Abstract
Background and aims
In 2008, Ontario initiated a population-based colorectal screening program using guaiac fecal occult blood testing. This work was undertaken to fill a major gap in knowledge by estimating serious post-operative complications and mortality following major large bowel resection of colorectal cancer detected by a population-based screening program.
Methods
We identified persons with a first positive fecal occult blood result between 2008 and 2016, at the age of 50–74 years, who underwent a colonoscopy within 6 months, and proceeded to major large bowel resection for colon cancer within 6 months or rectosigmoid/rectal cancer within 12 months, and identified an unscreened cohort of resected cases diagnosed during the same years at the age of 50–74 years. We identified serious postoperative complications and readmissions ≤30 days following resection, and postoperative mortality ≤30 days, and between 31 and 90 days among the screen-detected and the unscreened cohorts.
Results
Serious post-operative complications or readmissions within 30 days were observed among 1476/4999 (29.5%) cases in the screen-detected cohort, and among 3060/8848 (34.6%) unscreened cases. Mortality within 30 days was 43/4999 (0.9%) among the screen-detected cohort, and 208/8848 (2.4%) among the unscreened cohort. Among 30 day survivors, mortality between 31 and 90 days was 28/4956 (0.6%) and 111/8640 (1.3%), respectively.
Conclusion
Serious post-operative complications, readmissions, and mortality may be more common following major large bowel resection for colorectal cancer between the ages of 50 and 74 among unscreened compared to screen-detected cases.
Keywords
Introduction
Two guidelines were issued in Canada 1 , 2 recommending biennial guaiac fecal occult blood testing (gFOBT) for colorectal screening of asymptomatic persons aged 50–74, based on the 1998 meta-analysis 3 of trials of gFOBT, showing reduced mortality from colorectal cancer (CRC). Subsequently, the Ontario government established a population-based colorectal screening program, ColonCancerCheck (CCC), recommending biennial gFOBT for persons aged 50–74 who do not have a first degree relative affected by CRC. Information about CCC and performance measures have been published elsewhere. 4 , 5 In general, persons screened by gFOBT were offered stool testing kits by their primary care practitioner.
Early detection and treatment of CRC reduce the risk of death from the disease, 3 but also expose those whose CRC is screen-detected in the asymptomatic state to risks which they otherwise would not have experienced until presenting symptomatically. The randomized trials of gFOBT versus controls did not detect sufficient CRCs to provide robust estimates of serious post-operative complications or post-operative deaths following major large bowel resection for CRC detected by a screening program, and would not have been relevant to contemporary surgical outcomes. The objective of this work is to quantify the morbidity and mortality associated with major large bowel resection of CRC detected by CCC, because there is little information on these outcomes published by population-based colorectal screening programs, and to quantify these measures in an unscreened contemporaneous cohort from the same underlying population.
Methods
We extracted the first record of a positive gFOBT result per patient between 2008 and 2017 from the CCC gFOBT database, and linked the records to the Registered Persons Database (RPDB) of the Ontario Health Insurance Plan (OHIP) to identify those between 50 and 74 years of age. Using the Ontario Cancer Registry, the Ontario Crohn's and Colitis Cohort dataset, the OHIP billing claims database, and the RPDB, we identified and excluded those with a prior diagnosis of CRC or inflammatory bowel disease, a prior history of total colectomy, or who were not residing in Ontario.
We linked the gFOBT-positives to the OHIP billing claims database to identify those who underwent colonoscopy ≤6 months following the date of the first gFOBT result, and identified those with a first ever diagnosis of rectosigmoid or rectal cancer in the Ontario Cancer Registry associated with a billing claim for major large bowel resection 6 in the OHIP billing claims database ≤12 months following colonoscopy, and those with colon cancer, ≤6 months following colonoscopy. Major bowel resection was defined as right hemicolectomy, resection of large intestine with anastomosis or mucous fistula, anterior resection, proctosigmoidoscopy, Hartmann procedure, abdomino-perineal resection, or total colectomy. In the case of simultaneous rectosigmoid or rectal cancer plus colon cancer, the case was categorized as rectosigmoid or rectal cancer. We extracted cancer stage from the Ontario Cancer Registry. These persons comprise the gFOBT-screen detected and resected CRC cohort, the index date for which is the date of the positive gFOBT result. In order to estimate morbidity and mortality in an unscreened cohort, we identified first ever cases of CRC 2008–2017 at age 50–74, without a history of inflammatory bowel disease, total colectomy, gFOBT at any time on or before the diagnosis date, and without a record of colonoscopy at any time prior to the 6 months before the diagnosis date, among whom we identified major large bowel resection ≤6 months after diagnosis date of colon cancer and ≤12 months after the diagnosis of rectosigmoid plus rectal cancer. The diagnosis date is the index date for the unscreened cohort.
We used the Deyo adaptation of the Charlson score 7 modified for Canadian administrative data, containing International Classification of Diseases version 10 (ICD10) diagnosis codes contained in the Canadian Institute for Health Information (CIHI) Discharge Abstract Database (DAD), to compute five-year cumulative comorbidity prior to the date of the gFOBT. The score was dichotomized as zero versus ≥one. We used Johns Hopkins Ambulatory Care Group software to compute the two-year cumulative Resource Utilization Band (RUB) 8 from the OHIP billing claims CIHI databases. The RUB has six categories, which were collapsed into three, to avoid reporting covariate levels or outcomes with cell counts <6: 0, or non-user, 1–3 (healthy user, plus user with low morbidity, plus user with moderate morbidity) versus 4–5 (user with high or very high morbidity). We identified persons as residing in a rural area or in one of the five urban quintiles of median household income as a surrogate estimate of socio-economic status. We combined the three lowest income quintiles into one category and the two highest income quintiles into another, to avoid cell counts <6.
For both the gFOBT screen-detected cohort and the unscreened cohort, we extracted information about serious post-operative complications and readmissions ≤30 days following resection. From CIHI DAD, we identified the index admission record containing the resection date and all other admissions at any Ontario hospital with admission date ≤30 days following the resection date. Following the algorithm for identifying complications and readmissions within 30 days following major large bowel resection defined by Baxter et al., 9 we extracted its components (CIHI intervention codes, ICD10 diagnosis codes, and OHIP billing claim fee codes) from the CIHI DAD and OHIP billing claims databases. From the RPDB, we identified the date of death for all those who died ≤90 days following the date of major large bowel resection.
Within each cohort, we determined the number of persons with and without ≥1 serious postoperative complication or readmission ≤30 days following major large bowel resection, overall, and by anatomic site of CRC, cancer stage, time period (2008–2012 vs. 2013–2017), and baseline descriptors (age, sex, two-year cumulative RUB, five-year cumulative Charlson score, and socioeconomic status), and obtained p-values from Chi-square tests performed within each variable among the screen-detected cohort and among the unscreened cohort.
We compared those who died ≤30 days following resection to those who did not, by site of CRC, cancer stage, sex, and baseline covariates, separately within each cohort, obtaining p-values from Chi-square tests performed within each variable. We did not build adjusted models of death ≤30 days because of the low numbers of events in the screen-detected cohort. Using the same approach among those who survived 30 days, we compared those who died between 31 and 90 days following resection to those who survived >90 days by location of CRC, sex, and baseline covariates. We did not build an adjusted model of death because of the low numbers of events in the gFOBT screen-detected cohort. All analyses were conducted using SAS version 9.3. In compliance with the Personal Health Information Protection Act legislated in Ontario in 2004, the Institute for Clinical Evaluative Studies (ICES) does not allow us to report cell counts <6.
Results
We identified 172,870 persons aged 50–74 with a first record of positive gFOBT in the CCC gFOBT database between April 2008 and September 2017. We excluded 112 persons whose address was not in Ontario, 683 who had a prior diagnosis of CRC in the Ontario Cancer Registry, 1314 with a prior diagnosis of inflammatory bowel disease, and 91 with a record of prior total colectomy, to define an underlying population of 170,670 persons with a first time positive gFOBT, of whom 127,872 (74.9%) proceeded to colonoscopy ≤6 months following the date of the first positive gFOBT.
CRC was diagnosed and treated with major large bowel resection (Table 1) among 4999/127,872 (3.9%) between 2008 and 2017. Among those with screen-detected and resected CRC, 2397/4999 (48.0%) were aged 50–64, 1906/4999 (47.3%) were women, and 2366/4999 (47.3%) were diagnosed and resected between 2008 and 2012. The unscreened cohort included 8848 resected cases of CRC, 5387/8848 (60.9%) of whom were 50–64 years, 3316/8848 (37.5%) of whom were women, and 4901/8848 (55.4%) were diagnosed and resected between 2008 and 2012. The baseline descriptors of each cohort are presented in Table 2.
Surgical codes for major large bowel resection.

Characteristics of gFOBT-detected and unscreened cohorts.
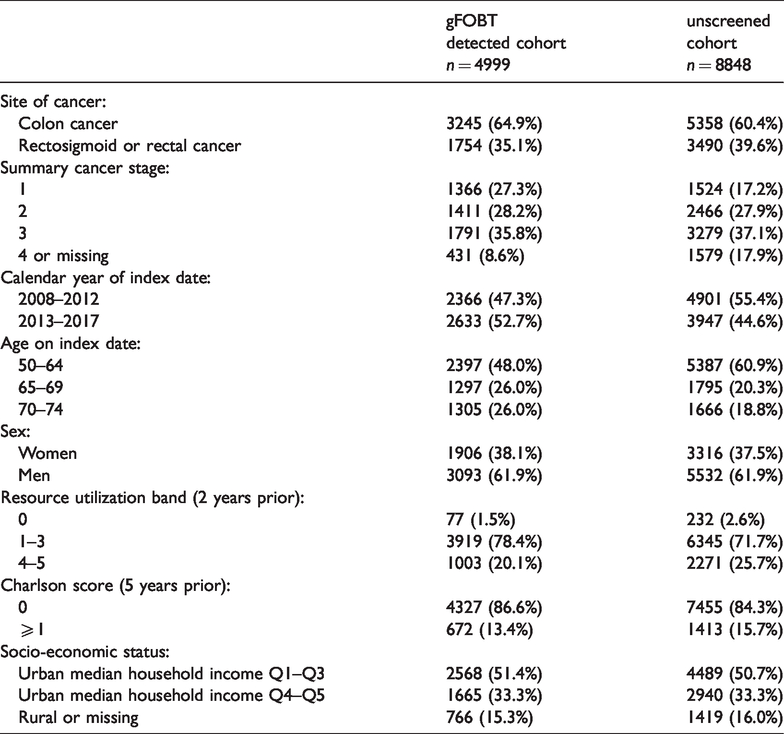
Among the gFOBT screen-detected and resected cohort, at least one post-operative complication or readmission was experienced by 1476/4999 (29.5%) ≤30 days following resection, compared to 3060/8848 (34.6%) among the unscreened cohort. Readmission within 30 days after major large bowel resection occurred among 515/4999 (10.3%) of the screen-detected and 1022/8848 (11.3%) of the unscreened. Major wound disruption (occurrence of diagnosis code for dehiscence or rupture of wound or procedure code for repair of abdominal muscles) was the most common complication among the screen-detected (597/4999, 11.9%) and among the unscreened (1107/8848, 12.5%); followed by reoperation (evidenced by procedure codes for laparotomy for drainage, resection or stoma creation after the index resection) for 325/4999 (6.5%) of the screen-detected and 757/8848 (8.6%) of the unscreened; and infections other than sepsis (evidenced by diagnosis code for intra-abdominal, stitch, subphrenic or wound abscesses, not submitted to percutaneous drainage) for 389/1476 (4.8%) of the screen-detected and 898/3060 (10.1%) of the unscreened. The frequency of other serious postoperative complications ranged from 0.2% to 3.7% among the screen-detected and 0.4% to 4.9% among the unscreened (Table 3). Any individual might have more than one serious complication recorded.
Frequency of readmission and complications ≤30 days after major large bowel resection.
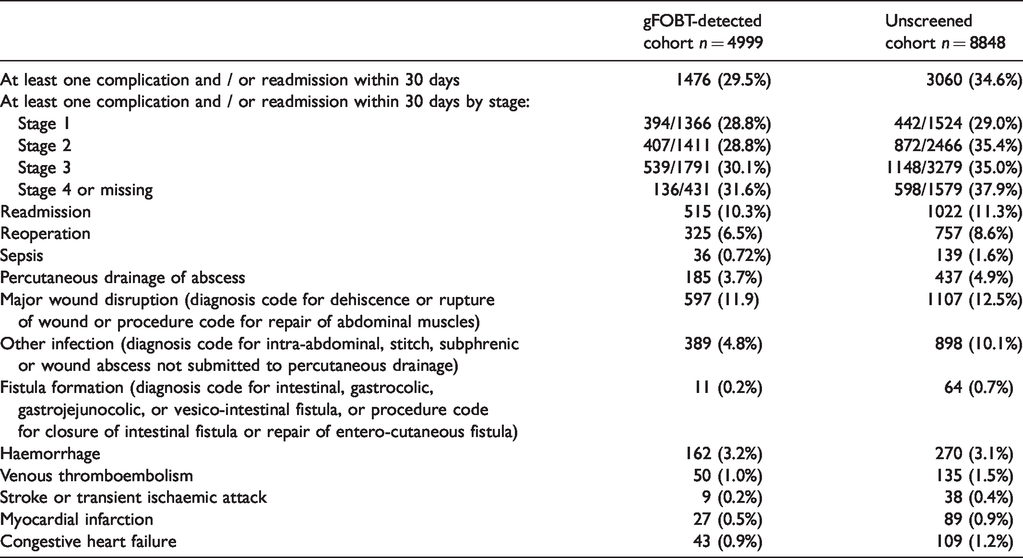
The occurrence of any serious postoperative complication or readmission within 30 days following resection among the screen detected was 1015/3093 (32.8%) among men compared to 461/1906 (24.2%) among women, and among the unscreened, 2032/5532 (36.7%) among men compared to 1028/3316 (31.0%) among women. At least one serious postoperative complication or readmission occurred among 904/3254 (27.8%) of those with screen-detected colon cancer and 572/1754 (32.6%) with screen-detected rectosigmoid or rectal cancer, compared to 1800/5383 (33.6%) and 1260/3490 (36.1%) respectively among the unscreened. Among those with baseline five-year cumulative Charlson comorbidity score of zero, 1240/4327 (28.7%) screen-detected and 2472/7455 (33.2%) unscreened experienced one or more serious postoperative complications or readmissions, compared to 236/672 (35.1%) screen-detected and 488/1413 (41.6%) unscreened with baseline score ≥1. Table 4 presents p-values derived from Chi-square tests comparing those with and without serious complications and/or readmission for each variable within each cohort. In both cohorts, p-values were <0.05 for RUB and Charlson score, and among the unscreened, for age and stage.
Complications and/or readmission ≤30 days after major large bowel resection by baseline characteristics, with standardized differences.
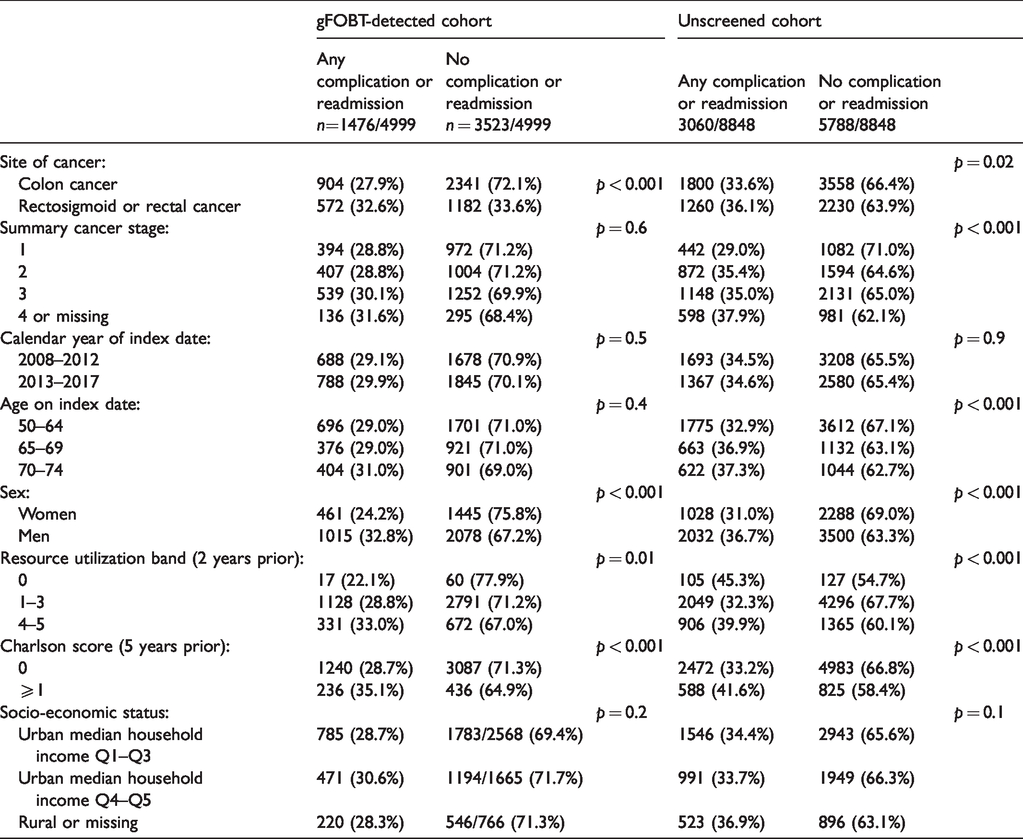
Death within 30 days following resection was the outcome for 43/4999 (0.9%) screen-detected and 208/8848 (2.4%) unscreened. Table 5 presents p-values derived from Chi-square tests comparing those who did and those who did not die within 30 days following resection, for each variable within each cohort. P-values were >0.05 for all variables in the screen-detected cohort, but <0.05 for age, RUB, Charlson score, cancer site, and stage in the unscreened. We did not build an adjusted model of death ≤30 days because of the small number of deaths in the screen-detected cohort.
Mortality within 30 days after major large bowel resection by baseline characteristics, with standardized differences.
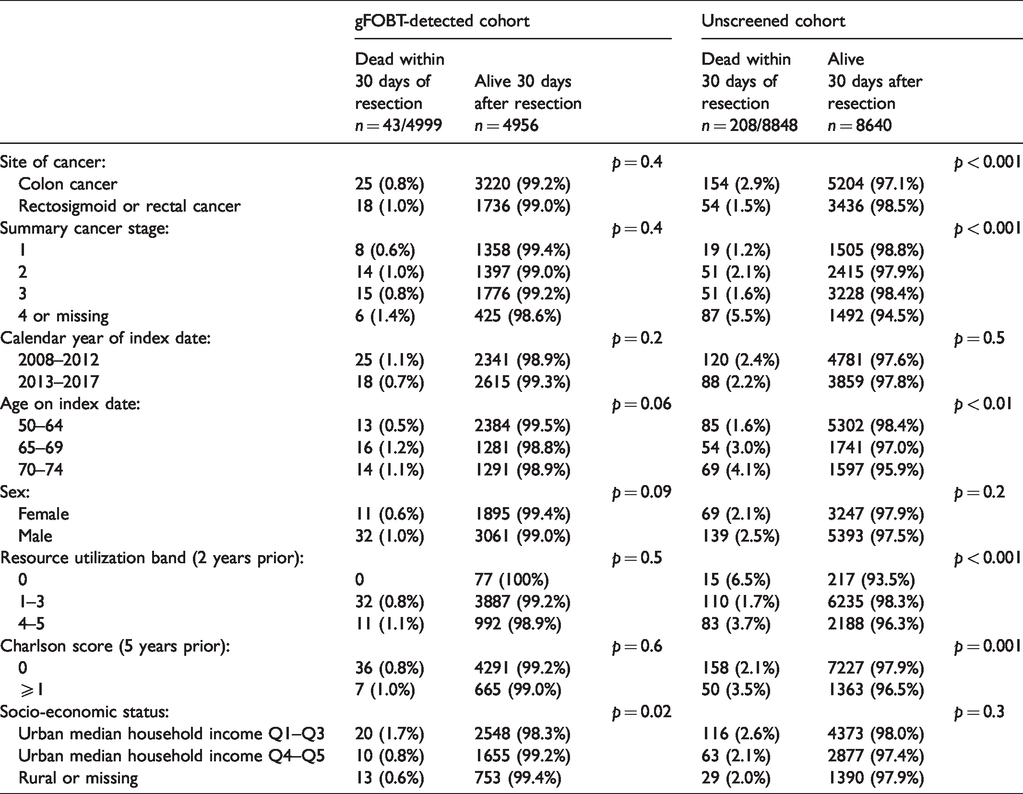
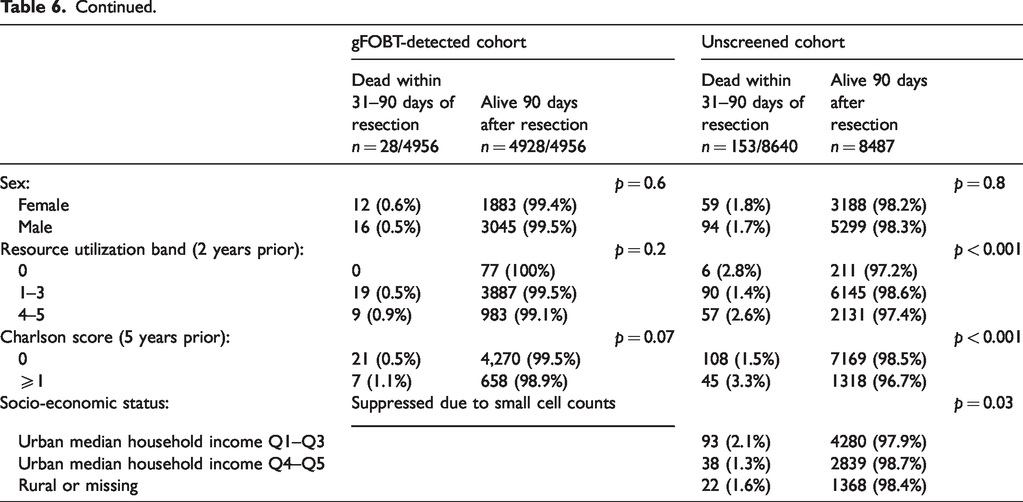
Among those surviving 30 days, 28/4956 (0.6%) screened and 111/8640 (1.3%) unscreened died between 31 and 90 days following major large bowel resection. Table 6 presents p-values derived from Chi-square tests comparing those who did and those who did not die 31–90 days following resection, for each variable within each cohort. In both cohorts, p values were <0.05 for age and Charlson score, and for stage and RUB among the unscreened. Among the screen-detected we could not report mortality stratified by socioeconomic status and cancer stage because of the presence of cell counts <6.
Mortality 31–90 days after major large bowel resection by baseline characteristics, with standardized differences.
Overall, there is little difference in mortality between 0 and 90 days following major large bowel resection between the period 2008 and 2012 (34/2,366 or 1.4%) and the period 2013 and 2017 (37/2,633 or 1.4%) among the screen-detected; among the unscreened, mortality between 0 and 90 days during the two periods was 214/4901 (4.4%) and 147/3,947 (3.7%) respectively.
Discussion
This description of serious post-operative morbidity and mortality following major large bowel resection for CRC detected by a population-based colorectal screening program provides important information not generally available from population-based colorectal screening programs. Although we have identified substantial major postoperative morbidity, it appears to be somewhat less among screen-detected resected cases than among the unscreened resected cases observed from the same underlying population. Mortality 0–30 days and 31–90 days postoperatively appears to be somewhat lower among the screen-detected. While these observations provide some reassurance, they also signify that there have been rare but major risks associated with the program. The frequency of major postoperative morbidity and readmission among screened and unscreened resected cases serves as an incentive for future research and quality improvement effort aimed at its reduction.
This work was able to draw on complete population-based health services utilization databases and the cancer registry. A major strength is the CIHI DAD database, from which most of the diagnostic and procedure codes used to ascertain major surgical complications were derived; these codes have been assigned at each hospital by well trained and experienced health records technologists following the explicit instructions for coding diagnoses according to the International Classification of Diseases, version 10, and by the rules governing the coding of procedures using the Canadian Classification of Interventions. Nevertheless, there is a possibility of missing codes or records, or miscoding.
We identified one published report comparing post-operative complications following resection in 80 patients with CRC detected by a screening program and 106 patients not screen detected, who were between the ages of 50–69. Complications occurred among 38.8% of screen-detected versus 63.2% of those who were not screen-detected. 10
Contemporary jurisdiction-wide reporting of postoperative serious complications, readmission and mortality following major large bowel resection for CRC does not distinguish resections for screen-detected CRC from CRC detected by symptomatic presentation, and generally include cases younger and older than the screening-eligible age range. Definitions of outcomes vary among reports, probably reflecting the variability in the recording and coding of complications among institutions and jurisdictions, as well as institutionally or jurisdictionally preferred definitions of post-operative mortality versus international consensus definitions. Many reports are structured around comparison of cases treated at institutions categorized by volume or by geographic setting, and present a range of frequencies of serious postoperative complications rather than overall frequency.
Aquina et al. 11 reported major post-operative complications after resection of colon cancer between 2004 and 2011 stratified by age: among those <65 years of age these occurred in 1852/8176 (22.6%) and among those aged 65–74 in 1798/6252 (28.8%). In our study, 904/3245 (27.9%) screened and 1800/5383 (33.6%) unscreened cases aged 52–74 experienced a serious post-operative complication after colon cancer resection. A US study of colon cancer resection in 2011 12 reported a slightly lower range of serious postoperative complications (19.4% in urban populations and 24.9% in rural populations) among all cases undergoing resection at age ≥18 years; however, among US colorectal cancer resections in 2014–2015, serious complications were reported in 21.3% to 33.9%, depending on hospital volume, of all resections among those aged ≥18. 13
Reports of postoperative mortality following major large bowel resection for CRC in the contemporary era are characterized by substantial variability in terminology, which causes difficulties in making comparisons between reports, or between our study and other reports. Some of the reports cited above define 30-day and/or 90-day mortality, while others define a composite measure comprised of 30-day mortality plus in-hospital mortality occurring at any time during the index admission regardless of length of stay prior to death, or in-hospital mortality only without any dimension of time since resection. In our study of screen-detected CRC treated by major large bowel resection, postoperative mortality is at a relatively low level throughout the observation period, lower than generally reported in most other contemporary studies, and lower than among our contemporaneous unscreened cohort derived from the same underlying population. It is unclear if the low postoperative mortality among the screened is related to unmeasured tumour-related factors which are less favourably distributed in an overall population which includes many symptomatically detected cases, or due to unmeasured health factors other than age which are also more favourably distributed in a screen-detected population compared to those with CRC not detected by screening.
Guida et al. 14 reported 30-day mortality in 2013–2014 across Italy as 4.07% for colon cancer and 1.95% for rectal cancer, among cases aged between 18 and 100, compared to our study which determined 30-day mortality to be 25/3245 (0.8%) for screen-detected cases of colon cancer and 154/5358 (2.9%) for the unscreened, among those 50–74 years, and 18/1754 (1.0%) for screen-detected and 54/3490 (1.5%) for unscreened cases of rectosigmoid and rectal cancer. Among three recent studies from the Netherlands, two reported higher mortality than our study: the first reported 30-day mortality for colon cancer as 3.9%–4.7% and rectal cancer 2.3%–3.4%, 15 and the second 16 reported a composite of 30 day plus inpatient mortality of 3.3% observed between 2011 and 2014 in a population among whom 4544/23593 (19.3%) were ≥81 years old. 16 However, de Neree 17 reported the composite 30 day plus inpatient mortality in 2015–2016 as 1.8% for colon cancer and 1.0% for rectal cancer, similar to screen-detected cases in our study. Diers et al. 18 reported nationwide postoperative inpatient mortality between 4.8% and 6.9% in Germany in 2012–2015; 59,515/129,196 (46.1%) of cases were aged ≥75. However, among the 57,948/129,196 (44.9%) aged 55–74, the mortality was 3.2%. One report from France covering the years 2010–2014 reported 3.8% 30 day postoperative mortality after colon resection among all cases ≥18 years old, 19 and for the years 2012–2017, 90 day post-operative mortality of 5.7% after colon resection and 3.1% after rectal resection, in a population of whom 35,223/113,283 (31.1%) were aged ≥80. 20 From the US National In-Patient Database, in-hospital mortality in 2011 was reported as 2.9% to 3.1%, and during 2014 to 2015 as 2.2% to 4.5%, among all cases aged ≥18. 12
Conclusion
There is wide variation in the definitions of postoperative complications and mortality following major large bowel resection for CRC. We have described serious postoperative complications, readmissions, and mortality in a resected CRC cohort gFOBT-screen detected by the population-based colorectal screening program in Ontario, Canada, and in an unscreened contemporaneous cohort derived from the same underlying population. Complications, readmissions, and mortality may be more common following major large bowel resection for CRC between the ages of 50 and 74 years among unscreened compared to screen-detected cases.
Footnotes
Acknowledgements
This study was supported by ICES, which is funded by an annual grant from the Ontario Ministry of Health and Long-Term Care (MOHLTC). The opinions, results and conclusions reported in this paper are those of the authors and are independent from the funding sources. No endorsement by ICES or the Ontario MOHLTC is intended or should be inferred. Datasets were linked using unique encoded identifiers and analyzed at ICES from the Ministry of Health and Long Term Care of Ontario (RPDB and OHIP), and Cancer Care Ontario (OCR). The opinions, results, views, and conclusions reported in this paper are those of the authors and do not necessarily reflect those of the ICES, the Ministry of Health and Long Term Care of Ontario, or Cancer Care Ontario, and no endorsement by these bodies is intended or should be inferred.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: At the time of data analysis, Rinku Sutradhar, Nancy Baxter, Jill Tinmouth, and Linda Rabeneck were employees of Cancer Care Ontario, which operates ColonCancerCheck. Lawrence Paszat received an operating grant from Cancer Care Ontario to conduct this work. Jin Luo has no declaration
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was funded by Cancer Care Ontario. The funder had no role in the design of the study or collection, analysis and interpretation of the data or in writing the manuscript.
