Abstract
Background
Newborn screening for propionic acidemia and methylmalonic acidurias using the marker propionylcarnitine (C3) is neither sensitive nor specific. Using C3 to acetylcarnitine (C3/C2) ratio, together with conservative C3 cut-offs, can improve screening sensitivity, but the false positive rate remains high. Incorporating the marker 2-methylcitric acid has been suggested, to improve the positive predictive value for these disorders without compromising the sensitivity.
Methods
Between July 2011 and December 2012 at the Newborn Screening Ontario laboratory, all neonatal dried blood spot samples that were reported as screen positive for propionic acidemia or methylmalonic acidurias based on elevated C3 and C3/C2 ratio were analyzed for 2-methylcitric acid, using liquid chromatography tandem mass spectrometry.
Results
Of 222,420 samples screened, 103 were positive for methylmalonic acidurias or propionic acidemia using C3 and C3/C2 ratio as markers. There were nine true positives: propionic acidemia (n = 3), Cobalamin (Cbl) A (n=1), and Cbl C (n = 5). Among false positives there were 72 neonates not affected, 20 with maternal B12 deficiency, and two incidental finding (transcobalamin II and unclassified Cbl defect). 2-Methylcitric acid was analyzed in all 103 samples and ranged between 0.1 and 89.4 µmol/l (reference range 0.04–0.36). Only 14 samples exceeded the set 2-methylcitric acid cut-off of 1.0 µmol/l, including the samples from all nine true positives.
Conclusion
By including 2-methylcitric acid in the screening algorithm, the positive predictive value of our primary and secondary screening targets improved from 8.7 to 64.3%. This would have eliminated 89 unnecessary referrals while maintaining 100% sensitivity.
Keywords
Introduction
In newborn screening for congenital or inherited disorders, the widespread introduction of tandem mass spectrometry (MS/MS) has enabled multiplexed detection of a significant number of neonatal metabolic disorders. Several defects of propionate metabolism are included in many screening programs, 1 for example, methylmalonyl CoA mutase deficiency (OMIM ID: 251000), propionic acidemia (PA, OMIM ID: 606054), and other disorders involved in cobalamin (Cbl) metabolism.1–5 Affected infants have a significant risk of neurological damage or life-threatening episodes due to metabolic acidosis, ketosis, and hyperammonemia. Improved outcomes are anticipated should treatment of affected babies start before symptoms appear.6,7
PA and methylmalonic acidurias (MMA), caused by defects in methylmalonyl CoA mutase or Cbl metabolism, are characterized by accumulation of propionylcarnitine (C3), a marker that can be detected by standard MS/MS screening methods. C3 concentrations in affected and unaffected newborns overlap significantly. 8 While conservative C3 cut-offs combined with C3 to acetylcarnitine (C3/C2) ratio can provide adequate screening sensitivity, the false positive rate is high. To mitigate this, we recently described a simple method to measure 2-methylcitric acid (MCA) in dried blood spots (DBS) that can be easily incorporated as a second-tier test for samples with elevated C3 and C3/C2 ratio. 9 MCA is generally accepted as a pathognomonic marker for propionate metabolic defects and has been shown to improve newborn screening for PA, MMA, and Cbl defects.9,10 In an 18-month study, we measured MCA in the original screening DBS samples as a second-tier test.
Methods
DBS specimens (n = 222,420) received at the Newborn Screening Ontario laboratory between July 2011 and December 2012 were tested for amino acids, acylcarnitines, and succinylacetone using standard MS/MS methodology that involves derivatization with n-butanol. Among these, 103 samples were screen positive for PA and MMA. At our laboratory, after extensive assessment of various analytes and potential ratios in previously proven positive cases, screening results are considered positive for PA and MMA if C3 to acetylcarnitine (C3/C2) ratio ≥ 0.27 and C3 ≥ 4.0 µmol/l, or C3/C2 ≥ 0.23 and C3 ≥ 5.5 µmol/l, in samples collected at or before seven days of age. If the neonate is older than seven days at sample collection, a C3/C2 ≥ 0.27 and C3 ≥ 2.6 µmol/l, or C3/C2 ≥ 0.23 and C3 ≥ 4.0 µmol/l is considered screen positive. The study was approved by the Research Ethics Board of the Children’s Hospital of Eastern Ontario.
Samples that screened positive for PA and MMA were submitted for MCA determination. Extraction and derivatization were achieved in a single step, as described previously. 9 Briefly, d3-MCA (20 µl in 50% acetonitrile), 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide (EDC) (25 µl in water), 4 -(dimethylamino)pyridine (DMAP) (25 µl in acetonitrile), and 4-[2-(N,N-dimethylamino)ethylaminosulfonyl]-7 -(2-aminoethylamino)-2,1,3-benzoxadiazole (DAABD-AE) (50 µl in acetonitrile) were successively added into a 2.0 ml polypropylene tube containing a single 3.2 mm DBS and incubated for 45 min at 65℃. After stopping the reaction by adding 120 µl of 10% methanol containing perfluorooctanoic acid (PFOA) at a concentration of 0.5 g/l 10 µl portions were subjected to analysis. Analysis was performed on an ACQUITY UPLC BEH C8 column (2.1 mm × 50 mm, 1.7 µm, Waters) using a Waters ACQUITY Ultra Performance LC system (Waters, Milford, MA, USA) equipped with a Xevo XE tandem mass spectrometer (Micromass, Manchester, UK). MCA and internal standard were detected by positive ion electrospray ionization (ESI)–MS/MS using transitions of mass to charge (m/z) of 499 to 151 and 502 to 151, respectively.
MCA and d3-MCA as internal standard were from CDN Isotopes (Pointe-Claire, QC, Canada). DAABD-AE was purchased from Sigma-Aldrich (St. Louis, MO, USA). DMAP, EDC, and PFOA were purchased from Sigma-Aldrich. HPLC grade acetonitrile was obtained from Burdick’s and Jackson (Muskegon, MI, USA). Water used throughout this study was generated by a Milli-QUV Plus Ultra pure water system (Millipore SA, Molsheim, France).
Results
Median and range levels of MCA, C3, and C3/C2 obtained in this study.
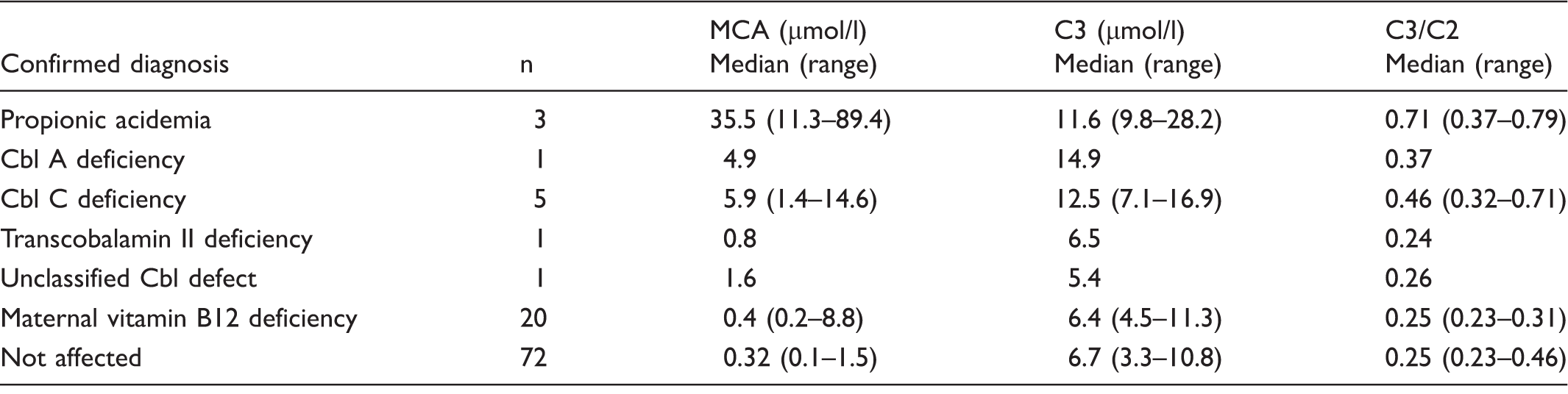
Cbl: cobalamin; MCA: 2-methylcitric acid.
Discussion
While C3 is known to be primarily elevated in disorders of propionate metabolism, overlap in levels between healthy and affected newborns makes this marker a poor discriminator that is associated with unfavourable balance between testing specificity and sensitivity.8,11 The use of acylcarnitine ratios such as C3/C2 improves this balance,12,13 but routine screening methods do not provide further differentiation, and the false positive rate for C3-related disorders remains high. This necessitates further diagnostic workup and may lead to an unbalanced cost–benefit ratio of newborn screening, as well as placing unnecessary emotional burden on the affected families.
Of 222,420 DBS specimens received at our laboratory in the study period, 103 screened positive for PA or MMA and patients were referred for further evaluation. Diagnostic testing revealed PA (n = 3), Cbl A (n = 1), Cbl C (n = 5), transcobalamin II deficiency (n = 1), and unclassified Cbl defect (n = 1). Maternal vitamin B12 deficiency was a frequent finding (n = 20) and a C3-related disorder could not be confirmed in the rest of these infants (n = 72). PA and Cbl A belong to the primary screening panel. Cbl C is a secondary target, 1 and transcobalamin II and unclassified Cbl defects are considered incidental findings. Accordingly, the positive predictive value (PPV) of primary and secondary screening targets using C3 and C3/C2 as screening markers is 8.7% (9/103).
Second-tier testing is an efficient approach to improve specificity of newborn screening. Reflexive analysis of the same screening specimen for a more specific marker eliminates the need to contact the patient to have a second sample collected. Typically, the second-tier test overrides the initial screening result. We retrospectively measured MCA in the 103 DBS samples that were screened positive for PA or MMA (Figure 1). Of these, 14 exceeded the MCA cut-off of 1.0 µmol/l, including all primary and secondary targets (n = 9), unclassified Cbl (n = 1), maternal vitamin B12 deficiency (n = 2), and two unaffected neonates. The PPV for primary and secondary screening targets was 64.3% (9/14), with 100% sensitivity. Incorporating MCA testing as a marker for PA and MMA would have eliminated 95% (89/94) of unnecessary referrals. Lowering the MCA cut-off to 0.8 µmol/l would have identified the patient with transcobalamin II deficiency, but six additional recalls would have occurred, reducing the PPV to 45% (9/20). Given that this disorder is not a screening target, this reduction in PPV may not be justified. MCA analysis is unlikely to prolong turnaround time, as this assay is characterized by short sample preparation and chromatography time.
MCA concentrations in DBS specimens (n = 103) obtained in this study (▴ represents true positives, • represents incidental findings, ▪ represents maternal vitamin B12 deficiency and ○ represents the not affected patients). The dashed line shows the MCA cut-off applied in this study. DBS: dried blood spots; MCA: 2-methylcitric acid.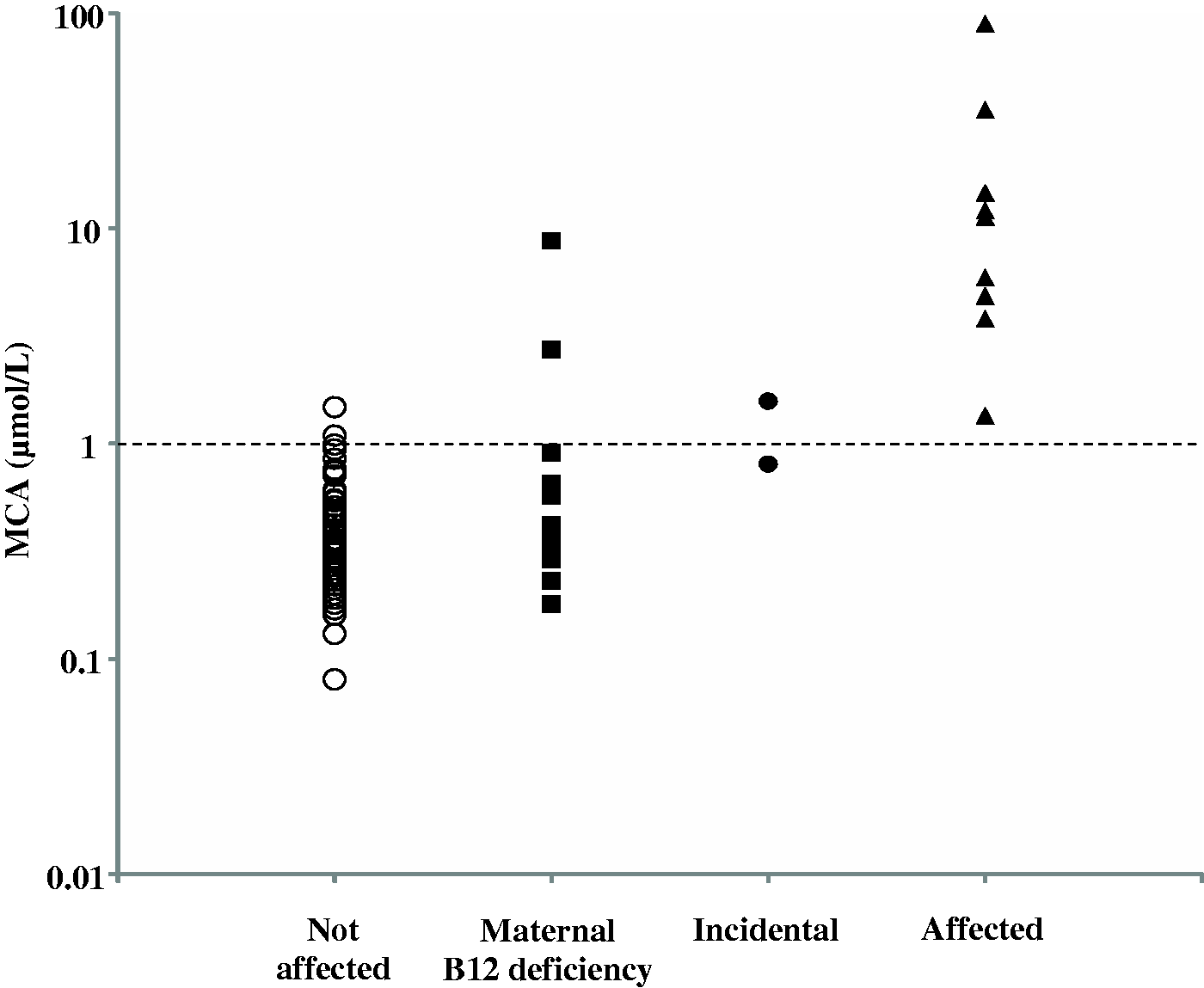
Our results are in line with other reported studies. Turgeon et al. described a method to measure MCA and other metabolites in DBS and found a similar improvement in PPV. 10 A different second-tier test that measures markers other than MCA improved the PPV of MMA to 21%, with 100% sensitivity.11,14
Conclusion
Our study successfully evaluated the integration of MCA analysis into routine newborn screening for PA and MMA. The use of MCA significantly improved our PPV of PA and MMA from 8.7 to 64.3% with 100% sensitivity. The improved specificity eliminated 95% of the false positives and incidental findings. This robust method determines MCA with high accuracy and may also be used to monitor patients under treatment.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
