a second sample for triage, avoiding reduced compliance.34,35 Compliance to one year retesting reached an acceptable 84%, although as higher compliance (>90%) has been observed in other Italian programmes 23 there is room for improvement. In particular, deviations from protocols can occur just after women receive the letter advising them of an HPV positive result. This letter can cause women to seek a gynaecological appointment and to move to private screening. Measures should be implemented at this turnstile, in addition to the point at which women are invited for retesting as was done in the pilot.
The complexity of the screening algorithm necessitated having software able to follow all the screening phases, connecting all the services involved in the process, automatically suggesting the next step for the woman, and preparing the informative requirements for subsequent tests. The software manages all invitation letters and appointments, connecting all family care clinics with the screening laboratory.
In contrast with most randomized trials, triage cytology in routine screening is interpreted with the knowledge that the woman is HPV positive. As the presence of HPVhr infection was known, atypical squamous cells were almost never considered of undetermined significance, 36 and so atypical squamous cells of undetermined significance (ASC-US) almost disappeared. This is in line with the observation that the positive predictive value of ASC-US and L-SIL are very similar among HPV+ women. 37 Informed cytology may also be the cause of higher positivity among HPV positive women (35.6%) compared with that observed in the Italian trial “New Technologies for Cervical Cancer (NTCC)”, in the same age group (25.3%).4,8,18 However, the detection rate in women HPV+/cytology positive was much higher when the HPV was performed as primary screening and cytology as triage than when cytology was the primary screening and HPV was the triage test (8.3/1000 v 3.7/1000). The European trials with blind cytology did not observe any relevant increase in detection rate in HPV+/cytology+ compared with cytology screening.1–3,8,18 The only reasonable explanation is that informed cytology is much more sensitive than blind cytology. A recent study found similar results in a randomized setting. 22
Notably, in the second round the positivity in cytology triage decreased to 24.8% (Table 1). This could be the consequence of screening women who were previously screen negative and so the infections were mostly recent infections, but it could also be due to a learning curve in triage cytology by cytologists.
The very high sensitivity of the baseline of first round test combinations (HPV+/cyto+) was compensated by a very low (lower than that observed in most trials) detection rate at one year follow-up. This observation is reassuring, suggesting that the higher detection rate at baseline corresponds to an earlier diagnosis of persistent lesions that are no longer found at one year follow-up.
The Italian guidelines 10 propose re-testing at one year, on the basis of the NTCC trial results 4 in which a reduction in cancer incidence was observed just three years after HPV screening in comparison with the Pap test. These results suggested it was too great a risk to wait more than one year for women HPV+/cytology negative, (i.e. exactly those women in which the identification of CIN3 could have determined the lower incidence of cancer three years later). From the results of pilot screening programmes with triage cytology, we know that many CIN3 that would be undetected in cytology screening are detected in the HPV+/cytology+ at baseline, because of the higher sensitivity of triage cytology. In contrast, after one year the prevalence of lesions is very low, implying a very low detection rate. In addition the virus clearance is quite low (about 50%), implying a very high colposcopy referral rate, and consequently a very low positive predictive value. It must be noted that we found only CIN2 in these colposcopies. Similar results have been found in other Italian pilot studies.23,24
If triage has very high sensitivity, due either to the high performance of cytology itself or because we adopt biomarkers such as P16ink4a or E6/E7 mRNA expression, it is reasonable to adopt longer intervals, i.e. 18 or 24 months, for retesting HPV positive/triage negative women. A longer interval will probably allow more clearance,8,38 reducing the burden of colposcopies at one year follow-up. Another solution could be to adopt a test with higher specificity than HPV DNA at one year follow-up, i.e. cytology again as in the Dutch protocol, 9 but the interval to retest women whose results are negative at this test should be evaluated for safety.
The overall CIN2+ detection rate at baseline and at one year follow-up in the first round was 10‰ (Table 4), more than double that of the detection rate with the Pap test in previous years. This increase is higher than that observed in trials, but similar to that observed in other pilot studies. This could be due to two factors: some of the trials obtained a quite low compliance to follow-up in HPV positive cytology negative women, 39 and the pre-analytic and analytic phases in these pilot studies were more automated than those adopted in trials, probably increasing the analytical accuracy of tests.
The results of the second round show that women with previous HPV DNA negative test results have half the probability of infection, two-thirds lower colposcopy referrals, and 20 times lower detection rate compared with women at their first HPV DNA screening round. These results clearly show that the three-year interval is too short for rescreening previously HPV-negative women. In fact PPV for CIN2+ is much lower than that considered acceptable in a screening programme (i.e. the PPV of L-SIL cytology). Longer intervals are already recommended by Italian and European guidelines10,40 and routine Italian screening programmes now adopt a five year interval. Nevertheless, triage tests will remain necessary to distinguish very new and/or transient infections. The most promising are the E6 and E7 oncogene activation biomarkers, including p16.
In contrast with the results of previous cohort studies, 41 the clearance of infection after one year in HPV positive/cytology negative women in the second round, when infections are supposed to be more recent, was not higher than in the prevalence round, when infections are a mix, including very old ones. As a result, the colposcopy workload was 2.3 times higher than that of cytology screening if we only consider women with HPV-negative results at the previous round, and 3.7 times higher if we consider the whole population of the second screening round, that included approximately 60% of women with a previous negative test and 40% of women at first HPV screening (Table 4). The referral rate of the Umbria cytology-based screening programme was one of the lowest in Italy, and so the increase is very high compared with the recall rate with cytology in this programme, but less impressive if we compare it with the average referral rate (2.4%) of Italian screening programmes. 42 In any case, such a low clearance in women with relatively recent infections and with a very low risk of cancer should lead to the reconsideration of retesting HPV positive and triage negative women after only one year.
Conclusion
Participation in HPV-based screening was not lower than participation in Pap-based screening, and compliance with protocols for HPV positive women was comparable with or higher than compliance with protocols for management of abnormal cytology. The colposcopy workload strongly increased in the first round. At the second round the decrease in HPV positivity did not compensate for the burden of colposcopies due to follow-up of HPV positive/cytology negative women. On the other hand, the prevalence of high grade lesions was extremely low, and the detection rate and positive predictive value were also low. A five-year interval is necessary to reduce colposcopies and to have an acceptable detection rate. Triage cytology was more sensitive than cytology as a first level screening test, allowing intervals for HPV positive/cytology negative women to be longer than one year.
Footnotes
Acknowledgements
We thank all the midwives and coordinators of the screening programme in the district of the Umbria Local Health Unit 1 who made this study possible.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The study was funded by the Umbria Region.
