Abstract
Bronchiolitis obliterans syndrome (BOS) is a severe pulmonary complication following allogeneic hematopoietic stem cell transplantation (allo-HSCT) with limited therapeutic options once refractory to standard immunosuppression. We conducted a pilot study from January 2018 to December 2024, enrolling consecutive patients with BOS defined by NIH criteria who failed glucocorticoids and calcineurin inhibitors for ≥4 weeks. Sixteen patients received salvage therapy with ruxolitinib 5 mg twice daily and nintedanib 150 mg twice daily (RN cohort) in continuous 28-day cycles for up to six cycles, while 37 contemporary patients served as controls. At baseline, NIH lung scores in the RN cohort were 18.8% NIH 1, 18.8% NIH 2, and 62.5% NIH 3. The median number of treatment cycles was 3.5 (range, 1–6). At 3 months, 11 patients (68.8%) achieved ≥10% improvement in %FEV1 from baseline (median = 26.5%, range = 15.6%–58.2%). By NIH lung response criteria, the overall response rate (ORR) was 62.5% (12.5% complete response, 50.0% partial response) in the RN cohort versus 13.5% (5.4% complete, 8.1% partial) in controls. Notably, hematologic toxicities were less frequent with RN therapy than in controls. These findings suggest that low-dose ruxolitinib combined with nintedanib is an effective and well-tolerated salvage regimen for BOS after allo-HSCT and warrant confirmation in a prospective phase II study.

Introduction
Allogeneic hematopoietic stem cell transplantation (allo-HSCT) is often complicated by transplant-related morbidities, among which graft-versus-host disease (GVHD) remains the most significant1–6. Pulmonary manifestations of chronic GVHD are common and frequently culminate in bronchiolitis obliterans syndrome (BOS), a progressive and typically irreversible small airway disorder characterized by fibrotic obstruction of terminal bronchioles7–9. The immunopathogenesis of BOS after allo-HSCT is not fully understood; however, it is thought to reflect the interplay between donor T and B lymphocytes, autoantibody production, and dysregulated fibrotic pathways, resembling features of autoimmunity and aberrant wound healing10–12. Diagnosis of BOS is particularly challenging given its nonspecific clinical presentation and the absence of sensitive early biomarkers13,14. First-line therapies, including systemic corticosteroids and calcineurin inhibitors (CNIs), which remain the mainstay of chronic GVHD management, often fail to provide durable benefit in patients with BOS. This therapeutic gap underscores the urgent need for novel and effective treatment strategies15–21.
Several emerging interventions have been evaluated beyond supportive therapies, including inhaled corticosteroids and the fluticasone–azithromycin–montelukast (FAM) regimen. Second-line therapies are being introduced earlier in the disease course, often after a short nonresponsive course to systemic corticosteroids. These include Janus kinase (JAK) inhibitors such as ruxolitinib, Rho-associated kinase inhibitors such as belumosudil, and CSF1R inhibitors such as axatilimab22–25. Among these, ruxolitinib has shown particular promise23,26–32. In a recent phase II multicenter trial (NCT03674047), ruxolitinib at 10 mg twice daily achieved encouraging best lung-specific overall response rate (ORR) 34.7% and allowed most patients to taper or discontinue corticosteroids 27 .
Parallel to immunomodulation, antifibrotic therapy has emerged as a promising strategy for inflammatory and fibrotic lung complications in the post-transplant setting. The STOP-BOS trial of pirfenidone in BOS demonstrated acceptable safety and signals of lung function stabilization in patients following allo-HSCT, with 41.3% of participants showing improvement in individual pulmonary function test trajectories, supporting the biological rationale for targeting fibrotic pathways33,34.
Nintedanib, an intracellular tyrosine kinase inhibitor targeting multiple pro-fibrotic pathways, has been demonstrated to slow the decline of lung function in idiopathic pulmonary fibrosis and other fibrosing interstitial lung diseases35–37. Preliminary clinical experience, including a case report of BOS improved with nintedanib, supports its potential role in the transplant setting 38 .
Since the formal approval of ruxolitinib in China in 2017, we conducted a pilot study to evaluate the combination of low-dose ruxolitinib (5 mg orally twice daily) with nintedanib in patients with BOS refractory to glucocorticoids and CNIs after allo-HSCT. Our objective was to explore whether dual targeting of immune-mediated injury and fibrotic progression could provide a more effective salvage approach for this devastating complication.
Methods
Patients
From January 2018 to December 2024, consecutive patients with BOS who provided written informed consent were enrolled at Peking University People’s Hospital. The inclusion criteria were: (1) a diagnosis of BOS according to the EBMT guidelines; (2) refractoriness to at least 4 weeks of standard therapy with glucocorticoids and CNIs 12 ; (3) not enrolled in investigational trials for chronic GVHD—such as those evaluating ROCK2, ROCK/JAK, or CSF1R inhibitors; and (4) no evidence of recurrent primary hematologic disease. Exclusion criteria included uncontrolled infections.
Study design
Enrolled eligible patients received salvage treatment with low-dose ruxolitinib (5 mg orally twice daily) combined with nintedanib (150 mg orally twice daily) continuously in 28-day cycles for up to six cycles (RN cohort). Contemporary patients with refractory BOS who did not enroll in RN cohort nor other cGvHD trials received the best available treatments (BATs) served as controls. The FAM regimen (fluticasone, azithromycin, and montelukast) was allowed as supportive therapy4,6,12,39 (Fig. 1).

Study design and patient enrollment flowchart. Schematic overview of the study design and patient selection. A total of 53 eligible patients were enrolled and divided into two cohorts: the RN cohort (ruxolitinib plus nintedanib, n = 16) and the control cohort (n = 37). Patients were evaluated for pulmonary function, treatment response, adverse events, and survival outcomes.
End points and definitions
The primary endpoint was the 3-month ORR after initiation of salvage treatment or best available therapies (BATs), defined as a ≥10% improvement in forced expiratory volume in 1 second (FEV1). Secondary endpoints included the best overall response rate (BOR), 3-year non-relapse mortality (NRM), 3-year overall survival (OS), and treatment-related toxicities (TRTs). The start date for all endpoints was defined as the first day of salvage treatment or BATs. ORR was determined by the proportion of patients achieving a complete or partial response, based on the percentage change in FEV1 relative to baseline. BOR was defined as the best response achieved during treatment. NRM was defined as death without evidence of relapse or disease progression, while OS was defined as the time from treatment initiation to death from any cause. TRTs referred to adverse events attributed to the study regimen.
Statistical analysis
Categorical variables were compared using the chi-square test or Fisher’s exact test, as appropriate. Continuous variables were analyzed using either the independent-samples t-test or the Mann–Whitney U test, depending on the data distribution. Overall survival was estimated using the Kaplan–Meier method, and differences between groups were assessed by the log-rank test. The cumulative incidences of ORR, relapse, and NRM were evaluated within a competing risks framework. For ORR, relapse and death were treated as competing events. For relapse and NRM analyses, each event was considered in turn with the other treated as a competing risk. All statistical tests were two-sided, with P values <0.05 considered significant. Results are presented with 95% confidence intervals (95% CI). Analyses were performed using R software (version 4.4.3).
Results
Patient characteristics
Between January 2018 and December 2024, a total of 53 patients with BOS were enrolled, including 16 in the ruxolitinib plus nintedanib (RN) cohort and 37 who received BATs in the control group. Baseline characteristics are summarized in Table 1 and Fig. 2a. The median age of the entire cohort was 33 years (range, 7–57 years). Survivors had a median follow-up of 21.3 months after BOS diagnosis (range, 1.8–57.1 months) as of the data cutoff on 10 August 2025.
Characteristics of the enrolled patients.

HCT-CI: hematopoietic cell transplantation comorbidity index; HLA: human leukocyte antigen; AML: acute myeloid leukemia; ALL: acute lymphoblastic leukemia; MDS: myelodysplastic syndromes; MNC: mononuclear cells; NIH: National Institutes of Health; GvHD: graft-versus-host disease; FEV1: forced expiratory volume in 1 second; MMF: mycophenolate mofetil.

Swimmer plot in the RN group. Treatment course and outcomes of the 16 patients in the RN cohort. Three deaths occurred during follow-up: two due to non-relapse mortality and one due to disease relapse. The swimmer plot illustrates treatment duration, timing of response, and long-term disease control.
The median interval from transplantation to BOS diagnosis was comparable between the RN and control cohorts, at 11.1 months (range, 3.2–35.1 months) versus 10.9 months (range, 2.6–32.3 months), respectively (P = 0.581). The median baseline FEV1 as a percentage of predicted was 38.9% (range, 22.4%–70.3%) in the RN cohort and 36.0% (range, 15.6%–79.5%) in the control cohort (P = 0.257). Based on NIH lung score, patients in the RN cohort were distributed as NIH 1 (18.8%), NIH 2 (18.8%), and NIH 3 (62.5%), while in the control cohort the distribution was NIH 1 (10.8%), NIH 2 (32.4%), and NIH 3 (56.8%) (P = 0.480) (Fig. 3a).

Primary outcomes. (a) NIH lung score based on %FEV1 predicted at baseline in RN and control cohorts. (b) Three-month change in %FEV1 from baseline in the RN cohort. (c) Three-month NIH lung-specific overall response rate (ORR) in RN and control cohorts. (d) Best %FEV1 change from baseline in the RN cohort. (e) Best NIH lung-specific ORR in RN and control cohorts.
Patients in the RN cohort received a median of 3.5 treatment cycles (range, 1–6). In the control cohort, BATs primarily consisted of CNIs (100%), with additional use of mycophenolate mofetil (27%), ruxolitinib (41%), and imatinib (8%).
A direct comparison between ruxolitinib (n = 14) and RN (n = 16) was summarized in Supplemental Table 1. The two groups were well balanced with respect to baseline pulmonary disease severity, showing similar distributions of NIH BOS grades and comparable lung function at study entry. Median baseline FEV1 was 38.9% (range, 22.4–70.3) in the RN group and 38.6% (range, 26.9–79.5) in the ruxolitinib group (P = 0.755) (Supplemental Figure 1A). The median interval from BOS diagnosis to treatment initiation was also similar between groups. Other baseline characteristics were generally comparable.
Key efficacy outcome parameters
At 3 months, 11 patients in the RN cohort (68.8%) achieved a ≥10% improvement in FEV1 compared with baseline, with a median increase of 26.5% (range, 15.6%–58.2%). Two patients experienced a ≥5% decline (Patient 2: baseline 38.2%, 3-month 31.0%; Patient 7: baseline 39.6%, 3-month 23.9%), and three patients remained stable (Patients 9, 15, and 16: baseline 60.0%, 68.9%, and 23.6%; 3-month 64.6%, 68.4%, and 23.6%, respectively) (Fig. 3c). Based on NIH lung response criteria, the 3-month ORR in the RN cohort was 62.5% (12.5% complete response [CR], 50.0% partial response [PR]), with 37.5% achieving stable disease and no patients showing progression. In contrast, the control cohort had an ORR of only 13.5% (5.4% CR, 8.1% PR), with 64.9% remaining stable and 21.6% worsening (Fig. 3). RN was associated with a numerically higher lung-specific response than ruxolitinib alone, with CR/PR rates of 12.5%/50.0% versus 7.1%/7.1%, respectively; stable disease occurred in 37.5% versus 57.1%, and lung function worsening in 0% versus 28.7% (Supplemental Figure 1B).
When the best FEV1 improvement during treatment was considered, 11 patients in the RN cohort demonstrated a ≥10% increase from baseline (median 30.6%, range, 15.6%–58.2%), 2 patients showed a ≥5% decline, and 3 remained stable. According to NIH lung response criteria, the best ORR in the RN cohort was 62.6% (18.8% CR, 43.8% PR), with 37.5% remaining stable and no patients worsening. The median best improvements in FEV1 during treatment for the NIH 1, 2, and 3 groups were 15.6%, 33.6%, and 21.6%, respectively (Supplemental Table 2). By comparison, the control cohort again achieved an ORR of only 13.5% (5.4% CR, 8.1% PR), with 64.9% (24/37) remaining stable and 21.6% (8/37) progressing (Fig. 3d, e and Supplemental Figure 1C).
Long-term survival outcomes
At 1 year, the overall survival (OS) rate was 83.5% (95% CI, 72.3–96.5) in the control cohort compared with 93.3% (95% CI, 81.5–100) in the RN cohort. At 3 years, OS declined to 60.4% (95% CI, 46.2–79.0) in the control group and 70.0% (95% CI, 45.9–100) in the RN group (P = 0.4). In the control cohort (n = 37), 16 events occurred, with the lower bound of the 95% CI for median OS at 29.4 months. In the RN cohort (n = 14), three events were recorded, with the lower bound at 19.7 months. Median OS was not reached in either group (Fig. 4a).
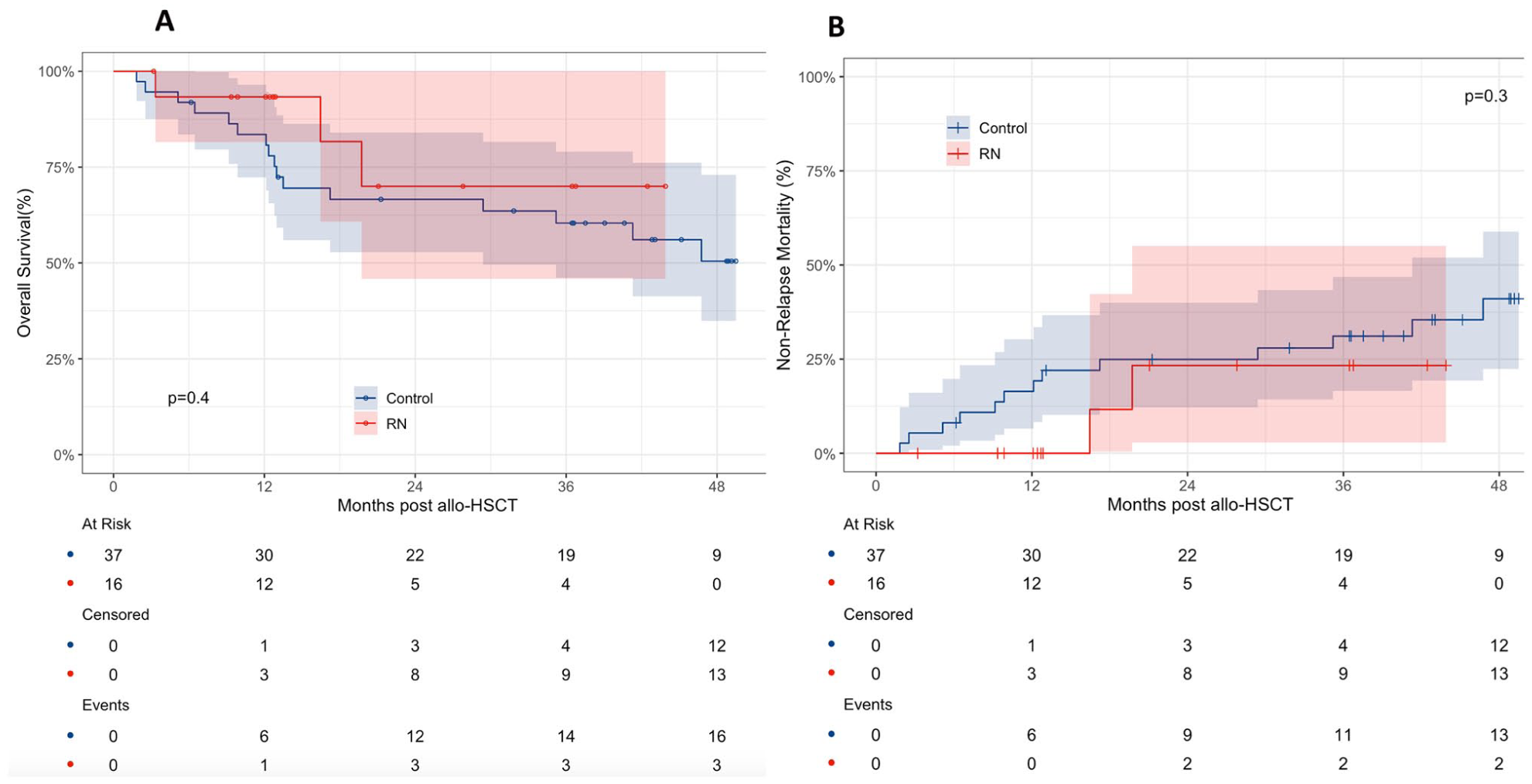
Survival. (a) Kaplan–Meier curves for overall survival (OS) in RN and control cohorts. (b) Cumulative incidence of non-relapse mortality (NRM) in RN and control cohorts.
At 1 year, the non-relapse mortality (NRM) rate was 16.5% (95% CI, 6.6–30.3) in the control cohort versus 0% in the RN cohort. By 2 years, NRM increased to 24.9% (95% CI, 12.2–40.0) in the control cohort and 23.3% (95% CI, 2.8–55.1) in the RN cohort, with no further rise through 4 years in the RN group. At 3 years, cumulative NRM reached 31.1% (95% CI, 16.6–46.8) in the control cohort (P = 0.3) (Fig. 4b). Notably, two non-relapse deaths occurred in the RN cohort during the second year after discontinuation of RN regimen, both attributed to acute COVID-19 infection. Given the small sample size of the RN cohort (n = 16), these events had a disproportionate impact on cumulative NRM estimates and likely contributed to the apparent reduction in OS in this group.
Toxicities
Treatment-related toxicities are summarized in Table 2. Patients in the RN cohort received low-dose ruxolitinib, which was associated with a lower incidence of hematologic adverse events compared with the control cohort. Rates of any-grade anemia, thrombocytopenia, and neutropenia were 18.7%, 31.2%, and 18.7% in the RN cohort, versus 46.0%, 48.6%, and 21.6% in the control cohort, respectively. Grade ≥3 hematologic toxicities were also less frequent in the RN cohort: 6.0% for anemia and neutropenia and 18.7% for thrombocytopenia, compared with 16.2%, 10.8%, and 27.0% in the control cohort.
Toxicity profile of RN regimen versus best available therapies.

Non-hematologic toxicities, including diarrhea, vomiting, and pneumonia, were generally comparable between the two cohorts and were predominantly low grade. Hyperbilirubinemia was observed in 25.0% of patients in the RN cohort and 43.0% in the control cohort, with grade ≥3 events occurring in 6.0% and 10.8% of patients, respectively.
Discussion
BOS is a devastating, progressive, late-onset pulmonary complication following allo-HSCT. It is characterized by irreversible airflow obstruction and is associated with poor prognosis. During the period when this study was initiated, therapeutic strategies for pulmonary chronic GVHD, including BOS, were highly heterogeneous across centers, and no universally accepted standard of care was established. Conventional immunosuppressive approaches at that time frequently included systemic or inhaled corticosteroids, sometimes combined with azithromycin and/or montelukast, as well as other agents used for chronic GVHD. However, these treatments were largely empirical and often associated with limited efficacy, substantial toxicities, and high failure rates, reflecting the incomplete understanding of the underlying immunopathological mechanisms of pulmonary chronic GVHD. Consequently, the development of novel, effective, and well-tolerated salvage therapies for BOS represents an urgent unmet clinical need7–10,38,40,41.
This study is, to our knowledge, the first to evaluate the combination of ruxolitinib and nintedanib (RN regimen) for the treatment of steroid-refractory BOS. Ruxolitinib, a selective JAK1/2 inhibitor, exerts potent anti-inflammatory and immunomodulatory effects and has demonstrated clinical activity in chronic GVHD23,26–30. Nintedanib, a multi-targeted tyrosine kinase inhibitor of VEGFR, FGFR, and PDGFR, has shown antifibrotic and anti-inflammatory efficacy in idiopathic pulmonary fibrosis and systemic sclerosis–related interstitial lung disease. While both agents have mechanistic plausibility and have been independently investigated in the settings of chronic GVHD and fibrotic lung disease, their concurrent use in BOS has not previously been reported. Given that BOS involves both immune-mediated inflammation and progressive airway fibrosis, the rationale for combining these two agents is strong and mechanistically complementary.
The efficacy of ruxolitinib in BOS has been explored in several clinical settings. Zhao et al. 30 firstly reported in a retrospective study that ruxolitinib as salvage therapy for BOS achieved a best overall response rate of 66.7%, with three patients (10.0%) attaining complete remission and 17 patients (56.7%) achieving partial remission. In the REACH3 trial of steroid-refractory chronic GVHD, ruxolitinib 10 mg twice daily achieved a superior ORR at week 24 compared with control (49.7% vs. 25.6%), but the lung-specific benefit was limited, with improvement observed in only 8.6% of patients with pulmonary involvement 29 . More recently, a multicenter phase II trial (NCT03674047) in BOS reported a best lung-specific ORR of 34.7% among 49 participants (16.3% complete responses and 18.4% partial responses) 27 . These findings indicate that while ruxolitinib has activity in BOS, its efficacy as monotherapy remains modest, particularly for fibrotic airway disease.
Evidence for the use of nintedanib in BOS is even more limited. Nintedanib has been extensively validated in idiopathic pulmonary fibrosis and other progressive fibrosing interstitial lung diseases, where it consistently slows functional decline 35 . Of particular note, a single case report described significant improvement in lung function in a post-transplant BOS patient treated with nintedanib monotherapy, including a marked increase in FEV1 38 . While anecdotal, this observation supports the potential role of nintedanib in managing fibrotic airway disease after transplantation, although robust clinical evidence is still lacking.
In contrast to the monotherapy approaches described above, our study demonstrates that combining low-dose ruxolitinib with nintedanib can simultaneously address both the inflammatory and fibrotic components of BOS pathophysiology. This dual-targeted strategy was associated not only with meaningful improvements in lung function but also with an improved safety profile. Hematologic toxicities were reduced in the RN cohort, with any-grade anemia, thrombocytopenia, and neutropenia occurring in 18.7%, 31.2%, and 18.7% of patients, respectively, compared with 46.0%, 48.6%, and 21.6% in the control group. Rates of grade ≥3 hematologic adverse events were likewise lower in the RN cohort. Collectively, these findings highlight the therapeutic potential of a dose-optimized, multi-targeted combination regimen as a promising salvage strategy for BOS.
The RN regimen demonstrated encouraging pulmonary benefits, with 68.8% of patients achieving a ≥10% increase in FEV1 at 3 months and an NIH-defined lung-specific ORR of 62.5%, compared with 13.5% in the control group. No patients in the RN cohort deteriorated, whereas about one-fifth in the control group showed worsening disease, suggesting that this regimen may effectively stabilize or even reverse pulmonary decline in steroid-refractory BOS. We acknowledge that the ORR observed in the ruxolitinib-alone cohort in our study was lower than that reported in the phase II trial 27 , in which an overall lung response rate of 34.7% (including 16.3% CR and 18.4% PR) was achieved. This discrepancy may be explained, at least in part, by differences in treatment exposure and study design. In the phase II trial, patients were prospectively treated with ruxolitinib at a standardized dose of 10 mg twice daily for up to 12 months, whereas in our retrospective real-world cohort, the median duration of ruxolitinib therapy was approximately 4 months, which may have limited the opportunity to achieve maximal pulmonary response.
Regarding long-term survival, the RN cohort showed a lower 1-year NRM rate (0.0%) compared with 16.5% in the control group. Two deaths in the RN cohort occurred during the second year, both related to acute COVID-19 infection, which disproportionately influenced cumulative NRM estimates given the small sample size (n = 16). By 3 years, cumulative NRM was 31.1% in the control group versus 23.3% in the RN group, with no statistically significant difference. These findings emphasize the need for cautious interpretation of long-term outcomes in small cohorts, particularly when external factors such as infections may confound survival analyses.
Although the results of this study are encouraging, several limitations must be acknowledged. The trial was conducted at a single center with a relatively small sample size, which limits the application of propensity score matching or weighting approaches, as such methods would have further reduced the effective number of analyzable patients and compromised statistical power. Consequently, potential baseline imbalances between treatment groups cannot be fully excluded. Notably, the proportion of patients with NIH Score 1 was higher in the RN cohort, suggesting possible enrichment for milder disease at baseline. These imbalances may have influenced the observed outcomes and should be interpreted with caution. Treatment duration was not uniform, ranging from three to six cycles, and follow-up intervals were variable. In addition, pulmonary function was assessed over a relatively short observation window. Therefore, our findings should be regarded as hypothesis-generating and warrant validation in larger, prospective studies with appropriate baseline adjustment or stratified design.
Conclusions
Our pilot study is the first to demonstrate that concurrent administration of low-dose ruxolitinib and nintedanib is a feasible, well-tolerated, and potentially effective salvage therapy for BOS. By simultaneously targeting immune-mediated inflammation and progressive fibrosis, this dual-target regimen may provide synergistic clinical benefit. The favorable safety profile observed with the reduced ruxolitinib dose (5 mg twice daily) underscores its suitability for post-transplant patients. Future prospective phase II trials with larger sample sizes and incorporation of quality-of-life assessments are warranted to validate these finding.
Supplemental Material
sj-docx-1-cll-10.1177_09636897261438731 – Supplemental material for Salvage nintedanib plus low-dose ruxolitinib therapy for bronchiolitis obliterans syndrome refractory to calcineurin inhibitors and glucocorticoids after allogeneic transplantation
Supplemental material, sj-docx-1-cll-10.1177_09636897261438731 for Salvage nintedanib plus low-dose ruxolitinib therapy for bronchiolitis obliterans syndrome refractory to calcineurin inhibitors and glucocorticoids after allogeneic transplantation by Ya Luo, Ying Wu, Qiu-Sha Huang, Xian-Ying Yin, Qian-Nan Shang, Yi-Han Yang, Ao-Ran Zhang, Tong Su, Xiao-Han Su, Su-Xuan Liu, Zheng-Li Xu, Jing Liu, Xiao-Dong Mo, Yu-Qian Sun, Yu Wang and Meng Lv in Cell Transplantation
Footnotes
Acknowledgements
We sincerely thank Prof. Xiao-Jun Huang, Prof. Xiao-Hui Zhang, and Prof. Lan-Ping Xu for their supervision of patient care and consultation throughout the study.
Authors’ Note
Part of the data included in this manuscript was previously presented as an oral presentation at the Annual Meeting of the European Society for Blood and Marrow Transplantation (EBMT) 2024.
Ethical Considerations
The study protocol was approved by the Ethics Committee of Peking University People’s Hospital (2017PHB221) on 2 December 2017.
Consent for Publication
Written informed consent for publication of clinical data was obtained from all participants.
Author Contributions
ML led the clinical trial, oversaw patient enrollment, performed data analysis, and drafted the manuscript. YL collected and curated the data and contributed to manuscript writing. YW and Q-SH were responsible for data collection. All other authors contributed to patient care and clinical management. All authors reviewed the manuscript and approved the final version for publication.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Beijing Research Ward Excellence Program (BRWEP2024W134080100), National key research and development plan of China (2021YFA1100902), Peking University People’s Hospital (RZ2024-01), and Joint Research Project of the Shijiazhuang-Peking University Cooperation Program.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used and/or analyzed in the current study are available from the corresponding authors on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
