Abstract
Objective
Tandem mass spectrometry is increasingly used in the Middle East in newborn screening for inborn errors of metabolism using dried blood spots. The sensitivity and specificity of this system for analyzing fatty and amino acids, screening for more than 40 metabolic conditions, is known. However, the short term stability of acylcarnitines and amino acids in dried blood spots in extreme heat and humid conditions is not well documented. We examined the short term effect of heat and humidity on the levels of 7 amino acids and 10 acylcarnitines used in newborn screening for inherited metabolic disorders.
Methods
Dried blood spots were exposed with humidity <30% to temperatures of 4℃, room temperature, 37° C, and 45℃, and also with humidity >70% at 37° C and 45℃. Amino acids and acylcarnitines in the dried blood spots were analyzed by tandem mass spectrometry.
Results
During the eight days of the study in high temperature and high humidity storage, most acylcarnitines and amino acids lost almost 50% of initial concentration. After eight days’ exposure at 37℃ and 45℃ with humidity >70%, methionine was determined to be the most sensitive, and phenylalanine and leucine were the least sensitive amino acids. At 37℃ with humidity >70% C6 was the most sensitive and free carnitine (C0) was the least sensitive acylcarnitine; at 45℃ with humidity >70% C16 was the most sensitive and C0 was the least sensitive.
Conclusion
Low humidity and low temperature conditions are required for transportation and storage of dried blood spots.
Introduction
Inborn errors of metabolism can lead to serious clinical consequences for affected neonates or young infants, and screening and early detection and treatment of inborn errors of metabolism can reduce mortality and minimize morbidity. 1 Studies conducted in some parts of the Middle East suggest that the incidences of inborn errors of metabolism are higher here than anywhere else in the world, due to the more common occurrence of consanguineous marriages.2--4
Over the last two decades, liquid chromatography tandem mass spectrometry (LC-MS/MS) has become the predominant technique for analyzing dried blood spots on Guthrie filter cards, to screen for inborn errors of metabolism, including amino acid, fatty acid, and organic acid disorders.5,6 These biomarkers in the blood spot are susceptible to environmental conditions, such as heat and humidity. Their stability is important in maintaining sample integrity for high quality analysis and measurements of amino acids and acylcarnitines in the blood spot. 7 The degradation of some of these unstable compounds presents a challenge for dried blood analysis during sample collection and storage. In many parts of the Middle East, the Guthrie cards are exposed to high temperatures (37–50℃) and high humidity (>70%) during collection and transportation, especially in remote areas, where refrigeration and immediate sample processing are unavailable. In Bahrain in summer, for example, the temperature ranges from 37℃ to 45℃, and relative humidity ranges from 30% to 73%. Temperature, humidity, and time-dependent degradation of metabolites may contribute to metabolite variability.8–12
Although many studies have reported on the long-term stability of dried blood spots in high temperatures and humidity, information regarding the short-term stability of amino acids and acylcarnitines in the blood spot is limited. We investigated the short-term stability of amino acids and acylcarnitines in environmental extremes, such as high temperatures and high humidity, which can be encountered during the collection and delivery of samples to our newborn screening laboratory in Bahrain.
Methods
All blood spots on the filter cards were prepared from a single fresh sample of whole human blood, adjusted to a hematocrit of 50 ± 2.0%, similar to the hematocrit of newborns. The whole blood then was dispensed in 75 -µL portions onto the Guthrie filter cards (903 Whatman paper, Germany) using an automatic pipette. The blood was allowed to thoroughly saturate the paper, and was air dried for a minimum of three hours, then separated and kept in zip-closure plastic bags and exposed to conditions that might be encountered in transit and storage in the laboratory. To study the stability of metabolic markers at room temperature and at 4℃, with no control of humidity, the blood spots were kept in the zip-closure bag without desiccant packets. At 37℃ and 45℃, with controlled humidity of less than 30%, the blood spots were kept in the zip-closure plastic bags with desiccant packets, with humidity indicator cards (blue, below 30%). At 37℃ and 45℃ with high humidity, the blood spots were kept in open bags in a high-humidity chamber, and the humidity was monitored by periodic hygrometer readings to maintain relative humidity above 70%.
The blood spots were first analyzed at baseline, before being exposed to different conditions, and then subsequently analyzed by LC/MS-MS at 24 hour intervals in the eight days after being kept at different conditions. Analysis of the blood spots was performed using commercially available LC-MS/MS kits (MassChrom®, Chromsystems, Germany).
The concentrations of some of the acylcarnitines in the blood were very low. Therefore 10 acylcarnitines were investigated, representing short, medium, and long chain fatty acids disorders. Low and high controls were used with each batch of samples. All samples in each complete time and temperature set were analyzed in triplicate.
Statistical analysis
The intra-assay and inter-assay (eight days) coefficients of variation for determination of all amino acids in the blood spot were less than 3.4% and 11.8% for low controls, and 6.6% and 11.6% for high controls. The intra-assay and inter-assay (eight days) coefficients of variation for determination of free carnitine (C0) and acylcarnitines in the blood spot were less than 6.6% and 16.4% for low controls and 3.7% and 10.7% for high controls.
The effect of time and storage of the blood spots on the concentration of amino acids and acylcarnitines in the refrigerator (4℃), at room temperature (25℃), at 37℃, and 45℃ with humidity less than <30%, and at 37℃ and 45℃ with humidity >70%, were assessed by comparing the mean percentage losses of amino acids and acylcarnitines after one and eight days.
Results
Figure 1a and b present the thermal stability data observed for amino acids and acylcarnitines, illustrating the degradation of these metabolites at 45℃ with humidity >70% within eight days. The percentage of most amino acids and all acylcarnitines recovered declined sharply during the first day of storage, but this decline was not observed during remaining eight days of the study.
Degradation of (a) amino acids and (b) acylcarnitines at 45℃ with humidity >70% within eight days.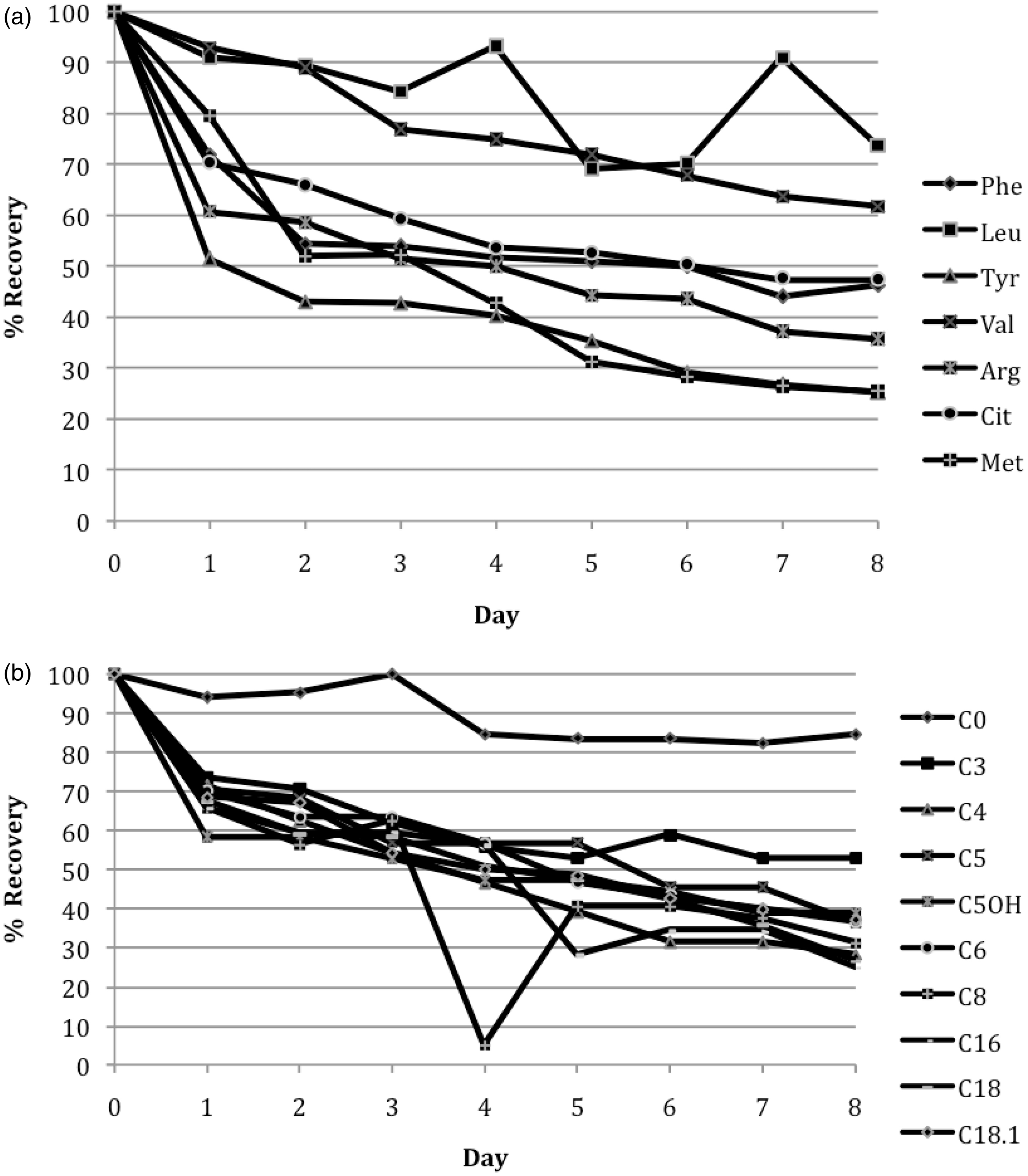
The mean percentage loss of amino acids and acylcarnitines after one and eight days’ exposure of the blood spots to different temperatures and humidity levels.
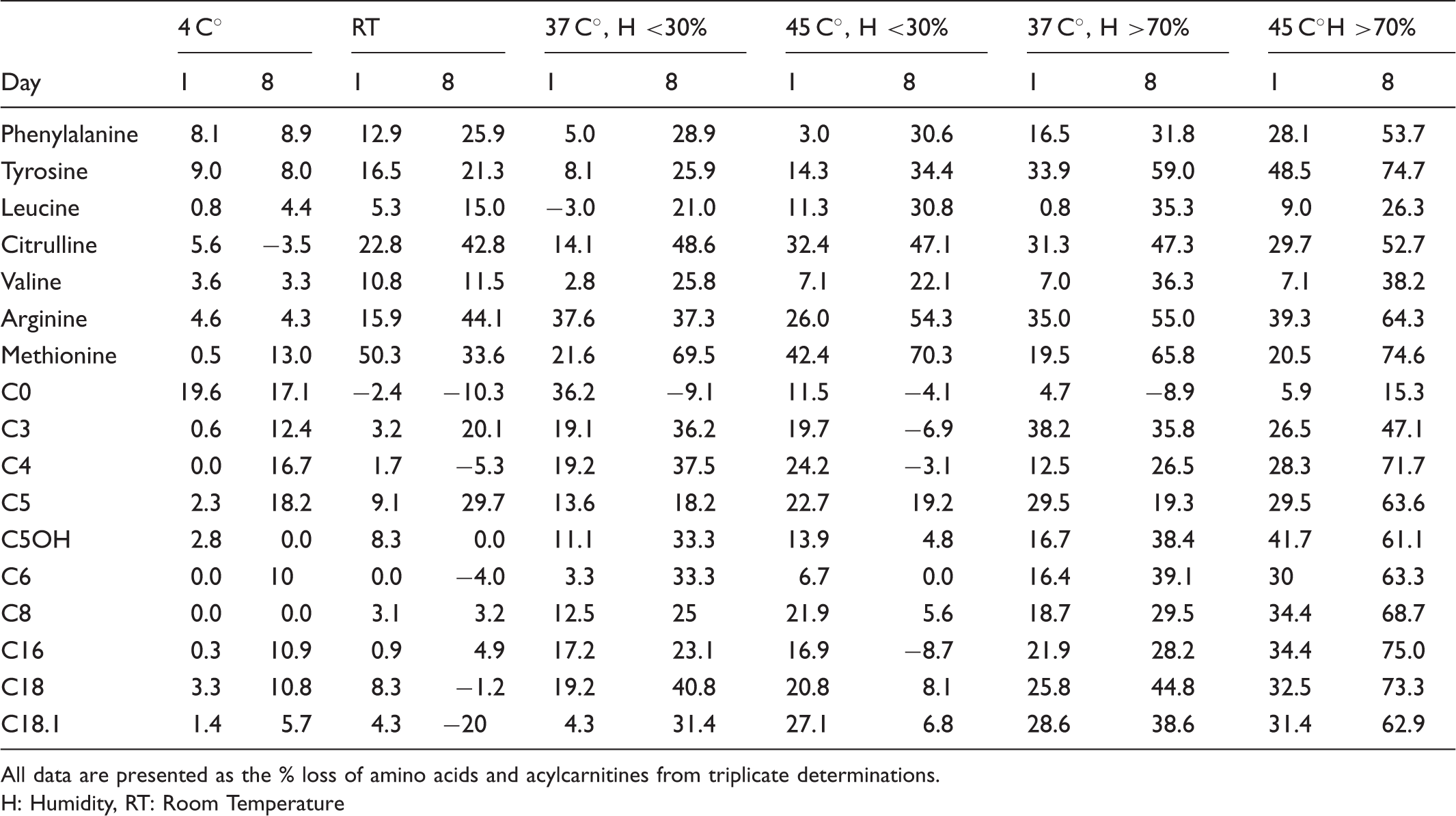
All data are presented as the % loss of amino acids and acylcarnitines from triplicate determinations.
H: Humidity, RT: Room Temperature
One day of exposure
After one day of exposure to 4℃, tyrosine was the most sensitive, and methionine was the least sensitive; at room temperature, methionine was the most sensitive, and leucine was the least sensitive; at 37℃ with <30% and >70% humidity, arginine was the most sensitive, and leucine was the least sensitive; at 45℃ with humidity <30, citrulline was the most sensitive, and phenylalanine was the least sensitive; and at 45℃ with humidity >70%, tyrosine was the most sensitive, and valine was the least sensitive amino acid.
Results on losses of acylcarnitine concentrations after one day of exposure showed that at 4℃, C0 was the most sensitive and C4, C5, and C6 were the least sensitive; at room temperature, C5 was the most sensitive, and C0 was the least sensitive; at 37℃ with humidity <30%, C0 was the most sensitive, and C6 was the least sensitive; at 37℃ with humidity >70%, C3 was the most sensitivem and C0 was the least sensitive; at 45℃ with humidity <30%, C18.1 was the most sensitive, and C6 was the least sensitive; and at 45℃ with humidity >70%, C5OH was the most sensitive, and C0 was the least sensitive.
Eight days of exposure
After eight days of exposure at 4℃, methionine was the most sensitive, and citrulline was the least sensitive; at room temperature, citrulline was the most sensitive, and valine was the least sensitive; at 37℃ with <30% humidity, methionine was the most sensitive, and valine was the least sensitive; at 37℃ with humidity >70% methionine was the most sensitive, and phenylalanine was the least sensitive; at 45℃ with humidity <30 methionine was the most sensitive, and valine was the least sensitive; and at 45℃ with humidity >70% methionine was the most sensitive, and leucine was the least sensitive amino acid.
Results on losses of acylcarnitine concentrations after eight days of exposure show that C5 was the most sensitive, and C5OH and C8 were the least sensitive; at room temperature C0 was the most sensitive, and C6 was the least sensitive; at 37℃ with humidity <30%, C18 was the most sensitive, and C0 was the least sensitive; at 37℃ with humidity >70%, C6 was the most sensitive, and C0 was the least sensitive. At 45℃ with humidity <30%, C5 was the most sensitive, and C16 was the least sensitive; at 45℃ with humidity >70%, C16 was the most sensitive, and C0 was the least sensitive.
Discussion
Time-dependent degradation of metabolites during short-term storage of dried blood filter cards at room and high temperatures, with low or high humidity, may contribute to false negative results. Although there are number of studies reporting the long-term stability of amino acids and acylcarnitines,8,9,12,13 there is limited data available on the short-time stability of these metabolites in dried blood filter cards at extreme heat (37℃–45℃) and humidity (>70%), conditions encountered during summer transit of dried blood spots in the Middle East.
In this study most of the degradation of amino acids and acylcarnitines was attributable to the adverse effects of high temperature and humidity. As illustrated in Table 1, degradation of all amino acids was greater at high humidity compared with low humidity, irrespective of storage temperature. Consistent with the results from this study, methionine has been reported to be the least stable amino acid. 9 Degradation of amino acids with high humidity is consistent with other studies reported that high humidity can influence dried blood spots.14–15
Our results suggest that free carnitine (C0) was the least sensitive to 37℃ and 45℃ temperatures combined with humidity >70% over a period of eight days. The stability of free carnitine could be due the slight increase of its concentration by hydrolysis of esterified acylcarnitines. A previous study has reported a 20–40% increase in free carnitine after one year of storage.16–17 In addition, Adam et al. 15 reported an increased C0 concentration during a 35-day study. However, the concentration of free carnitine could also be affected by hydrolysis of acylcarnitines during sample preparation. 16
Although it was expected that the degradation of acylcarnitines would increase with increasing storage temperature, our results suggest that degradation of acylcarnitines was significantly greater at high humidity than low humidity, irrespective of the storage temperature. The instability of acylcarnitines at 37℃ in a high-humidity environment has also been previously observed.13,15 Moisture is reported to affect the quality of blood samples, by altering extraction efficiency during analysis, or facilitating degradation of unstable analytes. 7
As illustrated in Figure 1, our results suggest a significant degradation of amino acids and acylcarnitines in the blood spot at high temperature (45℃) and humidity (>70%), particularly in the first day of storage. These conditions mimic the climate in much of the Middle East during summer, and the degradation of amino acids and acylcarnitines at high temperature and humidity could occur in the first day transit of blood spots. It could be avoided by utilizing climate controlled cargo compartments, or insulated delivery boxes containing cool packs. However, if there is no practical way to avoid these adverse conditions during blood spot transit, there should be guidelines for any newborn screening programme, to cope with these seasonal factors and the effect they may have on the integrity of the blood spots, and the accuracy of specimen classification.
One of the most important observations from the results of this study is the disproportional loss of amino acids and acylcarnitines at high humidity and temperature in the early phase after collection. Seasonal variations in the transit environment could have an impact on the informative ratios of these metabolic markers. As illustrated in Figure 1a, during one day of storage at 45℃ and high humidity, the amount of phenylalanine declined by about 20%, whereas tyrosine declined by about 50%. This difference between the stability of phenylalanine and tyrosine could affect the phenylalanine/tyrosine ratio used to identify phenylketonuria (PKU) carriers and to differentiate between PKU and non-PKU hyperphenylalanemia. 18 In addition, high temperatures and humidity could have an impact on the ratios of C0/acylcarnitines (e.g. C0/C16 + C18, carnitine palmitoyl transferase deficiency), and also the ratios of acylcarnitine/acylcarnitine (e.g C4/C3, short chain acyl-CoA dehydrogenase deficiency), used as parameters in the interpretation of abnormal results and differential diagnosis of fatty acid and organic acid disorders. 19
One of the limitations of this study is the lack of stability data in the blood spot samples with high amino acids and acylcarnitines and low C0, as this study was only carried out on a single blood sample from a normal individual. Further study may be required to investigate the stability of these metabolic markers in blood spot samples, to see whether the same rate of amino acid and acylcarnitine degradation is seen with increasing temperature and humidity.
In conclusion, our results suggest that the integrity of dried blood spots can be compromised within a short time frame by humidity and temperature during transportation of the samples. Blood spots transferred to the newborn screening laboratory should be properly packed and kept cool, minimizing both humidity and temperature, and should be transferred to the laboratory immediately.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
