Abstract
Objective
Using quantitative Faecal Immunochemical Test (FIT) in colorectal cancer screening enables adjustment of the cut-off for a positive test. As men have higher stool blood levels and higher prevalence of colorectal neoplasia, different cut-off levels can be chosen for men and women. We evaluated participation and positivity rates switching from guaiac-based faecal occult blood test (gFOBT) (Hemoccult®) to FIT (OC-Sensor), using gender-specific cut-offs.
Methods
The colorectal cancer screening programme of Stockholm-Gotland, Sweden, started in 2008 and invited individuals aged 60–69 to biennial testing using gFOBT. From 1 October 2015 the test was switched to FIT, with positivity cut-offs of 40 (200) and 80 (400) µg Hb/g (ng/mL) faeces for women and men, respectively. The first year was evaluated for compliance and positivity, number of reminders and incorrect/inadequate tests, compared with gFOBT in the preceding 12-month period.
Results
There were 127,030 and 87,269 individuals invited to screening with gFOBT and FIT, respectively. The change of test increased overall participation by 11.9% (95% confidence interval 11.5%–12.3%) from 56.5% to 68.4% (
Conclusions
Within a well-organised colorectal cancer screening programme, changing the test from gFOBT to FIT markedly increased participation, especially among men, and in the younger age group. With a lower cut-off in women than men, the positivity rate was similar.
Keywords
European Union quality assurance guidelines for colorectal cancer screening and diagnosis recommend primary screening using the Faecal Immunochemical Test (FIT). 1 FIT uses an antibody to detect human haemoglobin, and the analysis is either qualitative or quantitative, with higher precision than guaiac-based faecal occult blood test (gFOBT).2–5 With quantitative FIT, the cut-off for a positive test can be adjusted and, hence, also the sensitivity. The sampling procedure is easier for the individual and does not require dietary restriction, so the simplicity of FIT compared with gFOBT could be a motivator for compliance among invitees. 6 Switching from gFOBT to FIT could result in increased compliance, higher cancer detection rates and the possibility of improving quality assurance.
Based on randomized controlled trial results demonstrating a CRC mortality reduction with gFOBT in population-based screening,7–11 the programme of the Stockholm-Gotland region of Sweden used gFOBT Hemoccult® when starting in 2008. 12 As evidence accumulated regarding the precision of FIT, as well as experiences from countries with population-based screening programmes using FIT, a change from Hemoccult® to FIT was planned and implemented in the screening programme.
No general recommendation exists regarding cut-off levels for men and women when using FIT in a screening programme targeting an average risk population. When determining the cut-off level, the expected positivity rates and their consequences in terms of the balance between colorectal cancer detection rates, false positive (positive test with no cancer), and false negative (negative test with cancer) rates must be considered, and colonoscopy resources must be available for the diagnostic follow-up of a positive test. The initial cut-off level of the Dutch population-based screening programme resulted in a higher positivity rate and a lower positive predicted value than expected and, subsequently, the cut-off level was raised. 13 In the Stockholm-Gotland programme, the positivity rate of Hemoccult® has been around 2%, with adequate colonoscopy resources for follow-up assessments.
Lower cancer detection rates and higher rates of interval cancers have been reported in women compared with men when screening using both gFOBT and FIT,14–16 possibly due to lower positivity rates among women and, therefore, fewer follow-up diagnostic endoscopies. To equalize the short-term outcomes, in terms of cancer detection and interval cancer rates, using different cut-off levels for men and women has been suggested. 17
This study aimed to evaluate the effect of switching from guaiac-based Hemoccult® to FIT on compliance and positivity rates, using gender specific cut-off levels in the context of an established and well-organised colorectal cancer screening programme targeting an average risk population.
Methods
The population-based colorectal cancer screening programme of Stockholm-Gotland invites all individuals aged 60–69 to biennial screening. 12 The programme is centrally organised at the Regional Cancer Centre of Stockholm-Gotland, which is responsible for sending personal invitations including the test-kit, electronic requisition forms to the laboratory, and electronically receiving the laboratory results for the analysed samples. Individuals with normal results receive a standardised letter, and those with positive test results are electronically referred for follow-up diagnostic colonoscopy at a contracted endoscopy clinic responsible for the population of the catchment area.
In December 2012, it was decided to switch the programme from gFOBT (Hemoccult®) to FIT. As the existing programme had 100% invitation coverage, a compliance rate of approximately 60%, 12 and an established quality assurance organisation, it was decided that the change of test would be made without modifying the organisation or infrastructure of the programme. A decision was also made to continue to send the test kits with the mailed invitation and the County Council carried out a tender process for the FIT-tubes, separate from the analysis of the change of test process. The separate tender process was a cost-saving measure that allowed for the continuation of sending the test with the invitation. The programme operations IT system was developed to accommodate the new format of the electronic requisition forms sent to the laboratory from the screening centre and the quantitative FIT test results received from the laboratory. The IT system had to handle both gFOBT and FIT results, as gFOBT had been continuously sent out prior to the switch on 1 October 2015.
Using data on positivity rates and cut-off values from a series studies on the OC-Sensor system,17–21 it was possible to estimate the association between the concentration of faecal haemoglobin (f-Hb) and the positivity rate. A distance weighted least squares regression curve of f-Hb concentration cut-off (x-axis) and positivity rate (y-axis) suggested a positivity rate for women and men together of about 2.5–3.0% at approximately 40–80 µg/g (Figure 1). In a screening-based population, it has been demonstrated that cut-offs of 40 µg/g for women and 80 µg/g for men gave similar age dependent positivity rates of 2.7–4.9% and 1.9–4.5%, respectively. 17
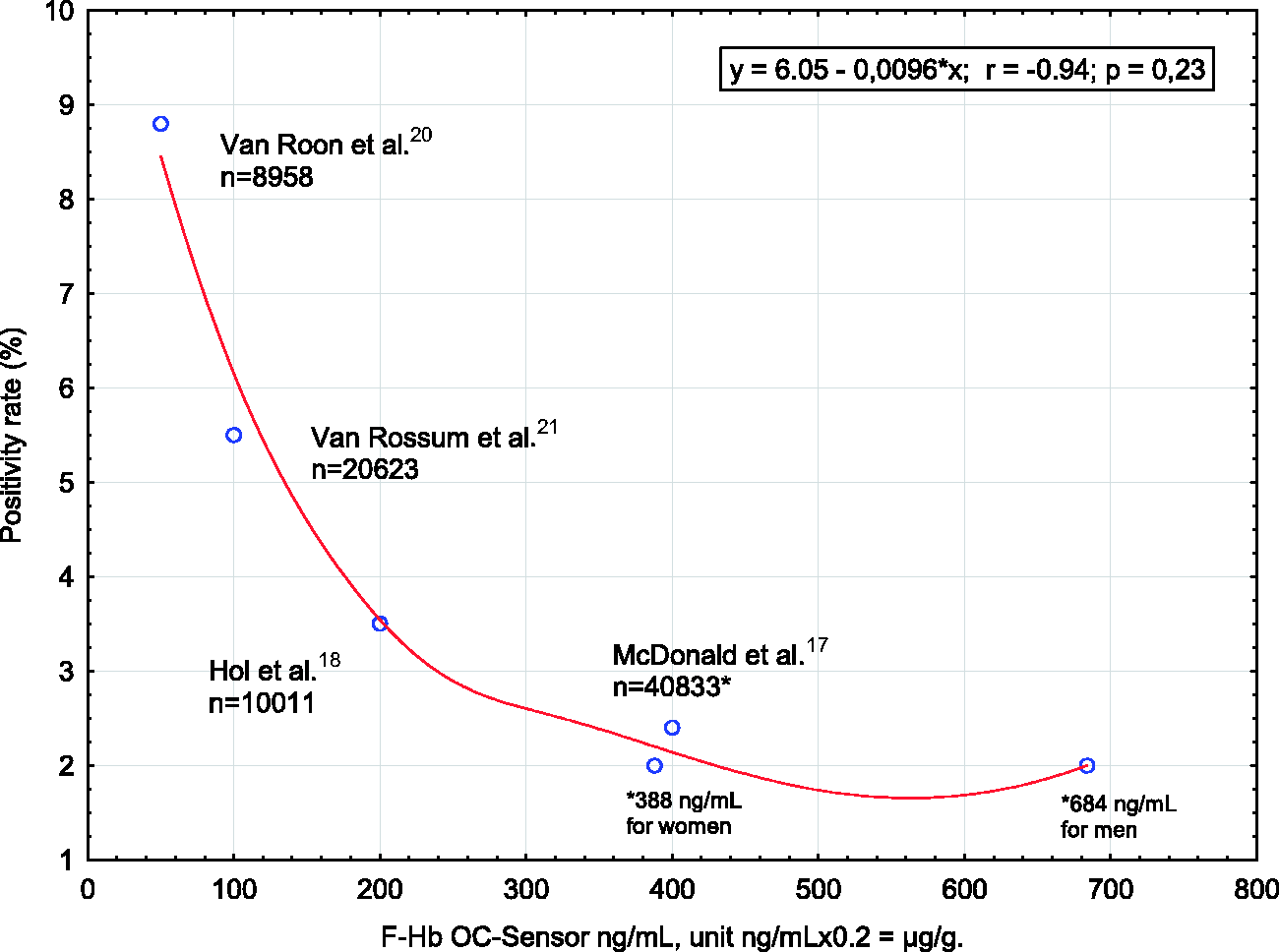
Literature with different cut-offs for positivity guiding the set cut-offs in the Stockholm-Gotland screening programme 40 µg/g (200 ng/mL) for women and 80 µg/g (400 ng/mL) for men.
One test tube OC-Sensor FIT (PTA, OC-Auto Sampling Bottle 3, Eiken) was included in the invitation, collecting approximately 10 mg faeces with a serrated probe when inserted at random into different parts of the passed stool. The probe is then re-inserted in 2.0 mL preservative buffer, the date of sample collection is marked by the participant on the FIT label, and the sample is sent directly to the laboratory (Aleris Medilab, Täby, Sweden) in a prepaid envelope. At the laboratory, the OC-Sensor FIT samples were analysed the same day or stored at 4°C until analysis.
The f-Hb was quantitatively measured using the OC-Sensor Diana Latex reagent on one single OC-Sensor Diana analyser (Eiken Chemical Co., Ltd, Tokyo, Japan).22–25 Performance characteristics of the OC-Sensor assay and other FITs have recently been reviewed and summarized. 26 F-Hb results were reported as µg Hb/g faeces (i.e. µg/g), as recommended by the Expert Working Group on FIT for Screening, the Colorectal Cancer Screening Committee, World Endoscopy Organisation. 27 Gender-specific cut-off values were used: f-Hb concentrations ≥40 µg/g for women and ≥80 µg/g for men were considered positive test results. All stool samples collected with an unexpired OC-Sensor tube were analysed. Positive tests that arrived at the laboratory >10 days from the date of sample or were missing the sampling date were deemed positive, and the participant was referred for follow-up colonoscopy. However, if the result was negative and the tube was expired or missing the sampling date, the result was deemed inconclusive and the participant received an informative letter with a new OC-Sensor FIT.
Electronic requisition forms were generated automatically for all individuals invited to screening, and retrieved by the central laboratory data information system when samples were registered at the laboratory. The f-Hb concentrations were recorded electronically by the analyser. The analyser presents the results in ng/mL units and, therefore, all results were recalculated by the central laboratory data information system to µg/g, using the algorithm ng/mL*0.2=µg/g. Analysis results were then sent electronically to the Regional Cancer Centre of Stockholm-Gotland centralised data administrative system in reply to the requisition forms sent to the laboratory for each invited individual.
This study includes invitations to the Stockholm-Gotland colorectal cancer screening programme sent from 1 October 2014 to 30 September 2016, and comprises 127,030 individuals invited for screening with gFOBT, and 87,269 individuals invited for screening with FIT. All invitations after 1 October 2015 included an OC-Sensor FIT. Invitees who had received a Hemoccult® test-kit prior to 1 October were regarded as participants, and the tests analysed if the tests were returned and analyzed within 180 days.
Participation, defined as submitting a OC-Sensor FIT to the laboratory for analysis within 180 days, was estimated as a proportion of all those invited, and compared with the preceding one year period when Hemoccult® was used. Test positivity in the FIT period (f-Hb concentrations ≥40 µg/g for women and ≥80 µg/g for men) was reported and compared with Hemoccult® positivity rates for the preceding period. Other programme parameters, including the proportion of individuals who participated after a reminder, and the proportion of inadequate/incorrectly completed stool samples, were monitored, reported, and compared with Hemoccult®.
All data were extracted from the centralised data administrative system at the Regional Cancer Centre of Stockholm-Gotland and analysed using the software package R. Statistical significance in differences between stool tests regarding participation by gender, age group, and positivity were estimated with Chi-square test of proportions at a significance level of 5% (
The study was approved by the Regional Ethical Review Board in Stockholm.
Results
Overall, participation increased by 11.9% (95% confidence interval (CI) 11.5%–12.3%) from 56.5% to 68.4% (
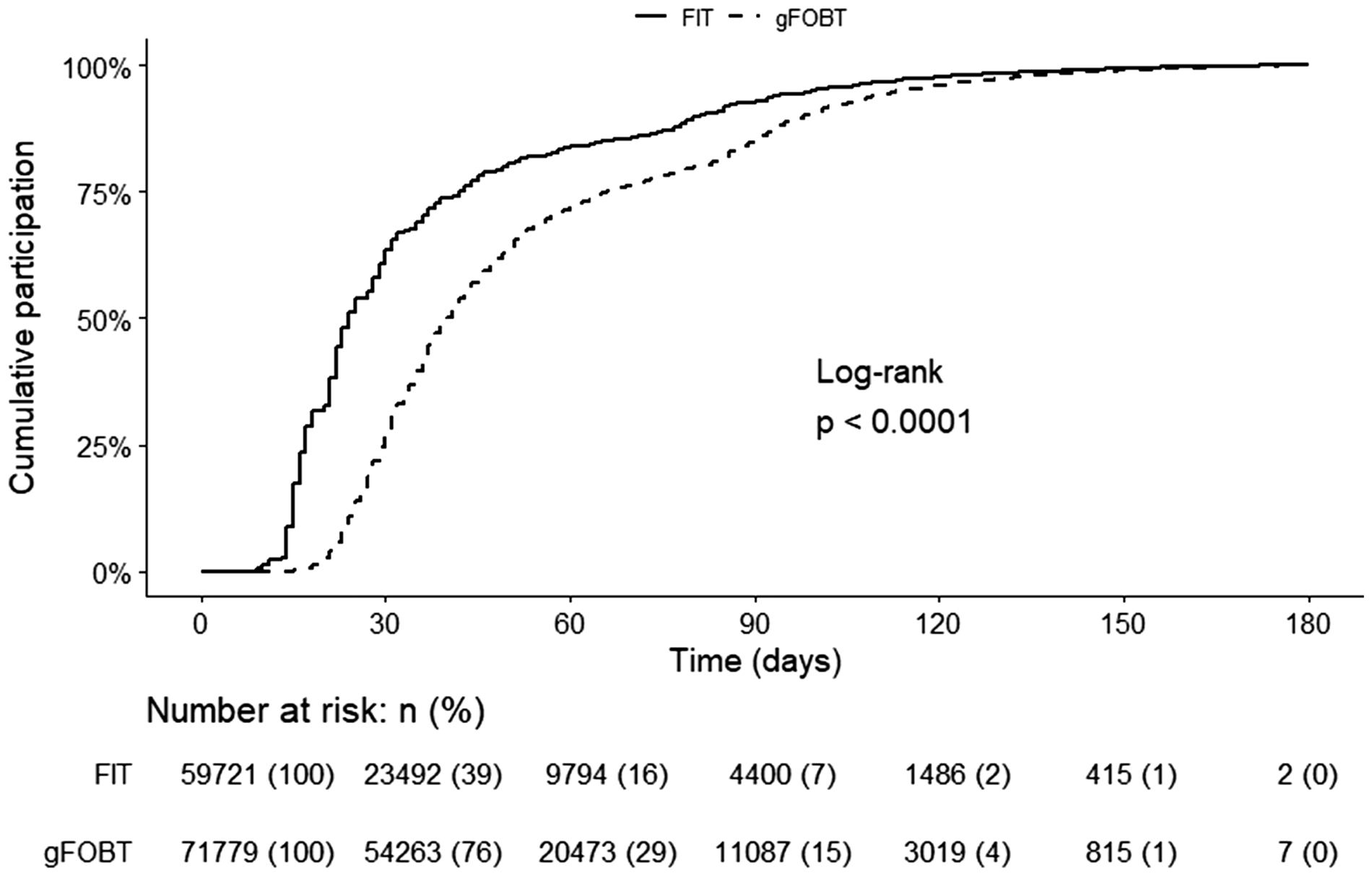
Time to participation in days since test-kits were sent, by stool test. FIT: Faecal Immunochemical Test; gFOBT: guaiac-based faecal occult blood test.
Invitations and participation in the Stockholm-Gotland screening programme, by stool test and gender.
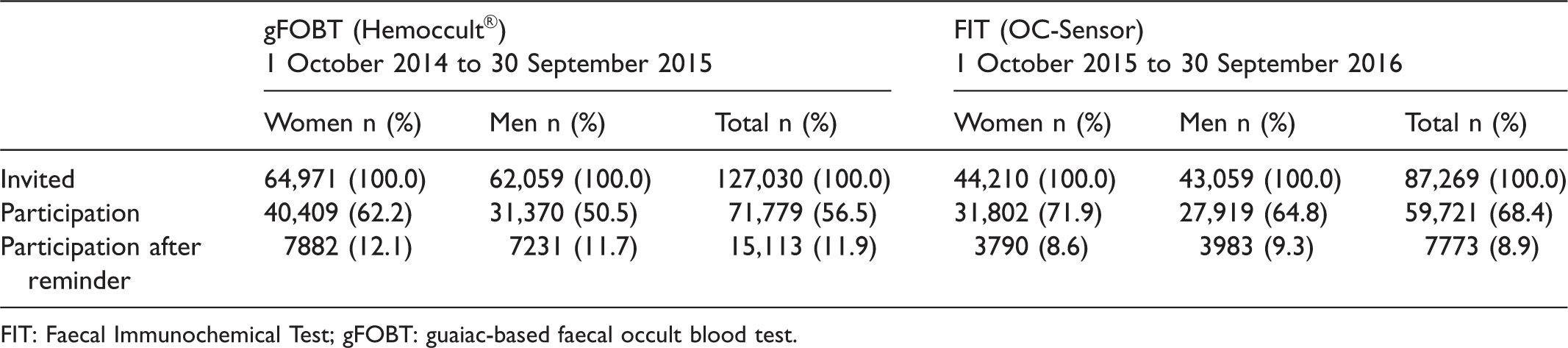
FIT: Faecal Immunochemical Test; gFOBT: guaiac-based faecal occult blood test.
Invitations and participation in the Stockholm-Gotland screening programme, by stool test and age group.
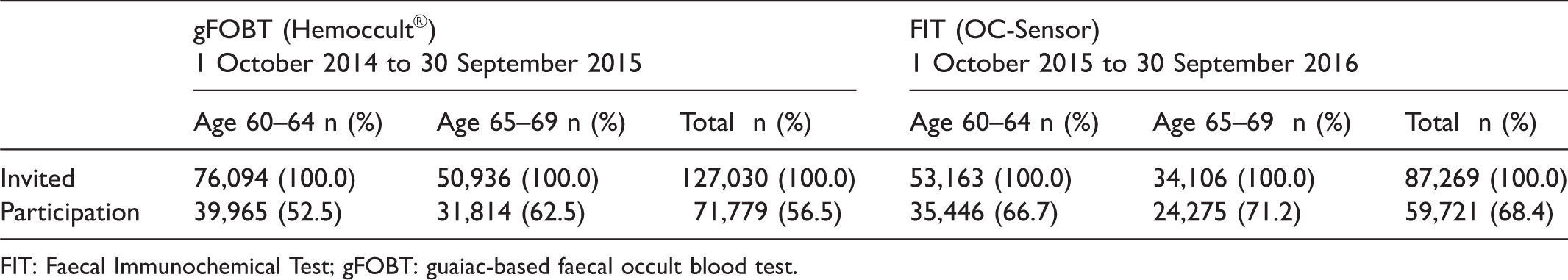
FIT: Faecal Immunochemical Test; gFOBT: guaiac-based faecal occult blood test.
As expected with the gender-specific cut-offs implemented, the positivity rate among women was higher with OC-Sensor (2.6%) than with Hemoccult® (1.3%). In men, the positivity rate was approximately the same with gFOBT and FIT with a cut-off of 80 µg/g, 2.4% and 2.5%, respectively. Fewer individuals needed retesting due to incorrect or incomplete stool sampling with FIT than with gFOBT (Table 3). The positivity rate was less than 2% in both age groups with gFOBT, and between 2% and 3% with FIT (Table 4).
Positivity rates in the Stockholm-Gotland screening programme, by stool test and gender (cut-off women 40 µg/g, men 80 µg/g).
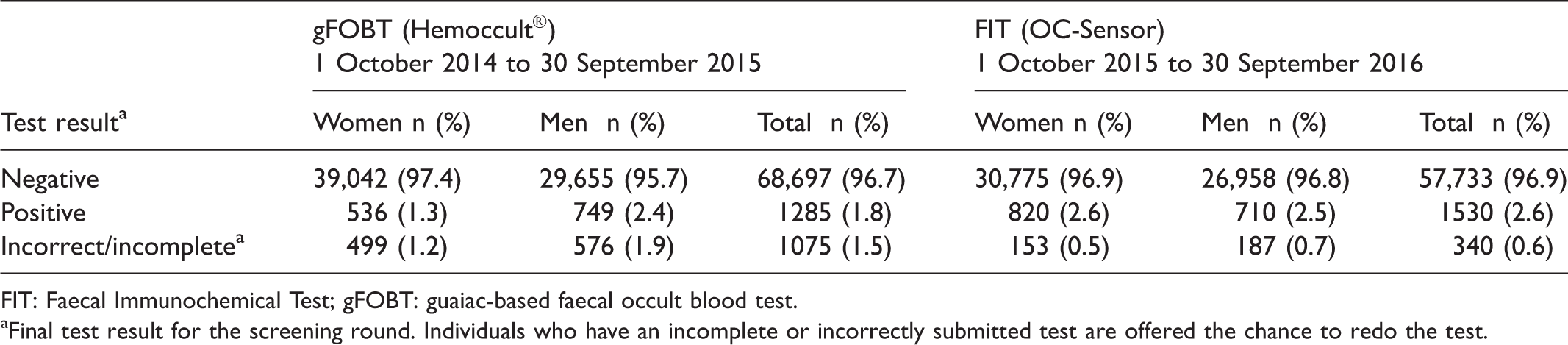
FIT: Faecal Immunochemical Test; gFOBT: guaiac-based faecal occult blood test.
aFinal test result for the screening round. Individuals who have an incomplete or incorrectly submitted test are offered the chance to redo the test.
Positivity rates in the Stockholm-Gotland screening programme, by stool test and age group.

FIT: Faecal Immunochemical Test; gFOBT: guaiac-based faecal occult blood test.
Discussion
In the Stockholm-Gotland CRC screening programme of an average risk population, switching from gFOBT (Hemoccult®) to FIT (OC-Sensor) increased the overall participation by 12%, more in men than women, and more in the group aged 60–64 than the group aged 65–69. With a cut-off for a positive test of 40 µg/g (equals 200 ng/mL) for women and 80 µg/g (400 ng/mL) for men, the positivity rate was very similar, 2.6% in women and 2.5% in men. The lower rate of reminders and incorrectly performed tests with FIT could be indirect outcome measures of the simplicity of testing compared with testing using Hemoccult®.
One strength of our study is the centrally administered programme with 100% invitation coverage of the target group, where a change of test could be made without modifying the infrastructure, or jeopardizing quality assurance. The only adjustement made to the programme during the study period was the change of test (and corresponding instruction manual); therefore, it is possible that differences in participation and positivity observed are explained by this.
An overall increase in screening participation of approximately 7%, similar to that seen in our programme, was observed in the pilot study of switching from gFOBT to FIT in the English Bowel Screening Programme. 28 The increase was higher in men than women. The overall screening participation with gFOBT was approximately 59%, so our increase of 12% with change of stool test was therefore more than expected, given these prior results. The increase observed in the Stockholm-Gotland programme in men was as high as 14%, compared with 8% in the English pilot. 28 Speculatively, information about random allocation to a study of FIT could have somewhat hampered the increase in the pilot.
The amount of blood in stool is higher in men than in women, 17 as is the prevalence of advanced colorectal neoplasia. 29 Within the German colonoscopy screening programme, Brenner et al. evaluated quantitative and qualitative FIT and gFOBT, and summarized that at all cut-off levels of quantitative FIT and the qualitative tests, the sensitivity and positive predictive value were substantially higher among men than women. 29 On the other hand, the specificity and negative predictive value were higher among women. In the Stockholm-Gotland programme, the positivity rate among women was close to half that of men (1.3% vs. 2.4%) using gFOBT, with a lower rate of diagnostic colonoscopies to follow. If FIT was introduced with the same cut-off for a positive test for both genders, there would be a higher sensitivity and lower specificity for advanced neoplasia in men than in women. 30
In a pilot study within the Scottish Bowel Cancer Screening programme of 50–74 year olds, 66,000 average risk individuals in two geographical areas received FIT (OC-Sensor). With a cut-off at 80 µg/g, the positivity rate was 2.4% among the approximately 40,000 returned test-kits (56.4% from men), 31 which is in line with the Stockholm-Gotland results, although with a lower cut-off in women. The accuracy of FIT by gender was not evaluated, but the correlation of quantity of blood and advanced neoplasia was examined. 31 The study demonstrated that 75% of CRC with positive FIT at a cut-off of 80 µg/g would have been positive with a cut-off as high as 200 µg/g. 31 Therefore, when screening primarily for CRC (and secondarily for advanced adenoma), it is probably the false positive tests in women at a cut-off of 40 µg/g, rather the potential false negative tests in men at a cut-off of 80 µg/g, that are of concern and must be evaluated in future studies in population-based screening programmes.
When adjusting cut-offs based on gender, endoscopy resources available are the limiting factor, and an approximate 2% positivity rate is optimal in the Stockholm-Gotland setting. With the cut-off of 80 µg/g for men, the positivity rate was approximately the same with FIT as with gFOBT, 2.5% and 2.4%, respectively, and so the same proportion of advanced adenomas would be expected. On the other hand, generating the same positivity rate in women with a cut-off of 40 µg/g (2.6%) as in men results in a positivity rate twice as high as with gFOBT (1.3%). The 100% increase in positive tests in women would probably include more advanced adenomas, but might be at the expense of a higher rate of false positives. Due to the short study period, available colonoscopy outcome data could be over- or under-estimations of surrogate effect measures of the programme. Analyses of the colonoscopy results after switching to FIT are expected in 2019.
Conclusion
Switching from gFOBT to FIT in an organised CRC screening programme increased the overall participation, more in men than in women, and more in the younger age group than the older. Gender-specific cut-offs for a positive test of 40 and 80 µg/g for women and men, respectively, resulted in similar positivity rates in both genders. Further studies will evaluate the screening-detected cancers and interval cancers at these cut-offs.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
