Abstract
Objectives
To estimate the cost-effectiveness of the public colorectal cancer screening program in the Abruzzo region, Italy.
Methods
Cost-effectiveness was analysed using a two-armed Markov model comparing: (1) Abruzzo screening program based on biennial faecal immunochemical occult blood testing, with colonoscopy as second level test for individuals with positive results, with (2) Treatment of symptomatic patients according to the stage of the neoplasm. Transition probabilities were adjusted for accuracy of tests and incidence of colorectal cancer. Diagnosis-related groups’ charges and field collected data were used to estimate costs. Costs and benefits were discounted by 3.5%. Monte Carlo simulation confirmed the robustness of the model results.
Results
Assuming a compliance rate of 64.7%, the incremental cost-effectiveness ratio for the current colorectal screening program was €433.06/quality adjusted life year gained, considerably lower than conventional thresholds (around €30,000).
Conclusion
Early detection and intervention programs help to avoid a large number of highly debilitating and expensive cancer treatments. These results show that the screening program currently implemented in Abruzzo should be considered as a good investment in health.
Introduction
Colorectal cancer (CRC) has one of the highest cancer mortality rates in Western countries, and is the second leading cause of cancer deaths in Italy.1–3 In 2013, there were 58,679 new CRC cases diagnosed in Italy; in the Abruzzo region, there were 1213 new cases, 474 deaths, and 7324 prevalent cases. In Italy, the lifetime risk of a CRC diagnosis is 50.9% for males and of 31.3% for females. 4 The distribution of CRC in Abruzzo is in line with the national average (mortality rate = 0.035%, incidence = 0.09%, prevalence = 0.49%). 2 CRC develops over a period of about 10 years. 5 Early symptoms are often overlooked. Surgery to eradicate the whole macroscopically detectable lesion is recognized as the only treatment which gives the patient a chance of recovering. 6
CRC screening tests suitable for people at average risk are faecal immunochemical occult blood testing (FIT), flexible sigmoidoscopy (FS), and total colonoscopy (TC). FIT, which allows detection of medium-large polyps which cause bleeding, is a less accurate but non-invasive test, and is recommended as a first level test to be performed every two years in the general population. 7 In Italy, the immunochemical examination of faecal samples8,9 is currently considered the preferred test. The FS test allows direct visualization of the intestine and the endoscopic polypectomy of lesions with a diameter of less than 1 cm, and is characterized by a high sensitivity and specificity for the polyps of the distal colon. FS is generally preferred over rigid sigmoidoscopy in clinical practice. It can detect ulcers, polyps, or cancer of the colon or rectum. FS is the only endoscopic screening modality that has been shown to reduce mortality in randomized clinical trials.10,11 TC provides a view of the entire colon, allowing the detection and the removal of polyps and the biopsy of suspicious tumour lesions. 12 Given its high diagnostic accuracy, and the time span that polyps may take to turn into cancer, TC screening every 10 years is considered adequately protective if there are no polyps or cancers. Guidelines therefore recommend TC every 10 years in asymptomatic, average risk people aged over 50.
If diagnostic efficacy were the only consideration, colonoscopy would be the ideal screening test, but colonoscopy also has considerable costs, for both the individual and the health service. Colonoscopy may be less acceptable to individuals as it is an invasive test that involves discomfort, and although rare, there are also health risks (e.g. perforation of the intestine). The effectiveness of the test may be undermined by poor uptake. In addition, the time taken to carry out the test and, consequently, the number of specialists needed to perform it, make it an expensive examination. 13 Colonoscopy is therefore not always used in organized screening programs (although virtual colonoscopy, providing a three-dimensional view of the inner wall of the intestine, could become the gold standard in the near future).
In Italy, several national and regional health care plans have recognized the urgent need to allocate resources for systematic programs to provide early diagnosis of the cancers with greatest epidemiological impact. In response to this, Italian regions have progressively committed to CRC screening programs, mostly based on biennial FIT, with TC as a second level assessment for the general risk population. Italian regions are autonomous in the choice of screening programs and tests. Typically, regions provide the FS test every two years to men and women aged between 50 and 70 or 74, although in some regions (e.g. Piedmont) only FS is offered once for people aged 58, and biennial FIT is offered until age 69 for those who refuse the invitation for FS.
The Abruzzo region CRC screening program, implemented in 2007, is directed at the general risk population aged between 50 and 70 (n = approximately 320,000): it uses biennial FIT as a first level examination, and total TC as a second level test for those with positive FIT results.
Evidence on the affordability of colorectal screening programs in Italy is limited, and the federal structure of the Italian NHS raises issues concerning the transferability of findings from one regional context to another. There is, therefore, a requirement for cost-effectiveness evaluations of regional screening programs. We aimed to investigate the cost-effectiveness of the Abruzzo CRC screening program, and to determine which variables most affect its success. The results may provide a generalizable decisional support tool to steer any revision of the current program.
Methods
We performed an economic evaluation based on a Markov model made up of two arms.14,15 The hypothetical cohorts in the model consisted of asymptomatic individuals aged 50–70 (i.e. the age group considered at risk in national and international guidelines6,16,17). We did not consider patients at high risk of CRC for reasons such as familiarity, inherited syndromes, chronic intestinal diseases; according to the regional guidelines, these individuals follow a different protocol. Data used to populate the model are shown in Table 1. The two arms of the model (see Figure 1) were:
Model data input.
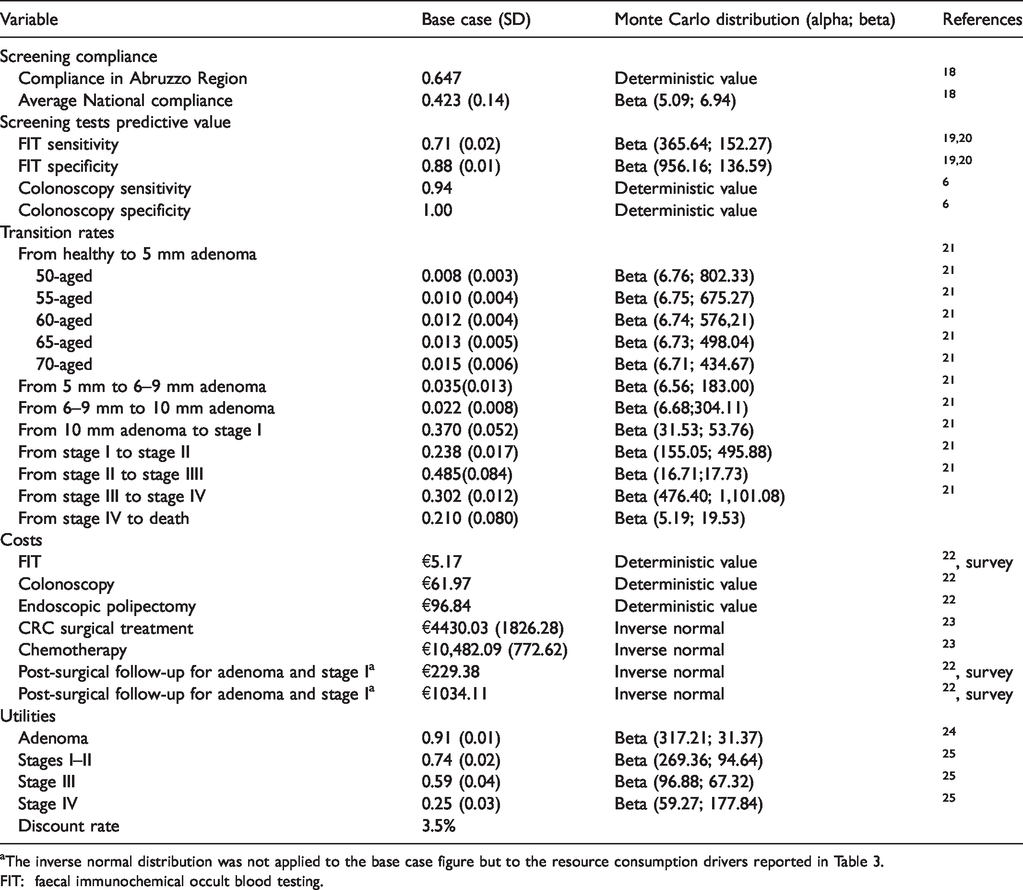
The inverse normal distribution was not applied to the base case figure but to the resource consumption drivers reported in Table 3.
FIT: faecal immunochemical occult blood testing.
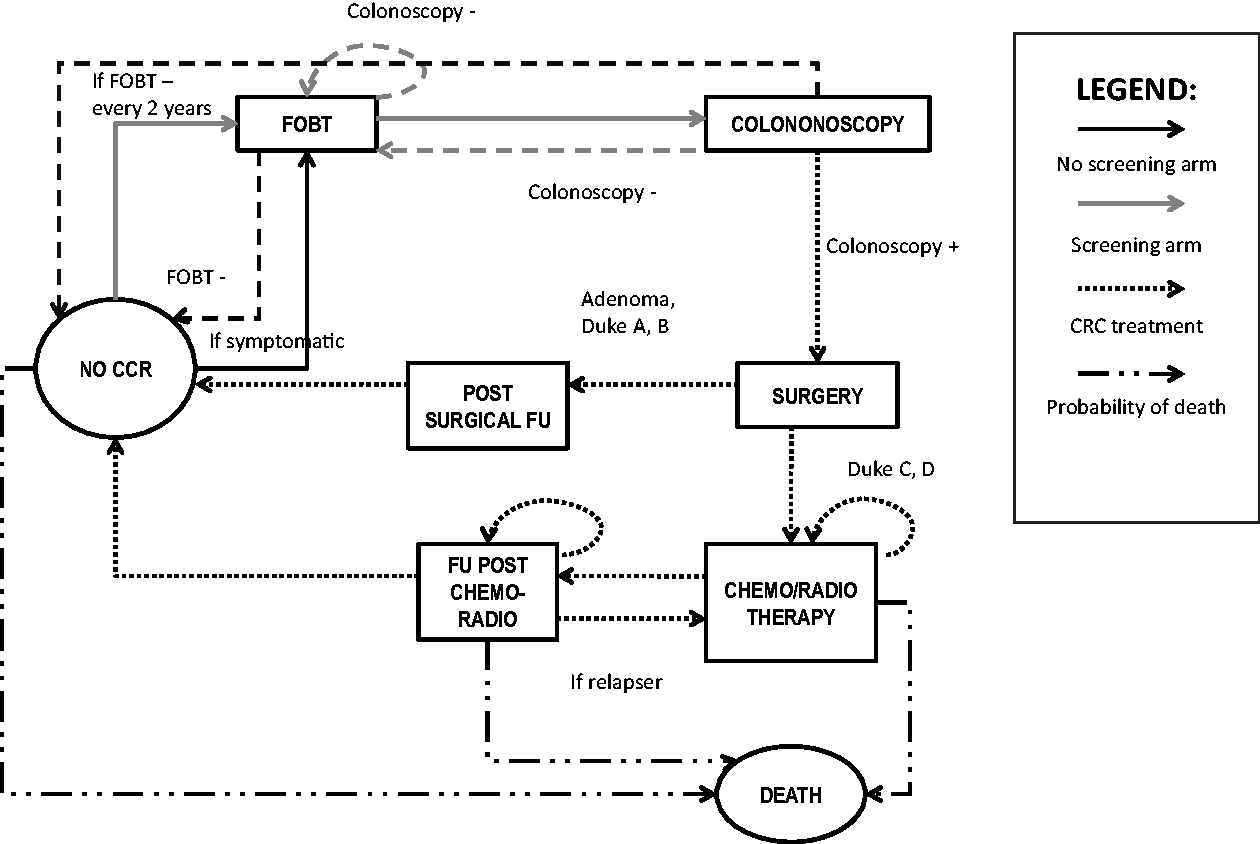
Screening and treatment of the CRC: Markov process. CRC: colorectal cancer.
“FIT + TC”: a screening programme involving FIT for the target population and TC for those with positive FIT results (≥100 ng/ml is generally considered the threshold for positivity);
“NS”: the alternative protocol, in which no screening (NS) is performed and patients are treated at the onset of symptoms.
We then performed a probabilistic sensitivity analysis to test the robustness of the results.
Each cycle of the Markov model lasts one year and the overall time horizon is lifetime. The states covered by the model are: the absence of symptoms (No CRC), FIT, colonoscopy, laparoscopic surgery, resection, follow-up after surgery, chemotherapy, follow-up after cancer treatment and death.
The effectiveness of screening tests was measured through sensitivity (to identify patients with the disease) and specificity (to identify healthy individuals).26,27 Sensitivity and specificity of FIT were obtained performing a fixed effect meta-analysis of two previous published studies,19,20 whereas sensitivity and specificity of TC were derived from Italian guidelines. 6
At the beginning of each cycle, the percentage of people who die from causes other than CRC (derived from ISTAT mortality tables 28 ) is excluded from the target population. The prevalence of the disease was not considered in identifying the target population, assuming that already symptomatic patients were not involved in the program.
Following the protocol of the Abruzzo region screening program, in period 0, men and women aged 50–70 were invited by personal letter to undergo FIT. Participants received online reporting; those with negative test results were asked to repeat the test every two years until age 70. Participants with positive test results were interviewed by a gastroenterologist who administered a TC. Participants with negative TC results were asked to repeat the FIT every two years; those with positive results were treated according to the level of disease severity. Patients with adenomas (precursors of CRC) were treated with resection, and followed up during the next cycle. 29 Patients with stage I or II CRC underwent resection by open surgery, and attended follow-up visits thereafter. Patients with stage III CRC underwent resection, followed by adjuvant chemotherapy. Patients with stage IV CRC were treated only with systemic chemotherapy and then followed up.18,30–32
Patients not compliant with early diagnosis enter the model when symptoms occur. Because the symptoms manifest when CRC is already at an advanced stage, it is assumed that positive patients undergo resection with adjuvant chemotherapy or radiation therapy if they are diagnosed with a stage III CRC, and directly pass to systemic chemotherapy (recommended for metastatic disease) if they are diagnosed with a stage IV CRC.
The transition probabilities were derived from a previous published study. 21 In the screening arm, these values were weighted to the predictive value of diagnostic tests. At the time of diagnosis, or at the symptomatic manifestation of the disease, the patient receives one of the treatments depicted in Figure 1, depending on the stage of the disease. The allocation of treatments to each disease stage was based on answers to the field survey, and on regional guidelines for CRC screening and treatment.
General organizational expenses to run the screening programme (€632,000), including invitations, recalls, and dedicated personnel and care for patients undergoing screening, were attributed to members of the target population, regardless of their participation in the program. Data on general costs, as well as the cost of the FIT kit to the Regional healthcare service, were collected in a face-to-face interview with the manager of the screening program. Data on cost drivers were derived from regional guidelines for screening. 6
Outpatient tariffs were used to estimate the cost per person of TC and immunological examinations, 22 whereas cost of hospital admissions, mainly due to adenoma or cancer resection, was quantified by means of diagnosis-related groups (DRG) tariffs currently in force in Abruzzo region.22,23 It was assumed that the drugs used in chemotherapy were included in the same DRG reimbursed for chemotherapy. The choice to quantify the cost of chemotherapy through the DRG was made because it was impossible to obtain reliable data, which implies an underestimation of the cost for patients in stages III and IV. However, available evidence does not allow an estimation of the volume of consumption of biological drugs for the chemotherapy for CRC in Abruzzo. Costs per person, cost drivers, and total costs referring to each disease stage were also collected.
In Europe, five-year relative survival of patients with CRC was 56.2% in 2002,31 though this will have improved in the intervening years. We used quality adjusted life years (QALYs), combining both survival and quality of life, as outcome measures, which enabled us to compare the estimated incremental cost-effectiveness ratio with conventional cost-effectiveness acceptability thresholds. 33 The quality of life coefficients for each health condition covered by the model were obtained from previous published studies.25,29 Costs and consequences of the two alternatives were discounted at an annual rate of 3.5%.
The study results were expressed in terms of incremental cost-effectiveness ratio (ICER). The reliability of the model results was assessed using deterministic and probabilistic sensitivity analysis. A one-way deterministic sensitivity analysis was conducted to identify credible ranges for critical parameters, and appraising the subsequent ICER’s variations. In addition, a multivariate probabilistic analysis was run to appraise the effect of the simultaneous variation of model’s parameters on the ICER. Appropriate random distributions were attached to each model parameter, 34 and 10,000 Monte Carlo simulations were generated to assess the distribution of these possible scenarios around the base case estimate. A beta distribution was attached to transition rates, utilities, predictive value of screening tests, and an inverse normal distribution was attached to costs and drivers of resources consumption (see Table 2). For cost items, the random distributions were attached to cost drivers.
Detailed costs of CRC screening and treatment.
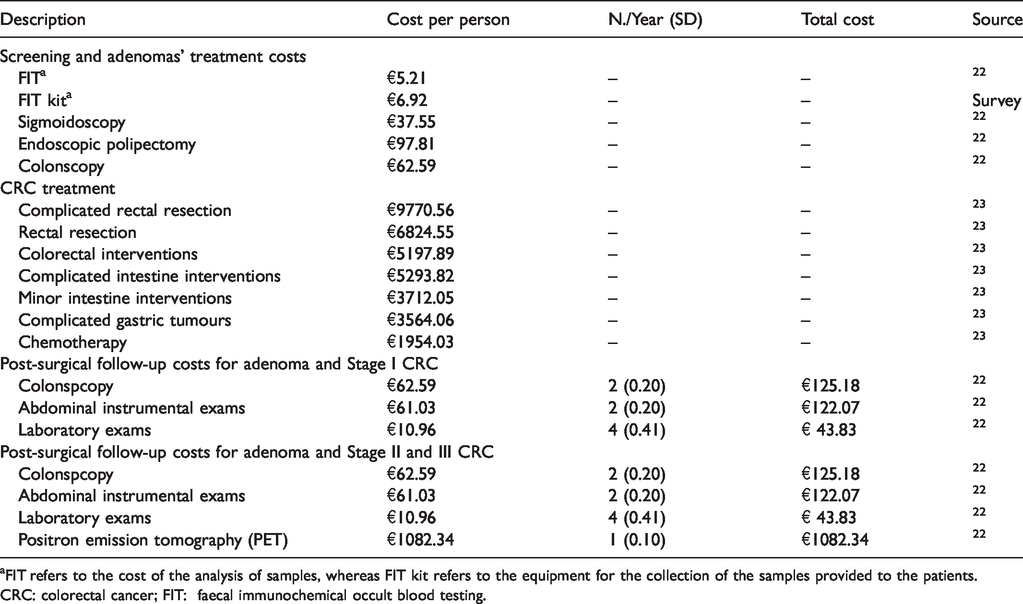
FIT refers to the cost of the analysis of samples, whereas FIT kit refers to the equipment for the collection of the samples provided to the patients.
CRC: colorectal cancer; FIT: faecal immunochemical occult blood testing.
A scenario analysis was conducted probabilistically to assess the impact of different levels of compliance on the model results. Minimum, maximum, and median compliance rate observed across Italian regions were utilized to build scenarios. 18
Results
In the base case, a healthy population of 320,000 asymptomatic individuals is undergoing FIT + TC every two years from age 50 to 70. The adjusted compliance rate observed in Abruzzo region (64.7% of invited individuals) was utilized to determine the number of people participating in the screening programme. Results, expressed in terms of cost per additional QALY gained with the screening program compared with an alternative protocol which only entails the treatment of symptomatic patients, are summarized in Table 3, which shows per patient values.
Base case results: “FIT + TC” vs. “No screening.”
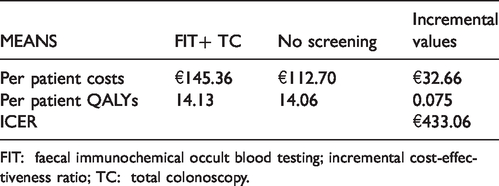
FIT: faecal immunochemical occult blood testing; incremental cost-effectiveness ratio; TC: total colonoscopy.
Overall, the screening program of FIT as the first level examination and TC as the second level diagnostic test yields an additional cost of €13,036,292.44 to the regional healthcare service, compared with no screening and the treatment of only symptomatic patients, along a lifetime horizon. On the other hand, the screening program produces 30,102.95 additional QALYs calculated for the whole cohort of patients. As a result, the screening produces a gain of and results in 0.64 QALYs (about seven months in perfect health) for each positive patient. The ICER for the screening strategy compared with no screening is equal to €433.6/QALY.
A one-way sensitivity analysis was performed, varying screening coverage from 10% to 100%. Predictive value of FIT, and FIT sensitivity and specificity were varied within their confidence intervals. For the outpatient services received during the follow-up, minimum and maximum numbers of visits and examinations were derived from the survey.
A sensitivity analysis on discount rate was also performed, by applying different discount rates to costs and QALYs. The tornado chart depicted in the Supplemental Appendix summarizes the results of deterministic sensitivity analysis. This analysis shows that the discount rate applied to costs is the variable that mostly affects the model results. Not surprisingly, regardless of the rate utilized to discount utility, a higher discount rate for costs disfavours the screening programme. While the screening programme requires an immediate investment and expenses concentrated in the first few years of screening activity, the alternative protocol, which only entails the treatment of CRC at the disease onset, does not produce immediate costs and is characterized by expenses spread out along the time horizon. The screening programme is dominant when costs are not discounted.
Compliance rate also strongly affects the model results. Hypothesizing a compliance rate of 100% produces an ICER of €367.16. A compliance rate of 10% results in an ICER of €747.86 per QALY gained. The other variables do not seem to affect the model results significantly.
A multivariate sensitivity analysis was performed though a Monte Carlo simulation. Results are depicted in Figure 2, in which the dispersion of simulations obtained around the base case is represented on a cost-effectiveness plan. The Monte Carlo results were ordered on a cost-effectiveness acceptability curve, which shows the probability of the screening being cost-effective for each level of incremental cost-effectiveness ratio. The figure shows that in several cases the screening program was dominant compared with the no screening scenario. In 10% of the scenarios produced, screening was more effective and less expensive than the no-screen option. There were no scenarios in which the screening option was less effective. The cost-effectiveness acceptability curve shows that the screening programme exhibits a cost per QALY gained smaller than €3500 with a probability of 100%.
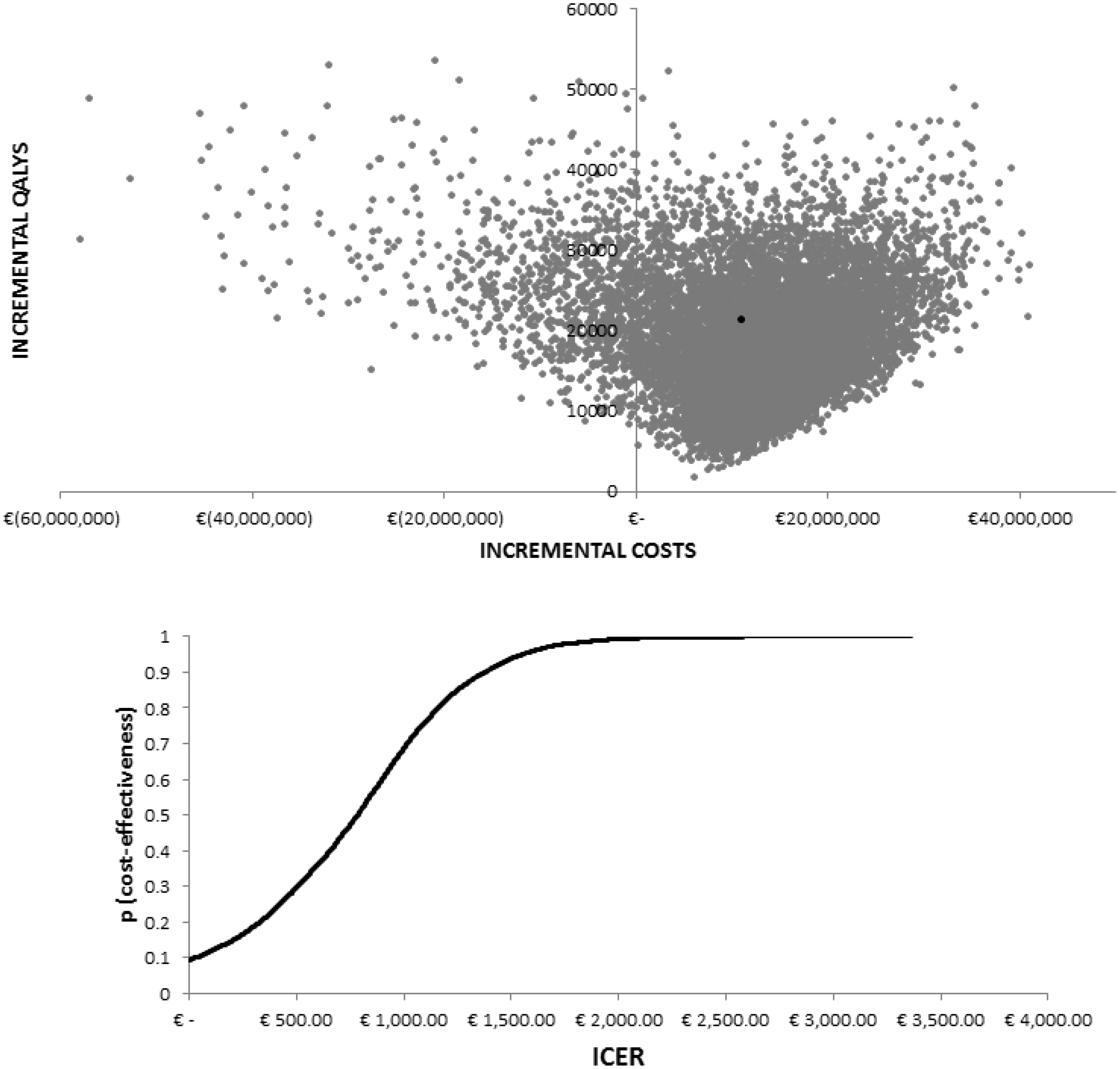
Monte Carlo analysis results.
The scenario analysis confirmed findings of the deterministic sensitivity analysis concerning adherence rate affecting the model results. The best scenario was obtained using the adherence rate of Abruzzo region. In 12.39% of scenarios, the screening programme was dominant and its maximum ICER equals €2172/QALY gained.
Discussion
In this study, the FIT+TC scenario was dominant, regardless of the adherence rate. The results show that the screening program currently implemented in Abruzzo should be considered as a good investment in health. The early intervention on patients with adenomas helps to avoid a large number of highly debilitating and expensive cancer treatments in the long term.
The sensitivity analysis confirmed the robustness of the result. The ICER for the screening strategy compared with no screening is equal to €433.6/QALY. This value is far below the conventional National Institute for Health and Care Excellence threshold of £20,000–£30,000 (equal to about €24,400–€36,700)/QALY.
The cost-effectiveness acceptability curve showed that the screening programme exhibits a cost per QALY gained smaller than €3500 with a probability of 100%. This figure is abundantly affordable if compared with the acceptability threshold conventionally taken into account by Italian decision makers. Because there is substantial cross-region variability in compliance with CRC screening programmes in Italy, a scenario analysis investigating extreme adherence rates has been addressed. There is some homogeneity in terms of screening schemes and costs across the whole of Italy, and so the scenario analysis could provide interesting insights to decision makers operating in other regional contexts.
Because cost savings and QALY gains are proportional to the compliance level, it is necessary to ensure that the culture of early detection is as widespread much as possible. Greater involvement of general practitioners in promoting screening procedures might prove to be an effective tool to reduce socio-economic-related inequalities. The contribution of general practitioners and specialists involved in the program should include encouraging citizens to undergo screening, and in cases with a positive FIT result, to undergo TC. A recent survey 24 demonstrated that the most effective way to obtain high levels of compliance is sending newsletters to the target population. A deeper investigation of communication methods might be useful to improve compliance.
Our results are in line with those of a previous study estimating the cost effectiveness of a similar screening programme in the United States. Although no cost-utility analysis was implemented, the authors found that with an estimated 8.7–9.4 million individuals screened, at a cost of $32 to $39 per person, an organized program prevented 2900 to 3100 deaths annually. 35 Several studies have shown that CRC screening is either cost-effective or cost-saving compared with no screening, regardless of the lack of agreement in the literature on the most cost-effective screening strategy. 36
Our study had some limitations. As we were unable to find reliable information on resource consumption, the costs of chemotherapy were valued using the relevant DRG, assuming that patients were treated according to the Abruzzo region guidelines on CRC screening and treatment. Using this approach, the costs attributed to each patient could have been underestimated, but had this been the case, the results of the model would have been favourable to the program.
In addition, the Markov model shows the progression of disease assuming that the patient moves from one health state to another in a one-year time span, although in reality the progression of the disease can occur at different time intervals. To overcome this limitation, we introduced a “half-cycle” correction, which evaluates the results of the model every six months. We also assumed that patients who were not undergoing screening only became aware of their CRC diagnosis in the later stages of the disease. In actuality, the time of diagnosis is usually strongly influenced by subjective factors, for example, individual habits, or the presence of other gastrointestinal diseases, which lead to more frequent medical checks. The transition probabilities used to populate the model were extrapolated from literature studies that utilized transition rates very similar to those considered in our model.37,38 As they were based on a different disease staging scale, however, we were unable to use them to populate our model.
We did not test any positivity cut-off for cost effectiveness; however, our sensitivity analysis took into account a wide range of possible values of prevalence and test accuracy. Conforming with guidelines for economic evaluation of healthcare programs, in our probabilistic sensitivity analyses, we attached random distribution to these parameters, which change simultaneously in 10,000 Monte Carlo simulations. None of these simulations produced an ICER that made the screening program non cost-effective, keeping the compliance level constant. The maximum ICER was slightly more than 3000 Euros. A cost/QALY of 35,000 Euro is conventionally considered the cost-effectiveness acceptability threshold. We assessed the effect of the accuracy of FIT in a one-way sensitivity analysis, the results of which are summarized in the tornado diagram in the Supplemental Appendix. The variation of sensitivity and predictive value of FIT, leaving all other considerations unchanged, does not significantly affect our ICER in either direction. With the lowest performance, FIT can induce an increase of the ICER up to 700 Euro per QALY gained, well below the cost-effectiveness acceptability threshold. Conversely, assuming the highest value of specificity and predictive value, the ICER stays positive and small.
Supplemental Material
MSC898732 Appendix - Supplemental material for Economic evaluation of colorectal cancer screening programs: Affordability for the health service
Supplemental material, MSC898732 Appendix for Economic evaluation of colorectal cancer screening programs: Affordability for the health service by Silvia Coretti, Matteo Ruggeri, Rossella Dibidino, Lara Gitto, Andrea Marcellusi, Francesco Saverio Mennini and Americo Cicchetti in Journal of Medical Screening
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study has been possible thanks to the funding of the Abruzzo region, in reference to a project under Article. D.Lgs.502/92 12, supplemented by the 12/bis D.Lgs.229/99. The views expressed here are those of the authors and not necessarily those of the funders.
Supplemental material
Supplemental material is available for this article online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
