Abstract
Objective
To use the results of the first five years of a cystic fibrosis newborn screening program to estimate the cystic fibrosis birth prevalence and spectrum of cystic fibrosis transmembrane conductance regulator (
Methods
Screening was performed from 2010 to 2015, using two-tier immunoreactive trypsinogen testing, followed by a sweat test. When sweat test values were >30 mmol/L, the
Results
Of 96,071 newborns screened, a second sample was requested in 119 cases. A sweat test was performed in 30 newborns, and 9 possible cases were detected (seven confirmed cystic fibrosis and two inconclusive). The most frequently detected
Conclusions
Cystic fibrosis birth prevalence in southeastern Mexico is 1:13,724 newborns. Immunoreactive trypsinogen blood concentration is influenced by gestational age and by the time of sampling. The spectrum of
Introduction
Cystic fibrosis (CF) has an estimated newborn prevalence of 1:4000. 1 It is infrequent among Indian (1:40,000–100,000) and Japanese (1:350,000–1:1,000,000) newborn populations, 2 but in the Hispanic population of California, birth prevalence is 1:9259. 3 In Latin America, only Costa Rica, Paraguay, and Uruguay have implemented nationwide mandatory newborn screening (NBS) for this disease, and in Mexico, screening is available only through some regional NBS programs or privately, 4 hence CF birth prevalence in this region is poorly characterized. In developed countries, CF patients currently have a life expectancy of ∼40 years, 5 an outcome achieved through combining better treatments and NBS strategies that allow earlier medical management.6-8 However, even in most developed countries, where neonatal detection of CF has been recommended and adopted, testing procedures are complex, and analytical protocols are not uniform. 1
Immunoreactive trypsinogen (IRT) has been used as a CF biomarker since Crossley et al. first demonstrated that trypsinogen levels are higher in newborns with CF.
9
Interpretation of IRT tests is complex, due to confounding factors such as age-related declines in IRT blood levels, prematurity, and perinatal stress.1,10 All newborn CF screening strategies begin with IRT quantification as the first tier, followed by a second IRT determination or quantitation of pancreatitis-associated protein and/or CF transmembrane conductance regulator gene (
Molecular
This study aimed to present the results of the first five years of a Mexican CF screening program, showing for first time the birth prevalence and the spectrum of
Methods
A mandatory expanded NBS protocol testing for congenital hypothyroidism, aminoacidopathies, organic acidemias, fatty acid oxidation defects, congenital adrenal hyperplasia, biotinidase deficiency, galactosemia, hemoglobinopathies, and CF was applied to all newborns delivered at Health Facilities for Infant-Maternal Care at the Health State Ministry of Yucatan in southeastern Mexico, between 2010 and 2015. Five drops of blood were taken from the heel and collected on standardized filter paper (Guthrie cards). Demographic data were obtained, and all confirmed cases received professional genetic counseling.
The IRT/IRT/ST screening strategy is shown in Figure 1. For IRT blood values > 70 ng/mL and < 250 ng/mL, a second blood sample was requested. An ST was requested for all newborns with a second cutoff IRT blood concentration > 60 ng/mL. Cases with an original IRT of > 250 ng/mL were sent directly to ST.
Cystic fibrosis newborn screening algorithm and overview of results. CF: cystic fibrosis; NBs: newborns; IRT: immunoreactive trypsinogen.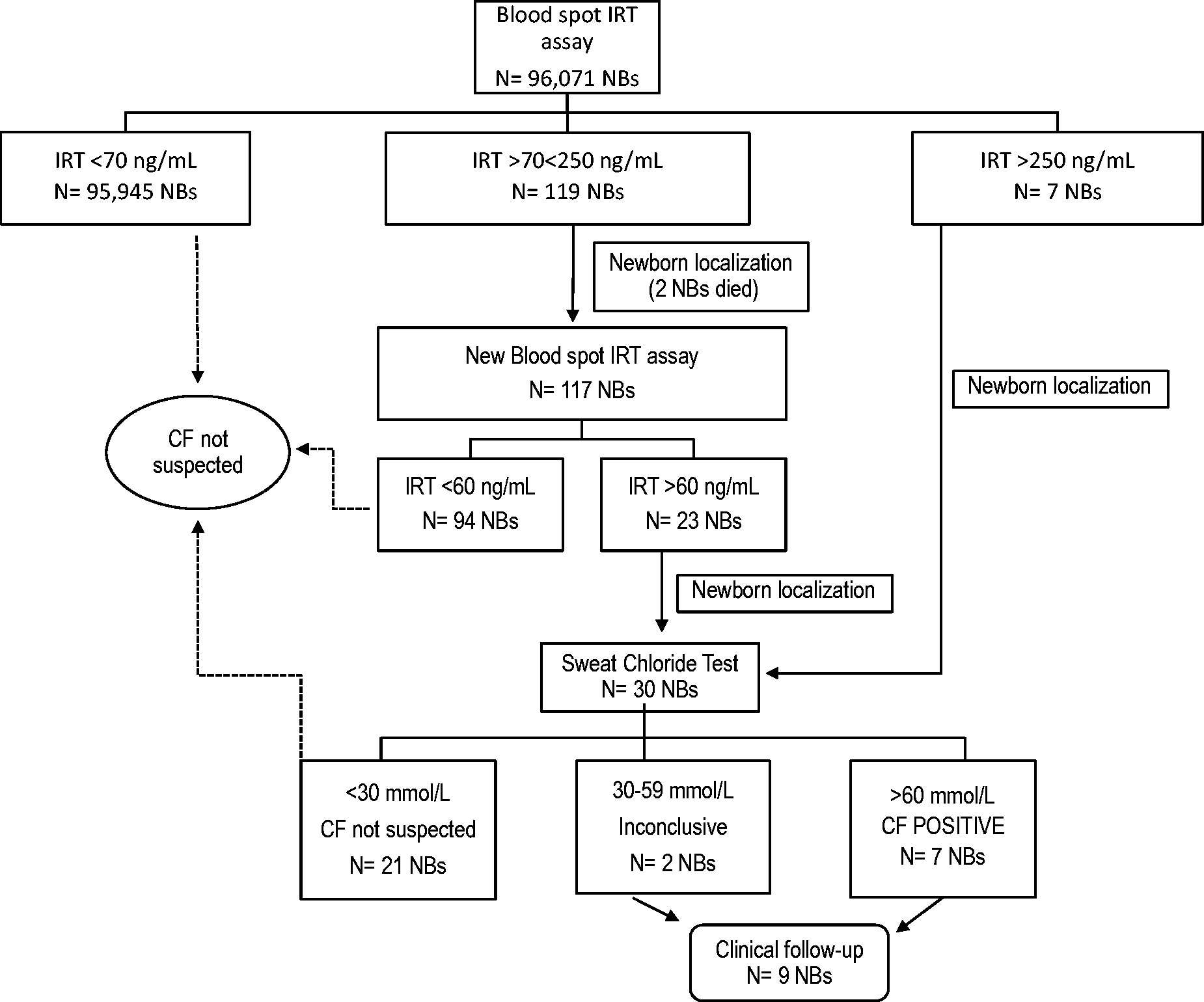
IRT was measured in blood spot samples by time-resolved immunofluorometric assay using an Autodelfia instrument and commercial kits (Perkin Elmer, Waltham, MA). Sweat samples were obtained using the Macroduct® (Webster Sweat Collection System 3700-SYS, Wescor). After cleansing the skin with water, pilocarpine iontophoretic was used to stimulate sweat production (pilogel pads). Sweat chloride concentration was measured in the laboratory according to the CLSI guidelines. 10 Newborns with sweat chloride concentrations > 60 mmol/L were considered true CF cases, and those with values between 30 and 59 mmol/L were considered inconclusive cases, according to 2015 CF Foundation diagnosis consensus conference recommendations on the diagnosis of CF in screened populations. 15 All true or inconclusive CF cases were subsequently referred to the Dr Agustín O’Horán Hospital for medical evaluation and follow-up.
DNA extracted from dried blood spots from nine newborns considered true or inconclusive CF cases was subjected to
Results
A total of 96,071 newborns in Yucatan (45.6% females and 54.4% males), representing 97% coverage of annual births, were screened for IRT over the five-year period. The modal age at sampling was five days (range, 1–28 days); 76.6% of samples were taken during the first week of life, 18.4% in the second, and 3.6% and 1.4% in the third and fourth weeks, respectively. A total of 7.7% of newborns were premature (<36 weeks gestation).
Figure 2(a) and (b) shows the IRT values in blood samples obtained in the first week of life from full-term and preterm newborns. The median IRT blood concentrations in first-week samples were 15.30 ng/mL in full-term newborns and 14.6 ng/mL in preterm newborns. A significant difference using Mann-Whitney Distribution of the neonatal IRT values in the first week of life by newborn gestational age (a) Full term, (b) Pre-term, and (c) IRT concentration values by gestational age according to the different weeks of age at the time of blood sampling. IRT: immunoreactive trypsinogen; T: full-term; P: pre-term.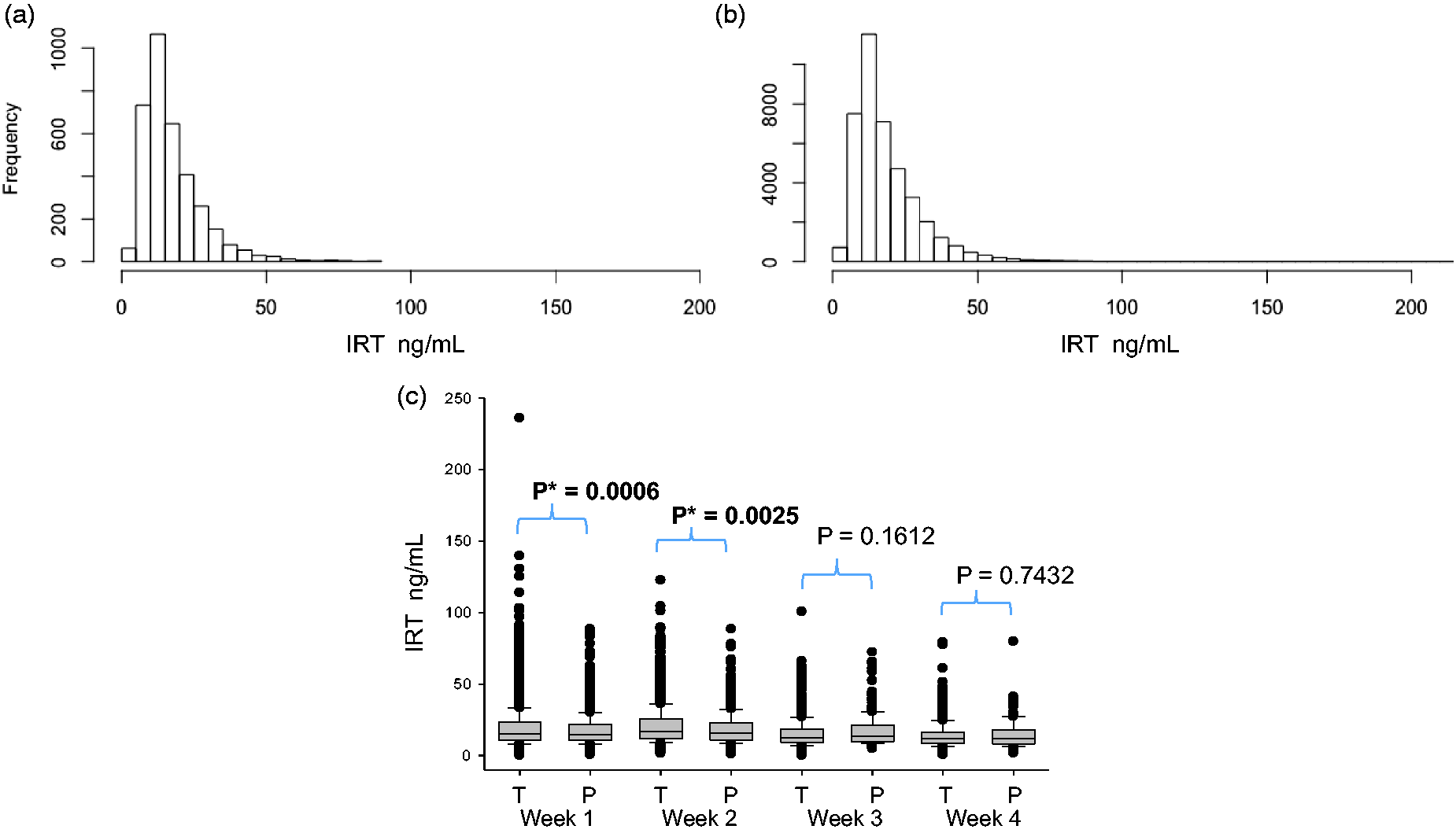
An overview of the screening strategy and results are shown in Figure 1. The cutoff value of 70 ng/mL represents the cumulative 99.5th percentile of the first 2500 studied newborns. The cutoff value for the second sample was determined based on the age of the newborn at sampling. Among the 96,071 screened newborns, blood samples from 126 were above the cutoff level (70 ng/mL), corresponding to a recall rate of 0.12%. In two newborns with high IRT blood concentrations (147 and 225 ng/L), a second sample was not obtained because the newborns died from unknown causes soon after the first sample was taken. In this study, seven CF cases were confirmed by the IRT/IRT/ST algorithm strategy, yielding a birth prevalence of 7.2 per 100,000 newborns (1:13,724 newborns).
IRT blood concentration, sweat chloride values, sweat test interpretation, NBS CF diagnosis, and spectrum of
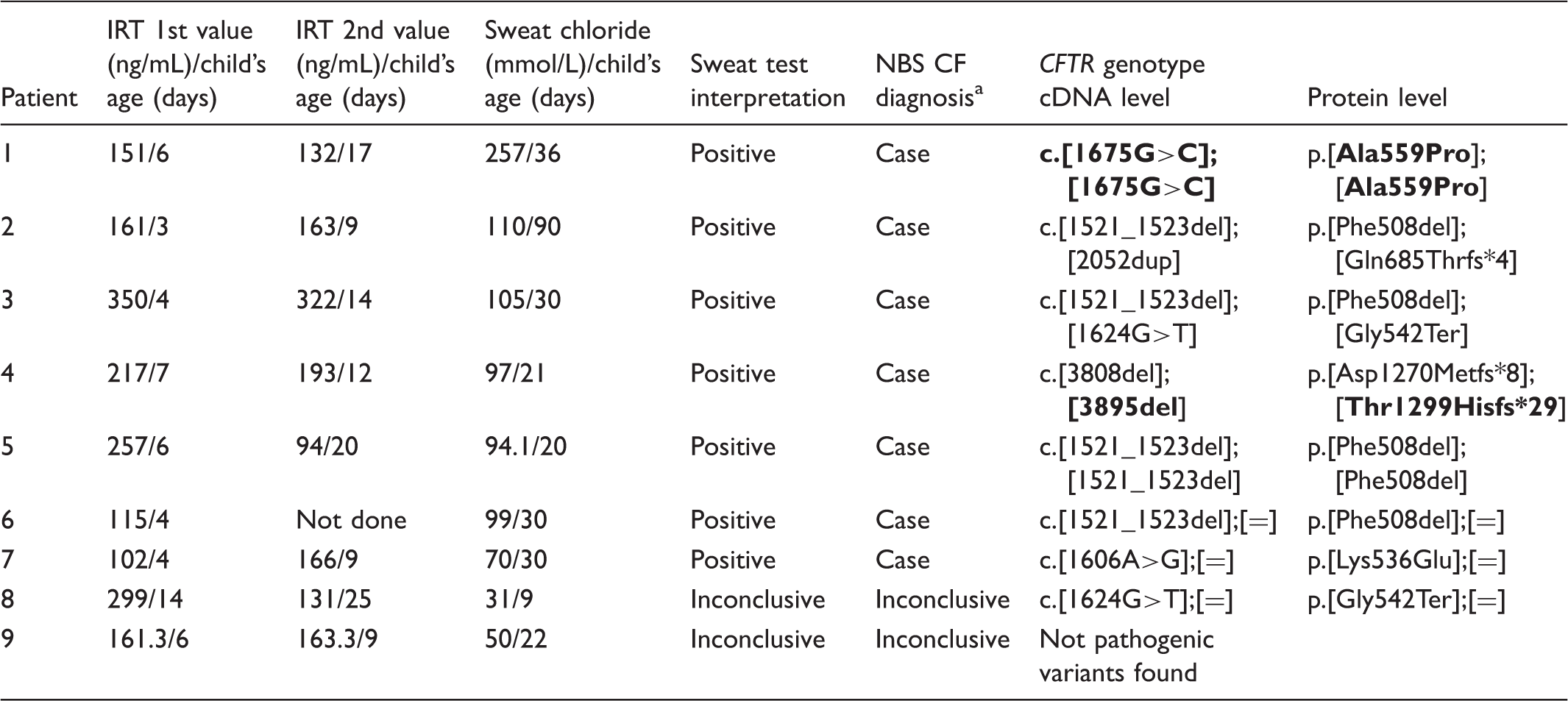
CF: cystic fibrosis; IRT: immunoreactive trypsinogen; NBS: newborn screening.
Complete sequencing of the
In accordance with the protocol algorithm (Figure 1) based on 2015 CF Foundation diagnosis consensus conference recommendations related to diagnosis of CF in the screening populations. 15
Discussion
We used a simple IRT/IRT/ST strategy (Figure 1) to detect seven CF cases. In this work, we had an extremely low recall rate (0.24%–0.9%) compared with that reported elsewhere.1,31,32 The explanation for this may be related to the ethnic characteristics of the population, with the small screened sample (less than 100,000 newborns), or with our cutoff value (70 ng/mL), which is higher than that used in other programs (50–62 ng/mL).1,3
Prior to our study, the occurrence of CF in Mexico had been estimated mainly based on the number of subjects diagnosed with symptoms (∼1:8,500)
2
, but details of the geographic origins of cases were not specified. In a large multiethnic NBS study in California, Kharrazi et al.
3
reported differing CF birth prevalences, with the lowest frequency in the Hispanic population (1:9,259), but the term “Hispanic” included not only the Mexican population but also individuals from Central and South America with different genetic backgrounds. Our work is the first determination of birth prevalence of CF using population-based NBS data, finding 7.2 affected per 100,000 newborns (1: 13,724 newborns) in Yucatan. The Mexican contemporary population represents a complex admixture of European, Native American, and African, with important ethnic variations throughout the country.
22
Accordingly, CF birth prevalence and
No false-negative results have been found to date, but not all late-diagnosed CF cases are relayed to the screening laboratory because of the absence of a national surveillance system for this disease.
The ideal time for blood sampling for CF NBS is in the first days of life, 1 but in some circumstances, newborns can only be screened later. In our study, 76.6% of newborns were screened during the first week of life. In Mexico, the official recommendation for NBS sampling is between age three and five days. Our results show that four of seven positive CF cases were sampled at those ages. In the remaining three newborns, the blood sample was obtained later (age six to seven days). In this study, the average age at CF diagnosis confirmation was 1.3 months (Table 1), which is acceptable according to the Laboratory Guide to NBS for CF in the UK and is similar to that in other programs.19,33
As previously described,1,14 we found that IRT blood concentrations were age dependent, with lower values at older ages (Figure 2(c)). Prematurity, low birth weight, and perinatal stress have been also identified as factors that affect IRT blood levels. 1
In our study, two ill newborns had an initially high IRT blood concentration and subsequently died, preventing further studies. In these two cases, the diagnosis of CF seems unlikely, because elevated IRT blood values have been observed among CF-unaffected newborns admitted to neonatal intensive care units, 1 although a diagnosis of CF cannot be discarded.
The only previous data about the
Given the diversity of known and novel
In this study, two novel
The p.(Lys536Glu) [rs148173473] variant was identified in one CF case (Table 1, patient 7) with abnormal and categorically positive ST (>70 mmol/L), but no other pathogenic
Molecular analyses also allowed us to detect one CF carrier (Table 1, patient 8), who subsequently received genetic counseling. Detecting carriers is a controversial issue, with some authors considering this situation to be an unwanted “side effect” of molecular testing of CF through NBS. 6
Conclusion
Using an IRT/IRT/ST strategy, we detected seven newborn CF patients, yielding a birth prevalence of 1:13,724 in southeastern Mexico. As with previous reports, IRT blood concentration was influenced by gestational age and by time of sampling. In the studied population, a
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: VAM and IGI have received honorarium as
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was by sponsored by
