Abstract
Objective
We present the results of the first screening round and the first year of the second round of the Valcamonica Human Papillomavirus (HPV) pilot screening project.
Setting
From 2010 to 2012, the entire target female population (aged 25–64) was invited to the first HPV screening round in an area where Pap test screening had been active since 2002.
Methods
For HPV-negative women, the interval was three years. For HPV-positive women, a cytological smear was stained and interpreted. Positive cytologies were referred to colposcopy; negatives were referred to repeat HPV after one year. If HPV was persistently positive, women were referred to colposcopy; if negative, to normal screening.
Results
In 2010–12 18728 women were screened, slightly higher participation than with Pap test (18233 64.7%); 1633 were HPV-positive (8.7%); 843 were positive at cytology triage (referral rate at baseline 4.5%). Of those referred at the one year HPV test, 84% complied (660/780); 356 were persistently positive (1.9%). The total referral rate was 6.4% compared with 3.7% for the Pap test. The detection rate was 9.2/1000 compared with 5.0% for the Pap test. The HPV positivity rate during the second round in women previously negative was 3.9% and the detection rate in HPV-positive cytology-positive women was 0.8/1000.
Conclusions
HPV-based screening increases colposcopies at the first round, but also strongly increases the detection rate. At the second round, HPV prevalence was much lower and the detection rate also fell, corroborating the need for longer screening intervals in HPV-negative women.
Keywords
Background
The identification of persistent infection with oncogenic types of human papillomavirus (HPV) as the necessary cause for cervical cancer 1 has led to the use of HPV DNA testing as a primary screening test. 2 Numerous studies have shown that testing for HPV is more sensitive than cytology for detecting cervical intraepithelial neoplasia (CIN2+ or CIN3+), but it is also less specific. 3 Evidence has also emerged that a negative HPV test leads to longer protection than a negative smear. 4 Randomized trials have shown that HPV screening is more effective than Pap test screening in the early diagnosis of CIN35–8 and consequently in reducing cervical cancer incidence.8–10
To increase the specificity of HPV screening, screen-positive women can be triaged using Pap tests; if they are cytologically negative, they can be rescreened by HPV testing one year later. This protocol has been used in the Italian pilot studies testing the feasibility of HPV-based screening, 11 and has now been adopted in the Italian recommendations on the use of HPV as a screening test. The protocol will also form part of the upcoming European recommendations, 12 and is similar to the other protocols adopted in Europe.13,14
The Italian pilot studies, sponsored by the Italian Ministry of Health, implemented HPV-based screening to test organizational impact, acceptability by women and operators, performance of cytology triage, impact on midwives, laboratory, cytology, and gynaecology workload, and costs,15–17 including budget impact and cost-effectiveness. 17
The aim of this study was to evaluate the performance of an HPV test-based strategy in a well consolidated screening programme in Northern Italy (the Valcamonica-Sebino Local Health Authority), relative to a complete round of screening, and to the first year of the second screening round.
Methods
Setting
Vallecamonica-Sebino is a community with a single Local Health Authority in the Lombardy Region of Northern Italy (99,776 inhabitants in 2010). A screening programme, in which all women aged 25–64 (27721 in 2010) are invited every three years for a Pap-test, was introduced in 2002. The designated appointment time in invitations can be changed by calling a toll-free number. Women who do not respond within three months receive a recall letter. All the tests and treatment are free. In 2010, the HPV strategy was introduced into the cervical screening programme. Invitations, appointments, answers and ascertainments management are computerized, with information on each woman, from invitation to histological results, collected and stored electronically. The database is checked for all women with positive tests and incomplete ascertainment records. Pap test coverage (including spontaneous screening activity) is about 80%, but data on tests performed outside the programme are not included in the database. In this paper we report screening results for women invited and screened during the first round (recruitment period 2010–12) and followed up until 31 March 31 2014. The entire target population was invited during the recruitment period.
Funding
This project started as part of a multicentre feasibility pilot study sponsored by the Italian Ministry of Health (Finanziamento CCM 2009) and the Agenzia Sanitaria Abruzzo (Lgs 138/09) that included five other screening programmes (Brescia, Ferrara, Florence, Monselice, Roma G); the Valcamonica Pilot project was also funded by the Regione Lombardia (DRG N.10813 del 16.12.2009).
Screening protocol
Pap test screening was conducted according to Italian guidelines.
18
Conventional smears were collected by midwives with spatula and brush using the 2001 Bethesda System;
19
women with ASC-US (atypical squamous cells of undetermined significance) or more severe cytology, as well as women with two unsatisfactory Pap smears if the reason for unsatisfactory smear was the same, were referred to colposcopy. The experimental HPV-based protocol was that proposed by the Italian Group for Cervical Cancer Screening.
11
Women aged 25–64 were invited for a new screening round with the HPV test; a double sampling (conventional smear and Specimen Transport Medium [Qiagen, Hilden Germany] for HPV) was performed; the slides of women who tested positive were dyed and interpreted by a single cytologist. Women with cytology ASC-US or more severe and those with unsatisfactory slides were referred for colposcopy, while women with negative cytology were referred to one-year HPV re-testing. All the HPV-negative women were referred to 3-year rescreening; new Ministry of Health recommendations to increase this interval to five years were issued in 2013. The adopted screening algorithm is described in Figure 1.
Flowchart and main data of the HPV test-based screening programme. Years 2010-2013. Results of the pilot project for controlled application of HPV testing as a primary screening (2010–2012) and the first year of implementation (2013).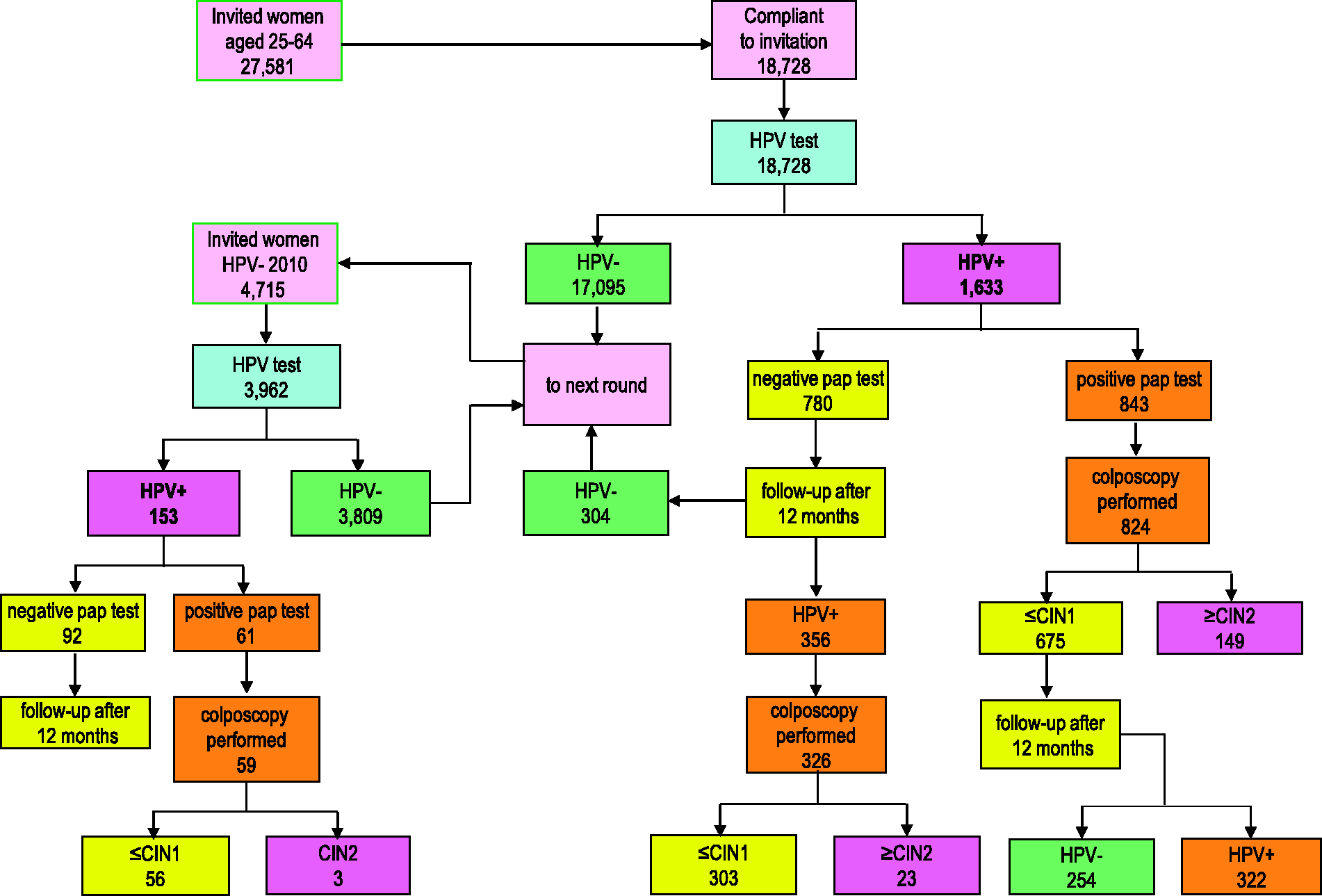
Laboratory procedures and quality control
Cervical cells were collected for both conventional cytology and the HPV test. HPV testing was performed using the Hybrid Capture 2 Assay (HC2; Qiagen, Hilden, Germany) to detect 13 high-risk HPV types (12 included in IARC group 1: 16,18,31,33,35,39,45,51,52,56,59; one included in IARC group 2a: 68). 20 All samples with a relative light unit/positive control ≥ratio1 were considered positive. Cytology was classified according to the 2001 Bethesda System. 19 An external quality control was scheduled based on a review by an external supervisor (M.C.) of all abnormal slides, inadequate slides, and 10% of negative slides.
Colposcopy and histology
Colposcopy was performed according to Italian guidelines. 18 Colposcopy-guided biopsies were taken in all positive colposcopies and almost always in cases of ASC-H, H-SIL, and glandular lesions. Histology was performed in the pathology laboratory of the Local Health Authority by a single pathologist, using Hematoxylin Eosin staining. P16 immunostaining was used as a diagnostic aid to distinguish between suspected CIN2 lesions and CIN1 or Atypical Immature Metaplasia.
Endpoints and analysis
To evaluate the performance of the HPV-based programme, we calculated the following parameters:
Participation in screening (screened women/invited women) Positivity on HPV test Rate of abnormal cytology among HPV positives Referral rate to colposcopy Positive predictive value (PPV) for CIN2+ at baseline Detection rate for CIN2+ at baseline Proportion of persistent HPV at one year Detection rate at one year follow up Detection rate in post colposcopy follow up Total detection rate
Participation, referral rate, and detection rate were compared with historical data obtained from Pap test screening in the previous round, calculating age-adjusted relative risk with 95% confidence intervals. For the second round of HPV screening, we present the indicators for the entire invited population (including those newly entered in the screening target age and those who did not respond to the first round invitation), and for the women who were negative at the first screening round. Including the latter group makes it possible to observe the reduction in the positivity and detection rates at subsequent rounds with a cohort design.
Ethics
The study was approved by the Ethics Committee of the ASL Vallecamonica-Sebino on 4 February 2010, decision number 0006141/10.
Results
Participation and compliance with screening protocols
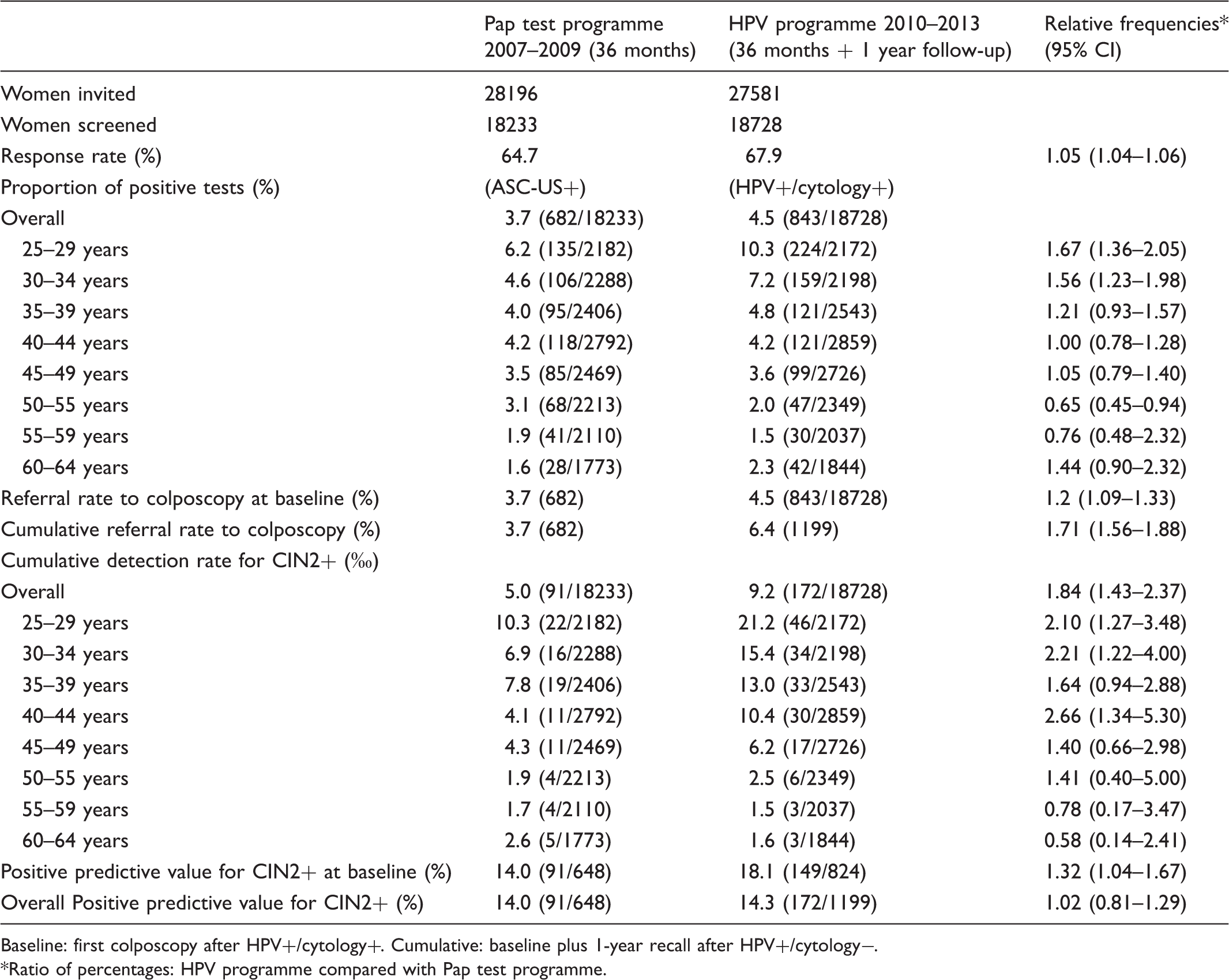
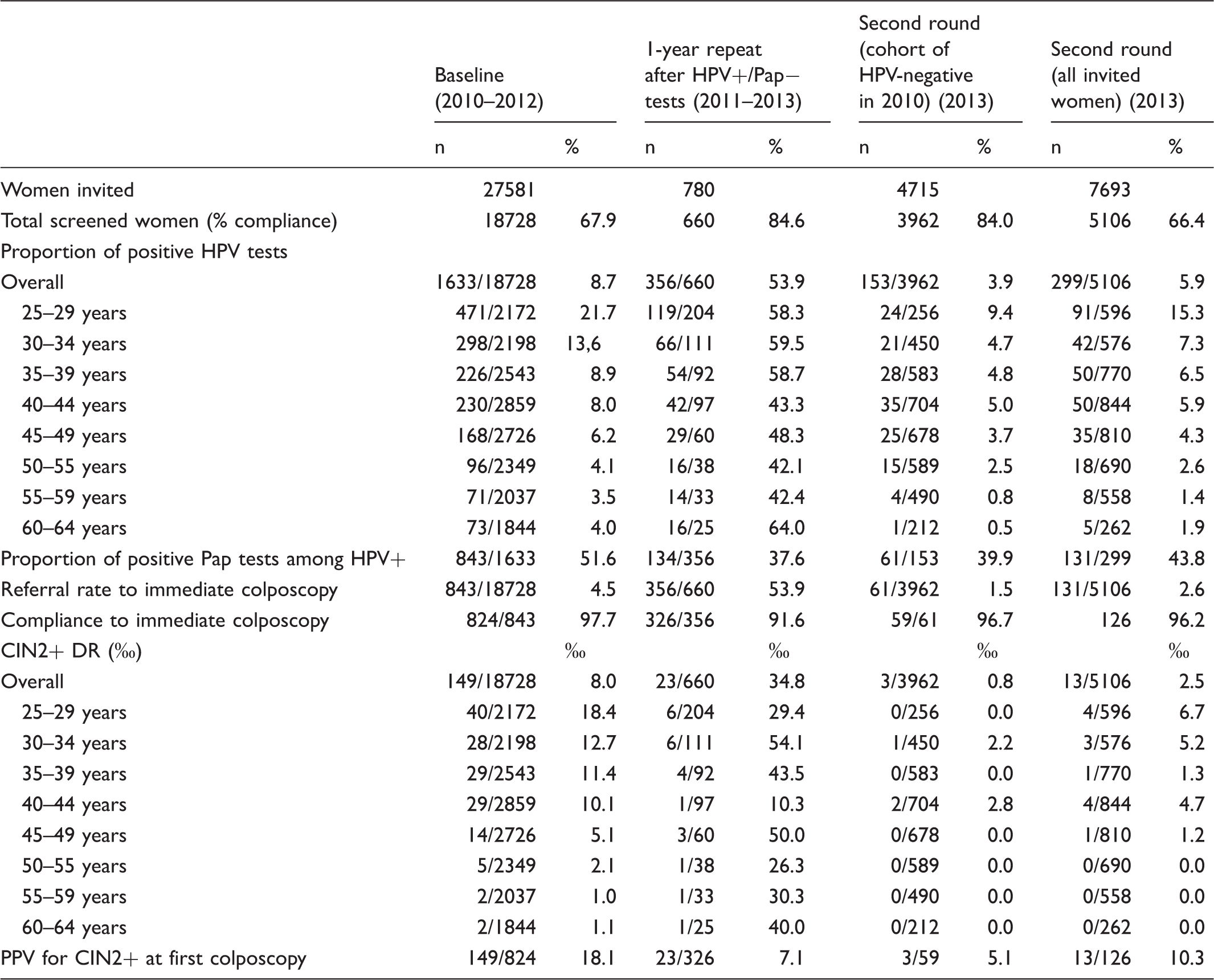
In the first round of the programme, from March 2010 to December 2012, 27581 women were invited to attend the new HPV screening; 18728 underwent HPV testing. Excluding letters undelivered due to wrong addresses, participation was 67.9%, higher than the 64.7% obtained with Pap test screening in the previous round (2007–09) (see Table 1 and Figure 2). Participation in the second round was 84% (3962 participants out of 4715 delivered letters). This figure includes only women aged 28–64 who had participated in the previous screening round. Among HPV-positive women, those who were cytology-negative (780) were referred to HPV retesting at one year. Compliance with this protocol was 84.6% (660). Compliance with colposcopy was 97.7% (824/843) in women with immediate referral, ie. those HPV-positives and cytology-positives at baseline, and 91.6% (326/356) of those referred for HPV persistence after one year.
Participation by age and mode of screening. Comparison of Pap test and HPV tests in organized screening programmes. Baseline: first colposcopy after HPV+/cytology+. Cumulative: baseline plus 1-year recall after HPV+/cytology−. *Ratio of percentages: HPV programme compared with Pap test programme.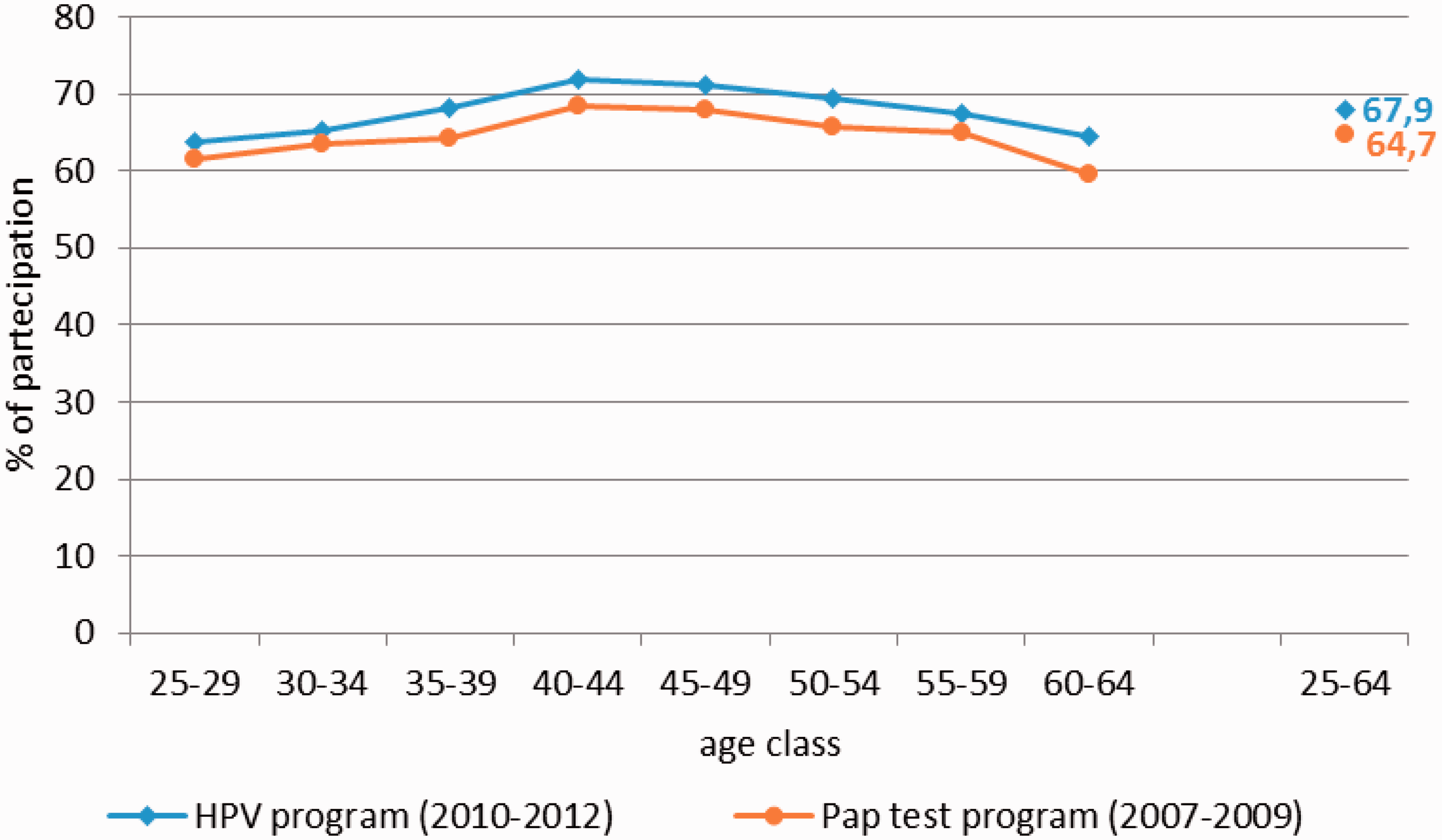
HPV test results
The HPV positivity rate was 8.7% (1633/18728, see Table 2 and Figure 3a), ranging from 21.7% in the youngest age group to about 4% in women over age 50. There were no samples unsuitable for testing. We did not register any missed link between HPV positive samples and the slide of the same woman, although we cannot rule out errors in labelling one or both, ie. the smear and the HPV sampling medium. The HPV positivity rate for the second round, including only women who were HPV-negative at first round, was 3.9% (153 out of 3962) and 5.9% including all the women invited during second round (299 out of 5106). The age curve reproduces substantially the same curve as the first round, but at a lower level. Only after the age of 55 did the HPV positivity reach values close to zero.
Positivity rate by age: A. Baseline test at first (2009–12) and second (2013) HPV screening round; B persistent positivity after one year (only for first round 2010–13). Main results of the HPV test-based screening programme at baseline (women screened from March 2010 to December 2012) and at the 1-year repeat of women found to be HPV+/cytology- at baseline (2011 to 2013) and the first year of the second round (2013).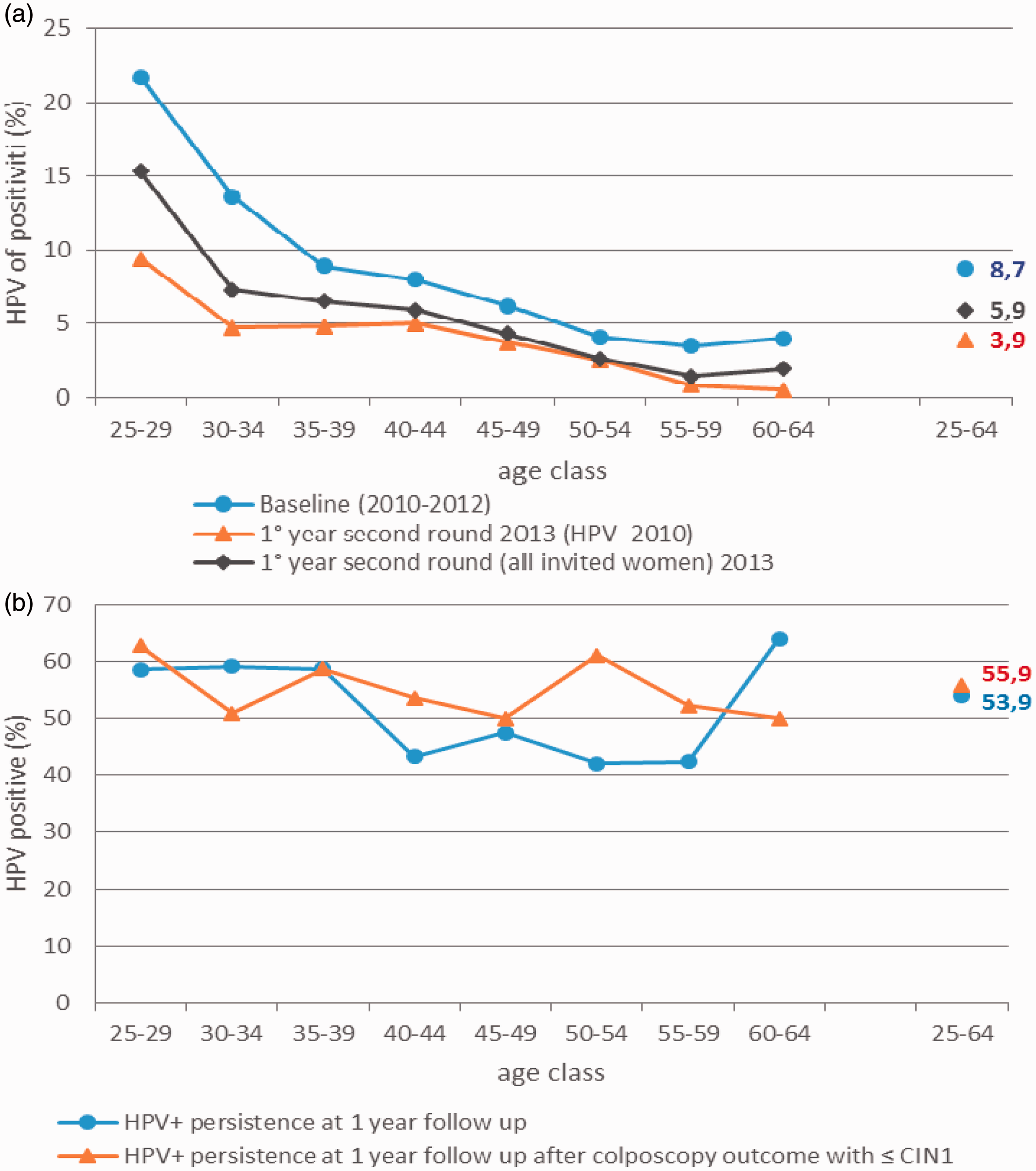
Cytology triage
Among the 1633 HPV-positive women, 44.4% (843) had a positive cytology, and 7.2% had an unsatisfactory smear. The positive slides were ASC-US, ASC-H, AGC, LSIL, and HSIL in 28.6%, 5.1%, 2.0%, 57.4%, and 6.9% of cases, respectively. The immediate referral to colposcopy rate was 51.6% among HPV-positives and 4.5% among all screened women (see Table 2). As the positivity to triage was substantially stable with age, the immediate colposcopy referral rapidly decreased with age. The proportion of cytology-positives was lower during the second round at 43.8% (131/299) for all screened women and 39.9% (61/153) in the cohort of those previously HPV negative, resulting in a much lower immediate referral to colposcopy (2.9% and 1.5% for the groups, respectively). Compared with the first round, this reduction was statistically significant (p < 0.0005).
Persistence of HPV at one year
Fifty-four percent of results from the women referred to HPV re-testing were positive (356/660). The persistence slightly decreased with age (see Table 2 and Figure 3b). The overall colposcopy referral rate was 6.4% (6.7% adjusting for non-compliance with one-year retesting). The colposcopy referral in the previous round, performed with Pap test, was 3.7% (Figure 4). The persistence of HPV infection was also similar (56%) in women who were HPV-positive and cytology-positive at baseline, but had a colposcopy negative result for CIN2+.
Colposcopy referral rate by age and screening round: A. Pap test programme (2007–2009); B. HPV screening first round (2010–2012), by baseline and one-year follow up; C. HPV screening second round (2013).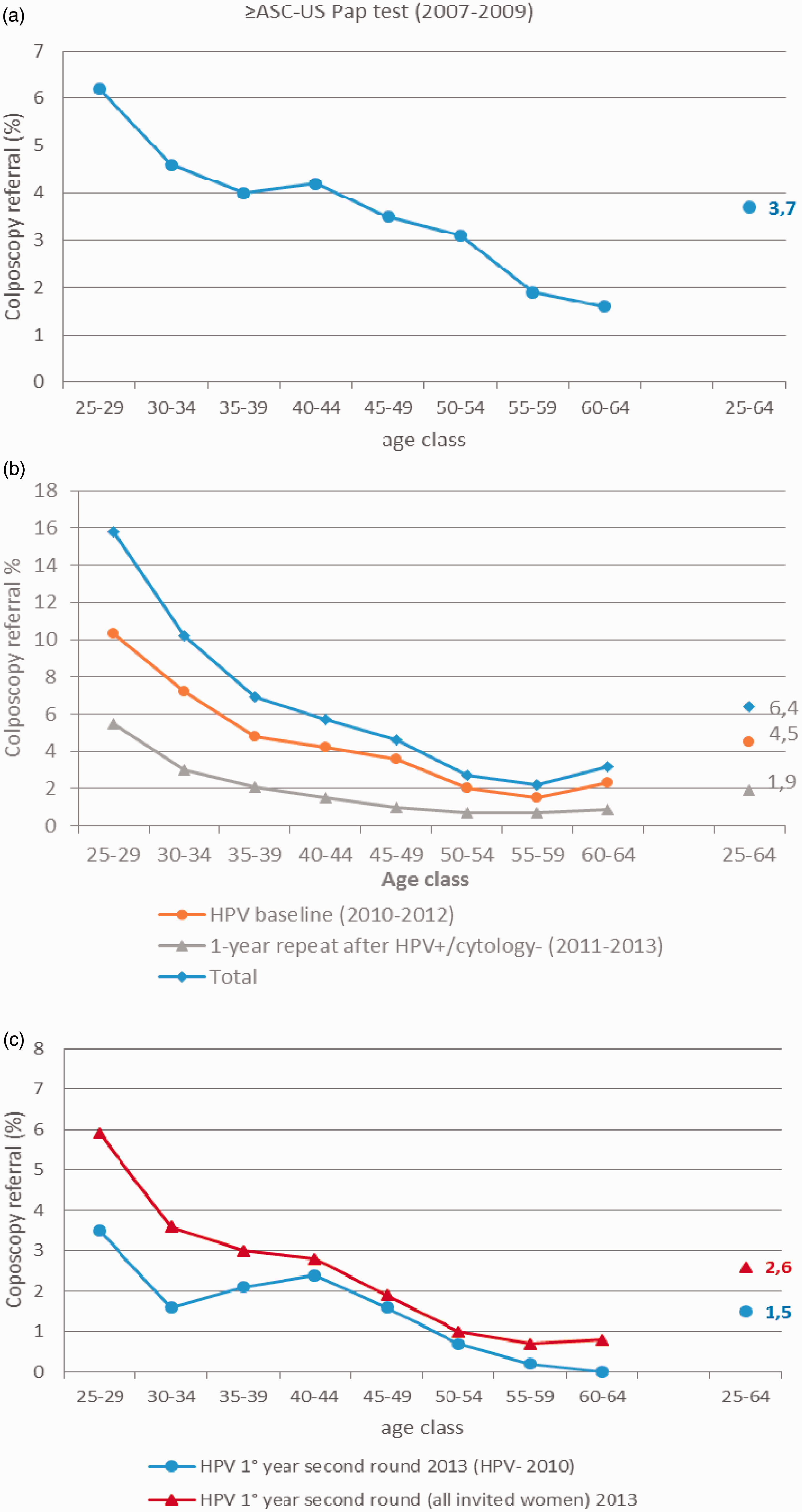
Detection rate and PPV
According to the screening algorithm, 824 women underwent immediate colposcopy, and 326 underwent colposcopy after one year. We found 149 and 23 CIN2+, respectively for an overall detection rate of 9.2/1000, compared with 5/1000 found in the previous round with the Pap test (relative detection rate 1.8; 95% CI 1.4–2.4). It must be noted that even the detection rate obtained at baseline with HPV screening (8.0/1000) was higher than that obtained in the previous round (relative detection rate 1.6; 95% CI 1.2–2.1) (see Table 2).
The PPV in HPV-based screening was 18.1% and 7.1% in HPV-positive cytology-positive and HPV-persistent women, respectively. The PPV was higher in women aged 35–44, and lower in women aged over 55. Of 356 women with persistent HPV, 134 (37.6%) had an ASC-US or more severe cytology, while the rest had a negative or inadequate cytology; 13 of the 23 CIN2+ were found in HPV-persistent cytology positive women, ie. 9.7% and 5.5% PPV, respectively.
For the second round, we only have results of the baseline testing, triage, and baseline colposcopies. Among the 126 colposcopies completed, we found only 13 CIN2+, for a partial detection rate of 2.5/1000 and a PPV of 10.3% (Figure 5). When the analysis was restricted to women with a previous negative test, we found only 3 CIN2, ie. 0.8/1000 detection rate and 5.1% PPV.
Detection rate by age and screening round: A. Pap test programme (2007–2009); B. HPV screening first round (2010–2012), by baseline and one-year follow up; C. HPV screening second round (2013).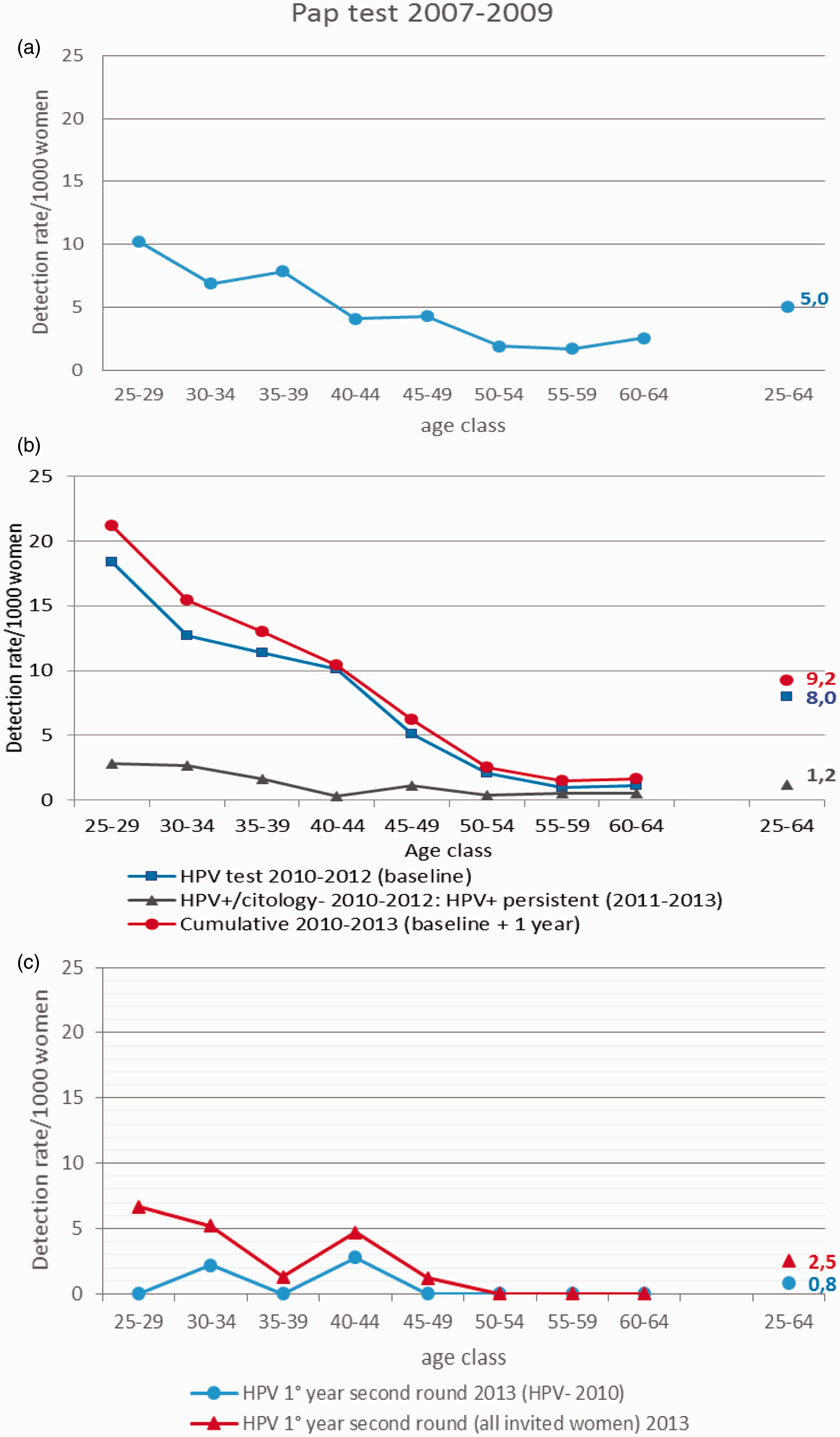
Wait times
The wait times for the communication of negative results with the HPV test were shorter compared with the Pap test: the proportion of women receiving results within three weeks changed from 62% to 98%, and the proportion receiving the results after more than one month changed from 6% to zero. There was, however, an increase in time from positive result of the first level test to colposcopy; the proportion of women undergoing colposcopy within four weeks changed from 48% to 25%, those from four to eight weeks increased from 21% to 31%, and those waiting more than eight weeks increased from 31% to 44%. In the case of HPV, these figures include the time needed for cytological triage.
Discussion
In this paper we report the results of one of the largest pilot studies of HPV-based cervical cancer screening studies in Italy. The study allowed us to assess the performance, impact on colposcopy workload, and acceptability by women and operators of the implementation of the screening algorithm, derived from the Italian guidelines and consistent with the upcoming European Guidelines. 12 Our study confirmed that this intervention is feasible and well received. The entire target population was invited during the three-year period, without any major logistical problems. The participation rate was slightly higher than the historical figures obtained with Pap test, and compliance with the proposed protocols was high, even to the one-year retesting in HPV-positive and cytology-negative women, which was expected to be problematic.21,22 It is worth noting that compliance to colposcopy in those with persistent HPV after one year was substantially lower than that in women positive to cytological triage. This finding has been already observed,16,17 and should be analysed to understand the reasons (eg. did women perceive their situation as low risk because the screening recommended waiting one year? Was the counselling by GPs or other physicians outside the screening programme confusing?).
The results show that most of the findings observed in clinical trials may be confirmed in routine practice. The main difference we noted in comparison with trials results was the detection rate in women HPV-positive cytology-positive at baseline, which was substantially higher in our study than in cytology-based screening, despite the fact that no cytology-negative women were referred to colposcopy. This finding is consistent with what has been observed in other studies15,16 that applied a real cytological triage. The cytology in most of the trials was interpreted blind to the HPV results,5,6,23–26 while in reality in an HPV-based screening programme, HPV positivity informs cytology. Our data suggest that the knowledge of HPV status by the cytologist increases sensitivity of Pap test, with a marginal effect on specificity. On the other hand, the detection rate in women HPV-positive cytology-negative at baseline and HPV-persistent at one year was lower than that observed in the trials. Given the non-randomized design of our trial, we cannot rule out the possibility that the two populations (that invited in the study period and that in the previous round) had different risk of disease, even though it is difficult to imagine such a different prevalence when most of the invited women in the two periods were the same.
During the first four years of the pilot study, the burden of colposcopies increased, due partly to the high level of cytology positivity at baseline, and particularly to low clearance at 1-year follow up, compared with the 50–70% observed in other studies.5,27,28,29 It was consistent, however, with the 40–50% observed in other Italian pilot studies.16,17 The finding is anomalous because persistence is higher in younger women, while the opposite was observed in all previous studies. 27 It is possible that the increase was only relevant for the first colposcopies, while colposcopies generated by the follow up after negative colposcopy and after treatment are reduced because of the introduction of HPV in the management protocols. 12 Currently, more than two thirds of colposcopies performed within screening programmes are follow ups to colposcopies that were negative for CIN2+ or for post-treatment follow up.
The increase in colposcopy referral compared with the Pap test will be somewhat lower in programmes adopting five-year intervals. However, this problem concerns only the first round, as in the subsequent round HPV positivity is rare and the positivity to cytology triage is much lower than in first round, leading to a colposcopy referral of about a third of that observed in the first round.
In the first year of the second round, we found not only a much lower HPV prevalence and colposcopy referral rate, but also an extremely low prevalence of CIN2+ lesions, about one tenth of that observed in the first round at baseline. This finding should further support the need for longer intervals in HPV-based screening.
Strengths and limitations
This pilot study was implemented within the real practice of a screening programme, and gives an idea of the problems a screening programme might face when adopting the recommendations of the upcoming European guidelines to shift from Pap testing to HPV testing. There are two notable differences in the protocol used in this study compared with what will probably be recommended. First, we adopted a three-year interval, while an interval of at least five years is now recommended. Second, our study included women from the age of 25, while the guidelines will probably recommend starting from age 30. In fact, using HPV testing for women under 30 may result in overdiagnosis and overtreatment of regressive lesions, as suggested by the results of the NTCC trial. 29 Our study also observed the strongest increase in detection rate with HPV compared with Pap test in this age group, although the preliminary results for the second round are showing a strong decrease in detection rate at all ages, including women who were 25–31 at first round: 1.4/1000 (1/706), compared with 18.4/1000 (40/2172) for women aged 25–29 in the first round. Unfortunately, while we suspect overdiagnosis caused by HPV, the Pap test has little or no efficacy in preventing cervical cancer in this age group.30–32
The pragmatic approach of our study implied that we did not have any randomized control, and the use of historical comparison may be biased by several factors. Nevertheless, it is impossible to evaluate the impact of a new population-based intervention, in terms of acceptability to the women and of organization, if a part of the programme continues to use the Pap test. Among other things, it would make public information campaigns (posters, brochures, local radio and TV, etc.) impossible, and it would be impossible to test the laboratory workload at the real volume of tests.
Finally, our main endpoint was histologically confirmed CIN2+, but we did not have an independent review of the pathology results.
Conclusions
Our study further confirms that HPV-based screening is feasible and well received by the target population. The main critical point may be the initial increase in colposcopy workload, but the adoption of longer intervals should partially compensate for this. After the first round, the positivity rate and the colposcopy burden will decrease dramatically, corroborating the need for longer intervals in HPV-negative women.
Data on the sensitivity of the screening protocol from real practice are even more optimistic than those from trials. Our data strongly suggest that triage cytology, when interpreted with knowledge of HPV positivity, has higher sensitivity than blind cytology.
Footnotes
Acknowledgements
We thank Jacqueline Costa for the English editing, all the screening programme operators, and all the women who participated in the screening.
Conflict of interest
PGR, as principal investigator of an independent study funded by the ministry of health, data owner, is making agreements with Roche diagnostics, Hologic Genprobe, Qiagen, Abbott, to obtain reagents at reduced price or for free.
Funding
This project started as part of a multicentre feasibility pilot study sponsored by the Italian Ministry of Health (Finanziamento CCM 2009) and the Agenzia Sanitaria Abruzzo (Lgs 138/09) that included 5 other screening programmes (Brescia, Ferrara, Florence, Monselice, Roma G); the Valcamonica Pilot project was also funded by the Regione Lombardia (DRG N.10813 del 16.12.2009).
