Abstract
Objective
To examine the diversity in paediatric vision and hearing screening programmes in Europe.
Methods
Themes for comparison of screening programmes derived from literature were used to compile three questionnaires on vision, hearing, and public health screening. Tests used, professions involved, age, and frequency of testing seem to influence sensitivity, specificity, and costs most. Questionnaires were sent to ophthalmologists, orthoptists, otolaryngologists, and audiologists involved in paediatric screening in all EU full-member, candidate, and associate states. Answers were cross-checked.
Results
Thirty-nine countries participated; 35 have a vision screening programme, 33 a nation-wide neonatal hearing screening programme. Visual acuity (VA) is measured in 35 countries, in 71% of these more than once. First measurement of VA varies from three to seven years of age, but is usually before age five. At age three and four, picture charts, including Lea Hyvarinen, are used most; in children over four, Tumbling-E and Snellen. As first hearing screening test, otoacoustic emission is used most in healthy neonates, and auditory brainstem response in premature newborns. The majority of hearing testing programmes are staged; children are referred after 1–4 abnormal tests. Vision screening is performed mostly by paediatricians, ophthalmologists, or nurses. Funding is mostly by health insurance or state. Coverage was reported as >95% in half of countries, but reporting was often not first-hand.
Conclusion
Largest differences were found in VA charts used (12), professions involved in vision screening (10), number of hearing screening tests before referral (1–4), and funding sources (8).
Introduction
Childhood sensory functions play a key role in intellectual and social development. Vision or hearing impairment affects both personal and societal health of children. Earlier detection of visual or hearing deficits improve outcome.1–4 Vision and hearing screening programmes are based on the same general principles, but vary both within and across European Union (EU) countries, regarding tests used, age of testing, frequency of testing, professions involved in screening, referral procedure, funding, and coverage. Such differences can result in health inequities. No screening, or screening with little population coverage, can result in delayed provision of the correct treatment and increased disease burden. Excessive screening can result in inappropriate interventions and increased costs for health care systems.
Vision screening and subsequent treatment has reduced the occurrence of insufficiently detected and treated amblyopia. 5 In the Netherlands, amblyopia is now detected more than two years earlier than in the 1970s. 6 Early screening and detection of hearing disorders, and timely intervention (eg. cochlear implantation or hearing aid) largely prevents delayed language development,1,2,7 and also improves general developmental outcome at age 3–5. 3
Despite increased consciousness that vision and hearing screening is effective, differences exist in implementation between countries. A 2002 survey of vision screening programmes in 190 countries found that screening was often state funded, visual acuity (VA) was always tested, and that in the EU screening was predominantly voluntary. 8 In a 2012 survey of the International Orthoptic Association, 98% of responders indicated that vision screening programmes existed in their country, (44% were national programmes). Screening was performed by a wide range of professionals. 9 An overview of universal newborn hearing screening (UNHS) in 24 European countries from 2004-2006 showed that in several countries UNHS programmes reached more than 95% of all neonates, but in many other countries programmes were recently introduced or were only partially functioning. 10 Other reports on national neonatal hearing screening programmes raise issues on implementation, test procedures, type of tests, coverage, detected cases of hearing loss, and costs.11–19
A Health Technology Assessment review in 200820 re-examining the cost-effectiveness of vision screening up to age 4-5 (following previous report in 1997 21 ) found that, based on the accepted value of a Quality-adjusted life year, the cost-effectiveness of screening for amblyopia depends on the long-term utility effects of unilateral vision loss and that there was currently no sustainable evidence of utility loss that would render any form of screening likely to be cost-effective. 20 Keren et al. concluded that UNHS in general has the potential for long-term cost savings compared with selective hearing screening and no screening. 22 Burke et al found that the cost-effectiveness of hearing screening depended mainly on the cost of the screening intervention per patient and on the prevalence of hearing loss in the population. 23
We aimed to compile an inventory of population-based vision and hearing screening programmes for children in Europe, and to quantify and examine the differences. This study should assist those countries without a screening programme and new EU member states in selecting which screening protocol to adopt. If large differences between programmes in EU countries are found, further study on the relative costs and effectiveness of the different approaches to screening will be necessary.
Methods
Drawing particularly from five major cost-effectiveness analyses,20,21,23–25 we selected the following items to formulate vision (Q1) and hearing (Q2) screening questionnaires:
Type of tests, eg. visual acuity chart or hearing screening device used (otoacoustic emission [OAE], automated auditory brainstem response [aABR]) Professions involved in screening, eg. nurses, orthoptists, doctors Funding, eg. State, health insurance Coverage, percentage of screened children.
Questions formulated in a focus group were structured as multiple-choice with room for comments and multiple answers (See Appendix, Q1, Q2). All forms of screening for vision or hearing problems were included (eg. inspection of the eyes was also counted as form of vision screening). To obtain a broader perspective of screening systems, a short public health questionnaire (Appendix Q3) to provide background information on screening and screening systems in all countries was developed through extrapolation of the vision and hearing questionnaires.
In each of the 28 EU full member states, five candidate states, potential candidate state Albania and associated states Israel, Moldova, Norway, and Switzerland, a paediatric ophthalmologist, orthoptist, otolaryngologist, audiologist, and screening professional were selected, based on their involvement in paediatric vision and hearing screening, and asked to complete the questionnaires for their own country. Public health representatives were identified through the Ministries of health or recommendation from the vision and hearing representatives.
Glossary.

Questionnaires were emailed from December 2013 until April 2014. Clinicians involved in population-based screening were identified and their answers were cross-checked with those given by general screening professionals. If answers were ambiguous the questionnaires were returned to both the clinician and the screening professional and they were asked to contact each other to agree corrections. Overviews of the questionnaire answers were circulated three times to all representatives. All representatives were asked to review and correct any errors in the overviews for their own and neighbouring countries. The overviews were also checked by external experts, involved first-hand in vision and hearing screening.
Results
Eligible countries.
(
Vision
Information on vision screening programmes was obtained from 36 countries including two Belgian regions. No information could be obtained from Albania, Macedonia, and Moldova. Thirty-five countries have a vision screening programme in place. In Belgium, Bulgaria, Estonia, France, Romania, Spain, and Switzerland this is a regional programme. In several countries with a national vision screening programme in place, regional differences in screening protocols exist.
Infant and preverbal screening tests
Infant screening (age 0–4 months) included inspection, fixation, red reflex testing, Hirschberg test, Brückner test, Cover test, pupillary reflexes, and motility. Most countries perform a combination of two or more of these tests. In Bulgaria, Greece, and Poland no infant screening is performed. In Germany only eye inspection is performed. In Ireland, Montenegro, and Spain, eye inspection is combined with red reflex testing. In Cyprus, Italy, Lithuania, and Malta only red reflex testing is done. In Latvia this is combined with motility testing. Preverbal screening (age 6-30 months) includes the same tests. Preverbal screening is not performed in eight countries, but most countries combine two or more tests.
Visual acuity (VA) measurements
In all countries VA is tested, but the age of the first measurement varies from 3--7 years. In a third of countries VA is tested once, one third twice, and in one third more than twice. In most countries, VA measurements are repeated at an older age. In children aged 4 and younger VA charts are most commonly picture charts and the Lea Hyvarinen chart, above age 4, Tumbling-E and Snellen are most often used.
Personnel and referral
Screening is mostly performed by paediatricians, ophthalmologists, and/or nurses. In most countries children are referred to ophthalmologists for further examination; in Latvia they are referred to the General Practitioner (GP), in the UK they may also be referred to joint orthoptic and optometry clinics or optometrists, in Malta to either the orthoptist or optometrist, and in the Netherlands they are mostly referred via the GP to an orthoptist or ophthalmologist, but sometimes directly to an orthoptist, ophthalmologist, optometrist, or optician.
Funding
In most countries vision screening is free, except for the Czech Republic, Switzerland, and Turkey. Funding is 33% (partially) provided by Health Insurance and 53% (partially) by the State. Parents and charity pay (part of the) screening in the Czech Republic, Latvia, Romania, Slovakia, Spain and Turkey.
Coverage
Coverage varied from just starting (Estonia, Portugal, Turkey) to more than 95% in Austria, Czech Republic, Denmark, Finland, Flanders, Germany, Hungary, Iceland, Luxembourg, the Netherlands, Norway, Serbia, Slovenia, Sweden, and parts of the UK. Coverage of different testing time points varied, as did number of children screened, dependent on the age at which testing was performed. The highest coverage percentage was regarded as coverage for each particular country.
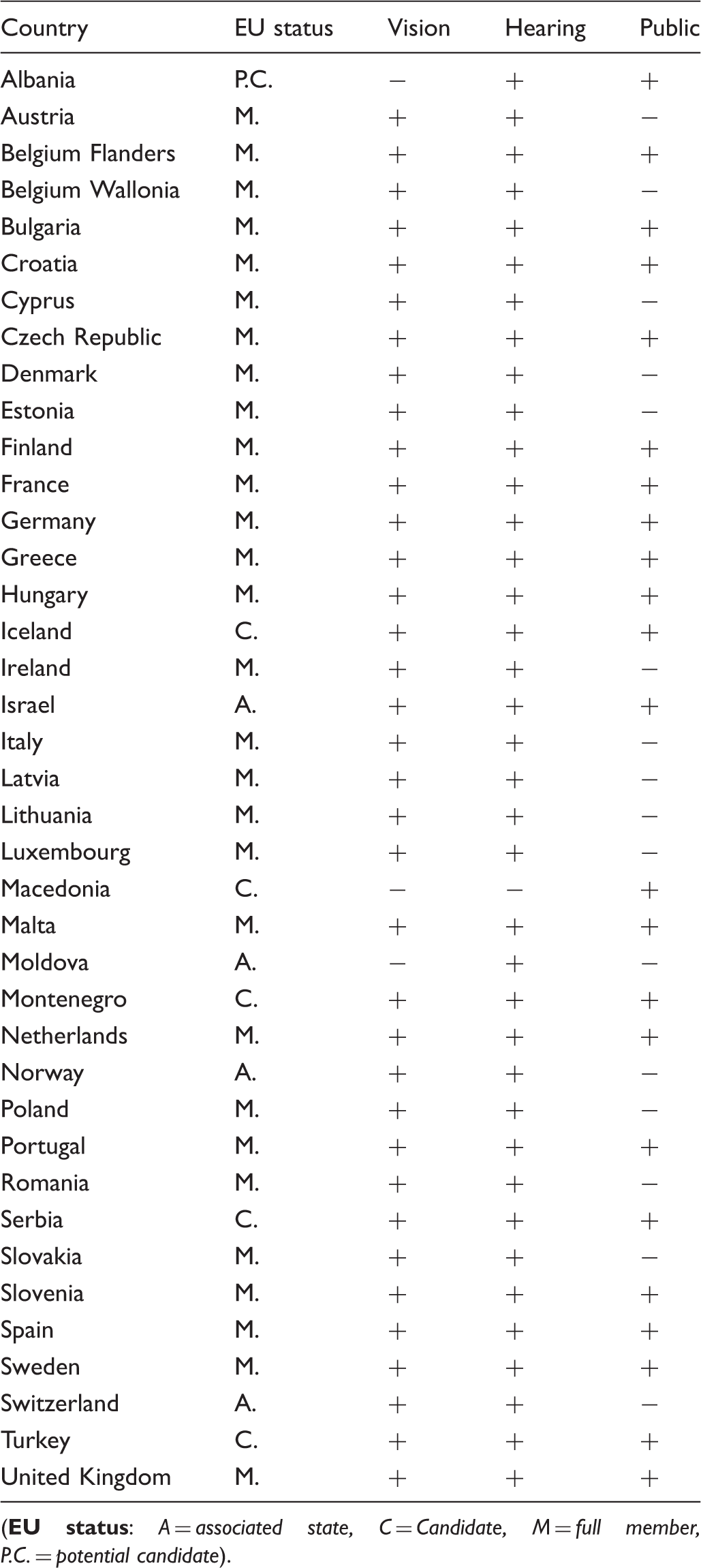
Vision screening programmes in 36 European countries.
Hearing
Information on neonatal hearing screening programmes was obtained from 38 countries (including two Belgian regions). No information could be obtained from Macedonia. Nationwide UNHS programmes exist in 33 of 38 countries. Malta has nationwide selective screening only for infants from neonatal and paediatric IC units. In Bulgaria, Moldova, and Serbia local selective screening programmes for high risk groups (premature newborns) exist. In Albania a pilot nationwide UNHS programme was discontinued due to lack of funds.
Tests
The most widely used audiometric test is OAE. Flanders has used aABR in all neonates, but in 2013 introduced additional auditory steady state responses (ASSR). Some regions in Denmark, Estonia, France, Germany, Spain, and Sweden use aABR, OAE, or both in the same infant as first test in healthy babies. In nearly all programmes both ears are tested, except in Finland and Switzerland, where one or two ears are tested, depending on the institution or the presence of risk factors. Testing is not staged in five countries, two-staged in 13 countries, three-staged in 19 countries, and 4-staged in one country. aABR is used as final stage in the majority of countries. In high risk groups, eg. premature newborns, most programmes use aABR or a combination of OAE and aABR, but in eight countries OAE only is used. In Wallonia (Belgium) all premature infants undergo full ABR. In less than half of the countries, a hearing test in pre-school or early school age children is a regular part of health screening programmes.
Referral
Neonates who do not pass the test are referred to a combined audiology/ ear, nose, throat (ENT) institution in most countries, in some countries to an audiologist, and in a few countries to an ENT specialist.
Funding
In most countries the government or health insurance finances the neonatal hearing screening programme. Other reported funding includes hospital, parents, and private funds.
Coverage
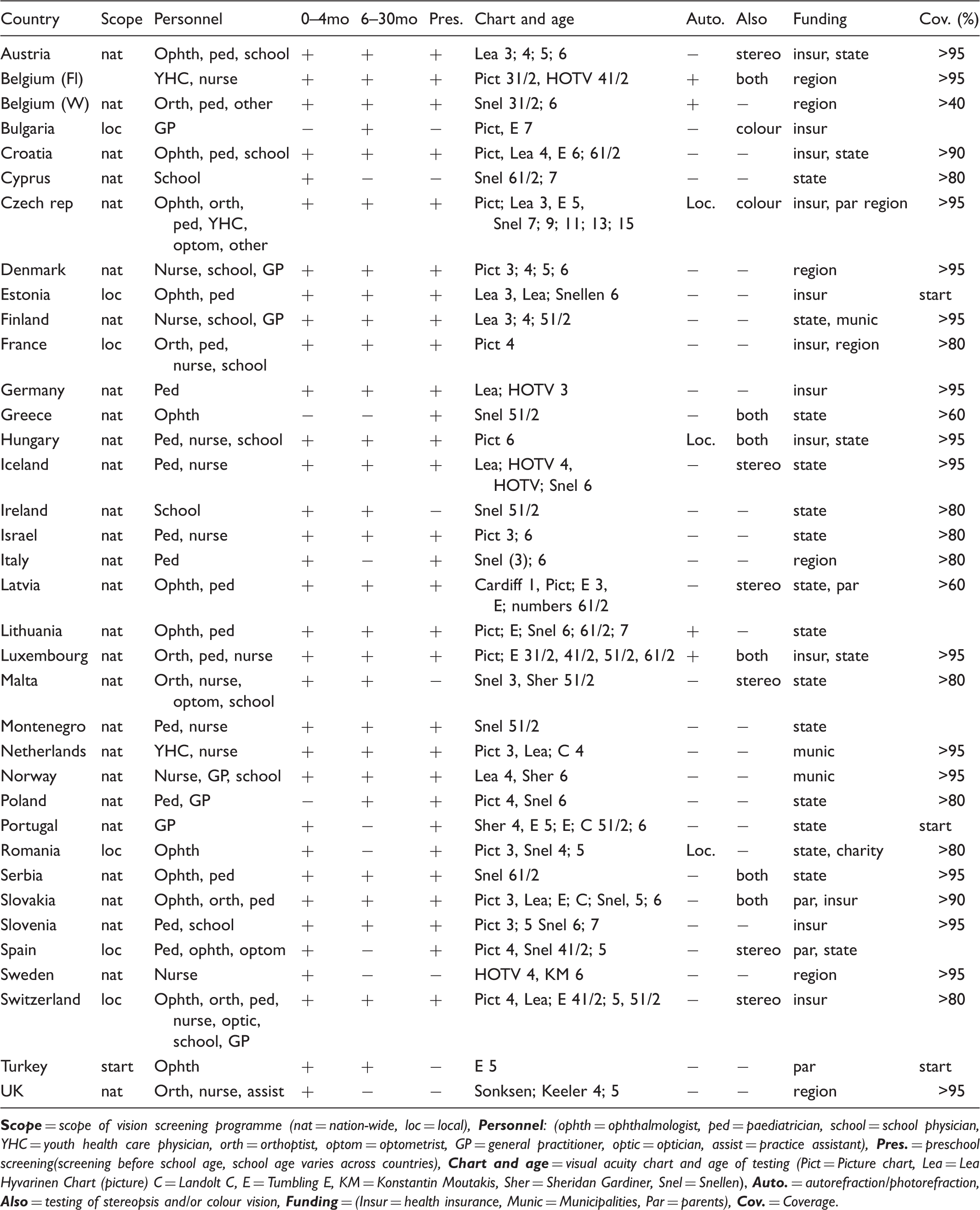
Overview of neonatal hearing screening programmes in 38 European countries.
Public health
Extra information on public health screening programmes was obtained from 23 countries including one Belgian region (Flanders). All have a public health screening programme, but in Albania, Belgium, and Spain this is a regional programme. In the Netherlands and Sweden a combination of national and regional programmes exists. Almost all countries have a programme for all children, except Albania, where screening is selective. Screening is not free in Albania, Bulgaria, and Czech Republic, and is compulsory in Bulgaria, Flanders, Greece, Hungary, and Turkey.
Tests
Weight, height and head circumference are measured in all countries, cardiac function in all but Albania, lung function in all but Albania and Flanders, vision in all but Albania and Turkey, hearing in all but Albania and Malta, motor skills in all but Czech Republic and the UK, speech and language development in all but Albania, Bulgaria, Czech Republic, and the UK, cognitive development in all but Albania, Czech Republic, Flanders, and the UK. Psychosocial development is assessed in all countries but Albania, Bulgaria, Czech Republic, Flanders, Germany, Israel, Sweden, and the UK.
Referral, funding and coverage
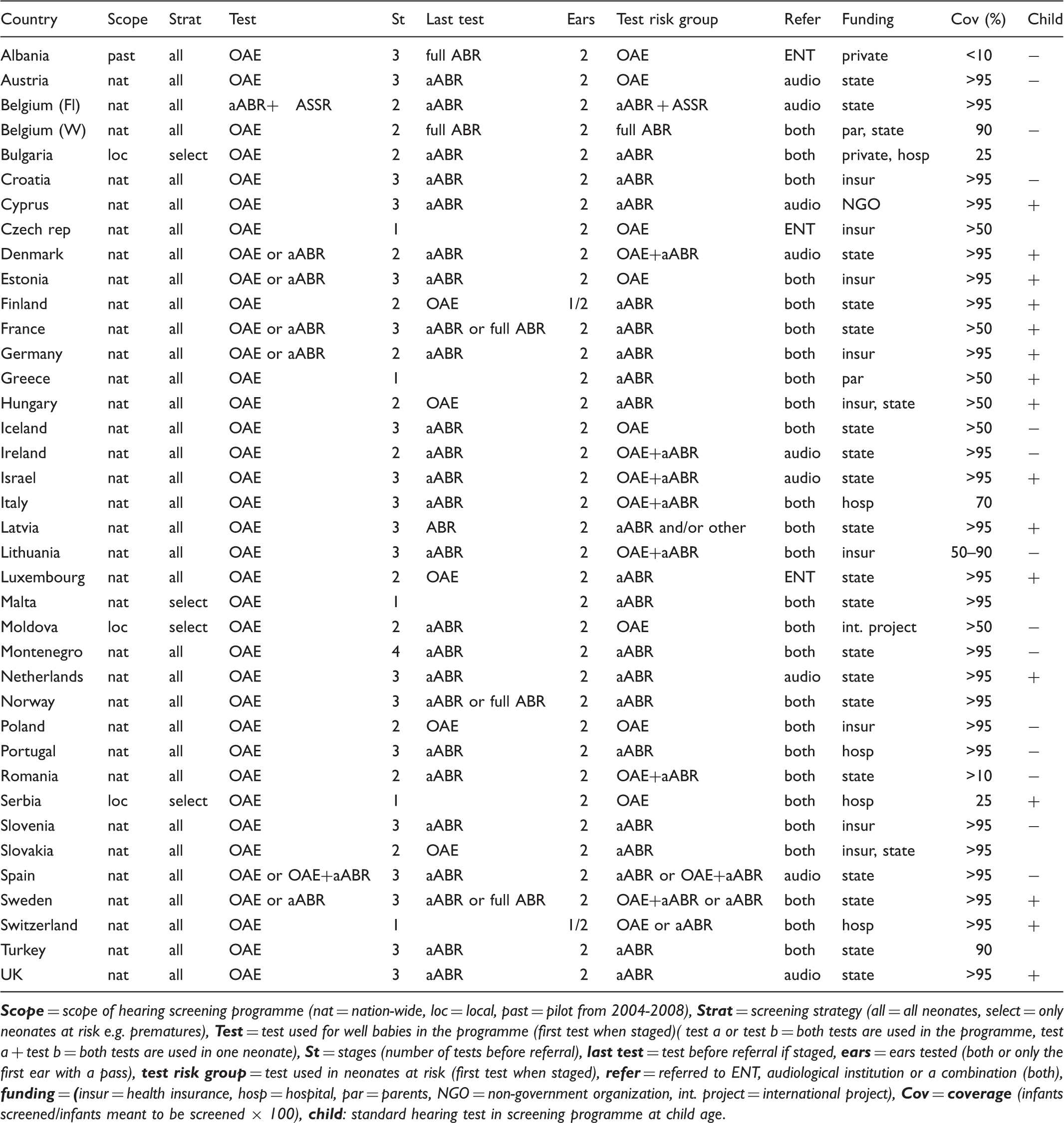
Public health screening programmes in 23 European countries.
Questionnaire answer check
Changes were made based on the first round of questionnaire answers. In hearing screening data: for Belgium (Flanders) the ASSR was added as test for neonates at risk; for Finland “testing one ear” was changed to “testing one ear or both ears”; for France “testing one ear and testing both ears” was changed to “always testing both ears”; for Italy coverage of “ >95%” was changed to “70%”; Malta selective screening, not population-wide screening, was confirmed; for Poland “non-staged screening” was changed to “staged screening”; for Israel, Italy, Lithuania, and Switzerland “only aABR testing” for neonates at risk was corrected to “OAE and/or aABR”.
Vision screening data was revised: for Austria funding was changed from “health insurance” to “health insurance and state”, for Belgium (Flanders) personnel was changed from “nurse” to “nurse and youth health care physician”, testing of stereopsis and colour vision was added and VA chart was changed from “Landolt C” to “Pictures and HOTV”; for Croatia VA chart was changed from “only Tumbling-E” to “Pictures, Lea and Tumbling-E”; for Czech Republic Pictures and Lea chart were added; for Denmark the “Snellen chart” was changed to “Pictures” and coverage was changed from “>80%” to “>95%”; for Iceland Snellen chart was added; for Israel coverage was changed from “>95%” to “>80%”; for Italy funding was changed from “state” to “regions”; for Latvia “Picture chart and Tumbling-E” was corrected to “Cardiff, Pictures, Tumbling-E and numbers”; for Norway the Sheridan Gardiner chart was added; for Slovenia autorefraction was corrected as in Slovenia autorefraction is only performed in ophthalmology clinics for referred children and not for screening; for Sweden the Konstantin Moutakis chart was added; for the UK funding was corrected from “state” to “regions”, and personnel were changed from “orthoptist, optician and optometrist” to “orthoptist, nurse and practice assistant”.
Discussion
This study showed that large differences exist in tests used, age, and frequency of testing in paediatric population-based vision and hearing screening programmes throughout the EU. First measurement of VA varies from ages 3–7, but in most countries it is measured before age 5. In children aged 3–4, picture charts, including Lea Hyvarinen, are used most; in children over 4 Tumbling-E and Snellen. Vision screening is performed mostly by paediatricians, ophthalmologists, or nurses. As a first hearing screening test, OAE is used most in healthy neonates, and ABR in premature newborns. The majority of hearing testing programmes are staged. Children are referred after one, two, three, or four abnormal tests. Funding is by health insurance, state, regions, municipalities, charity, hospital, parents, or private funding. A high coverage is reached in most countries for both vision and hearing screening.
Our study was limited by the difficulty in obtaining referenced or first-hand data sources from respondents. Where possible we tried to maintain the quality of our data by involving clinicians involved in population based screening, and cross-checking their answers with those from general screening professionals. Obtaining accurate information on funding and coverage was the most difficult. Information on tests, personnel, and age was easier to acquire. Coverage may have been overestimated by the country representatives.
Screening for vision and hearing deficits has similarities, but also differences. An essential difference is that objective tests are available for hearing screening at a very early age, enabling screening directly after birth. This is probably the reason for the more uniform approach and higher coverage reported for hearing screening compared with vision screening. We assumed that the personnel operating the screening apparatus at the hospital or during home visits would be a technician, so did not ask the profession explicitly. The only two tests for hearing screening are OAE and aABR, so the major difference in hearing screening is the number of stages before referral. Multiple stage screening is more expensive, but yields higher specificity, which reduces the number of false referrals to specialized and expensive audiological care centres.23,26 There are most frequently two or three stages of screening before referral, generally with OAE as the first test and aABR as last test. It has been suggested that three stages may be more cost-effective 26 , but this is not based on combined use of OAE and aABR. Pre-school or early school-age hearing tests may potentially discover hearing loss acquired during the years after birth, but this occurs rarely and these tests have been abolished in many European countries.
The wide differences between European screening programmes may have occurred because these programmes arose piecemeal, before robust evidence on effectiveness and cost-effectiveness was available to guide protocol design or implementation. In addition, most preventive health care programmes are government funded and, therefore, competition is lower than in curative health-care. Further assessment is needed on the influence of funding source (eg. state, health insurance, or municipalities) on the efficiency of screening.
Further study should also be undertaken into the relative costs and effectiveness of different approaches to screening, as in Europe, 12 different VA charts are used, 10 professions are involved in vision screening, one to four hearing screening tests take place before referral, and eight funding sources are involved. The large number of screening tests used in vision screening should be compared. Efficiency of screening (ie. sensitivity and specificity per euro) should be calculated for screening performed by different screening professions.
We now plan to include data sources in a much larger and more detailed questionnaire. The EUS€REEN study group, an EU-wide consortium (see list at end of paper), is currently preparing a Europe-wide study to compare and optimize the cost-effectiveness of vision and hearing screening, and give country-specific advice in all candidate, associate, and full EU-member states.
Footnotes
Acknowledgements
We thank Ferdinando Grandori, Gwen Carr, Anneke Meuwese-Jongejeugd, Bert van Zanten, Cécile Guérin, Karel Hoppenbrouwers for their information, comments and corrections.
Financial disclosure
None of the authors have financial relationships relevant to this article to disclose. No funding was secured for this study. The authors had no commercial or propriety interest.
Conflict of interest
None of the authors have conflicts of interest to disclose.
Consortium member list
EUS€REEN consortium: EU vision:
Austria: Langmann A; Lindner S; Gaugl H (Medical University Graz), Belgium Flanders: ten Tusscher M (University Clinic Brussel), Guérin C; Hoppenbrouwers K, van Lammeren M ( University Leuven), Boelaert K (Kind en gezin, Brussel), Godts D (University Hospital Antwerp), Belgium Wallonia: Paris V (CHU de Liège), Bauwens A (Bastogne), Bulgaria: Stateva D (Medical University Pleven), Croatia: Petrinovic-Doresic J; Bjelos M (University Eye Clinic University Hospital “Sveti Duh”, Zagreb), Novak-Stroligo M (Clinical Hospital Rijeka), Alpeza-Dunato Z (Rijeka University Hospital Center), Cyprus: Gavrielides Michaeloudes M, Czech Republic: Dostálek M (Center of Paediatric Ophthalmology, BINOCULAR s.r.o. Litomysl & Masaryk's University, Brno), Zobanova A (Prague), Jerabkova A, Denmark: Hesgaard H, Welinder LG (Aalborg University Hospital), Sandfeld L (University of Copenhagen, Roskilde Hospital), Larsen S (Squinting Eyes, Copenhagen), Estonia: Levin M (Ida-Tallinn Central Hospital), Klett A; Somma K (Tallinn), Ismagilova S, Finland: Hyvärinen L (Developmental Neuropsychology, University of Helsinki), France: Thouvenin D (Purpan University Hospital, Toulouse), Coursager K, Germany: Elflein H; Pitz S (University Hospital, Johannes Gutenberg-University, Mainz), Lenk-Schaefer M (Nürnberg), Van-Waveren M (Tübingen), Greece: Ziakas NG (Aristotle University Thessaloniki), Polychroniadis Scouros S (Hygeia Hospital Athens), Hungary: Knezy K; Nemeth J (Semmelweis University, Budapest), Soproni A (Anna Soproni's Private Eye Clinic Budapest), Facskó A; Berkes S (University of Szeged), Iceland: Gudmundsdottir E (Landspitalinn, University of Iceland, Reykjavik), Ireland: McCreery K (Blackrock Clinic, Dublin), Israel: Morad Y (Assaf Harofeh Medical Center, Tel Aviv University, Zrifin), Ancri O (Goldschleger Eye Institute, Sheba Medical Center, Sackler Faculty of Medicine, Tel Aviv University), Italy: Nucci P; Serafino M; Lembo A (University Hospital San Giuseppe, Milan), Bottin D (Hospital of Bolzano), Latvia: Valeina S (Riga Children's University Hospital), Lithuania: Misevice A (Clinic of Ophthalmology, Kaunas University of Medicine), Asoklis RS (ENT and Eye Clinic, Vilnius, Lithuania), Luxembourg: Planata-Bogdan B (Service Orthoptique et Pléoptique, Esch-sur-Alzette), Malta: Francalanza M (Mater Dei Hospital, Malta) MJ Gouder, Montenegro: Jovovic N; Pojuzina N (Children's Hospital, Podgorica), Netherlands: Sjoerdsma T (Municipal Health Service, Amsterdam), van Rijn R (VU University Medical Center, Amsterdam), Norway: Osnes-Ringen O, Moe M (Center for Eye Research, Oslo University Hospital), Poland: Bakunowicz-Lazarczyk A (Medical University of Bialystok), Portugal: Reich-d'Almeida F (Faculty of Medical Sciences New University Lisbon), Marques Neves C (Centro Hospitalar de Lisboa Norte, Lisbon), Reich d' Almeida I, Oliveira M, Romania: Vladutiu C (Clinica Oftalmologică Cluj-Napoca), Serbia: Stankovic B (Faculty of Medicine University of Belgrade, Institute of Ophthalmology, Clinical Center of Serbia); Djokić V (Institute of Ophthalmology, Clinical Center of Serbia) Slovakia: Gerinec A (Klinika Detskej Oftalmológie DFNsP-LF UK, Bratislava), Slovenia: Stirn Kranjc B (University Medical Centre, Ljubljana), Spain: Gomez-de-Liano Sanchez R (Hospital Clínico San Carlos, Madrid), Rajmil L; Prats B (Catalan Agency for Health Quality and Assessment), Sweden: Nilsson J (Institute of Neuroscience and Physiology, The Sahlgrenska Academy, University Gothenburg), Flodin S (The Sahlgrenska Academy, University Gothenburg), Switzerland: Landau K (University of Zurich), Sturm V (Kantonhospital St. Gallen), Zuber C (Cabinet orthoptie Neuchâtel et La Chaux-de-Fonds), Glauser V, Turkey: Atilla H (Ankara University), UK: Horwood AM (University of Reading), Williams C (University of Bristol), Shea S (Orthoptic Department, North West Wales NHS Trust, Ysbyty Gwynedd, Bangor), Griffiths H; J Carlton (University of Sheffield).
EUS€REEN consortium: EU hearing:
Albania: Birkena Qirjazi, (University of Medicine of Tirana), Austria: Markus Gugatschka, (Medical university Graz), Belgium Flanders: Luc Stappaerts (Kind en Gezin, Brussels), Belgium Wallonia: Bénédicte Vos (Centre d'Epidémiologie Périnatale-School of Public Health/Université libre de Bruxelles, Brussels), Bulgaria: Mario Milkov (St. Petka Eye and Ear Clinic, Varna), Croatia: Marko Velepic (Rijeka University Hospital Center, Rijeka), Cyprus: Chryssoula Thodi (European University Cyprus, UNHS Programmeme), Czech Rep: Josef Syka (Czech Academy of Science, Prague), Denmark: Therese Ovesen (Aarhus University Hospital), Estonia: Liina Luht (East Tallinn Central Hospital), Finland: Riina Niemensivu; Antii Aarnisalo (Helsinki University Hospital), France: Françoise Denoyelle (Hopital Necker-Enfants Malades, Paris), Germany: Annerose Keilmann (Johannes Gutenberg-Universität Mainz), Katrin Neumann (Ruhr-University Bochum), Greece: Thomas Nikolopoulos (Athens University School of Medicine), Hungary: Zsolt Beke (Sanct Rokus Hospital, Baja), Iceland: Ingibjörg Hinriksdóttir (National Hearing and Speech Institute of Iceland, Sími), Ireland: Ann O'Connor (RCSI Surgery, Dublin),Israel: Lisa Rubin (Public Health Service, Ministry of Health, Jerusalem), Italy: Patrizia Trevisi; Alessandro Martini (University of Padova), Ferdinando Grandori, (Institute of Biomedical Engineering, Milan), Latvia: Sandra Kuške (Latvia Children Hearing center), Lithuania: Eugenijus Lesinskas (University Hospital Santariskiu Clinics, Vilnius), Luxembourg: Jean Marc Hild (Services Audiophonologiques, Strassen), Malta: Anthony Fenech, (Mater Dei Hospital, Msida), Moldova: Anghelina Chiaburu (Republican Center of Audiology, Chisinau), Montenegro: Ognjen Jovicevic, (Institute for Children's disease, Clinical Center of Montenegro, Podgorica), Norway: Karl Nordfalk; Sverre Medbø (Universitetssykehus, Oslo), Poland: Witold Szyfter, Grażyna Greczka (University of Medical Sciences, Poznań), Portugal: Luisa Monteiro (Lisbon), Romania: Madalina Georgescu, (University of Medicine and Pharmacy, Bucharest),Serbia: Snezana Andric Filipovic (Clinical Center of Serbia, Clinic of ENT and Maxillofacial Surgery, Belgrade), Slovakia: Gabriela Pavlovcinova, Milan Profant (University Hospital Bratislava), Slovenia: Saba Battelino; Irena Hocevar Boletezar (University Medical Center, Ljubljana), Spain: Faustino Núñez-Batalla (Hospital Universitario Central de Asturias), Oviedo Javier Cervera (Hospital Infantil Universitario Niño Jesús, Madrid), Sweden: Inger Uhlén, (Karolinska University Hospital, Stockholm), Switzerland: Dorothe Veraguth, (University Hospital Zürich), Turkey: Huban Atilla (University Ankara), UK: Gwen Carr; Adrian Davis; Adam Bruderer (UCL Ear Institute, London), Tony Sirimanna, (Great Ormond Street Hospital for Children, London)
EUS€REEN consortium: EU Public health:
Albania: Qirjazi B (Faculty of Medicine, Science Medical University of Tirana), Roshi E (Faculty of Public Health Medical University of Tirana) Belgium Flanders: Hoppenbrouwers K; Guérin C (KU University Leuven), Bulgaria: Georgieva L (Faculty of Public Health, Medical University, Sofia), Croatia: Rukavina T (University Rijeka School Medicine), Czech Republic: Bourek A (Masaryk University), Finland: Hietanen-Peltola M (The National Institute of Health and Welfare), France: Jégat C (Association Nationale pour l'Amélioration de la Vue Paris), Germany: Ottová-Jordan V(University Medical Center Hamburg-Eppendorf), Greece: Polychroniadis Scouros S (Hygeia Hospital Athens), Hungary: Kovacs A (Scientific Committee at Association of Primary Care Paediatricians), Iceland: Jónsdóttir LS (Directorate of Health), Israel: Morad Y (Department of Ophthalmology, Assaf Harofeh Medical Center, Tel Aviv University, Zrifin); Grotto I (Israel Ministry of Health), Malta: Farrugia Sant'Angelo V (Ministry for Health, Floriana, Malta), Macedonia: Memeti S (Institute of Public Health of the Republic of Macedonia), Montenegro: Mugosa B, (Institute of Public Health of Montenegro, Podgorica), Netherlands: Raat H (Department of Public Health, Erasmus Medical Center Rotterdam), Portugal: Gaspar T (Institute of Psychology and Educational Sciences, Lusiada University, Lisbon), Serbia: Zivkovic Sulovic M (Institute of public health of Serbia), Slovenia: Juricic M (University of Ljubljana, Medical Faculty), Spain: Rajmil L (Catalan Agency for Health Quality and Assessment), Sweden: Hjern A, (Karolinska Institutet), Turkey: Atilla H (Ankara University), UK: Dahlmann-Noor A (NIHR Biomedical Research Centre at Moorfields Eye Hospital and UCL Institute of Ophthalmology)
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
