Abstract
Objectives
Faecal immunochemical test accuracy may be adversely affected when samples are exposed to high temperatures. This study evaluated the effect of two sample collection buffer formulations (OC-Sensor, Eiken) and storage temperatures on faecal haemoglobin readings.
Methods
Faecal immunochemical test samples returned in a screening programme and with ≥10 µg Hb/g faeces in either the original or new formulation haemoglobin stabilizing buffer were stored in the freezer, refrigerator, or at room temperature (22℃–24℃), and reanalysed after 1–14 days. Samples in the new buffer were also reanalysed after storage at 35℃ and 50℃. Results were expressed as percentage of the initial concentration, and the number of days that levels were maintained to at least 80% was calculated.
Results
Haemoglobin concentrations were maintained above 80% of their initial concentration with both freezer and refrigerator storage, regardless of buffer formulation or storage duration. Stability at room temperature was significantly better in the new buffer, with haemoglobin remaining above 80% for 20 days compared with six days in the original buffer. Storage at 35℃ or 50℃ in the new buffer maintained haemoglobin above 80% for eight and two days, respectively.
Conclusion
The new formulation buffer has enhanced haemoglobin stabilizing properties when samples are exposed to temperatures greater than 22℃.
Keywords
Introduction
Early detection of colorectal cancer (CRC), or its precursor stage of adenoma, can reduce mortality from CRC. Screening using faecal immunochemical tests (FIT) for haemoglobin, where a positive test result triggers colonoscopic investigation, can decrease incidence of CRC, 1 allow detection at an earlier stage, 2 and reduce mortality. 1 The relatively recent replacement of guaiac faecal occult blood testing (gFOBT) with FIT has improved both cancer and adenoma detection. 3 Use of FIT circumvents many of the disadvantages of the gFOBT, including the risk of false positive results from upper gastrointestinal bleeding, from dietary intake of red meat, or from fruits and vegetables that contain peroxidases. 4 False-positive test results can lead to unnecessary colonoscopy, which increases risk and cost of screening programmes. 3 The introduction of FIT, which is specific for the globin moiety of human haemoglobin, has reduced, but not eliminated, the false positive rate for neoplasia, as benign conditions can also bleed. Another limitation of FIT may be the risk of false negatives through prolonged or inadequate sample storage, as the globin chains in haemoglobin molecules degrade more rapidly than haem. 5 These factors may contribute to the range of positivity rates reported between different studies and different countries. 6
In liquid-based FIT technology, potential exists for haemoglobin to be partially or completely degraded by faecal proteases or bacterial processes, or for epitopes required for antibody recognition to be altered over time. Degradation of haemoglobin may be accelerated through exposure to higher ambient temperature, which may explain reports that FITs completed in the summer months have a lower positivity rate compared with those completed in winter.5–8 While FIT participants are advised to keep their completed samples cool or refrigerated until they are returned to the testing laboratory, these storage recommendations are not necessarily adhered to, and we have previously found that only 62% of participants indicate storage of samples in the refrigerator. 6 In many countries, including Australia, samples are posted back to the laboratory, so may be exposed to elevated temperatures during transit. One FIT manufacturer has recognized this problem and has devised a new sample buffer formulation to improve haemoglobin stability at higher ambient temperatures. The aim of the study was to evaluate the effectiveness of the new Eiken OC-Sensor FIT buffer formulation to maintain faecal haemoglobin stability over a range of faecal sample storage temperatures and times.
Methods
Through the Bowel Health Service (Repatriation General Hospital, Daw Park, South Australia), participants in a personalized screening programme 9 were sent a FIT kit. Each kit contained two identical sample collection tubes (OC-Sensor, Eiken Chemical Co. Ltd, Tokyo, Japan), both with either the original formulation or the new formulation sample buffer. Participants collected samples from two different bowel motions, avoiding contamination with urine. Dietary or medicine restrictions were not requested. Participants were instructed to keep the kit in a cool place, and mail it back to the laboratory within 14 days of the first collection.
OC-Sensor is a quantitative FIT, with chemistry based on human haemoglobin antibody-mediated latex agglutination. The sample collection device is a grooved probe that holds approximately 10 mg of faeces collected from the outer surface of the faeces, and contains 2 mL of haemoglobin stabilization buffer. Faecal samples in buffer were tested for the presence of haemoglobin using the automated analyzer (OC-Sensor DIANA, Eiken Chemical Co. Ltd). Prior to analysis, the auto-analyzer was calibrated with a standard curve of known haemoglobin concentration, and low- and high-haemoglobin concentration quality control samples were run with each batch to ensure machine accuracy (as per manufacturer instructions).
Only a small fraction of the 2 mL faecal sample in buffer is required for haemoglobin determination using OC-Sensor. Following initial faecal haemoglobin determination, returned sample collection tubes with a haemoglobin concentration ≥10 µg/g faeces (50 ng/mL buffer) were re-sealed with parafilm and stored at room temperature (22℃–24℃), in the refrigerator (1℃–6℃), or in the freezer (−20℃). Samples were re-analysed once following 1–14 days in storage, with a minimum of five different samples analysed at each time point (total samples analysed: freezer original buffer = 76, new buffer = 71; refrigerator original buffer = 95, new buffer = 71; room temperature original buffer = 95, new buffer = 71). Haemoglobin concentrations were expressed as percentage of original concentrations. Due to the findings, additional storage conditions were performed on samples in new formulation buffer, where samples were stored for up to 10 days at 35℃ or 50℃ in a dry oven (total samples analysed: 35℃ = 59, 50℃ = 56). FIT collection tubes were identified as containing the original or new buffer based on tube batch numbers. Collection and analysis of faecal samples within the original buffer formulation was conducted between May 2013 and April 2014, while samples within the new buffer formulation were analysed between February and December 2015.
All procedures were approved by the Southern Adelaide Clinical Human Research Ethics Committee, with all participants providing informed written consent (approval number 422.13).
Statistical analysis
Data from the in vitro study were modelled with univariate linear regression analysis, to examine the effect of time (days) on the haemoglobin percentage of original concentration. This allowed the determination of the number of days until the haemoglobin concentration dropped to less than 80% of the original value. While there appears to be no international standard for acceptable haemoglobin measurement levels with FITs, other in vitro device standards specify that results must be within ± 20% of laboratory values (ISO: 15197:2013), which is why 80% was chosen as the lower limit of acceptable variability. Haemoglobin concentrations recorded throughout the screening programme between the different buffer formulations were compared with a Mann–Whitney U Test, and screening programme characteristics were compared with Chi-squared tests. Haemoglobin concentration changes with different storage temperatures and durations between the different buffers were compared with two-way ANOVAs. Statistical analyses were carried out using Stata (version 13, StataCorp). A p value of less than 0.05 was considered statistically significant.
Results
Participant and test characteristics associated with samples received from the personalized screening programme during the assessed collection period.
Sample storage denominator was for the participants who had recorded this variable with return of samples.
Analysis was performed with either Mann–Whitney U Test or a Chi-squared test.
Duration of collection for the original formulation faecal immunochemical test (FIT) sample tubes was between May 2013 and April 2014. Duration of collection for the new formulation FIT sample tubes was between February 2015 and December 2015.
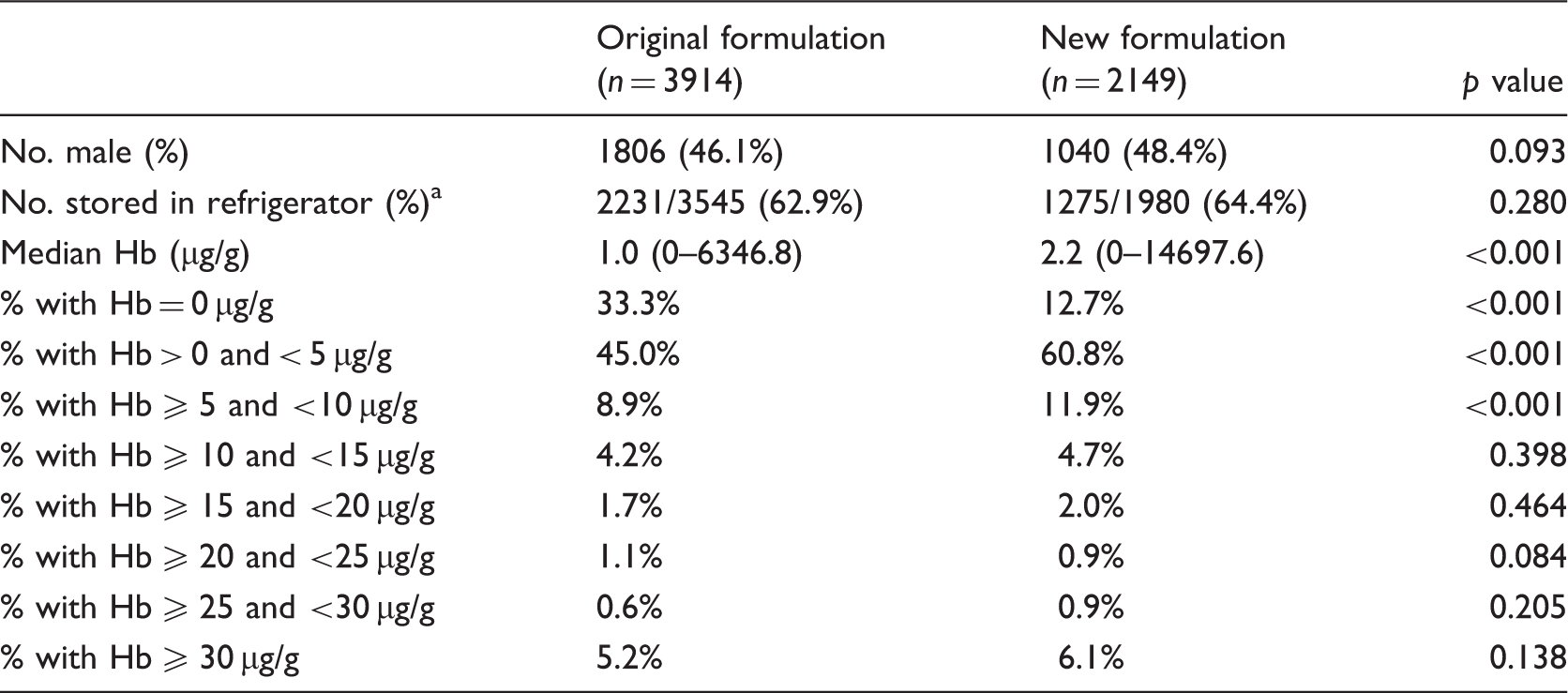
Storage in the freezer with both the original and new buffer caused an initial decrease in haemoglobin concentration which then remained constant over time (Figure 1(a), p = 0.916). There were no differences between haemoglobin stability at −20℃ when comparing the two sample buffers (p = 0.983), with an average decrease in the haemoglobin concentration of 12.0% with the original buffer, and 12.2% with the new buffer.
In vitro assessment of haemoglobin concentrations following storage of OC-Sensor FIT sample tubes in the freezer (a), refrigerator (b) and at room temperature (c) for up to 14 days, with two different faecal immunochemical test sample buffer formulations. Data are means ± standard error and expressed as a percentage of the original haemoglobin concentration. A minimum of five samples with original haemoglobin ≥ 10 µg/g faeces have been measured at each time point (total samples analysed: freezer original buffer = 76, new buffer = 71; refrigerator original buffer = 95, new buffer = 71; room temperature original buffer = 95, new buffer = 71). The solid squares represent the original formulation buffer; the open triangles represent the new formulation buffer. The solid horizontal bar shows the original haemoglobin concentration. *p < 0.05 when comparing the two buffer formulations.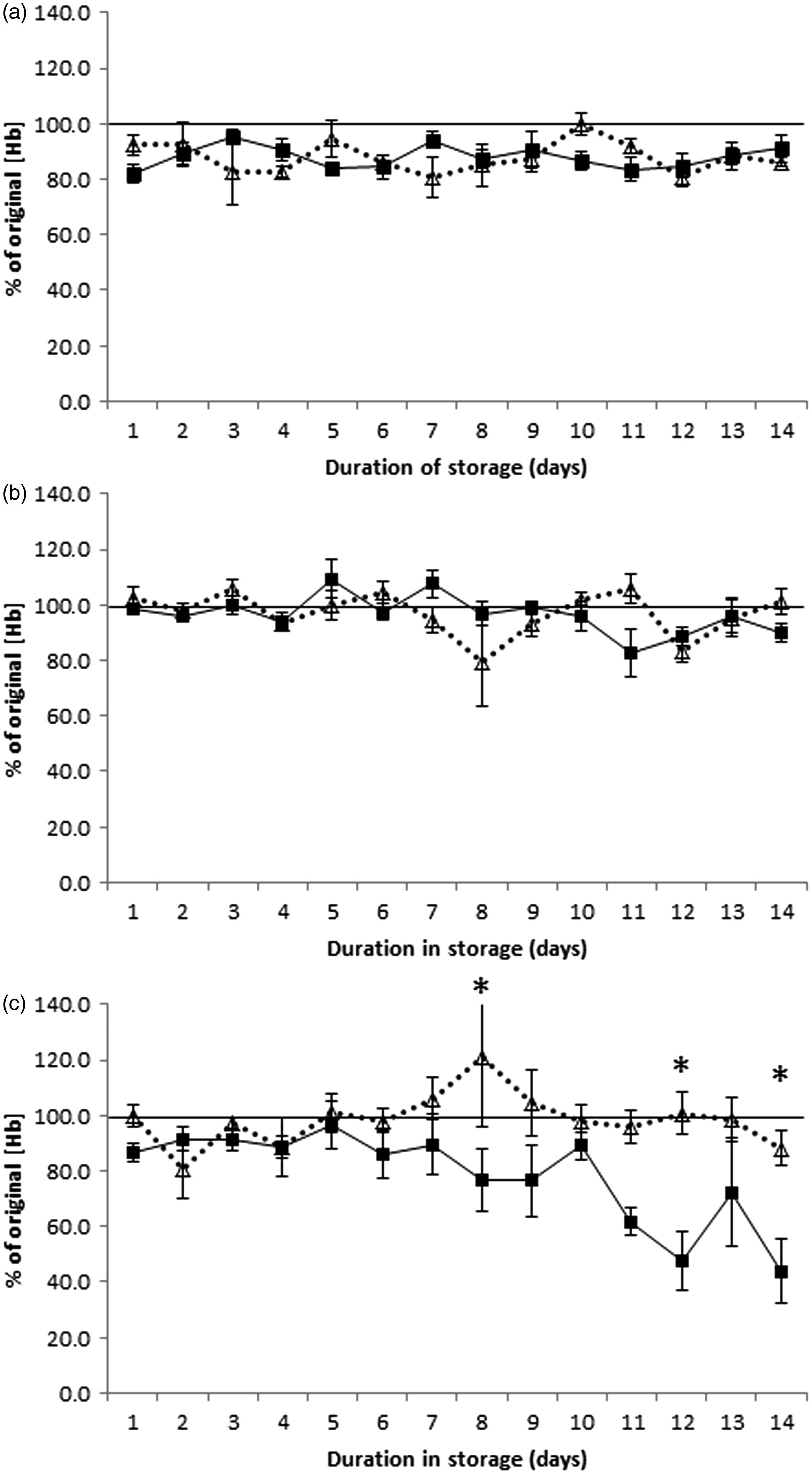
Within the first 14 days of refrigerator storage, haemoglobin concentration changes were not significantly different between the buffers (Figure 1(b), p = 0.826), or affected by storage duration (p = 0.070). Linear regression analysis indicated that haemoglobin concentration would drop below 80% after 36 days in the refrigerator with the original buffer, and after 106 days with the new buffer.
Room temperature storage of samples within the original buffer was associated with a 4.0% loss per day of haemoglobin, with measurement levels dropping to less than 80% of the original haemoglobin concentration after six days of storage. The new buffer significantly improved stability (p < 0.001), with a 0.8% per day decrease in measured haemoglobin concentrations, and the maintenance of concentration above 80% of the original level for 20 days (Figure 1(c)).
Linear regression analysis indicated that storage at 35℃ with the new formulation sample buffer would retain haemoglobin concentrations at more than 80% of the original value for eight days (Figure 2(a)). Storage at 50℃ produced a biphasic response, with haemoglobin concentration decreasing to less than 80% of the original value after two days, but then increasing above original concentrations at day 5, and remaining elevated for a further two days (Figure 2(b)).
In vitro assessment of haemoglobin concentrations following storage of OC-Sensor FIT sample tubes at 35℃ (a) and 50℃ (b) for up to 10 days, with the new sample buffer formulation. Data are means ± standard error and expressed as a percentage of the original haemoglobin concentration. A minimum of five samples with original haemoglobin ≥ 10 µg/g faeces have been measured at each time point (total samples analysed: 35℃ = 59, 50℃ = 56). The solid horizontal bar shows the original haemoglobin concentration.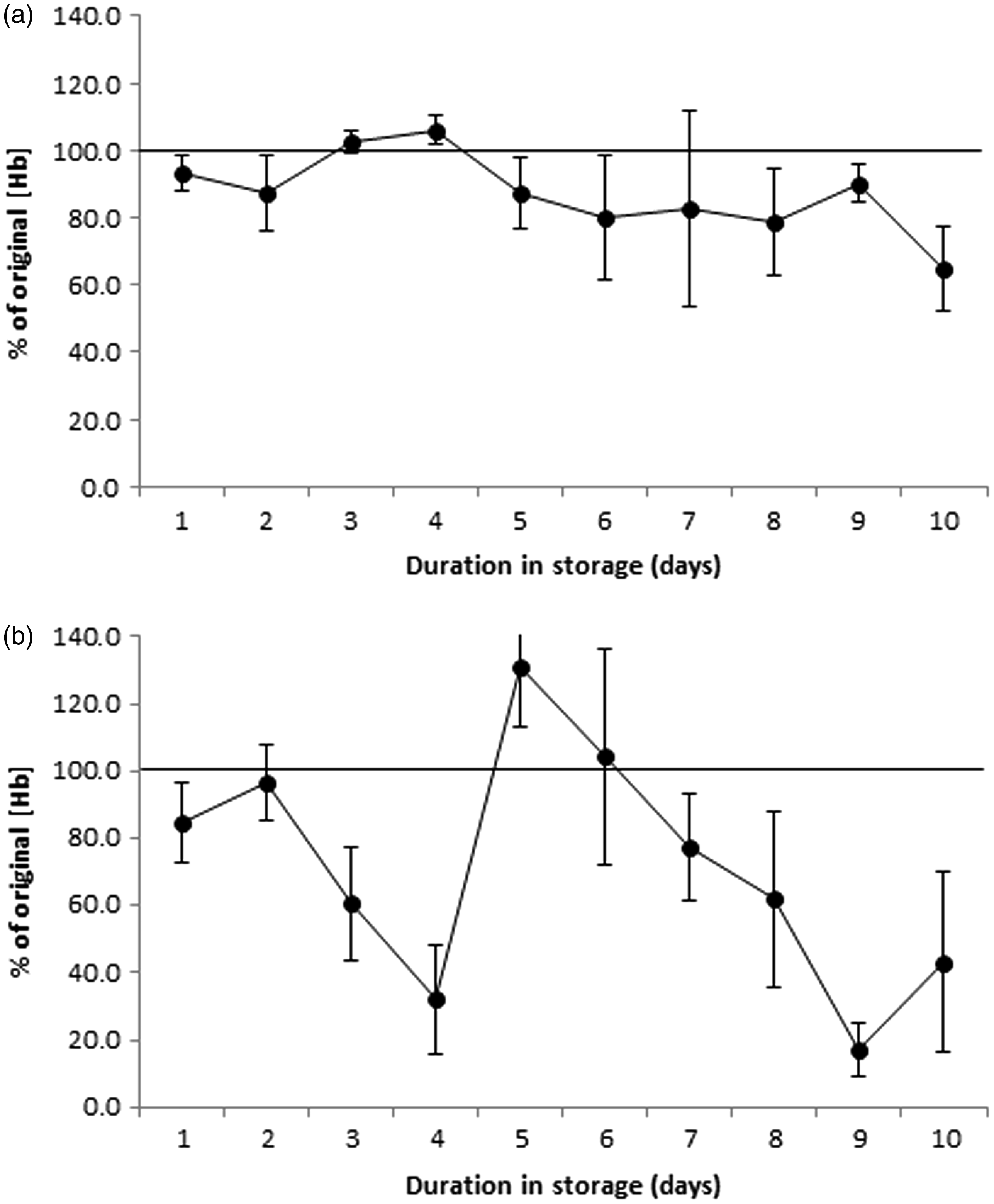
Discussion
This study assessed the effect of storage temperatures on faecal haemoglobin stability in different OC-Sensor FIT sample buffers and over time and found that the haemoglobin concentration in faecal samples stored within the refrigerator or freezer is stable for 14 days irrespective of buffer formulation. Our results also showed that at room temperature storage, there is reduced haemoglobin detection with the original OC-Sensor buffer after six days, but with the new buffer, there was no significant loss of detection up to three weeks, and at 35℃, the new buffer facilitated reasonable detection to five days. These findings have major implications on storage for FIT-based screening programmes, especially in countries such as Australia where FIT samples may be exposed to high day time temperatures during transport to the testing laboratory.
Our results suggest that the best short-term storage option is in a refrigerator. When FITs were stored in a refrigerator, haemoglobin concentrations were comparable with fresh samples for over two weeks. The new formulation buffer may maintain haemoglobin above 80% of the original concentration for 106 days. This storage suggestion, however, has limited acceptability, and previous experience as well as the current study has shown that only two-thirds of participants will comply with this recommendation. 6 Sample tube storage in double zip-lock bags has been reported to increase the rate of refrigerator storage, 10 but other storage options are needed.
Few in vitro studies have been performed to assess the stability of FITs with liquid buffers at lower storage temperatures. One report indicated that haemoglobin concentrations could decrease to 67% of the initial concentration after being frozen. 11 We found that following one freeze–thaw cycle, OC-Sensor FITs decreased to an average of 88% of original haemoglobin concentrations, regardless of whether the original or new buffer was used. This drop in haemoglobin did not change with storage time and was found to be retained up to four weeks of storage (data not shown). Several research centres reportedly freeze FIT samples prior to analysis,12,13 which will result in lower haemoglobin concentrations compared with unfrozen samples. Our results suggest that analytical centres could adjust their FIT positivity threshold to allow for this reduction in haemoglobin detection.
Several studies have shown that temperatures above 20℃ decrease FIT haemoglobin concentrations over time,10,14 but this has mostly been assessed in vitro, for example, by spiking stool samples with blood lysate. 14 Our methods more accurately represent the conditions encountered by samples collected in screening programmes. After seven days at room temperature in the original FIT buffer formulation, haemoglobin concentration dropped to less than 80% of initial value, equivalent to a daily loss of haemoglobin of 4% per day, and consistent with previous reports.5,10,14 The new formulation buffer had superior haemoglobin stability compared with the original buffer, with more than 80% of the haemoglobin concentration retained after 20 days. At higher temperatures, faecal haemoglobin in the new buffer formulation appeared to be relatively stable for up to eight days at 35℃, and 50℃ for two days. Earlier work on quantitative and qualitative FITs and gFOBT showed that only gFOBT using dried faecal samples was able to detect faecal haemoglobin at 45℃. 11 Our data suggest that the new OC-Sensor buffer is an improvement over the original buffer, but that further development is required if test accuracy is to be retained at higher ambient temperatures, or when there is a prolonged time between sample collection and testing.
As a reduction in haemoglobin concentration due to heat exposure could lead to false negative test results, it is important to assess the impact this could have on screening programme accuracy. Previous studies have found that FIT samples with a haemoglobin concentration just above a set positivity threshold can fall below that threshold following heat exposure. 10 It has also been shown that when sample buffers did not provide an adequate environment for haemoglobin stability, 30% of samples became negative by two weeks after collection, 5 although this is not likely to affect programme sensitivity for CRC as these lesions bleed in excess of test positivity thresholds. 15 Heat exposure of 50℃ may lead to false positives for some samples, as we observed an increase in haemoglobin concentration to 131% at day 5 of heat exposure. The reason for this temporary increase is not clear. Many countries use 20 µg/g faeces as the FIT threshold for positivity. 16 Samples with an original concentration just below this threshold (15–19 µg/g faeces) may therefore become positive with high heat exposure. Within our study population only 2% of samples returned were within this concentration range, and therefore, the impact of heat-induced false positives on a screening programme will be small. In addition, it has been proposed that haemoglobin concentrations that are just below the positive threshold are likely to increase and become positive in the next screening round. 17 Any heat-activated increase in haemoglobin would result in earlier detection of the growing bleeding lesion.
A limitation of this study was the uncontrolled and likely differing storage conditions of the faecal samples prior to the first analysis. This means that some samples may have already changed between collection and measurement. However, as this variable was common between the two arms of the study, and samples were randomly allocated to temperature storage group, comparison of haemoglobin measurements using the original and new formulation buffers remains valid. In addition, as this study population was a higher risk population enrolled in a personalized screening programme, it is possible that the range of initial haemoglobin concentrations will be different from that observed in an average risk population.
Conclusion
The new formulation buffer for OC-Sensor appears to have superior haemoglobin stabilizing components compared with the original buffer. This should improve programme sensitivity for countries that have elevated summer temperatures, and where samples are mailed through the postal system. Based on our study, the best short-term storage condition for OC-Sensor is refrigeration at 4℃. It is important to emphasize the importance of sample storage conditions to screening programme participants, and to laboratories.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Parts of this research were supported by a project grant from NHMRC Australia (APP1006242). Eiken Chemical Company (Tokyo, Japan) provided all OC-Sensor faecal immunochemical tests and reagents, but they were not involved with the design of the study or the analysis of the results.
