Abstract
Objective
The prevalence of biotinidase deficiency and the frequency of biotinidase gene variants in Brazil are not documented. We aimed to determine the incidence of partial and profound biotinidase deficiency in the state of Minas Gerais, Brazil, and to calculate the frequency of biotinidase gene variants in the newborn screening program of Minas Gerais.
Methods
Neonates (1,168,385) were screened from May 2013 to June 2018. Those detected with abnormal biotinidase activity based on semi-quantitative assays underwent confirmatory serum tests. The biotinidase gene was sequenced in all confirmed cases.
Results
The combined incidence of partial and profound biotinidase deficiency was estimated at 1:13,909 live births (95% confidence limit 1:11,235–1:17,217), much higher than the incidence rates reported in other populations worldwide. The most frequent biotinidase gene variants were p.D444H (allele frequency, 0.016), haplotype c.1330G>C;c.511G>A (p.D444H;A171T), p.D543E, c.310-15delT (intronic), p.V199M, and p.H485Q. Together these accounted for 74.6% of the alleles analysed.
Conclusion
Newborn screening for biotinidase deficiency, which revealed a higher incidence in Minas Gerais, is feasible and plays a critical role in the early identification of affected neonates and prevention of symptoms and irreversible sequelae. Biotinidase gene sequencing is a useful tool to confirm the diagnosis, and also provides valuable information about genetic variability among different populations.
Introduction
Biotinidase deficiency (BD, OMIN#253260) is an autosomal recessive metabolic disorder caused by mutations in the biotinidase gene (BTD). This gene codes for biotinidase enzyme (BTD, EC 3.5.1.12), which plays a major role in biotin metabolism.1–3 Determination of BTD activity helps to identify individuals with profound BD (up to 10% of mean normal activity), and those with partial BD (10% to 30% of mean normal activity). 4 BD clinical features are highly variable, including neurological and cutaneous symptoms. 5 Administration of free biotin at pharmacological doses (usually 10 mg daily) throughout life is a simple, inexpensive, and highly effective form of treatment, that prevents the onset of symptoms when initiated in the first months of life. 6 BD was first included in a newborn screening program in 1984 in the United States, based on a colorimetric method used for semi-quantitative determination of BTD activity in dry blood samples on filter paper, 6 and several countries now include BD in their newborn screening programs. 7
Since 1993, the Newborn Screening Program of Minas Gerais has screened an average of 240,000 babies annually. A pilot study for BD screening conducted in 2007–2008 revealed that the combined incidence of partial and profound forms of BD in 182,891 babies was higher than that in several other parts of the world. 8 Based on these results, BD was officially included in the newborn screening program disease panel in 2013.
Diagnosis of neonatal BD by biochemical techniques can be challenging, as the enzymatic activity is influenced by external factors like improper handling of specimens, improper drying, delayed submission, or exposure to excess heat.9–11 In this context, BTD sequencing can be a useful tool to determine BD status, to support therapeutic decisions of supplementing biotin in these patients.11–13 Additionally, borderline biochemical results can be better evaluated if the underlying BTD mutations are known. More than 200 BTD variants have been described (see http://www.arup.utah.edu/database/BTD/BTD_display.php, http://www.ncbi.nlm.nih.gov/clinvar/, and http://www.hgmd.cf.ac.uk/ac/index.php). However, the accurate frequencies of these variants in Brazil have not been reported.8,12,14,15
This study aimed to determine the incidence of BD in Minas Gerais, as well as the frequency of BTD variants in babies diagnosed with partial or profound deficiency, including that of the most common variant, p.D444H (c.1330G>C),7,9 in the newborn population of Minas Gerais.
Methods
From June 2013 to May 2018, the Newborn Screening Program of Minas Gerais screened 1,168,385 newborns for BD. The biochemical and molecular tests were performed in the laboratories of the Center for Newborn Screening and Genetic Diagnostics (Nupad) of the Federal University of Minas Gerais, Brazil. The BD screening test was performed on dried blood samples on filter paper, collected between days 3 and 5 of life, using Neonatal Biotinidase Kit (Perkin Elmer, Wallac Oy, Turku, Finland) per the manufacturer's instructions. Babies were screened twice, and the results were considered abnormal if both tests on samples collected on filter paper at different times yielded values <60 nmols/min/dL. Additionally, results were considered abnormal when neonates provided inadequate samples for the first test and the second test yielded values <60 nmols/min/dL, when babies provided inadequate samples for both tests, or when the first test for preterm or in-hospital neonates yielded values >60 nmols/min/dL, but the second test yielded values <60 nmols/min/dL.
The families of babies with results considered abnormal were scheduled for clinical consultation, and serum samples were collected from the babies and, when possible, from their respective parents. Serum BTD quantitative assay was performed according to the protocol published by the American College of Medical Genetics Laboratory Quality Assurance Committee. 9 For the calculation of the relative BTD activity in the serum, the average of the reference range of values for BTD activity in Nupad lab, 7.25 nmol/min/mL, was used as 100%. Hence, the reference values for interpretation of the results were: (i) normal BTD activity, 5.2–9.5 nmol/min/mL, (ii) BTD activity suggestive of heterozygosis, 2.2–5.1 nmol/min/mL, (iii) BTD activity suggestive of partial deficiency, 0.8–2.1 nmol/min/mL, and (iv) BTD activity suggestive of profound deficiency, <0.8 nmol/min/mL. Values of 2.2 and 2.3 nmol/min/mL were considered as borderline results. For patients with more than one determination, the highest value was used for classification.
All infants diagnosed with profound or partial BD were given oral supplementation with 10 mg biotin daily, and have been followed up in a specialized outpatient clinic. Exons 1–4 of BTD as well as their flanking regions were sequenced, as described previously, 8 for all cases that were classified as profound or partial BD, or for those with borderline values, as well as for babies whose biochemical categorization changed along the course of our study. Allele-specific polymerase chain reaction for the variant p.D444H (c.1330G>C; rs13078881) was performed with DNA extracted from the dried blood samples on filter paper for 601/634 neonates (94.8%) for whom the fluorimetric test results were abnormal. Specific primers were designed for this study and are available on request. The amplified fragments were subjected to capillary electrophoresis using Qiaxcel DNA Screening Kit and QIAxcel Advanced Instrument (Qiagen, Hilden, Germany) according to the manufacturer's instructions.
This study was approved by the Research Ethics Committee of the Federal University of Minas Gerais, Brazil, approval # 1,532,490. Written informed consent was obtained from either the parents or guardians of the infants.
Results
For 634 out of 1,168,385 babies screened, the results for BD were abnormal. Families of these babies were invited to confirm the abnormal results with quantitative serum BTD activity determination. Of the 634 families, 620 (97.7%) attended a medical appointment and had serum samples collected. For 416 infants (67.1%), the serum BTD activity was also determined in both the parents; in 110 babies (17.7%), blood was drawn only from the mothers. In the remaining 94 families (15.2%), only samples from babies could be collected. We diagnosed a mother and a father from two distinct families with partial BD. Both were followed at the outpatient clinic, but both were asymptomatic during the course of this study.
Among the 620 newborns, 42 (6.8%) had BTD activity in the normal range (5.2–9.5 nmol/min/mL) and 442 (71.3%) had values in the range suggestive of heterozygosis (2.2–5.1 nmol/min/mL). In the remaining 136 (21.9%), the BTD gene was sequenced: 84 were classified as having BD (six with profound BD and 78 with partial BD), 6 presented borderline values, and 46 presented values indicating partial BD or suggestive of heterozygosis, as previously defined. All 136 children were followed up at the specialized outpatient clinic. During the study period, BTD activity was measured (1–6 times) at different times in these 136 children, and in 131 of them, the test was carried out at least twice.
Based on these data, the combined incidence of partial and profound BD was estimated as 1:13,909 live births (1:11,235 to 1:17,217, 95% confidence limit). The incidence of profound BD was 1:194,731 (1:89,286 to 1:424,809, 95% confidence limit) and that of partial BD, 1:14,979 (1:12,003 to 1:18,692, 95% confidence limit).
After sequencing BTD, 36 different mutations were identified, including 9 novel variants, and 27 of that have been previously reported (Table 1). There were 28 missense mutations, 5 base deletions, and 1 nonsense and 2 silent mutations; the majority of these were located in exon 4. In relation to the BTD variants identified in these 136 newborns, 264 alleles were included in the calculation of frequency (Table 2). Eight alleles were excluded because they were found in siblings. Parental BTD sequencing was done for only 16 children. In addition to the well-known haplotype c.1330G>C;c.511G>A (p.D444H;A171T), other inherited cis-mutations identified included one case of c.310-15delT;c.632G>T (intronic;p.R211L), one case of c.1330G>C;c.470G>A (p.D444H;R157H) and seven cases of c.595G>A; c.1413T>C (p.V199M;C471C). The cis configuration was confirmed by gene sequencing of the respective parents in all three haplotypes. In four cases of the well-known haplotype c.1489C>T; c.1284C>T (p.P497S;Y428Y), parental BTD was not sequenced. The silent variant c.1413T>C (p.C471C) was always detected together with c.595G>A (p.V199M), and never detected alone or in cis with any other variant. Notably, one child homozygous for this haplotype was identified.
Variants identified in newborn screening patients in Minas Gerais, Brazil.
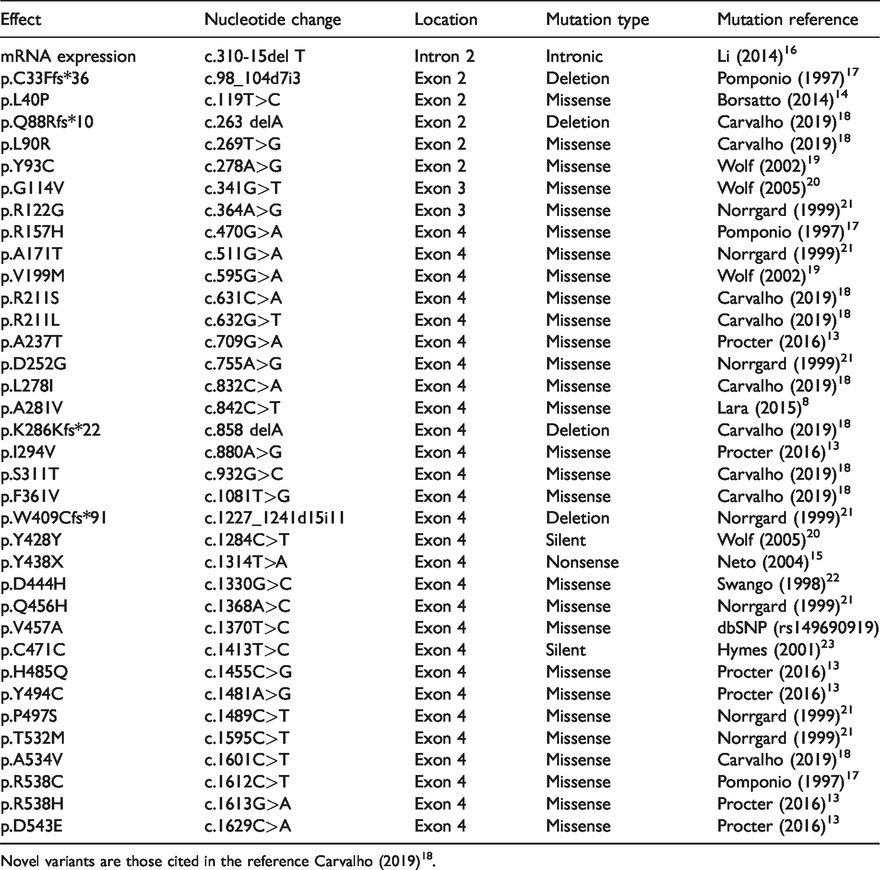
Novel variants are those cited in the reference Carvalho (2019)18.
Frequency of BTD variants identified in newborn screening from Minas Gerais, Brazil, during the 5-year study.
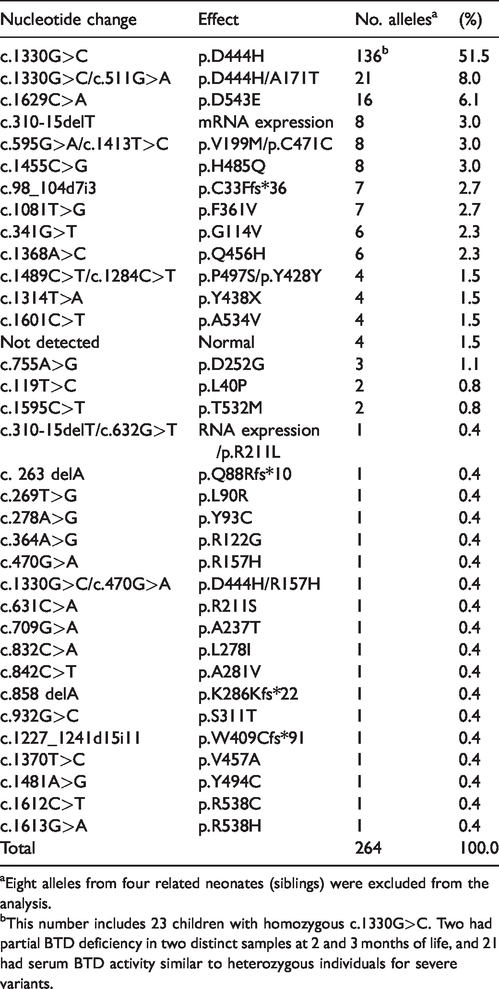
aEight alleles from four related neonates (siblings) were excluded from the analysis.
bThis number includes 23 children with homozygous c.1330G>C. Two had partial BTD deficiency in two distinct samples at 2 and 3 months of life, and 21 had serum BTD activity similar to heterozygous individuals for severe variants.
Among the 136 newborns, only eight were identified in homozygous state (except for p.D444H): three with profound BD (variants p.G114V, p.A534V and haplotype c.1330G>C;c.511G>A (p.D444H;A171T), and five with partial BD, two with p.F361V, two with p.D543E, and one with p.V199M. Clinical consanguinity was reported for only the last child, but genetic tests were not run in any of the couples.
Among the 634 newborns for whom the screening fluorimetric test detected abnormal values for BTD activity, allele-specific polymerase chain reaction for the detection of p.D444H (c.1330G>A) was performed on dried blood samples of 601 babies. We identified 296 newborns homozygous for the p.D444H allele, yielding a frequency of 0.000253 in the five-year period of this study (0.0253%). Based on the Hardy–Weinberg equilibrium, the estimated frequency of the mutant allele was 0.016 (1.6%), and that of the wild type allele was 0.984 (98.4%). The frequency of heterozygous individuals for p.D444H in the newborn population studied was 0.031 (3.1%).
The costs of running such a programme were US$460,000 a year, including screening tests and serum BTD determination, given the number of newborns in Minas Gerais and the incidence of BD detected in the present study. Molecular studies in 21.5% of children who had BTD serum determination added US$2,720 a year to the costs.
Discussion
From over one million newborns screened in the state of Minas Gerais in this study, 84 were diagnosed with BD. The estimated worldwide combined incidence of profound and partial BD is 1:60,089. 24 In the United States, the incidence of profound BD is 1:80,000, and partial BD between 1:31,000 and 1:40,000. 11 In Europe, a study including nine countries showed an incidence of combined BD of 1:47,486. 25 Few studies exist on the incidence of BD in Brazil. The first, in the state of Paraná, found two neonates with BD (one partial and one profound) among 125,000 babies screened during eight months. 26 This was close to the worldwide incidence, 24 but the sample size was about nine times smaller than that in the present study. A smaller study (20,529 neonates) in Maringá (Paraná) found a combined incidence of profound and partial BD of 1:6843, 27 but the diagnostic method was not clearly stated, and it is possible that only the semi-quantitative colorimetric test was used. In a study of 225,136 babies across several Brazilian states, the estimated combined BD incidence was 1:9000, 15 but the authors reported difficulties in the transport and storage of serum samples owing to the large size of the country. As BTD is temperature-sensitive, the possibility of false-positives should be considered. In the present study, BD incidence was estimated at 1:13,909, even higher than that reported in our previous pilot study of 182,891 neonates (1:22.861). 8 The strengths of our study are the population size, the methodologies used, and adherence to stringent recommended quality control processes.9,11 It is possible that BD incidence in Brazil may be among the highest in the world, and only lower than in countries with high consanguinity rates, such as Turkey, where the combined BD incidence is 1:7116. 28
In our study, 36 different mutations were identified in the BTD gene, except in exon 1. This can be attributed to the fact that BTD has two potential AUG translation start codons; the second and more frequently used codon is located in exon 2.5,23,29 All mutations identified in this study were exonic, except for a single intronic variant, c.310-15delT, which was not detected in any other study conducted in the Brazilian population. Cis mutations have also been identified in BTD, which has been reported for BD in several databases.
The most common BTD variants observed in newborn screening in the United States are p.D444H, p.Q456H, haplotype c.1330G>C;c.511G>A (p.D444H;A171T), p.C33Ff*36, and p.R538C, which account for about 60% of the mutant alleles. 21 In the present study, the most frequent variants were p.D444H, haplotype c.1330G>C;c.511G>A (p.D444H;A171T), p.D543E, c.310-15delT (intronic), p.V199M, and p.H485Q, which together account for 74.6% of the alleles analysed. With the exception of the first two, these markedly differ from those reported in the United States. 14 In other Brazilian studies,12,15 p.D444H was the most frequent variant, followed by p.C33Ffs*36 and p.D252G. These two variants were also detected in our study, but were not the most frequent. Conversely, two variants in exon 3 (seven alleles; Table 2) identified in our study have not been reported previously. Further, 11 novel variants were detected by our group.8,18 These observations illustrate the genetic diversity in the Brazilian population.
In four newborns with partial BD diagnosed based on their serum BTD activity, we detected only p.D444H variant in heterozygosis. BTD activity was repeated in samples collected one or two months apart. The most elevated values considered for BD classification of these four children were 1.5, 1.6, 2.1, and 2.2 nmol/min/mL, respectively. This is not compatible with the observed biochemical phenotype, because this variant usually yields BTD activity of approximately 75% of the reference value for normal individuals.11,30 On sequencing BTD in these patients, no mutations were found in the second allele. As described previously, gene deletions or epigenetic phenomena could be the underlying reason, as these children had at least two serum BTD determinations at distinct moments. However, gene deletions could not be detected by the method used in the present study. Alternative methods therefore would be necessary.31,32
In the present study, the allelic frequency of the most common variant, p.D444H, was 51.5% among patients with BD, similar to that reported by another study on the Brazilian population (46.7%). 12 The frequency of the p.D444H allele in our study was estimated to be 0.016. This frequency is lower than the first reported figure of 0.039 in the United States, 32 and also lower than that in the cited Brazilian report (0.04). 12 According to the 1000 Genomes Project, the frequency of rs13078881 (D444H) varies by population, being highest among those with European ancestry (0.043), and very low (0.0023) in those with African ancestry (www.ncbi.nlm.nih.gov/projects/SNP/snp_ref.cgi?do_not_redirect&rs=rs13078881). The slave trade from Africa to Minas Gerais in the 17th–19th centuries is historically compatible with the lower frequency of the D444H allele in our study.
Although newborn BD screening is not recommended in all countries, it meets 9 of the 10 criteria set out by Andermann et al., 33 especially in regions with a high incidence of BD, such as ours. The eighth principle that states “There should be an agreed policy on whom to treat as patients” is not entirely met, because it is not known for certain whether children with partial BD need biotin supplementation to remain symptom-free. However, treating these children, as is usually recommended, 10 was demonstrated to be cost-effective in a study in Spain, where the reported incidence of combined BD in 540,000 neonates was 1:22,500. 34
Conclusions
The incidence of partial BD in the State of Minas Gerais may be among the highest in the world, excluding countries with high rates of consanguineous marriage. Given the high BD incidence, newborn screening plays a major role in early identification of the disease in neonates and preventing the development of symptoms and irreversible sequelae. The genotypic variability in the present study is noteworthy, and reflects the multi-ethnic population of Minas Gerais. BTD sequencing is a useful tool, not only to confirm BD cases screened in this newborn program, but also to obtain valuable information about genetic variability among different populations. Although BD has been included in the newborn screening program only recently, our study demonstrates that such screening is feasible, useful, and probably cost-effective in Minas Gerais.
Footnotes
Author contributions
Study concept and design: NOC, JNJ, RCN, MBV; acquisition of data: NOC, RCN, ALPS; molecular and biochemical analysis: NOC, GLPF, DMN, RVPL, DMC; analysis and interpretation of date: NOC, RCN, MBV; drafting of the manuscript: NOC, RCN, MBV; critical revision of the manuscript for important intellectual content: NOC, JNJ, GLPF, DMN, RVPL, DMC, ALPS, RCN, MBV; final proofreading: MBV, RCN, NOC.
Acknowledgments
The authors thank the laboratory members and scientific staff of the Center for Newborn Screening and Genetic Diagnostics (Nupad/UFMG) for their commitment and logistics, and the team responsible for following up children in the outpatient clinic. We would like to thank Editage (![]() ) for English language editing.
) for English language editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Nupad-UFMG; Brazilian Ministry of Health; and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq, Brazil) to Marcos Borato Viana (grant number 305261/2017–7).
