Abstract
Objective
The national population-based colorectal cancer screening programme in Hungary was initiated in December 2018. We aimed to evaluate the current programme and investigate the costs and benefits of potential future changes to overcome the low coverage of the target population.
Methods
We performed an economic evaluation from a healthcare payer perspective using an established micro-simulation model (Microsimulation Screening Analysis-Colon). We simulated costs and benefits of screening with fecal immunochemical test in the Hungarian population aged 50–100, investigating also the impact of potential future scenarios which were assumed to increase invitation coverage: improvement of the IT platform currently used by GPs or distributing the tests through pharmacies instead of GPs.
Results
The model predicted that the current screening programme could lead to 6.2% colorectal cancer mortality reduction between 2018 and 2050 compared to no screening. Even higher reductions, up to 16.6%, were estimated when tests were distributed through pharmacies and higher coverage was assumed. This change in the programme was estimated to require up to 26 million performed fecal immunochemical tests and 1 million colonoscopies for the simulated period. These future scenarios have acceptable cost-benefit ratios of €8000–€8700 per life-years gained depending on the assumed adherence of invited individuals.
Conclusions
With its limitations, the current colorectal cancer screening programme in Hungary will have a modest impact on colorectal cancer mortality. Significant improvements in mortality reduction could be made at acceptable costs, if the tests were to be distributed by pharmacies allowing the entire target population to be invited.
Keywords
Introduction
Colorectal cancer (CRC) is a major health problem in Hungary, where mortality rates are among the highest in Europe and an increasing trend in incidence was projected due to the aging population.1–3 CRC screening can reduce cancer-specific mortality significantly and might also lead to a reduction in all-cause mortality.4,5 However, screening could also result in certain harms 6 and, therefore, expected net benefit should be assessed before implementing CRC screening at population level. 7 In Hungary, multiple pilot screening programmes were conducted with moderate success, considering screening and follow-up participation rates. 8 After these pilots, the national population-based organized CRC screening programme was initiated in December 2018, offering biennial fecal immunochemical test (FIT) screening to individuals aged 50–70 (positivity cut-off: 20 µg/g). 9 The invitation process for the target population is centrally coordinated by the National Public Health Institute. This involves sending invitations to all individuals associated with GPs who are participating in the programme. GPs volunteered to participate in the programme for extra funding, which is a fixed fee per screened individual. The invited individuals can collect the FIT kit from their GP.
Some organizational barriers might limit the performance of the current screening programme as shown by the EU-TOPIA project framework. 10 GPs are generally overwhelmed in Hungary as the country has been suffering from a significant workforce crisis in primary care in the past two decades, which is indicated by the decreased inflow of GPs and by the fact that almost half of GPs are aged over 55. 11 Hence, not all GPs have decided to have an active part in the organized CRC screening programme for reasons including the additional workload and the user-unfriendly IT platform of the programme. Thus, only eligible individuals whose GPs had volunteered to participate were invited in the first implementation of the screening programme. This resulted in a situation where a substantial part of the target population was not invited (invitation coverage approximatively 50%). Moreover, among the invited population, the willingness to perform the test and participate in diagnostic colonoscopy after a positive result was low. 8 Considering these major limitations, the short- and long-term outcomes of the current screening programme should be systematically evaluated in order to provide input for strategic health policy decisions. 12
In this study, we performed an economic evaluation of the Hungarian national CRC screening programme using an established micro-simulation model, investigating also the costs and benefits of potential future changes to the programme that may help to overcome the abovementioned barriers, including improvement of the IT platform currently used by GPs and distributing the FIT kits through pharmacies instead of GPs.
Materials and methods
MISCAN-colon model
We used the Microsimulation Screening Analysis-Colon (MISCAN-Colon) model (Erasmus University Medical Center, Rotterdam, The Netherlands) to simulate future outcomes of CRC screening in Hungary. MISCAN-colon is a well-established microsimulation model which has been used to inform public health policies in the US, Canada, Australia and Europe.6,13–15 The structure and underlying assumptions of the model are reported in the Supplementary Materials.
Study population
The model simulated the Hungarian population from 2015 to 2050. The age distribution was based on the observed age distribution in Hungary in 2018. 16 Supplementary Table 1 provides an overview of the main model assumptions. In this analysis, our model was specifically calibrated to replicate the age-specific CRC incidence observed in Hungary in 2008–2012 (period before introduction of screening, Supplementary Figure 1). 17 Incidence and age-specific CRC mortality data were obtained from the National Screening Registry. As data on CRC stage distribution were not available in Hungary, those model parameters were calibrated using pre-screening data from a neighboring country (Slovenia, period 2004–2008). 18 The model used all-cause mortality estimates from the 2014 Hungarian life tables. 19 Because age- and stage-specific information on CRC relative survival was not available in Hungary, we informed our model with the age- and stage-specific survival observed in The Netherlands during 1999–2003 (five-year CRC relative survival: 59%). Under these assumptions, the predicted CRC mortality rates showed a reasonable fit with the Hungarian CRC mortality rates during the period 2008–2012 (Supplementary Figure 1). 20
Simulated screening scenarios
In order to evaluate potential future improvements to the programme, we modeled the 2015–2050 Hungarian population under 17 specific screening scenarios as described in Table 1. First, we simulated no screening (as reference for computing all screening benefits; “No screening”). Second, we simulated the current screening scenario assuming biennial FIT screening from age 50 to 70 (starting in 2018), with the FIT kit collected from the GP, in which 50% of target population is invited to FIT screening, and of those 40% participate (“Current screening strategy”). 8
Overview of the assumptions for each simulated screening strategy.
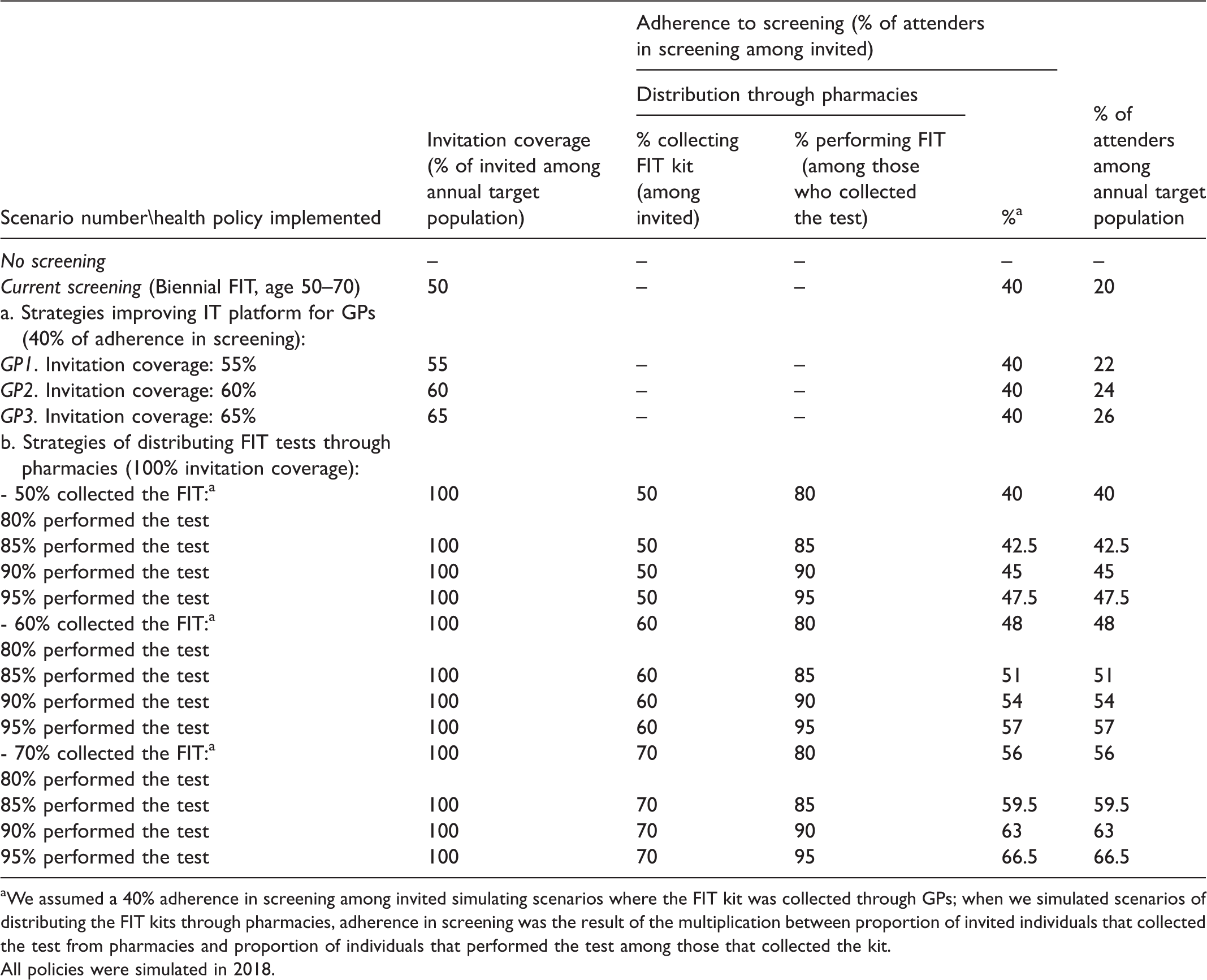
aWe assumed a 40% adherence in screening among invited simulating scenarios where the FIT kit was collected through GPs; when we simulated scenarios of distributing the FIT kits through pharmacies, adherence in screening was the result of the multiplication between proportion of invited individuals that collected the test from pharmacies and proportion of individuals that performed the test among those that collected the kit.
All policies were simulated in 2018.
Then, we investigated the impact of updating the IT platform of the organized screening programme used by GPs. We assumed such an improvement would increase GP participation. Specifically, we simulated three specific scenarios (see Table 1) where we assumed that this policy would result in a direct increase in invitation coverage because those individuals of the target population whose GPs would newly join the programme could now be invited for screening. In all these three scenarios, the other characteristics of the screening programme were simulated as in the current screening scenario. 8
Finally, we simulated 12 specific screening scenarios to investigate the potential impact of involving pharmacies instead of GPs in the distribution of the FIT kit. For all these scenarios, biennial FIT screening starting in 2018 was simulated assuming the entire target population aged from 50 to 70 years was invited (100% invitation coverage). Full invitation coverage was assumed because it is expected that pharmacies would collectively join the screening programme under the lead of their advocacy organization, instead of joining individually as the GPs did. We assumed that the impact of this policy relates to: (i) the proportion of invited individuals who collect the test from the pharmacies; and (ii) the proportion of individuals that perform the test once collected. Specific assumptions for these parameters are listed in Table 1.
All screening scenarios (except for no screening) were simulated assuming 60% adherence in diagnostic colonoscopy. 8 For individuals with adenomas detected during a diagnostic colonoscopy, surveillance colonoscopy was offered. Surveillance was simulated every one to five years depending on the number and size of adenomas, in line with the European guidelines, assuming an adherence of 60%. Assumptions for test characteristics for FIT and colonoscopy were based on scientific literature (Supplementary Table 1).
CRC screening costs
We performed a cost-effectiveness analysis from a healthcare payer perspective. Costs for CRC screening were obtained from the National Screening Coordination Department and costs for CRC treatment were extracted from a study that estimated the net cost of CRC patients’ care at patient-level in Hungary. 21 All costs were converted to Euro (Supplementary Table 1). Simulating FIT screening, screening costs were accounted differently according to the simulated screening policy. Simulating current screening, we accounted a FIT organizational cost (€9.6) for each invited individual, as well as a laboratory cost (€8.9) and GP reimbursement cost (€4.8) for each FIT performed. When we simulated scenarios with the updated IT platform, we accounted the same FIT cost as in the current screening. When we simulated the FIT kit distributed by pharmacies, we accounted a FIT organizational cost (€9.6) for each invited individual, a pharmacy reimbursement (€2.1) for each FIT kit collected, and a laboratory cost (€8.9) for each FIT performed. Finally, we included for each screening scenario (except ‘No screening’) two organizational public investments (€1.85 Million in 2018 and €1.9 Million in 2020) made by the Hungarian government to improve the facilities of health service providers performing colonoscopies.
Model outcomes
For each simulated scenario, we computed the effectiveness, i.e. prevented CRC deaths and life-years gained from screening (LYG), and costs of screening. LYG and costs were discounted by 3.7% annually, as indicated by the Hungarian guideline of performing economic evaluations. 22 In addition, we computed the cumulative reduction in CRC mortality due to screening over time and the total undiscounted net costs (compared to the current screening strategy) per calendar year during the period 2018–2050.
Cost-effectiveness analysis
Cost-effectiveness parameters were evaluated comparing each simulated screening scenario with no screening. However, incremental cost-effectiveness ratios (ICERs) were estimated as ratio of additional costs and additional LYG in comparison with the current screening scenario. Cost-effectiveness results were computed specifically among individuals aged 50 or older during the period 2018–2050 (cost-effectiveness outcomes in period 2018–2030 were also computed and reported in Supplementary Table 2).
Sensitivity analyses
We investigated the model parameter uncertainty by performing specific sensitivity analyses. In these, we assumed: (i) a higher participation in the follow-up diagnostic colonoscopy (80%); (ii) lower organizational costs for the FIT test (−10%, −20%, or −50%); (iii) higher or lower GP reimbursement costs (variations in the actual reimbursement assumed as follows: 50%/20% lower; or 20%/50% higher); and (iv) higher or lower pharmacy reimbursement costs (variations in the actual reimbursement assumed as follows: 50%/20% lower; or 20%/50% higher). We summarized the results of those analyses in Supplementary Table 3.
Results
In the absence of screening, the model predicted up to 189,600 CRC deaths in Hungary between 2018 and 2050. The current screening strategy was estimated to avoid 2.9% of CRC deaths in the 2018–2030 period and up to 6.2% in the period 2018–2050 (Table 2, Supplementary Table 2). Up to 7.9 million performed FITs and 0.3 million colonoscopies were required by the current screening scenario (Table 2).
Colorectal cancer screening simulated outcomes (×10,000, for individuals in the total Hungarian population aged 50–100 years-old in 2018–2050) per policy implemented.
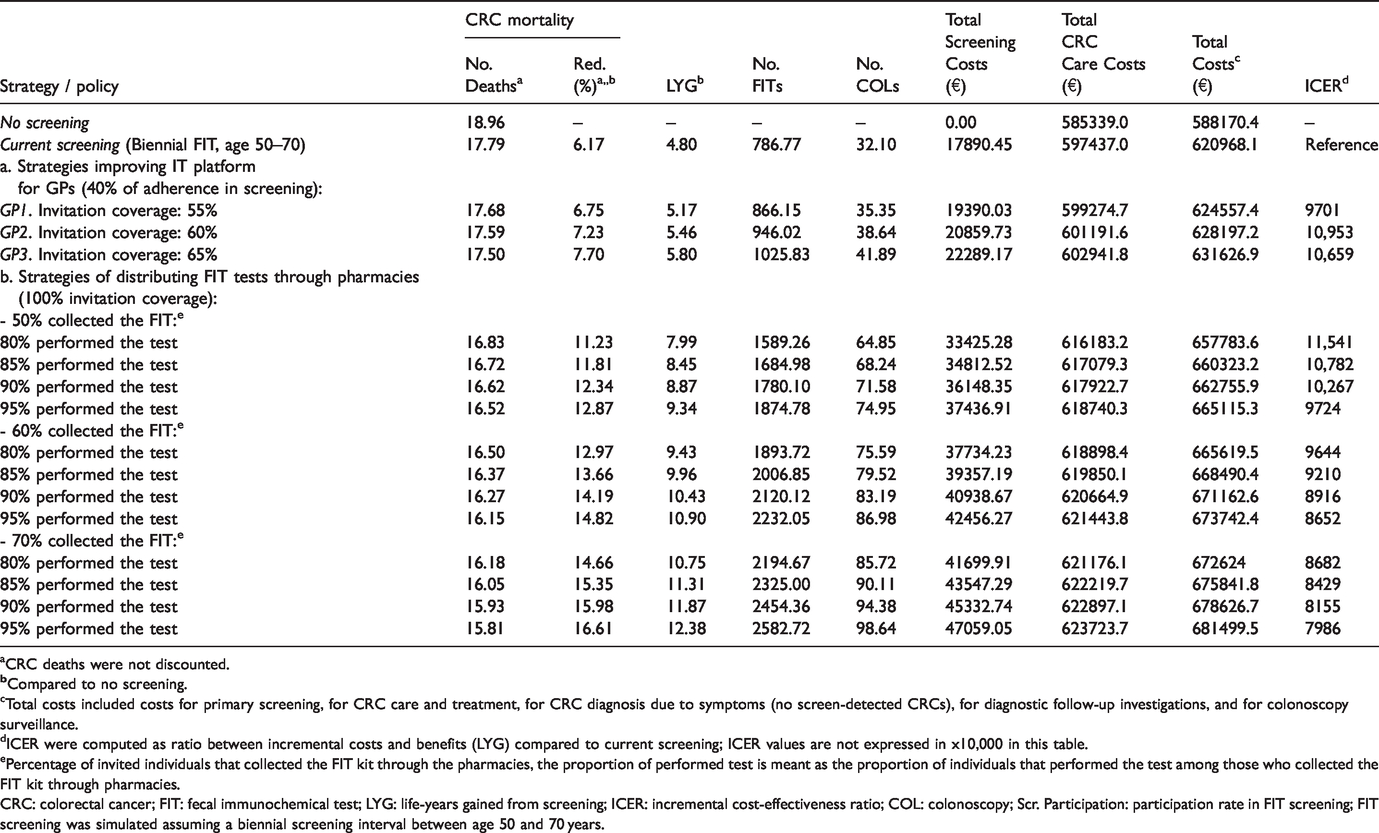
aCRC deaths were not discounted.
cTotal costs included costs for primary screening, for CRC care and treatment, for CRC diagnosis due to symptoms (no screen-detected CRCs), for diagnostic follow-up investigations, and for colonoscopy surveillance.
dICER were computed as ratio between incremental costs and benefits (LYG) compared to current screening; ICER values are not expressed in x10,000 in this table.
ePercentage of invited individuals that collected the FIT kit through the pharmacies, the proportion of performed test is meant as the proportion of individuals that performed the test among those who collected the FIT kit through pharmacies.
CRC: colorectal cancer; FIT: fecal immunochemical test; LYG: life-years gained from screening; ICER: incremental cost-effectiveness ratio; COL: colonoscopy; Scr. Participation: participation rate in FIT screening; FIT screening was simulated assuming a biennial screening interval between age 50 and 70 years.
Updating the IT platform for the GPs reduced CRC mortality in the 2018–2050 period up to 7.7% in the case of 65% invitation coverage (compared to No screening) (Figure 1), requiring up to 10.2 million performed FITs and 0.4 million colonoscopies. Screening-related costs of the programme increased from €179 million to €223 million in this scenario. Moreover, annual net costs were estimated to potentially reach €6.2 million in the period 2018–2030 (Figure 2). Compared to current screening, the incremental costs for every additional life-year (ICER) were €9701, €10,953 and €10,659 per LYG for the invitation coverages of 55%, 60% and 65%, respectively.
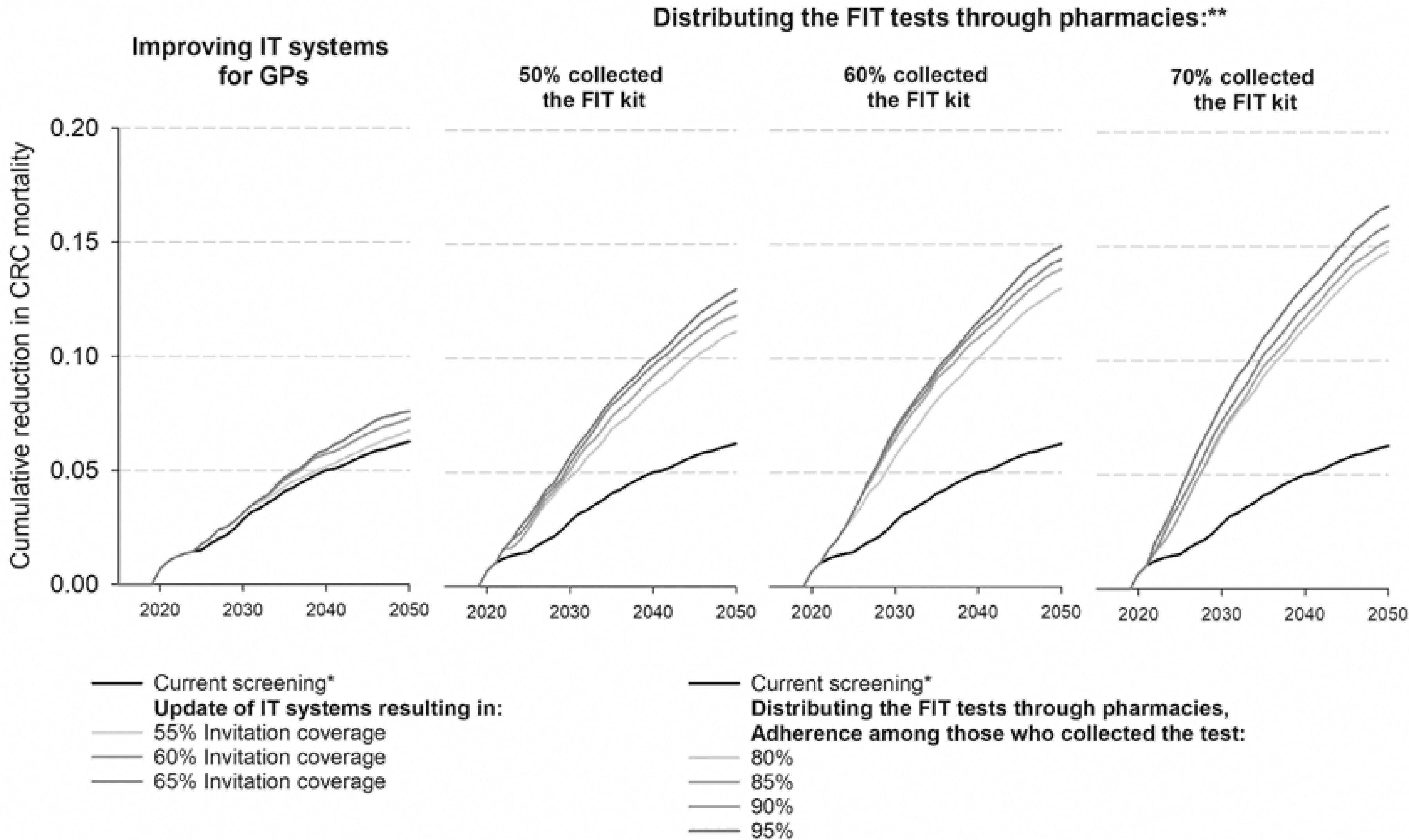
Cumulative colorectal cancer mortality reduction due to screening in Hungary for individuals aged 50 years or older and per simulated screening scenario. Note: *current screening: biennial FIT, 50–70, invitation coverage = 50% and screening adherence among invited = 40%; ** we assumed full invitation coverage in scenarios where the FIT kits were distributed through pharmacies.
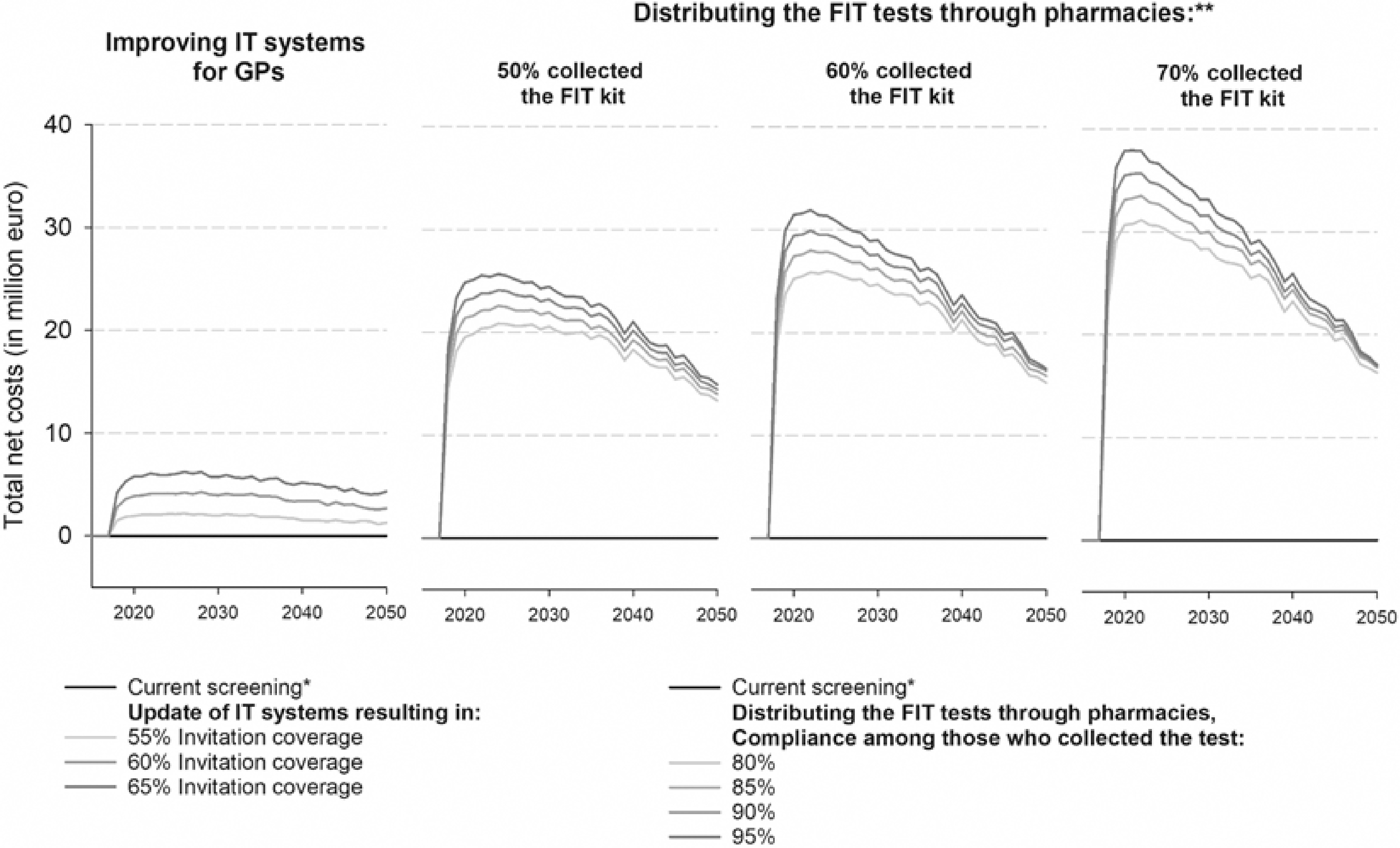
Estimated total annual net costs in Hungary among individuals aged 50 years or older and per simulated screening scenario (net costs compared to the current screening scenario). Note: *current screening: biennial FIT, 50–70, invitation coverage = 50% and screening adherence among invited = 40%; ** we assumed full invitation coverage in scenarios where the FIT kits were distributed through pharmacies.
The model predicted higher reductions in CRC mortality when pharmacies distributed the FIT test (Figure 1). During 2018–2050, the estimated mortality reduction ranged from 11.2% to 16.6% depending on expected rates of FIT collection and adherence. The highest reduction was observed when 70% of the invited individuals collected the test with 95% performing the test after picking it up. Distributing the test through pharmacies was also estimated to avert slightly more CRC deaths in the first decade (2018–2030) compared to the current screening scenario (Figure 1, Supplementary Table 2).
When 50% of the individuals were simulated to collect the FIT through pharmacies, the model estimated that up to 18.7 million FITs and 0.7 million colonoscopies were required by the programme, with predicted ICERs ranging from €9700 (95% adherence rate) to €11,500 (80% adherence rate) per LYG. When more individuals collected the FIT from pharmacies, the resources needed for the programme and the costs increased (Table 2). When 70% of invited individuals collected the test, up to 26 million performed FIT and 1 million colonoscopies were needed by the programme, with predicted ICERs ranging from €8000 (95% adherence rate) to €8700 (80% adherence rate) per LYG. The screening-related costs for this scenario were estimated to be €471 million. Annually, total net costs of the pharmacy scenarios (screening costs + CRC care costs) ranged from €13 to €37 million, with the highest annual net costs estimated in the first years after the introduction of the policy (2018–2025; Figure 2).
Compared to the scenarios of updating the IT platform, the scenarios of distributing the FIT tests through pharmacies resulted in more benefits and lower ICERs when at least 60% of invited individuals collected the tests. Therefore, these alternatives are more cost-effective options to improve the current system.
Sensitivity analyses
The impact of model parameter uncertainty was investigated for a selected number of scenarios in Figure 2 and for all simulated scenarios in Supplementary Table 3. ICERs were reduced by between €1000 and €3000 per LYG when we assumed a higher participation in diagnostic colonoscopy (i.e. 80%) or a 50% reduction in the cost of the FIT. Varying the GP reimbursement costs, pharmacy reimbursement costs, or reducing the FIT costs (by up to 10% or 20%) did not substantially increase or decrease the ICERs.
Discussion
This study provides the first comprehensive evaluation of the newly implemented CRC screening programme in Hungary, using a widely validated simulation model. We show that even with its important limitations, the current screening programme can ensure a modest mortality reduction in the long term for the Hungarian population aged 50–100. However, we also investigated a number of alternative scenarios in which the major barriers of the screening programme could (at least partly) be overcome, leading to even better outcomes: e.g. over 15% mortality reduction with ICERs estimated between €8000 and €8400 per LYG in some scenarios. These ICERs are well below the current Hungarian threshold for cost-effectiveness in drug reimbursement (health technologies with an ICER above three times the GDP per capita [∼€40,000]/quality-adjusted life years are considered not cost-effective). 22 The national guideline for health technology assessment does not define cost-effectiveness thresholds for other health technologies such as public health programmes.
Our study presumed that different invitation strategies lead to a variation in participation rate (i.e. different % of attenders among annual target population shown in Table 1). The key benefit of the pharmacy scenario would not necessarily be the increased attendance rate of the invited individuals, but the higher coverage, which could lead to the invitation of the full target population for the screening. An interesting trade-off was investigated in this respect, where we assumed a lower unit cost per FIT test for pharmacies compared to GPs, with a more frequent occurrence as pharmacies would receive the funding for every distributed KIT, whereas GPs received it after every performed test. The ICERs showed that most scenarios of distributing the FIT tests through pharmacies were more favorable, indicating that the costs of reimbursing more tests (even some unused) would be outweighed by the higher benefits expressed in LYG.
As the effectiveness of screening is directly associated with the level of screening participation, it is also reasonable to expect that screening costs (i.e. test and following investigation costs) increase as well. However, our model also estimated elevated costs due to CRC care (both in the short and long term). This is counterintuitive as detecting CRC at an earlier stage should be associated with less expensive treatments and hospitalization costs, as shown in previous cost-effectiveness analyses (also carried out with MISCAN-Colon).23–25 This result can be explained by the fact that costs did not vary substantially according to CRC care phase: costs accounted in the last year before death (from CRC or other causes) were in line with those for ongoing/continuous care. In previous cost-effectiveness analyses, terminal care costs were from 10- to 50-fold higher than those assumed for continuous care.23–25 Thus, when a CRC case was detected early at a lower stage in our analysis, the model accounted lower CRC terminal care costs (death averted) than in the previous analyses but higher costs for CRC ongoing care (because accounted for each LYG for those with a screen-detected CRC).
Besides the results concerning the cost-benefit ratio of the investigated scenarios, other estimations of our economic evaluation, such as number of tests performed or number of follow-up examinations executed, are important for capacity planning purposes (e.g. human resources, organizational capacities). Scenarios in our study also indicated an increasing number of colonoscopies to be performed in the future. These estimations could help healthcare policymakers to judge whether the two public investments made in 2018 and 2020 to improve the facilities for performing colonoscopies were sufficient, or whether further investment is required in the future.
In the literature, CRC screening programmes have been proven to be highly cost effective, ensuring major health gains at acceptable costs.7,26,27 However, it is not clear which screening strategy is preferable for a population-based CRC screening programme, as costs of screening, screening adherence, test sensitivity, and costs of CRC treatment have a substantial impact on overall cost-effectiveness and are highly dependent on country settings. 28 For example in a previous economic evaluation, Arrospide et al. 26 found that, in the first years of the CRC FIT screening programme in the Basque country (64.3% screening adherence), €69.2 million were necessary (on average) to annually fund the programme. In our budget analysis, we found that in Hungary the current FIT screening programme would need from €14 to €20 million of annual funds (Supplementary Table 4) during its first years assuming that 20% of the individuals in the target population participated in screening.
In the scenarios where pharmacies would distribute the tests, we projected a relatively large total net cost compared to the current screening strategy (ranging from an extra €13 to €37 million, see Figure 2). However, these estimates include not only the screening-related costs but also the increased costs in CRC care. Still, such an increase in costs would require a significant investment considering the Hungarian public health perspective. To put this amount into a local context, about €85–90 million were nominated for public health in the annual national budget in Hungary (not counting the less centralized regional or local-level spending on public health). This amount covered mainly national-level public health programmes, interventions and initiatives.
Our study has a number of limitations. First, some of the input data for the modeling were not available for Hungary, and therefore data from other countries were used. Second, our screening scenarios are based on the experience and knowledge of experts from Hungary, and it is difficult to judge whether they are realistic to achieve in real life. However, we performed extensive analysis with multiple scenarios and sensitivity analyses, so the results should be useful under a wide range of circumstances. Third, there were certain cost elements which were not possible to estimate and include in the calculations (i.e. additional costs of pharmacies to implement screening-related tasks or additional investments needed to improve the current IT platform used by the GPs), and therefore future screening scenarios might underestimate costs. Moreover, our analysis did not include additional alternative options of invitation, such as sending the FIT kit by post. However, with the low participation in CRC screening currently observed in Hungary, introducing this last option may not be opportune. Fourth, the benefits of screening in economic evaluations are frequently expressed in quality-adjusted life years.29,30 However, quality-of-life data were not available in our case and, therefore LYGs were used. Finally, the MISCAN-Colon model simulates the natural history of CRC through the adenoma-carcinoma sequence and does not consider adenoma histology (villous histology or advanced atypia) or sessile serrated polyps.
The results of our study should serve as the basis for further improvement of the current CRC screening programme in Hungary. Switching to the distribution of FIT kits through pharmacies instead of GPs in the organized screening programme seems to be a justifiable and important step towards achieving higher invitation coverage. However, it should be noted that such a change might be difficult to achieve without the collaboration and support of GPs who are currently the key stakeholders. Although the test kits would not be distributed by the GPs in this scenario, their role in the screening process would still be crucial as they would still be responsible for the coordination of the patient pathway from the screening to patient care. Thus, appropriate communication with the GPs is an important initial element for implementing future changes in the screening programme. On the other hand, pharmacies should also be prepared to implement screening-related activities into their current practice.
Conclusions
Despite the important organizational limitations, the current national CRC screening programme in Hungary can ensure modest mortality reduction in the long-term for the population aged between 50 and 100. However, this study shows that in order to fully exploit the benefits of the programme, further improvements are required; for instance, changes to the current IT platform or involving pharmacies in the test distribution mechanism. We estimated that alternative scenarios reflecting these changes have favorable cost-benefit ratios.
Supplemental Material
sj-pdf-1-msc-10.1177_0969141320968598 - Supplemental material for Modeling costs and benefits of the organized colorectal cancer screening programme and its potential future improvements in Hungary
Supplemental material, sj-pdf-1-msc-10.1177_0969141320968598 for Modeling costs and benefits of the organized colorectal cancer screening programme and its potential future improvements in Hungary by Marcell Csanádi, Andrea Gini, Harry de Koning, György Széles, János G Pitter, Beatrix Oroszi, Piroska Pataki, Petra Fadgyas-Freyler, Gyula Korponai, Zoltán Vokó and Iris Lansdorp-Vogelaar: for the Oxford Vascular Study Phenotyped Cohort in Journal of Medical Screening
Footnotes
Acknowledgements
The content of this publication reflects only the EU-TOPIA groups’ views and the European Commission is not liable for any use that may be made of the information contained herein. We would like to acknowledge all members of the EU-TOPIA consortium for their support in the development of the project and contributions to this publication. We also would like to acknowledge the valuable support of Dr. Attila Kovács in the manuscript preparation, who was an Advisory Board member of the EU-TOPIA project.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was part of the EU-TOPIA project, funded by the EU-Framework Programme (Horizon 2020) of the European Commission, project reference 634753.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
