Abstract
Objective
To assess the impact of ambient temperature on faecal immunochemical test (FIT) performance in the colorectal cancer screening programme of Emilia-Romagna (Italy).
Methods
A population-based retrospective cohort study on data from 2005 to 2011. Positive rate, detection rate, and positive predictive value rate for cancers and adenomas, and incidence rate of interval cancers after negative tests were analysed using Poisson regression models. In addition to ambient temperature, gender, age, screening history, and Local Health Unit were also considered.
Results
In 1,521,819 tests analysed, the probability of a positive result decreased linearly with increasing temperature. Point estimates and 95% Confidence Intervals were estimated for six temperature classes (<5, 5 |−10, 10 |−15, 15 |−20, 20|−25 and ≥25℃), and referred to the 5|-10℃ class. The positive rate ratio was significantly related to temperature increase: 0.99 (0.97–1.02), 1, 0.98 (0.96–1.00), 0.96 (0.94–0.99), 0.93 (0.91–0.96), 0.92 (0.89–0.95). A linear trend was also evident for advanced adenoma detection rate ratio: 1.00 (0.96–1.04), 1, 0.98 (0.93–1.02), 0.96 (0.92–1.00), 0.92 (0.88–0.96), 0.94 (0.88–1.01). The effect was less linear, but still important, for cancer detection rates: 0.95 (0.85–1.06), 1, 1.00 (0.90–1.10), 0.94 (0.85–1.05), 0.81 (0.72–0.92), 0.93 (0.80–1.09). No association or linear trend was found for positive predictive values or risk of interval cancer, despite an excess of +16% in the highest temperature class for interval cancer.
Conclusions
Ambient temperatures can affect screening performance. Continued monitoring is needed to verify the effect of introducing FIT tubes with a new buffer, which should guarantee a higher stability of haemoglobin.
Introduction
Faecal immunochemical testing (FIT) is the preferred initial test for many colorectal cancer screening programmes because of its many advantages over the guaiac test (gFOBT), including the possibility of avoiding dietary restriction, use of a single stool sample, a higher sensitivity and specificity, 1 and the availability of a quantitative result. FIT may, however, suffer from stability problems related to the possible denaturation of the globin, due to the time elapsed between collection and analysis of the sample, and/or exposure to temperatures that do not comply with manufacturer recommendations.2–5 Previous studies have assessed the potential effects of ambient temperature on the performance of FIT in population-based screening programmes by correlating screening indicators with the season of testing. Grazzini and colleagues 6 in Italy found decreasing average haemoglobin levels and positive test rates with increasing temperature, as well as lower detection rates of cancers and advanced adenomas during the summer compared with winter. In a similar study in Holland, Van Rossum 7 found no significant difference in the positivity rates across the seasons, but observed temperature-related detection rates for cancer and advanced adenomas with lower values in winter than in summer. Grazzini and colleagues 8 suggest that the differences between the two studies could be attributed to the significantly lower average temperature in the Netherlands, and that the relationship between ambient temperature and the degradation of the globin might have a non-linear relationship, with manifest effects even at low temperatures. Zorzi and colleagues 9 in a study of five screening centres in Italy found a higher incidence of interval cancers after tests that had been carried out in the summer compared with those performed in the winter. We investigated the influence of ambient temperature on the main early-impact indicators in the screening programme of Emilia-Romagna, a populous region of northern Italy (4,471,104 residents in 2012) with a sub-continental climate, significant annual temperature ranges, hot summers, and severe winters.
Methods
Setting
Since 2005, the Emilia-Romagna Department of Health Policy has coordinated local screening programmes through the 10 Local Health Units (LHU). These programmes cover every stage of screening, diagnosis, and treatment, including possible surgery and follow-up. 10 The eligible population aged 50–69 is asked by post every two years to perform a free, single sample test for occult blood in the faeces. In one LHU, the test kit is sent with the invitation letter. Test kits are returned to the laboratories through collection centres, health centres, and pharmacies, by dedicated courier delivery. Screening participants are requested to keep the sample collected in the device at 4–8℃ if it is not returned within the day, and to return it within two days. The LHUs actively maintain short schedules for sample collection, storage, and analysis. The first level test is performed by OC Sensor (Eiken Chemical, Tokyo, Japan), with a positive cut-off of 100 ng/ml buffer (equal to 20 µg haemoglobin per g faeces). Each local programme has adopted the same methodology, instrumentation, and Internal and External Quality Controls, to allow comparison of the outcomes from the entire region. At the regional level, data are collected in a standardised way, according to a record layout agreed with the National Monitoring Center for screening programmes (Osservatorio Nazionale Screening).
Ambient temperature estimates
The ambient temperature at the subjects’ municipalities was considered in our study. The Regional Agency for Prevention and Environment of Emilia-Romagna provided estimates of the average daily temperatures for each municipality in the region except, for technical reasons, for the eight municipalities of Rimini province. They used the software PRAGUE 11 to reconstruct the values of the main meteorological variables on a regular grid with cells of 5 km2. The meteorological data used for the interpolation were obtained from the Rirer network of real-time observation of Emilia-Romagna, consisting of approximately 100 weather stations in thermometric telemetry. As the time span between the collection, storage and analysis of the test was not available, a previous analysis carried out in the Modena LHU was utilised. The study indicates that sample analysis usually takes place within 48 hours of stool collection. Therefore, we attributed to each test an ambient temperature calculated as the average value of the temperatures estimated in the municipality of residence on the day of the FIT analysis and on the previous day. For each person involved in the study, the municipality of residence was considered as fixed over the whole study period.
Design
We conducted a retrospective population cohort study based on the results of screening tests carried out in the region in the period between 2005 and 2011. The outcomes investigated were the proportion of positive tests, the detection rate, and the positive predictive values for cancer, advanced adenoma, and early adenoma. For each positive FIT, only the worst lesion found by colonoscopy or surgery was considered. The detection rate, expressed as the proportion of participants with a specific lesion, was calculated according to European and Italian guidelines.3,12 To account for different levels of adherence to colonoscopy, we subtracted positive tests without subsequent colonoscopy from the total number of tests performed. Advanced adenoma was defined as an adenoma with size of 10 mm or more, or with at least 20% of villous component, or with high-grade dysplasia.3,12 An adenoma was classified as early if it showed no characteristics of advanced adenoma.
We also analysed the incidence of interval cancers in a large subset of negative test results. To identify interval cancers, data from negative tests were matched with the Regional Colorectal Cancer Registry and with the Regional Mortality Registry. As the cancer registry data only covered the years 2005–2007 for two LHUs (Bologna and Ferrara), and the years 2004–2008 for all the other LHUs, it was necessary to select negative FITs according to these restrictions. A malignant lesion was classified as an interval cancer if its incidence date was in the period at risk of developing it. The period at risk was calculated as the time difference between the date of the negative test report and the earliest date among the following: the date of a subsequent FIT, the end date of the cancer registry coverage, the date after two years of negative testing (730 days), death, or the incidence date of colorectal cancer. Cancer registry and screening programme operators reviewed each lesion detected through this method. We were unable to obtain information about possible losses to follow-up during the study period due to emigration out of the region.
The main determinant, in all the analyses, was the external ambient temperature estimated in the municipality of residence of the subject. Temperature was divided into six classes: <5, 5 |- 10, 10 |- 15, 15 |- 20, 20 |- 25 and > = 25℃. We used the 5 |- 10℃ class of temperature as the reference category, because this is the temperature at which people are instructed to store their samples until delivery, and because lower temperatures can also affect haemoglobin concentration. Other determinants, considered primarily as confounders, were age at the time of the test, gender of participants, and screening history (first examinations or later tests). The LHU was included as a proxy for local organisational differences that may themselves have an influence on the investigated indicators.
Statistical analysis
Statistical analyses were performed using STATA 11 SE software. Poisson regression models with robust variance estimator were used. Analyses were conducted to obtain, for each class of temperature, the positive test relative risk (PRR), the Detection Rate Ratio (DRR), and Positive Predictive Value Ratio (PPVR) for cancer and adenomas, as well as the interval cancer Incidence Rate Ratio (IRR) after a negative test. For each association measure, a test for linear trend, across classes of temperature, was performed.
Results
Distribution of FITs, participant attributes, and main outcomes by ambient temperature intervals.
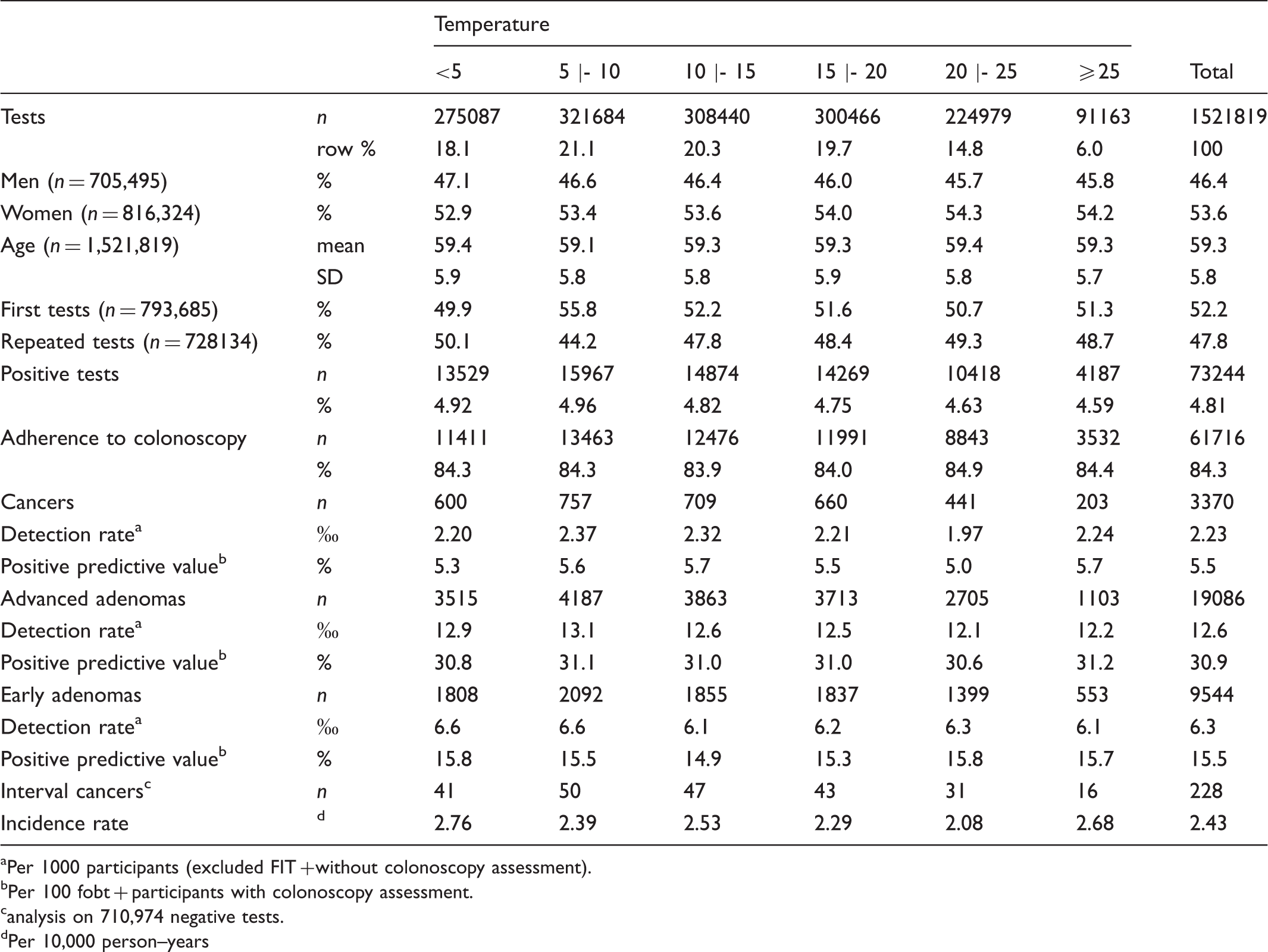
Per 1000 participants (excluded FIT + without colonoscopy assessment).
Per 100 fobt + participants with colonoscopy assessment.
analysis on 710,974 negative tests.
Per 10,000 person–years
Distribution of mean and percentiles of haemoglobin in positive tests, by type of lesion detected. a
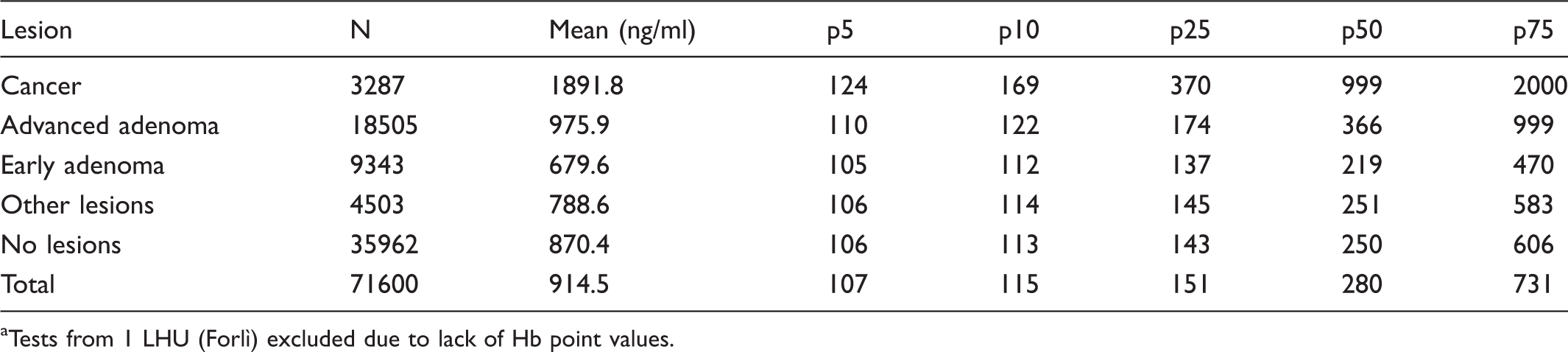
Tests from 1 LHU (Forlì) excluded due to lack of Hb point values.
In our study, 4.81% of tests were positive, with a neat linear decrease in positivity and in PRR with increasing temperature. The highest positivity was observed at 5 |- 10℃ (4.96%), the lowest at a temperature of 25℃ or above (4.59%). In 84.3% of the positive tests (61,716), the second and third level investigations led to the identification of 3370 cancers, 19,086 advanced adenomas and 9544 early adenomas.
Table 2 illustrates the results of a large subset of positive FITs (71,600), for which point Hb values were available. The distribution (mean and percentiles) of Hb concentration is presented by type of lesion. Hb levels were always higher in cancers, followed by advanced adenomas, and early adenomas. Of the tests with early or advanced adenomas, 5% showed Hb values equal to or lower than 105 and 110 ng/ml respectively, falling very close to the cut-off for test positivity (100 ng/ml).
As shown in Table 1, all detection rates were highest at 5 |- 10℃. Cancer detection rate at 5 |- 10℃ was 2.37 (per 1000 participants), and showed a minor drop at <5℃ and a major drop between 20 and 25℃ (1.97), with a following rise. The advanced adenoma detection rate showed a clear downward trend starting from 10℃. The early adenoma detection rate also dropped at 10℃, but remained approximately uniform across the subsequent temperature classes. The positive predictive values for the three neoplastic lesions did not show any trend with increasing temperature. Due to the restrictions mentioned in the Methods section, the analysis on interval cancers was performed for 710,974 negative FITs, corresponding to 49% of the total 1,448,575 negative FITs. The overall interval cancer incidence rate was 2.43 per 10,000 person–years. It did not show an increasing trend with temperature rise, as might be expected if ambient temperature was correlated with an increased rate of missed cancers. On the contrary, incidence rates showed decreasing values starting from 2.76 at <5℃ to 2.08 at 20–25℃, rising again to 2.68 in the highest temperature class of ≥25℃.
Estimates and confidence intervals at 95% of the main outcomes investigated, adjusted by age, sex, screening history, and local health unit (LHU).
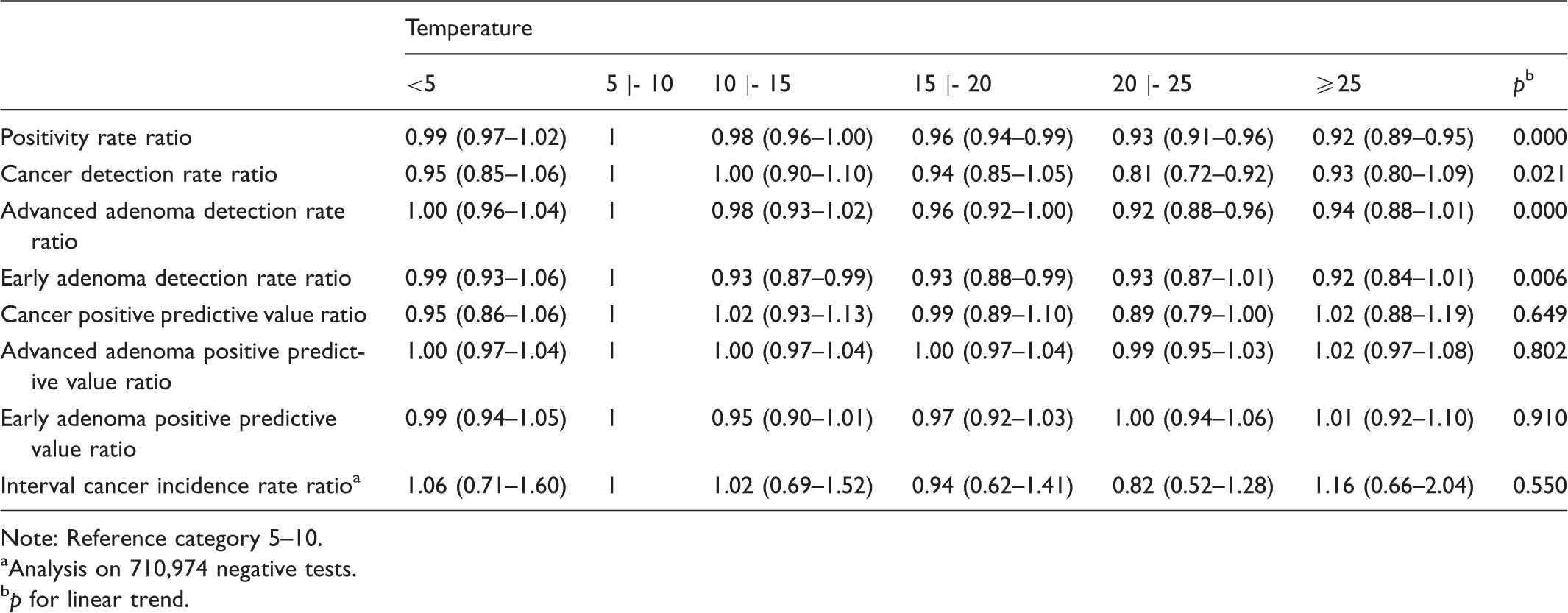
Note: Reference category 5–10.
Analysis on 710,974 negative tests.
Discussion
Our results confirm the conclusions of previous studies6,9 regarding the potential effects of ambient temperature on some indicators of screening programme based on FIT as first level exam. We found a linear decrease of the positivity rate with increasing temperature. The extent of such decrease seems narrow, with a maximum decrease of about 8% at temperatures of 25℃ or above (PRR: 0.92).
Our data, consistent with the existing literature,13,14 indicate that bleeding increases with the severity of the lesion. The concentration of haemoglobin at each percentile of the distribution was always higher in cancers, followed by advanced adenomas, and early adenomas (Table 2). This sort of findings led some authors to speculate that the effect of high temperatures, in terms of missed diagnosis, could be more significant on minor lesions compared with larger adenomas or invasive cancers. 6 Our findings do not completely agree with this hypothesis. We found a clear linear effect on advanced adenoma detection rates, while for early adenoma there seems to be a sharp decline starting from 10℃. For both the indicators, we observed a similar maximum decrease of 8%. The effect on cancer detection rates seems to be less linear, but perhaps more importantly, the detection rates are lower in most temperature classes compared with the reference category. The maximum effect was at 20|−25℃ (DRR: 0.81, −19%). Despite these findings, we did not find a clear correlation between the incidence of interval cancers and temperature. On the contrary, incidence rates seem to decrease with increasing temperature, and in the highest temperature class only we found an excess of + 16%, although this was not statistically significant.
Strengths and limitations
The assumption that the subject’s residence has not changed since the last data collection could result in some non-differential misclassification, with a probable underestimation of the measures of association investigated.15,16 However, a study performed on the health registry of the LHU of Modena found that, for each year of the period 2005–2011, on average only 3.8% of the residences were different from those reported in 2012. We presume that the residential mobility was similar in the entire region. Furthermore, it is highly probable that the lack of information on cases lost to follow-up has introduced an even more negligible bias. According to data from municipal registry offices, obtained from the National Statistics Institute and analysed by the Statistics and Geographical Information Service of the Emilia-Romagna region, in the same period and within the same age group considered in the search for interval cancers, the yearly percentage of people who emigrated from the region amounts, on average, to 0.3%. Due to the temporal and geographical limits in the coverage of the regional cancer registry, as outlined in the design section, we could not assess for interval cancer on the overall negative FITs, but only on a numerically large subset (710974 negative tests, 49%). There is no reason, however, to assume that a differential selection bias could be in action.
The strengths of this study include the analysis of early impact indicators for every stage of the screening programme, from the proportion of positive tests to the incidence rate of interval cancers, the considerable size of the population involved, the long period of observation, and the coordination of the screening programmes on a regional basis. This last choice has guaranteed the homogeneity of analytical methods (from the kit used for the sample collection to the laboratory instrumentation), as well as the adoption of a common record layout for personal data collection. Moreover, we believe that, compared with seasonal temperatures, average daily temperature estimates in the municipalities of residence constitute a better proxy of the actual temperature to which the kits are exposed.
Conclusions
Our study confirms that ambient temperature had an impact on performance of the Colorectal Cancer Screening Programme of Emilia-Romagna region in the period 2005–2011, both on the rate of positive tests, and on detection rates of adenomas and cancer. The loss of diagnosis of adenomas and malignant lesions related to the outdoor temperature does not seem to have led to a clear and consequent increase in interval cancers, although the excess found for the temperatures >25℃ does not enable us to completely exclude this possibility. It is likely that most of the lesions missed at screening are not so severe that they will become clinically manifest within two years of the missed diagnosis. Our results call for continued monitoring of the effect of ambient temperature. It will also be necessary to verify whether the recent introduction of OC Sensor tubes with an improved haemoglobin stabilisation buffer will have some positive effects on the performance of screening at higher ambient temperatures. Attention should also be focussed on the correct conservation of the kit after the sample collection, the need for a strict and short schedule between return of the kit and analysis of the sample, and the redistribution of invitations during colder periods, as is already implemented in some LHUs in the region.
Footnotes
Acknowledgement
We thank Francesca Francesconi, Department of Health Policy, Emilia-Romagna, Bologna, Italy, for her help to conduct the study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
